Abstract
The objectives of this study were to define the acute and chronic effects of 1-hour unilateral in vivo renal ischemia on renal function and histology in cats. Twenty-one adult purpose-bred research cats were anesthetized, and 1 kidney underwent renal artery and vein occlusion for 1 hour. Serum creatinine and urea concentrations, urine protein:creatinine ratio, urine-specific gravity, glomerular filtration rate, hematocrit, platelet concentration and function, and white blood cell count were measured at baseline and variable time points after ischemia. Renal histopathology was evaluated on days 3, 6, 12, 21, 42, and 70 postischemia; changes in smooth muscle actin and interstitial collagen were examined. Following ischemia, whole animal glomerular filtration rate was significantly reduced (57% of baseline on day 6; P < .05). At the early time points, the ischemic kidneys exhibited severe acute epithelial necrosis accompanied by evidence of regeneration of tubules predominantly within the corticomedullary junction. At later periods, postischemic kidneys had evidence of tubular atrophy and interstitial inflammation with significantly more smooth muscle actin and interstitial collagen staining and interstitial fibrosis when compared with the contralateral control kidneys. This study characterizes the course of ischemic acute kidney injury in cats and demonstrates that ischemic acute kidney injury triggers chronic fibrosis, interstitial inflammation, and tubular atrophy in feline kidneys. These late changes are typical of those observed in cats with naturally occurring chronic kidney disease.
Keywords
Acute kidney injury (AKI) in cats can be caused by toxic, ischemic, inflammatory, obstructive, or infectious insults. 27,40,45 Despite advances in patient care, mortality secondary to AKI is common in people, where AKI is often associated with renal ischemia. 6 In cats, mortality following AKI is similarly high, reported to be 47% to 64%. 27,40,45 The most common cause of feline AKI is reportedly nephrotoxicosis (lily ingestion, ethylene glycol, nonsteroidal anti-inflammatory drugs), 40,45 although in clinical practice the cause of AKI in affected cats is frequently unknown. 40,45
The pathophysiology of ischemic AKI in cats has been reviewed. 37 Briefly, ischemia results in depletion of intracellular ATP in tubular cells. The resultant failure of plasma membrane Na+-K+ ATPase, as well as other metabolic derangements, results in cytotoxic perturbations in ionic concentrations. Intracellular calcium concentrations increase, and cytoskeletal changes cause degradation of apical microvilli and loss of cellular polarity. Tubular cells may lose their attachments to the basement membrane and slough. There is evidence in other species that injured tubular cells release cytokines, which recruit leukocytes into the renal interstitium. 18 The resultant inflammation, which involves a number of signaling pathways, plays a critical role in the initiation and progression of AKI. Unfortunately, the sequence of events that occurs in the kidneys of cats with ischemic AKI has not been well characterized. Studies of feline AKI are limited, although prognostic factors have been proposed. 27,40,45 There is a paucity of information related to the underlying pathologic process in these patients, and little is known about the long-term consequences of AKI to renal structure in cats that survive. In people, AKI is implicated as a risk factor for subsequent development of chronic kidney disease (CKD). 1,6,12,13,33,43 Indeed, the relationship of AKI and CKD has been described as bidirectional, as (1) the presence of CKD predisposes people to AKI and (2) an occurrence of AKI is a risk factor for subsequent development of CKD. 33 The relationship between AKI and CKD in cats has not been established.
Previously, we evaluated a 60-minute bilateral renal ischemia model in a small number of cats. 39 This earlier model provided information on the pathologic consequences of renal ischemia in cats but was confounded by the presence of uremia and a coagulopathy. To allow long-term studies of changes associated with a single severe renal ischemic insult in a clinically normal cat, unilateral ischemia with a normally functioning contralateral kidney was employed. The objective of the present report is to describe the functional and histologic effects of unilateral renal ischemia in cats. Our hypothesis was that renal ischemia leads to both short- and long-term structural changes in the feline kidney.
Materials and Methods
Animals
Twenty-one adult intact male purpose-bred domestic shorthair cats were used. The cats were originally obtained from a US Department of Agriculture class A licensed dealer (Liberty Research, Waverly, NY). Approximately 10 months prior to entrance in the study, cats were inoculated with Brugia malayi as part of a project to produce microfilaria for research. 21 Cats included in the present study were selected from among those that failed to establish patent infections. In a dedicated animal care facility, cats were singly and group housed (2 or 3 cats per cage bank). For approximately 1 week following surgery, cats were housed singularly and were subsequently housed with 1 or 2 other cats, unless they were socially incompatible. A 12-hour light-dark cycle was utilized. Cats had access to free-choice water and were fed a standard feline dry laboratory diet ad libitum. Cats were socialized on a daily basis.
Cats were enrolled in this study from 2011 to 2013. As they became available, cats were enrolled nonrandomly and sequentially in groups with end points at 3, 6, 12, 21, 42, and 70 days postischemia (n = 3 cats per end point, except the 70-day end point where n = 6; Table 1). The mean ± SEM age of the cats at enrollment was 23.2 ± 1.3 months, and the mean weight was 4.6 ± 0.2 kg.
Sampling and Survival Times for Cats Following Unilateral Renal Ischemia.a

aEach × represents a time when renal functional data were collected from each cat: serum creatinine and urea concentrations, urine protein:creatinine ratio, urine-specific gravity, and plasma clearance of iohexol as an estimate of glomerular filtration rate.
For each cat, the last × in the row also corresponds to the day of euthanasia, necropsy examination, and postmortem tissue sample collection.
Prior to entrance into the study, the cats’ general health was confirmed with a physical examination, complete blood count (CBC), serum chemistry profile, and urinalysis. Animal use was approved by the University of Georgia Institutional Animal Care and Use Committee and carried out in accordance with the Animal Welfare Act in an American Association for Laboratory Animal Care–accredited facility.
Anesthesia and Surgery
Two days prior to abdominal surgery, cats were anesthetized for placement of an indwelling jugular catheter. Cats were fasted 12 hours prior to anesthesia; sedated with intramuscular acepromazine (0.01 mg/kg), buprenorphine (0.04 mg/kg), and ketamine (7 mg/kg); and induced and maintained on isoflurane gas and 100% oxygen delivered via facemask. An indwelling 20-gauge 12-cm catheter was placed in a jugular vein. This catheter was utilized for baseline glomerular filtration rate (GFR) measurements and surgical venous access and was left in place up to 6 days (cats in the 3- and 6-day groups).
Cats were again fasted 12 hours prior to abdominal surgery. Cats were premedicated with the regimen described above. In an adjacent research surgery suite, anesthesia was induced with isoflurane delivered by facemask. Cats were intubated and maintained on isoflurane delivered with 100% oxygen. Cats were moved into a sterile surgery suite after induction, and they received intravenous isotonic crystalloid fluids (0.9% NaCl) at 10 ml/kg/h throughout the procedure. Body temperature was supported with a hot water blanket.
Briefly, a ventral midline laparotomy was performed; both kidneys were inspected; and the vasculature of the left kidney was isolated near the aorta and vena cava. A nontraumatic vascular clamp was placed across the left renal artery and vein pair. Proper placement of the clamp was confirmed by visual inspection and absence of an arterial pulse distal to the clamp. A renal cortical wedge biopsy was taken from the body of the left kidney immediately after the onset of ischemia. The vascular clamp remained in place for 60 minutes. For cats in the 3-, 6-, 12-, and 21-day group, a second and third renal biopsy were obtained. The second biopsy was taken from the cranial pole of the left kidney just prior to release of the vascular clamps. Following release of the clamp, the restoration of renal blood flow was confirmed by palpation of a pulse in the renal artery distal to the site of clamping. Following 60 minutes of reperfusion, the arterial clamp was briefly applied again, and a third renal biopsy was taken from the caudal pole of the left kidney. These serial intraoperative superficial cortical biopsies comprised 36 total biopsies and represented preischemia (n = 12; 3 cats in 4 groups), 1-hour postischemia (n = 12), and 1-hour postreperfusion (n = 12) samples. Following the third biopsy, renal hemostasis was ensured and the abdomen closed routinely. Cats in the 42- and 70-day group had only a single preischemic biopsy taken to verify normal renal structure. Other than inspection to confirm normal size and shape, the contralateral (right) kidney was not disturbed.
Postoperatively, a 25-µg/h transdermal fentanyl patch (Fentanyl Transdermal System; Noven Pharmaceuticals, Weston, FL) was placed on the lateral abdomen; in cats <5 kg, half of the patch was folded on itself to allow only half of the absorptive area to contact the skin. Based on a discomfort score 38 assigned during monitoring, postoperative analgesia was augmented via transmucosal buprenorphine (0.03 mg/kg) as indicated in the 24 hours following fentanyl patch application. After that, subcutaneous hydromorphone (0.1 mg/kg) was provided, as indicated, for analgesia. Postoperatively, cats were assessed for attitude, appetite, thirst, urination, and defecation.
Renal Function
Renal function was assessed with serum creatinine and urea concentrations, urine protein:creatinine ratio, urine-specific gravity (USG), and plasma clearance of iohexol as an estimate of GFR. The above renal function data were obtained as presurgical baseline in all animals, but in cats in longer survival groups (ie, 42 and 70 days), data from early postischemia time points were not gathered (Table 1). Data were available for 6 cats at each time point for all renal function measurements, except that GFR on days 42 and 70 was performed in 3 cats each.
The GFR was determined with a 2-point decay curve.
20,22
For this procedure, iohexol (iodine, 300 mg/kg, intravenous) was administered to nonsedated cats via the medial saphenous or cephalic vein. Blood samples were drawn prior to iohexol administration and at 2 and 3 hours after administration from the indwelling jugular catheter. Plasma was separated within an hour of collection and stored at –80°C until analysis. Samples were analyzed by a commercial laboratory for determination of iohexol concentration (Diagnostic Center for Population and Animal Health, Michigan State University, East Lansing, MI). The ln[iohexol] vs time curve was plotted, and the extrapolated iohexol concentration at 0 minutes (y intercept) and the rate of elimination (Ke; the slope of the regression line) were determined by linear regression. The area under the curve (AUC) was calculated by the following equation:
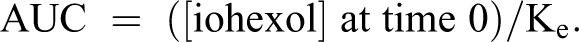
GFR (mL/min/kg) was then estimated per the calculation of iohexol clearance:
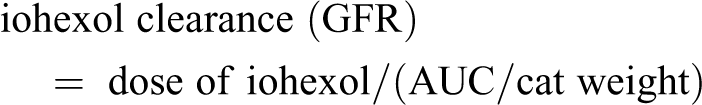
Blood Pressure
Systolic arterial blood pressure was measured by Doppler ultrasonography prior to surgery and on days 1, 3, and 6 after surgery in conscious cats in the 3-day (n = 3) and 6-day (n = 3) end point groups (6 cats total). The same cuff on the same forelimb was used on all cats, and the same operator collected these data for each cat. The mean of at least 3 readings was recorded as the value for systolic blood pressure.
CBC, Platelet Concentration, and Platelet Function
Blood was obtained for a CBC 1 day prior to surgery (baseline) and at the study end points in 15 of 21 cats. Blood was drawn into an EDTA tube or into 1.8-ml tubes containing 3.2% sodium citrate (final citrate: blood ratio of 1:9, collected in animals only when platelet function testing was performed). At each sampling period, the CBC, including a platelet count, was performed with an impedance-based methodology (CBC-Diff; Heska Corporation, Loveland, CO). Data utilized from the CBC included the hematocrit, platelet concentration, and total white blood cell count.
Whole blood (impedance) aggregometry was performed with citrated whole blood and 3 agonists (Chronolog Corp, Havertown, PA): adenosine diphosphate (ADP; 10 μM) and 2 concentrations of collagen (2 and 5 μg/ml). The blood was held at room temperature (22°C) for at least 30 minutes and no more than 60 minutes from collection, and the tubes were not agitated until gentle inversion just prior to testing. Then 500 μl of citrated whole blood and 500 μl of sterile physiologic saline for irrigation (0.9% sodium chloride) were added to a cuvette with a siliconized magnetic stir bar rotating at 1200 rpm. The cuvette and impedance probe were placed in the warmed aggregometer (37.0°C; model 700; Chronolog Corp) and observed until a stable baseline was achieved, after which 1 of the 3 agonists was added. Tracings were recorded for at least 6 minutes after addition of agonist, and at least 2 agonists were assessed in each cat. Data collected included the maximum aggregation (resistance, in Ω) and the slope of the aggregation curve.
Tissue Handling
Cats (6 cats at 70 days, 3 cats at all other periods) were euthanized by intravenous injection of euthanasia solution on postoperative days 3, 6, 12, 21, 42, and 70 for pathologic studies. A complete necropsy was performed, and samples were obtained for histopathology. All organs were evaluated grossly, and samples were taken for histopathologic evaluation. Intraoperative renal biopsies and tissue samples obtained at necropsy were fixed in 10% neutral-buffered formalin and embedded in paraffin, and 3-µm sections were stained with hematoxylin and eosin, periodic acid–Schiff and hematoxylin (PASH), Masson trichrome (TRI), and a PASH/TRI combination.
For smooth muscle actin (SMA) immunohistochemisty, the primary antibody was mouse monoclonal smooth muscle actin (1A4) antibody (202M-98; Cell Marque, Rocklin, CA). The antibody was provided as a ready-to-use reagent with a staining time of 60 minutes. The primary antibody was diluted (S0809 Antibody Diluent; Dako, Carpinteria, CA). Antigen retrieval was performed with heat-induced epitope retrieval via citrate buffer with a pH of 6.0 (HK086-9 K; Biogenex, San Ramon, CA). Endogenous peroxidase was quenched with 3% hydrogen peroxide (H312-500; Fisher Scientific, Fair Lawn, NJ). All other blocking was completed with a commercially available solution (Power Block, HK085-5 K; Biogenex). Positive tissue controls consisted of formalin-fixed, paraffin-embedded canine intestine. As a negative reagent control, the primary antibody was eliminated and substituted with purified mouse immunoglobulins in buffer (NC494 H; Biocare Medical LLC, Concord, CA). The secondary antibody used was biotinylated anti-mouse IgG, rat absorbed, made in horses (BA-2001; Vector Labs, Burlingame, CA). A streptavidin-based label was used for detection (Biocare 4Plus Streptavidin HRP Label, HP604 H; Biocare Medical LLC). The substrate chromogen system used was DAB (K3466; Dako). The tissue sections were counterstained with Gills II hematoxylin and bluing; dehydrated in alcohol levels of 70%, 95%, and 100%; cleared in xylene; and mounted with xylene-based mounting medium.
Histopathologic Analyses
Renal tissues were evaluated by a single pathologist (C.A.B.). Renal interstitial inflammatory cells were enumerated in kidneys from 21 cats with hematoxylin and eosin–stained histologic sections of the previously ischemic (left) kidney and the contralateral (right) control kidney. Plasma cells, macrophages/lymphocytes, neutrophils, and eosinophils were counted in 10 consecutive high-power fields (HPFs; 400×) within the outer cortex (OC) and midcortex (MC) and at the corticomedullary junction (CMJ). Macrophages, monocytes, and lymphocytes were counted as a single category, as they were typically present in clusters and difficult to quantitate individually. Additionally, tubular epithelial cell mitotic figures, atrophic tubular profiles, and tubules with evidence of ongoing necrosis were similarly quantified for each region. Atrophic tubular scores were determined on PASH or PASH/TRI sections and defined as tubules that were decreased in diameter with tubular basement membrane thickening and wrinkling. Tubular profiles were scored as necrotic if they exhibited acute coagulative epithelial cell necrosis with eosinophilia and cell detachment and/or subacute epithelial loss with basophilia and cell attenuation, anisokaryosis with large hyperchromatic nuclei, and variable hyperplasia with nuclear crowding. Renal arteries and arterioles were evaluated in each cat for evidence of vasculitis or hypertensive injury (hyaline or hyperplastic arteriosclerosis). For histomorphometry, sections of the ischemic and contralateral control kidney were placed in 1 block per cat to ensure similar staining intensity. Slides containing sections of the ischemic kidney and contralateral control kidney were prepared and stained with anti-SMA antibodies and PASH/TRI combination staining. The SMA staining was used to quantitate actin within cells (predominantly myofibroblasts) as a measure of profibrotic activity. The PASH/TRI combination staining was selected for quantification of interstitial collagen (IC) due to its differential staining of different types of collagen. 8 With this combination stain, basement membranes and mesangial matrix—which are composed of type IV collagen—stain purple, while fibrillar IC stains blue. Photomicrographs were taken from consecutive nonoverlapping fields of the ischemic and normal kidney sections at 100× magnification. Three images from the cortex, CMJ, and medulla were used as representative samples of each region of the ischemic and contralateral control kidneys from each cat. Histomorphometric analysis of SMA and IC was quantified with commercially available image analysis software (Image Pro Plus; Media Cybernetics, Rockville, MD). Measurements were made for each image to determine the percentage of tissue staining positive for SMA and IC in the previously ischemic and contralateral control kidneys of each cat.
Statistics
A repeated measures model was used to test for differences from baseline for various measurements that were obtained serially over time in each cat. The full model included a fixed factor of day (time after ischemia) and a random factor of cat. Multiple comparisons to baseline were adjusted with the Dunnett test. An unstructured covariance was used in all repeated measures models.
For the histomorphometric data, a 2-way analysis of variance (ANOVA) was used to test for differences in various parameters between days after ischemia and kidneys with and without ischemia (ie, ischemic kidneys vs contralateral controls). These data were assessed for each region of the kidney that had been evaluated (cortex, CMJ, and medulla) over time, and to increase power, these data were evaluated as pooled data from all 3 regions such that all regions of the kidney were included in a single analysis over time. The histomorphometric data for the various regions were also compared with an ANOVA. Post hoc analysis was performed with a Tukey test. Finally, regional data were compared between ischemic and contralateral control kidneys via a 2-sided unpaired Student t test.
For the inflammatory cell, atrophic, and necrotic tubule counts, a Student t test was used to compare the mean cell counts between ischemia and control kidneys. The folded-form F statistic was used to determine if variances were equal between ischemia and control kidneys. If unequal, then the Satterthwaite approximation for degrees of freedom for the Student t test was used.
A 2-way ANOVA was used to compare the mean cell counts between days (time after ischemia) and locations for the ischemic kidneys. If a significant interaction of days and location was present, than all individual day and location pairwise comparisons were examined.
All tests were 2-sided, and the significance threshold was set to 0.05. The repeated measures analysis was performed with PROC MIXED, and 2-way ANOVA was performed with PROC GLM in SAS 9.3 (SAS, Cary, NC). For descriptive statistics, nonparametric data were expressed as median (range), and parametric data were expressed as mean ± SEM.
Results
Clinical and Renal Function Data
The baseline physical examination results were normal in all cats, and there were no significant abnormalities in the baseline blood and urine test results. In the 6 cats in which it was measured, there was no significant difference in systolic blood pressure over time.
Renal function (Table 2) significantly declined following unilateral renal ischemia. Mean GFR dropped to 57% of baseline on day 6 and was significantly lower than baseline on days 1, 3, 6, and 12. Serum urea concentration increased significantly on days 1 and 3 to approximately 141% of baseline. Despite a significant decrease in mean GFR, the mean serum creatinine concentration was not significantly increased at these periods. A significant increase in mean serum creatinine concentration was present on day 70; cats at this point were mildly azotemic (Table 2). Similarly, the USG did not change significantly until days 42 and 70, when there was a reduction in urine concentration.
Renal Function Following Unilateral Renal Ischemia in Cats.a
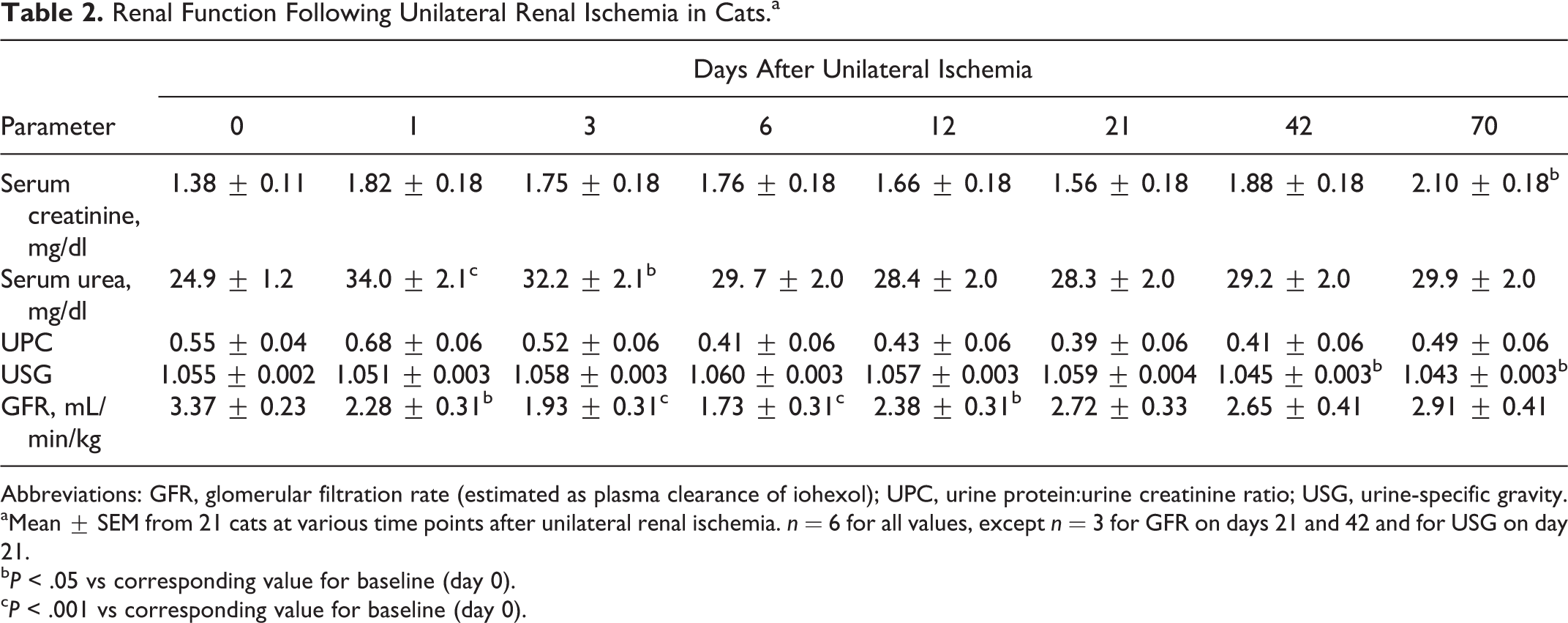
Abbreviations: GFR, glomerular filtration rate (estimated as plasma clearance of iohexol); UPC, urine protein:urine creatinine ratio; USG, urine-specific gravity.
aMean ± SEM from 21 cats at various time points after unilateral renal ischemia. n = 6 for all values, except n = 3 for GFR on days 21 and 42 and for USG on day 21.
bP < .05 vs corresponding value for baseline (day 0).
cP < .001 vs corresponding value for baseline (day 0).
There was a significant increase in white blood cell concentrations (Table 3) following surgery on days 1 and 3. The platelet count was significantly increased on day 21 compared with baseline. There was an increase (P < .05) in the slope (Ω/minute) of the ADP-induced aggregometry curve on day 3 (25.3 ± 4.1) versus baseline (15.5 ± 4.1). On day 6, there were increases (P < .05) in the slope in response to the collagen agonist at 2 µg/mL (35.7 ± 4.3 vs 22.6 ± 3.9) and 5 µg/mL (33.7 ± 3.5 vs 20.5 ± 3.1).
Hematology Results Following Unilateral Renal Ischemia in Cats.a

Abbreviation: WBC, white blood cell count.
aMean ± SEM data from 15 cats at various time points after unilateral renal ischemia.
bP < .05 vs corresponding value for baseline (day 0).
cP < .01 vs corresponding value for baseline (day 0).
Descriptive Pathology
Regarding perioperative samples, serial intraoperative superficial cortical biopsies (preischemia, 1 hour postischemia, and 1 hour postreperfusion) demonstrated subtle morphologic changes of proximal tubular injury characterized predominantly as brush border attenuation, as previously described. 39
At necropsy, the ischemic kidneys could be grossly distinguished from the contralateral controls at all periods. Ischemic kidneys on days 3, 6, and 12 were pale, yellow, and swollen (Fig. 1) with hyperemia at the CMJ. The pallor in the ischemic kidney was progressively less pronounced at days 21, 40, and 70. Additionally, ischemic kidneys at day 70 exhibited mild surface pitting. The renal papillae and pelves were grossly normal in all kidneys.
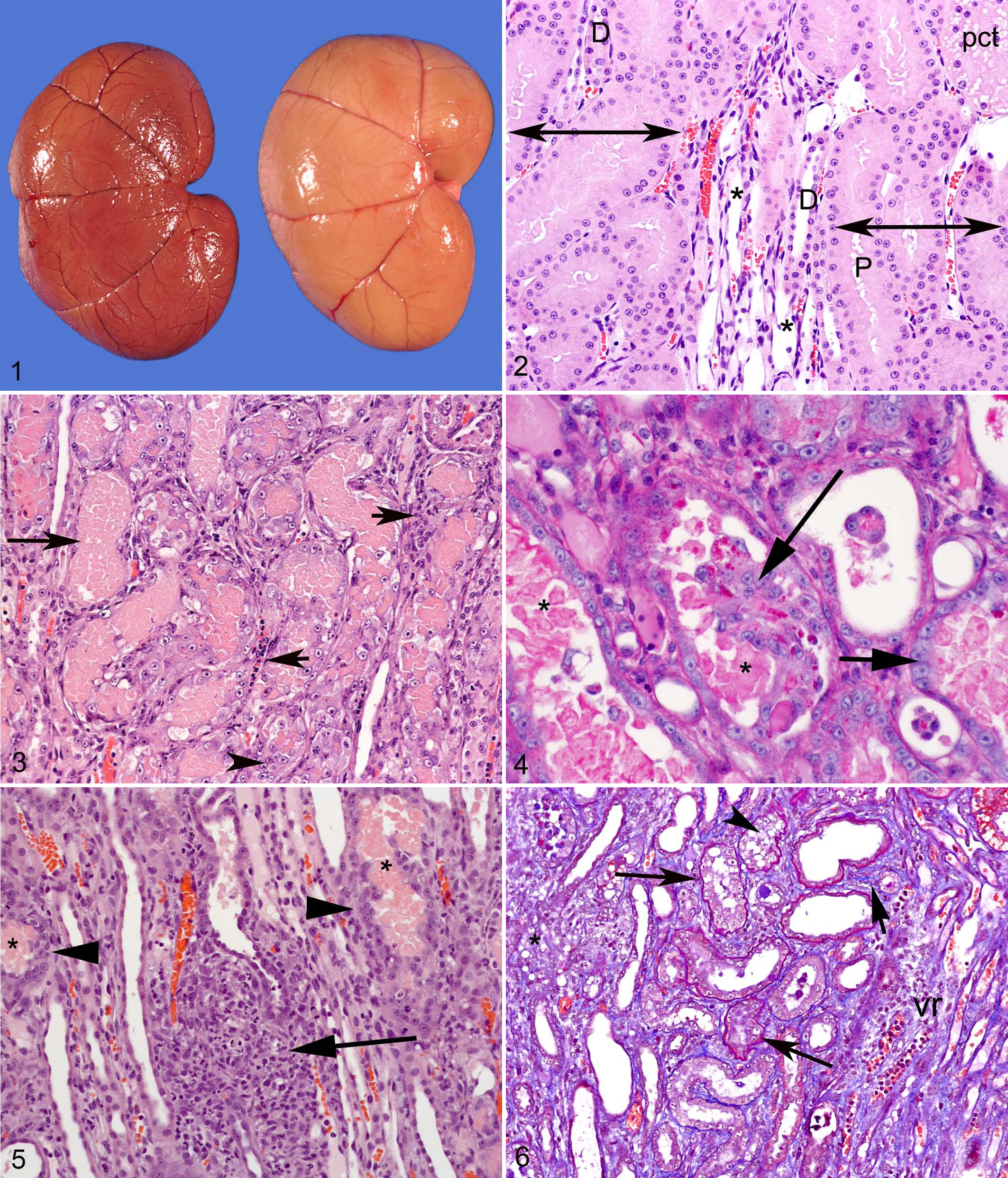
Kidney, cat, 6 days after unilateral ischemia. Pallor and swelling of ischemic kidney (right), compared with contralateral control kidney (left).
Histologically, kidneys subjected to 1 hour of ischemia had significantly more tubulointerstitial lesions than contralateral control tissue for all variables (Table 4). Renal lesions were consistent across animals euthanized on the same day postischemia, although there was variability in severity. At all periods postischemia, renal lesions were more severe within the CMJ than in the OC or MC.
Prevalence of Inflammation and Tubular Changes (Counts per 10 High-Power Fields) in Feline Kidneys: Previously Ischemic and Contralateral Control.a
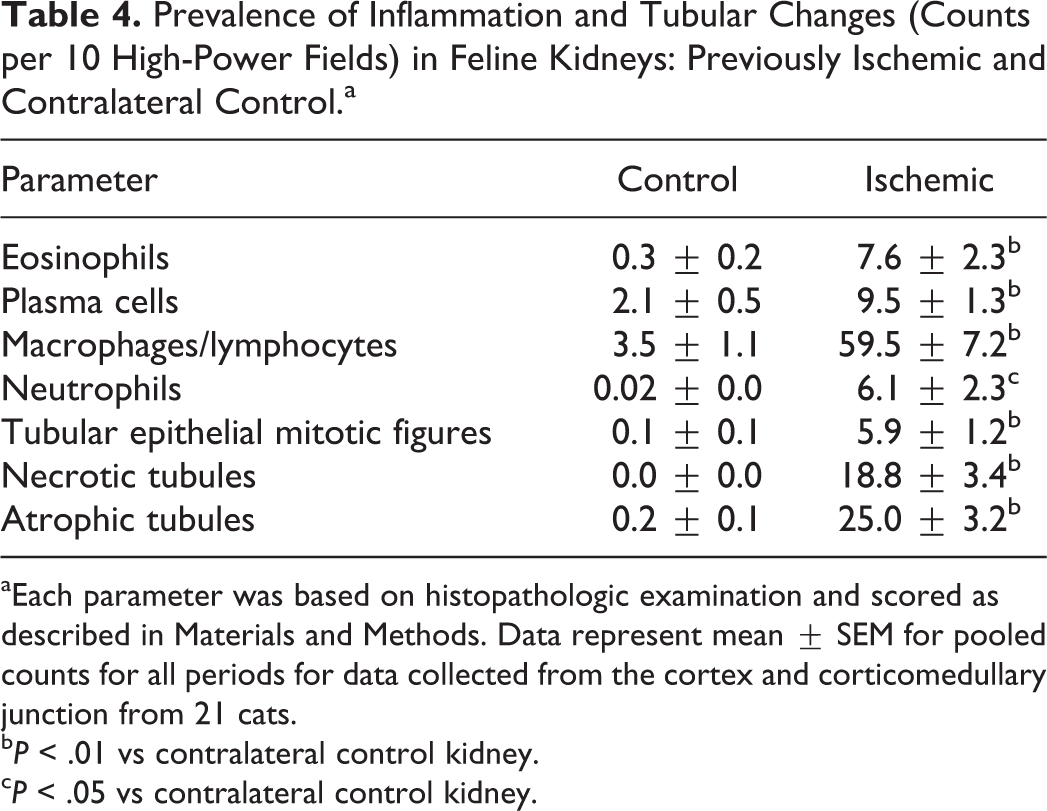
aEach parameter was based on histopathologic examination and scored as described in Materials and Methods. Data represent mean ± SEM for pooled counts for all periods for data collected from the cortex and corticomedullary junction from 21 cats.
bP < .01 vs contralateral control kidney.
cP < .05 vs contralateral control kidney.
Early Changes in the CMJ After Ischemia
Predominant lesions in the CMJ at days 3, 6, and 12 postischemia were tubular necrosis, tubular epithelial cell regeneration, and inflammation. Tubules within medullary rays—composed predominantly of the straight portion of the proximal tubules and the thick ascending limb of the loops of Henle (Fig. 2)—exhibited severe acute coagulative epithelial cell necrosis at day 3 (Fig. 3) and day 6. Affected tubules were filled with eosinophilic, sloughed epithelial cells and were partially lined by basophilic, attenuated epithelial cells. Coagulative necrosis of tubular epithelium persisted but was less severe on days 12 and 21; it was also limited to scattered individual cell necrosis on days 42 and 70. Statistically, at the CMJ and when compared at successive time points, there were significantly (P < .01) more necrotic tubules present on day 3 (88.0 ± 18.5/10 HPFs) than on days 12 (50.7 ± 10.5/10 HPFs), 21 (37.3 ± 1.9/10 HPFs), 42 (15.3 ± 2.2/10 HPFs), and 70 (5.0 ± 1.9/10 HPFs). Also at the CMJ, tubular necrosis scores at day 6 (57.7 ± 2.2/10 HPFs), 12, and 21 were significantly (P < .05) greater than at day 70 and significantly (P < .01) greater on day 6 than on day 42.
The early time points were also characterized by evidence of tubular regeneration, including tubular epithelial cell hyperplasia with nuclear crowding and increased numbers of epithelial mitotic figures. Marked tubular epithelial cell hyperplasia, often with clumps of cells extending into the lumina, was observed at day 3 but most prominently at day 6 postischemia (Fig. 4). Correspondingly, mitotic figures were observed at these early periods, beginning at day 3 (P < .05).
Inflammatory cells in the CMJ were observed at day 3, although this change was mild at this early time point (typically <10 inflammatory cells in 10 HPFs). Occasional peritubular and intraluminal neutrophils were present. By day 6, the inflammation was more severe and composed primarily of macrophages, lymphocytes, and fewer neutrophils and rare plasma cells in a diffuse to multifocal distribution (Fig. 5). Eosinophils were present in moderate numbers (up to 32 eosinophils in 10 HPFs) at days 6 and 12.
Macrophages and lymphocytes were present at day 6 and at all times thereafter. Regarding statistical evaluation of macrophage and lymphocyte counts in the CMJ and other locations, there was a significant interaction of days after ischemia with location (OC, MC, or CMJ; P < .05); thus, individual pairwise comparisons for days after ischemia/location combinations were examined rather than pooled day or location comparisons. The macrophage/lymphocyte counts were highest at the CMJ. On day 21, these counts at the CMJ were significantly (P < .05) higher than the corresponding data for OC and MC on days 3, 6, 12, 21, 42, and 70. Similarly, the macrophage/lymphocyte counts at the CMJ on day 70 were significantly (P < .05) higher than the MC counts for these cells on days 6, 12, 21, 42, and 70.
At days 3, 6, and 12, occasional peritubular capillaries and capillary bundles between the medullary rays (vasa recta) at the CMJ were congested and contained large numbers of inflammatory cells (Figs. 3, 5). These changes were not observed at later periods or in the contralateral control kidneys at earlier periods. Vascular changes suggestive of hypertension were not present at any sampled time.
Later Changes in the CMJ After Ischemia
The ischemic insult was followed in the later time points by irreversible degenerative lesions. Tubular atrophy (Fig. 6) was first observed in the CMJ at day 12 postischemia; it was a later finding than tubular cell mitosis and necrosis (Fig. 7); and it was present at all subsequent periods in the CMJ. Interstitial fibrosis, first observed at day 12, was present in these areas of tubular atrophy (Fig. 6). While atrophic tubules were absent or rare at days 3 and 6 (0.86 ± 0.4/10 HPFs and 0.44 ± 0.2 /10 HPFs, respectively), substantial numbers of atrophic tubules were noted at 12 days postischemia and were present at all subsequent periods. Atrophic tubules were significantly (P < .01) different when compared by day after ischemia but not by location within the ischemic kidney. Compared to day 3, significantly (P < .05) more atrophic tubules were present at days 12 (30.6 ± 8.6/10 HPFs), 21 (55.4 ± 10.6/10 HPFs), 42 (30.2 ± 5.7/10 HPFs), and 70 (25.9 ± 3.8/10 HPFs). Likewise, the number of atrophic tubules on day 6 was significantly (P < .05) lower than days 12, 21, 42, and 70.
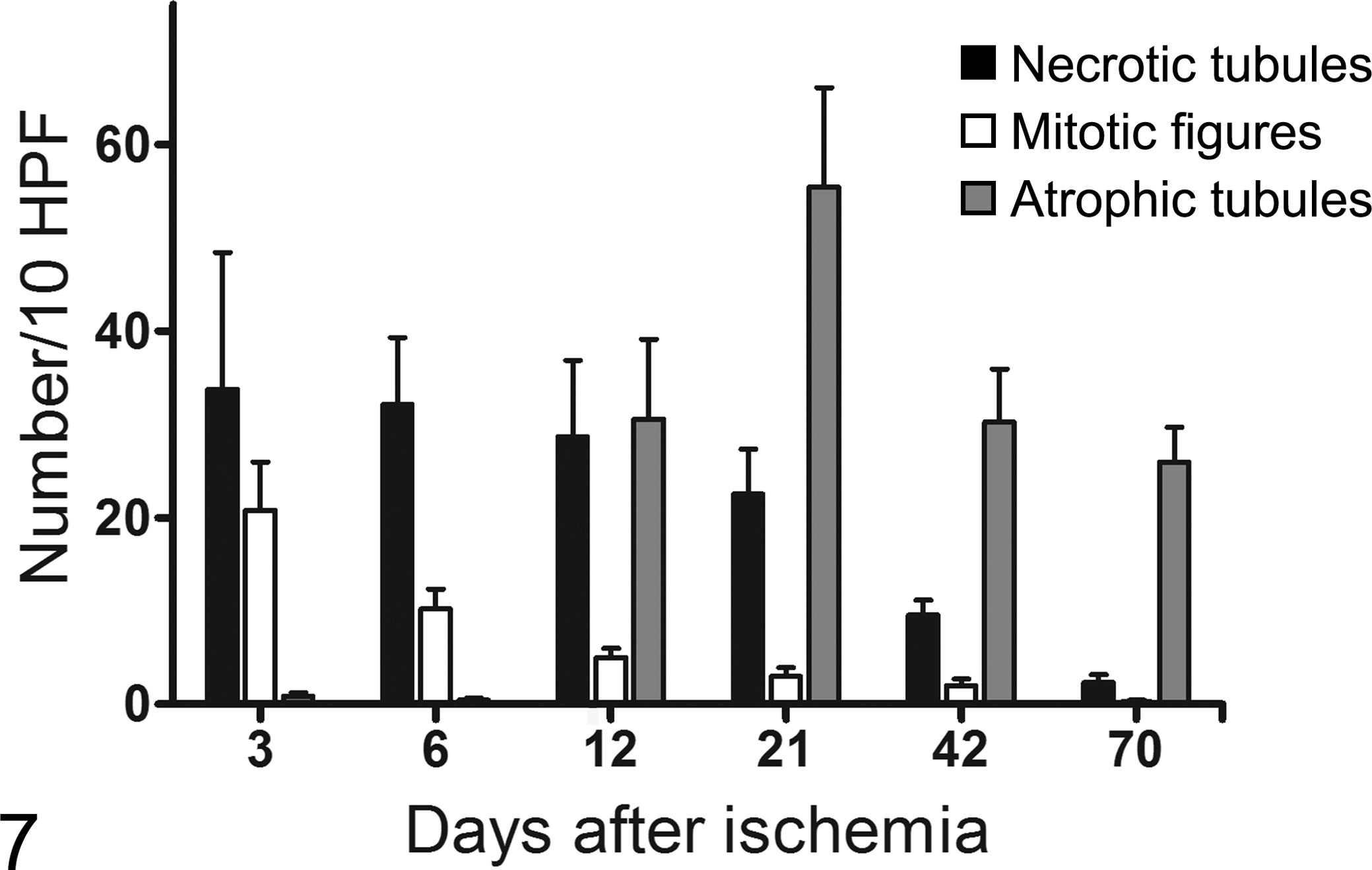
Time course of tubular epithelial necrosis, mitosis, and tubular atrophy in feline kidneys following a single unilateral renal ischemic insult. Mean ± SEM of the pooled counts per 10 high-powered fields (HPFs) from the outer cortex, midcortex, and corticomedullary junction (CMJ) in 21 cats. There were significant differences in some parameters over time and for location within the kidney (see text for details).
The epithelium within atrophic tubules was disorganized and often contained large lipid vacuoles. At day 12, interstitial lipid droplets in foci of granulomatous inflammation were first observed. Lipid-containing foci were seen in 2 of 3 cats at day 12 and in 3 of 3 cats at all later periods. These foci were widely scattered, with 1 or 2 per 10 HPFs. Some of these lipid-containing foci were clearly associated with ruptured tubules (Fig. 8). Disruption of normal corticomedullary architecture by IC and inflammatory cells with atrophy and loss of tubules was evident at days 21, 42, and 70 (Figs. 9, 10). Mononuclear cell inflammation predominated at later periods (days 21, 42, and 70). Plasma cells were a minor component of this inflammation, although there was a trend for increased numbers of plasma cells at later periods. There were no significant differences in plasma cell counts in days after ischemia or location in the ischemic kidney.
Kidney, corticomedullary junction (CMJ), cat, 42 days after unilateral ischemia. Disruption of the tubular basement membrane (tubulorrhexis; arrow) with release of lipid (arrowhead) and resultant granulomatous inflammation. Periodic acid–Schiff and hematoxylin (PASH).
Early Changes in the Cortex After Ischemia
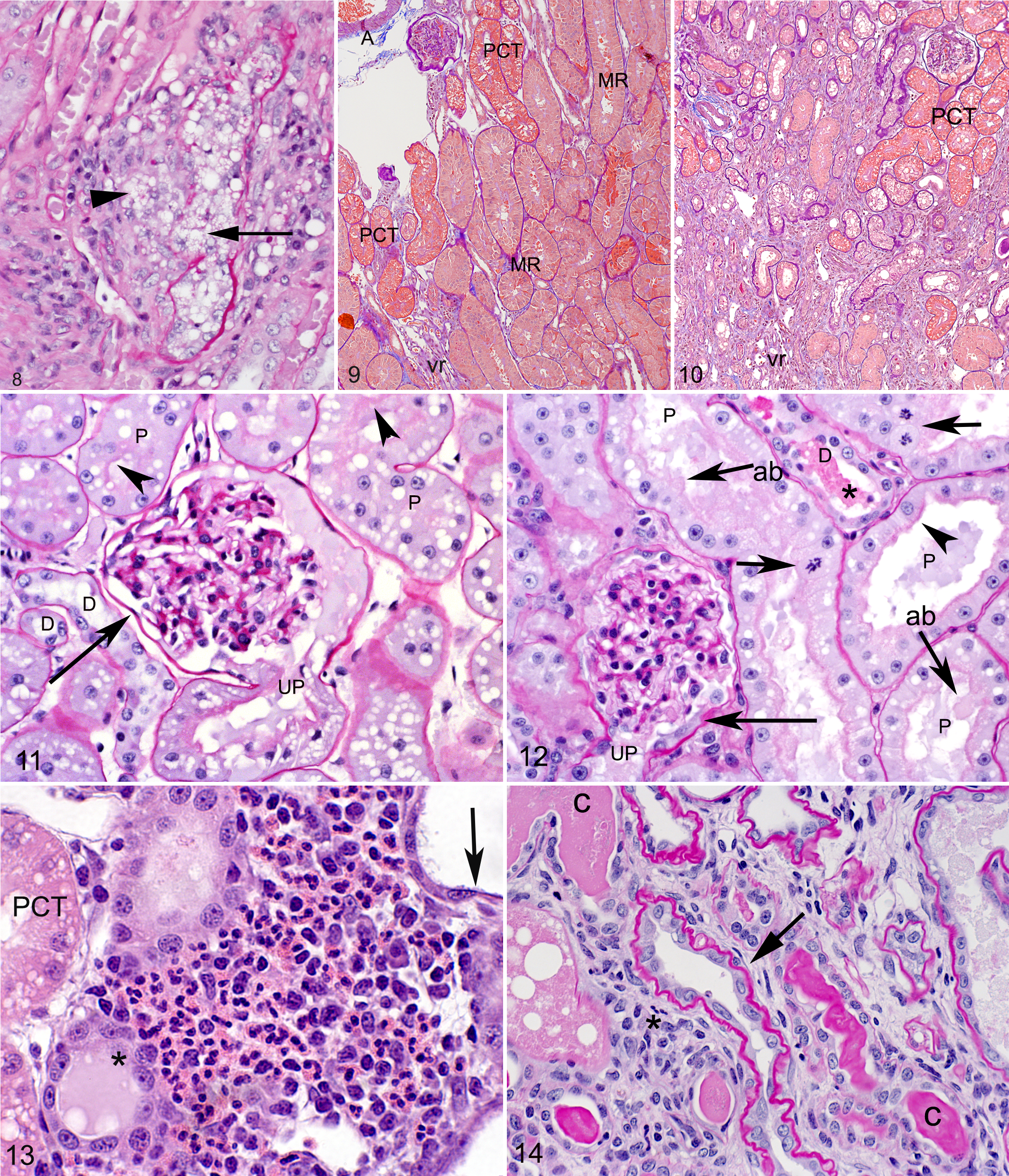
Lesions of acute tubular injury were less severe and less prevalent in the cortex compared with the CMJ. In the cortex on day 3, ischemic proximal convoluted tubules exhibited evidence of acute injury characterized by tubular dilation (ectasia), mild attenuation of the brush border, blebbing of the apical cytoplasm, and increased numbers of mitotic figures as compared with the contralateral control kidney (Figs. 11, 12). Glomerular lesions were mild, focal, and limited to extension of reactive changes in the proximal tubule from the urinary pole to include the parietal epithelial cells lining Bowman capsule. Affected parietal epithelial cells had a marked increase in cytoplasm compared with normal quiescent cells (hypertrophy or “tubularization” of parietal epithelial cells; Fig. 12). Rare mitotic figures were also observed in areas of reactive parietal epithelial cells. Tubular epithelial attenuation and coagulative necrosis of individual cells were predominantly located in scattered cortical straight tubules, consistent with the straight portion of the proximal tubule and thick ascending limbs of the loops of Henle. On day 3, the number of necrotic tubules was significantly (P < .01) less in the OC (7.33 ± 4.1/10 HPFs) and MC (6.00 ± 3.0/10 HPFs) compared with the CMJ. Similarly at day 6, significantly (P < .01) fewer necrotic tubules were present in the MC (15.67 ± 1.2/10 HPFs) compared with the CMJ (57.67 ± 2.2/10 HPFs). Finally, on day 21, the OC (11.7 ± 5.7) had significantly (P < .05) fewer necrotic tubules than the CMJ (37.3 ± 1.9).
As in the CMJ, mitoses of epithelial cells were numerous in the proximal tubules at day 3 (Fig. 12) and day 6. In both the CMJ and the cortex, significantly (P < .05) more mitotic figures were observed on day 3 (20.78 ± 5.2/10 HPFs) compared with days 12 (5.00 ± 1.0/10 HPFs), 21 (3.00 ± 0.9/10 HPFs), 42 (2.00 ± 0.7/10 HPFs), and 70 (0.56 ± 0.3/10 HPFs).
Inflammation was present but mild in the OC and MC at the early time points, when cortical inflammation was often present around subcapsular veins. An early eosinophilic inflammatory response was noted in the previously ischemic tissue in all cats. Eosinophilic inflammation was most intense at day 6, with moderate to large numbers of eosinophils (26–106 per 10 HPFs) observed around subcapsular veins and injured straight tubules (Fig. 13). On day 6, eosinophil counts (74.33 ± 24.5 cells per 10 HPFs) were significantly higher in the OC than all other day/location combinations (P < .01 for all). Eosinophils were not present (0 eosinophils per 10 HPFs) in the contralateral control kidneys. Mononuclear cell interstitial inflammation occurred early in the cortex and was more severe in the OC compared with the MC, as it was often present around subcapsular veins. Significantly (P < .05) fewer mononuclear cells were present in the cortex than in the CMJ at all periods.
Late Changes in the Cortex After Ischemia
The ischemic insult was followed by degenerative lesions in the cortex, although these changes were more segmental in distribution compared with the diffuse changes occurring in the CMJ. As in the CMJ, tubular atrophy was first observed on day 12 postischemia and persisted at subsequent periods (Fig. 14). Similar numbers of atrophic tubules were present in the OC and MC at day 12 and were present in similar numbers at all later periods. Atrophic tubules were typically present within linear streaks of fibrosis and inflammation in the area of medullary rays (Fig. 15).
Kidney, cortex, cat, 70 days after unilateral ischemia. Foci of tubular loss, tubular atrophy, mild inflammation, and fibrosis interspersed with areas containing normal-appearing proximal convoluted tubules (PCTs). L, interstitial lipid. Periodic acid–Schiff and hematoxylin/Masson trichrome.
While interstitial mononuclear inflammatory cells were present at all periods after day 3, there was a change in the distribution of the cells from locations around subcapsular veins at day 6 to areas of atrophic tubules at later time points. Numbers of inflammatory cells were greater in the superficial cortex at the earlier time points, reflecting distribution predominantly around subcapsular veins. At day 70, similar numbers of mononuclear inflammatory cells were present within the OC and MC (mean, 52 and 45 cells in 10 HPFs, respectively) as inflammation extended throughout the parenchyma in areas of fibrosis.
Histomorphometry
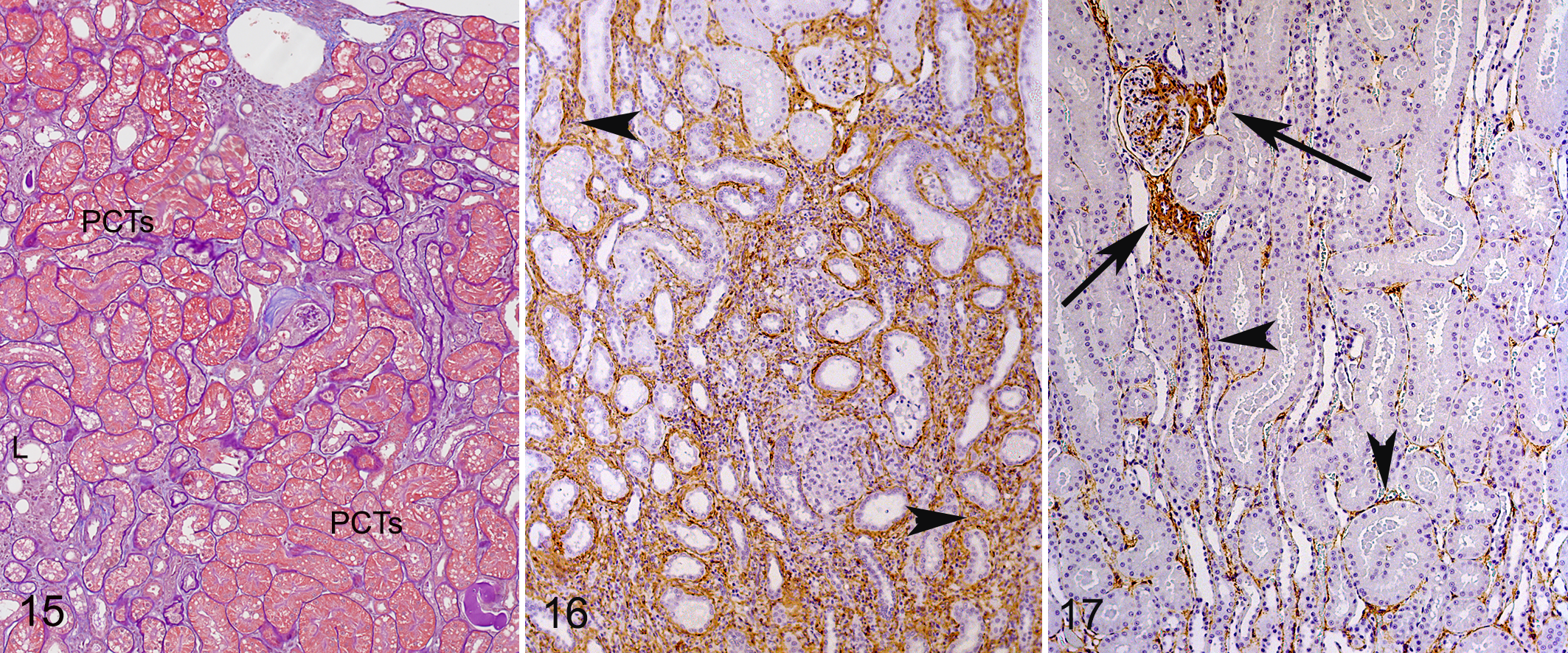
There was increased SMA staining postischemia compared with that observed in the contralateral control (Figs. 16, 17). When data were evaluated by region (Table 5), the ischemic insult led to significantly more SMA in the cortex, CMJ, and medulla compared with the same areas in the contralateral controls (Fig. 18; P < .01 for all regions). In the ischemic kidney, when all time points were considered together, the CMJ had significantly more positive staining (Fig. 18, P < .01) compared with the cortex and medulla. The SMA staining in postischemic tissue was significantly greater on days 21 and 42. In the CMJ from ischemic tissues, the SMA staining tended to increase through day 42 but was decreased by day 70 (Table 5). A similar pattern over time was not apparent in the cortex or medulla for SMA staining.
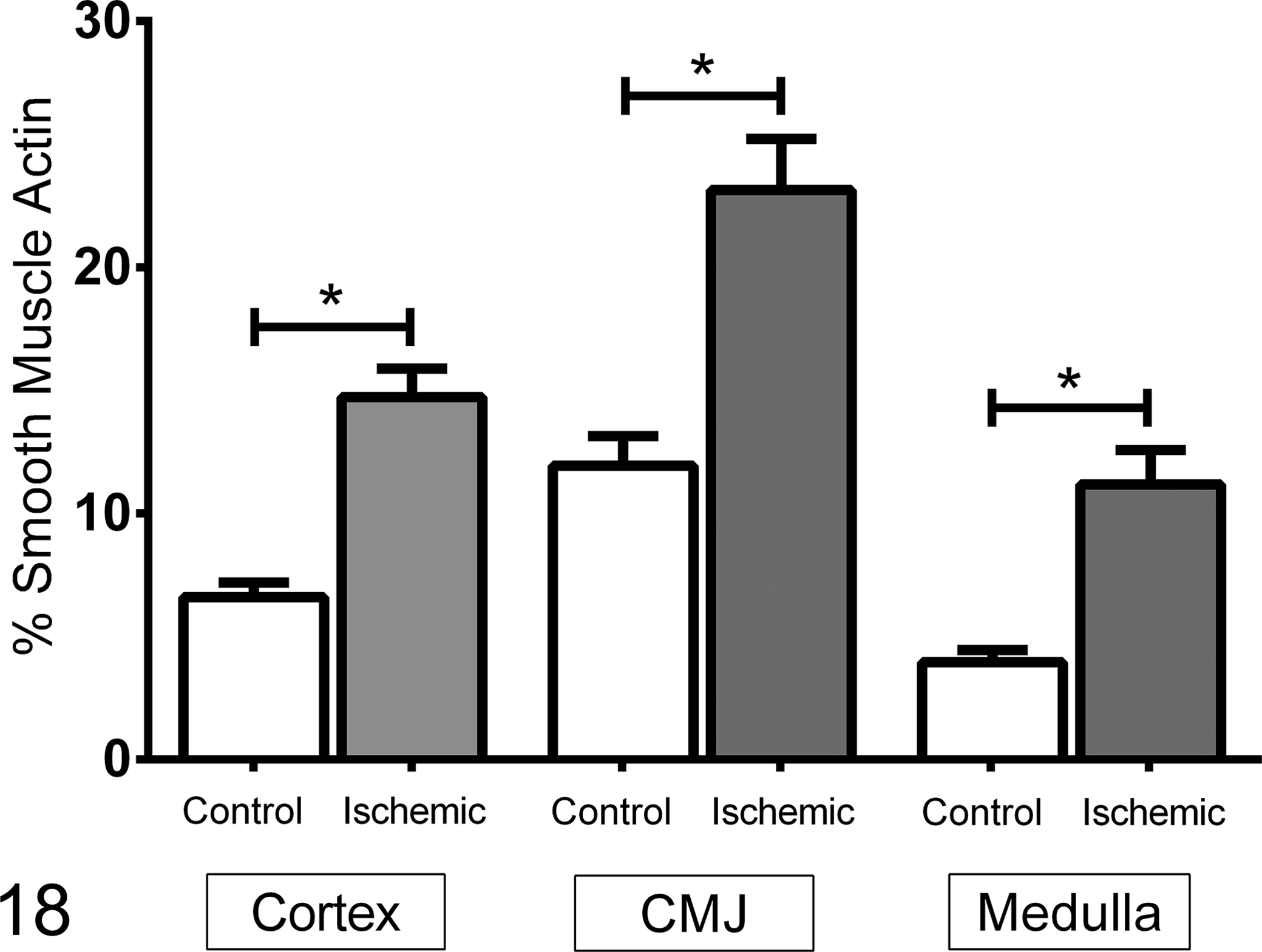
Smooth muscle actin staining of tissue from 21 cats at variable time points following 1 hour of unilateral renal ischemia. The data are mean ± SEM of percent of the tissue that is immunostained. Within each of the 3 regions, the previously ischemic kidney had more staining for smooth muscle actin than the contralateral control kidney. CMJ, corticomedullary junction. *P < .01.
Smooth Muscle Actin Staining in Regions of Feline Kidneys: Previously Ischemic and Contralateral Control (Percentages).
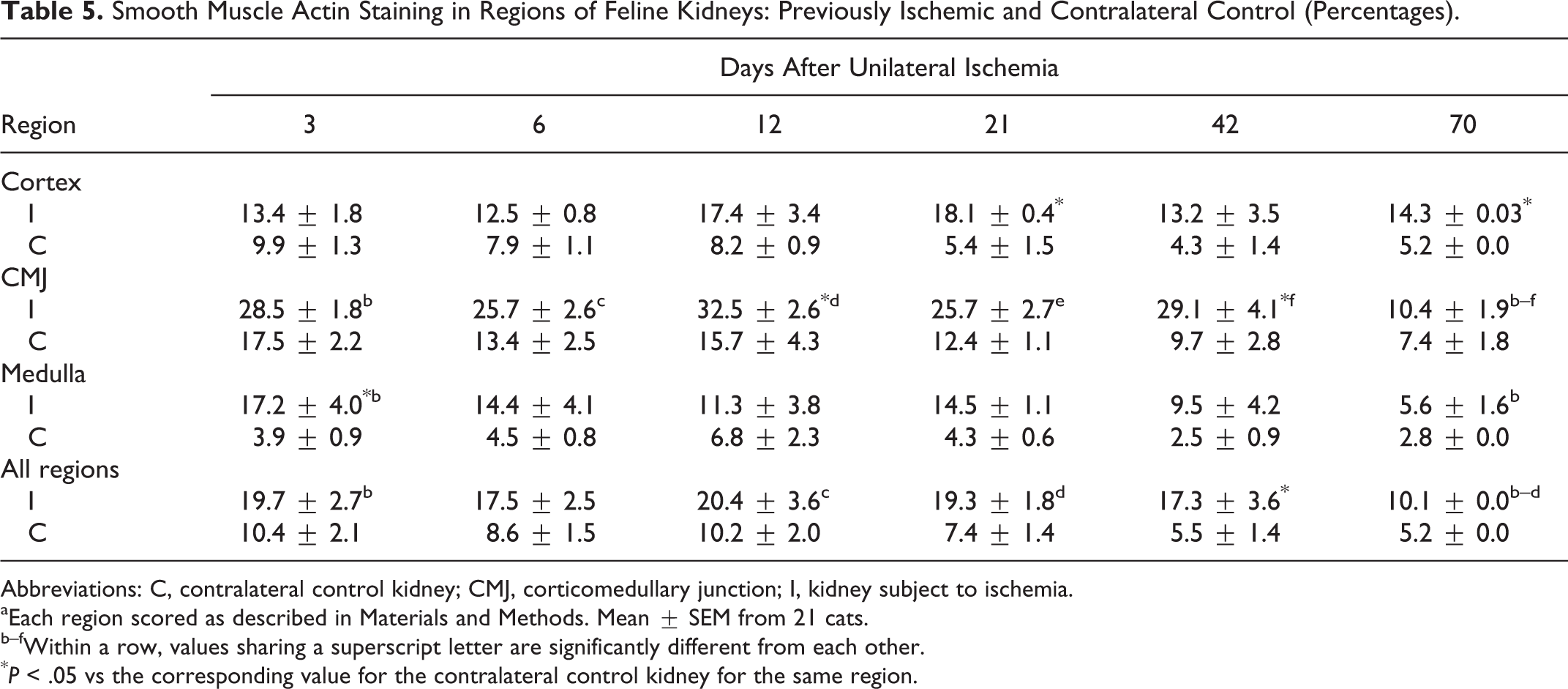
Abbreviations: C, contralateral control kidney; CMJ, corticomedullary junction; I, kidney subject to ischemia.
aEach region scored as described in Materials and Methods. Mean ± SEM from 21 cats.
b–fWithin a row, values sharing a superscript letter are significantly different from each other.
*P < .05 vs the corresponding value for the contralateral control kidney for the same region.
Combination staining with PASH/TRI demonstrated more (P < .01) IC in the cortex, CMJ, and medulla following ischemia when compared with the same areas in contralateral control kidneys (Fig. 19; Table 6). In the control kidney, IC staining was significantly (P < .01) less in the cortex compared with the CMJ and medulla. Increased IC staining at early time points in the ischemic kidney was attributed to expansion of the interstitium by basophilic edema. On days 12, 21, and 42, edema was no longer a confounding factor when staining for IC. At these time points, the ischemic kidney had significantly greater IC staining compared with the control.
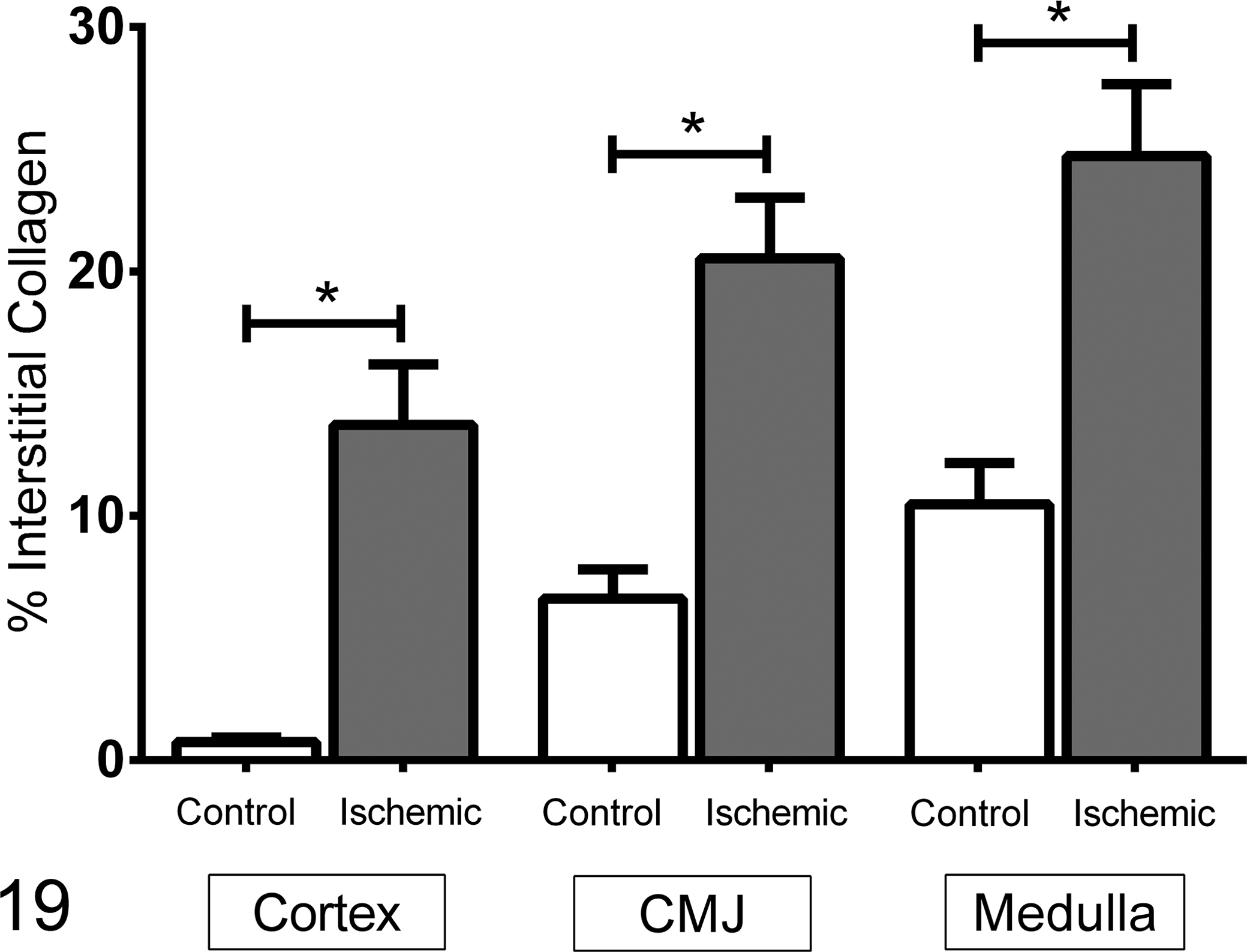
Interstitial collagen staining of tissue from 21 cats at variable time points following 1 hour of unilateral renal ischemia. The data are mean ± SEM of percentage of the tissue that is immunostained. Within each of the 3 regions, the previously ischemic kidney had more staining for interstitial collagen than the contralateral control kidney. CMJ, corticomedullary junction. *P < .01.
Interstitial Collagen Staining in Regions of Feline Kidneys: Previously Ischemic and Contralateral Control (Percentages).a
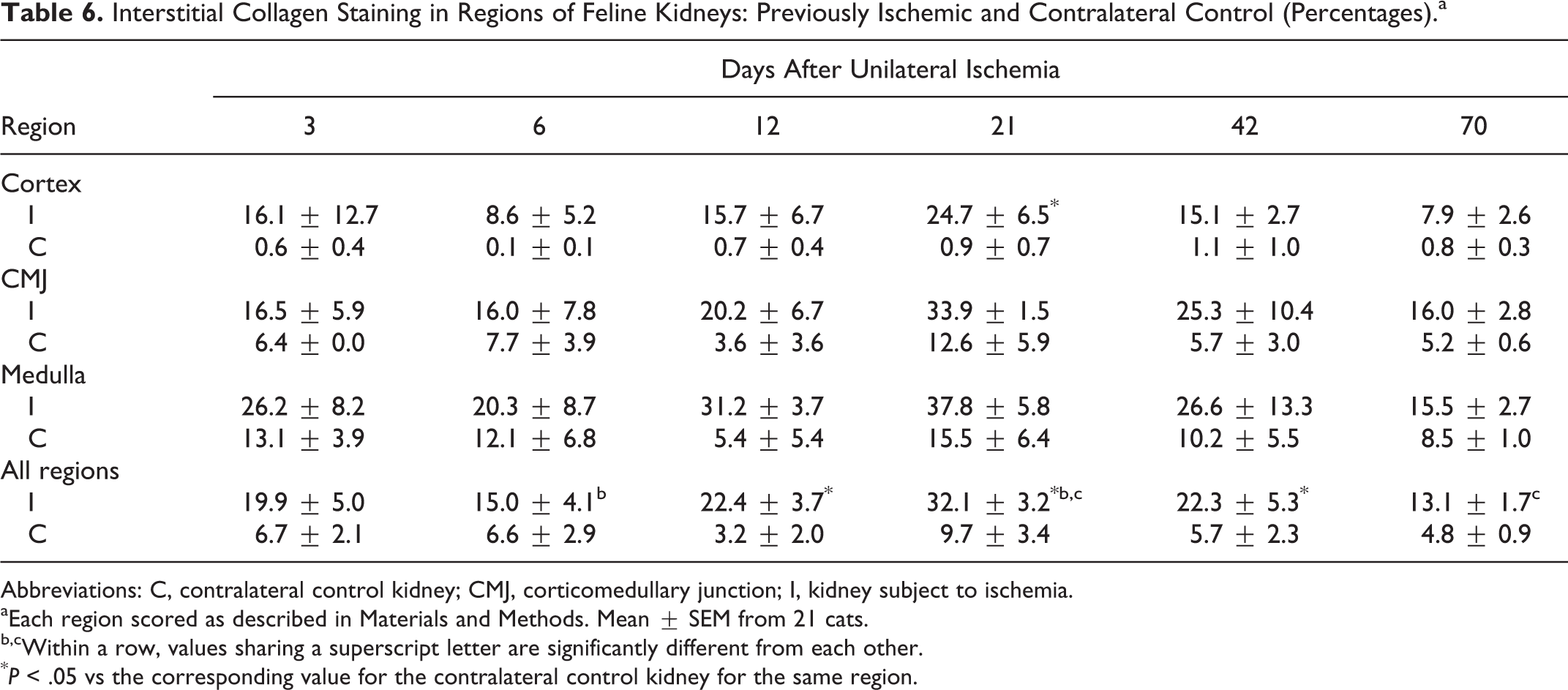
Abbreviations: C, contralateral control kidney; CMJ, corticomedullary junction; I, kidney subject to ischemia.
aEach region scored as described in Materials and Methods. Mean ± SEM from 21 cats.
b,cWithin a row, values sharing a superscript letter are significantly different from each other.
*P < .05 vs the corresponding value for the contralateral control kidney for the same region.
Discussion
In the cat, AKI induced by unilateral renal ischemia initiated a cascade of events leading to interstitial inflammation and fibrosis, with lesions that closely mimic those observed in naturally occurring feline CKD. This unilateral model was advantageous, as it allowed for maintenance of systemic homeostasis during the acute phases of AKI. Additionally, the contralateral kidney offers an important control for each cat, reducing or eliminating the requirement for sham-operated control animals. While the contralateral kidney is likely to be affected to some extent by sequelae of the contralateral ischemia and, in particular, is expected to be undergoing compensatory hypertrophy, similar processes would be expected in surviving nephrons of the previously ischemic kidney. Thus, the contralateral kidney offers a useful control in this model.
Unlike cats experiencing bilateral renal ischemia, 39 cats undergoing unilateral ischemia did not exhibit uremia or a coagulopathy. In the model described here, GFR was reduced by approximately 49%, with a nadir occurring 6 days postischemia. The serum creatinine concentration tended to increase, although this difference was statistically significant only late in this study. A 50% decline in GFR might be expected to cause an increase in serum creatinine, as was the case with unilateral nephrectomy in cats, 29 and it is likely that the lack of significant difference in serum creatinine concentrations at early time points reflects interanimal variability, the variable influence of nonrenal factors on creatinine kinetics, and the small numbers of animals in the present study. It is interesting to note the presence of a modest but statistically significant reduction in USG over time, which likely reflects the chronic loss of function of the ischemic kidney. Injury to the thick ascending limbs of the loops of Henle, in particular, would be expected to reduce medullary interstitial fluid osmolality and maximal urine concentrating ability. Although not investigated, it is possible that the increased serum creatinine concentration or the reduced USG at the later time points is a reflection of other factors, such as alterations in hemodynamic function of the contralateral kidney secondary to systemic exposure to proinflammatory cytokines arising from the injured kidneys. 1,3
In people, AKI and CKD are bidirectionally connected, as the presence of one predisposes to the development of the other. 33 In the model described here, while the bidirectional nature of this relationship was not investigated, acute tubular necrosis and associated inflammation were followed by chronic interstitial inflammation and fibrosis—characteristics of spontaneous feline CKD. 11 While the small number of cats in the present study and the 70-day end point preclude conclusions about the long-term course of these changes, our findings suggest that AKI and ischemia might be factors in the genesis or progression of CKD in cats. Similarly, ischemic AKI has been described in inbred strains of mice and rats as a model for renal fibrosis; however, the mechanism for this fibrosis is not clear. 2,16,35,42 –44,46
Four progressive phases have been proposed in the pathogenesis of renal fibrogenesis. 19 The first phase is the activation and initial injury phase, characterized by inflammation and appearance of activated myofibroblasts. The myofibroblasts increase in number as chemotactic recruitment of fibroblasts and monocytes begins in the signaling phase. The fibrogenic phase is characterized by accumulation of extracellular matrix. The final renal destructive phase is characterized by capillary destruction and consequential tubular ischemia and nephron loss. 19
The source of collagen-producing fibroblasts in CKD is uncertain, but these cells have been hypothesized to originate from pericytes and perivascular fibroblasts found in association with the intrarenal vasculature, 28 epithelial–mesenchymal transition, 25 or bone marrow–derived cells. 26 Sustained activation of epidermal growth factor receptor appears to be an important mediator of renal fibrogenesis following ischemic AKI. 16,30,42
Unlike bilateral renal ischemia, 39 significant decreases in platelet number or function were not observed in these cats following unilateral ischemia. An increase in the speed of aggregation (as reflected in the slope of the aggregation curve) was seen for both ADP (on day 3) and collagen (on day 6). This increased platelet reactivity may be a product of mild systemic inflammation induced by the ischemic event or by the surgical procedure, or it may be related to the presence of an indwelling jugular venous catheter. Regardless, the changes are modest; as a result, thrombosis—as observed in the bilateral ischemia model—was not a confounding factor.
This study was the first to characterize the time course of structural changes of ischemic AKI in cats. Acute glomerular lesions following ischemia were limited to “tubularization” of the parietal epithelial cells lining Bowman capsule, as has been described in human kidneys following ischemia. 32 This parietal cell hypertrophy is presumed to reflect reactive changes occurring in the proximal tubule. Vascular lesions were present at early periods in all cats and were limited to increased nucleated cells within vasa recta and scattered peritubular capillaries at the CMJ. Accumulations of nucleated cells (lymphocytes, monocytes, and neutrophils) have been described in the vasa recta of rats with nephrotoxic AKI 41 and are a common finding in people, where it may be the sole histologic indication of AKI. 32
In ischemic AKI, lesions are predominantly those of tubular damage. Ischemic injury is typically patchy, as observed in this study, affecting a short segment of the proximal tubule at the glomerular junction (the “tubular takeoff”), portions of the straight segment of the proximal tubule, and focal areas of the ascending limbs of the loop of Henle. 32 The morphologic lesions of ischemic AKI are typically subtle, compared with the more obvious necrosis occurring in toxic tubular injury. In this study, the severity of tubular injury following ischemia varied by tubular segment and by time after ischemia. Ischemia caused acute coagulative epithelial necrosis in tubules at the CMJ, representing portions of the straight (S3) region of the proximal tubule and the thick ascending limbs of the loops of Henle. More subtle and therefore more typical lesions of ischemic injury were present in tubules within the cortex—including brush border thinning, tubular ectasia, apical blebbing, and regenerative changes with nuclear crowding, high nuclear:cytoplasmic ratio, anisokaryosis, and mitoses.
The severe necrosis present in ischemic tubules at the CMJ in this study is likely a reflection of the severity of the ischemic insult and regional susceptibility of tubular segments to ischemia. The CMJ has a high concentration of metabolically active tubule segments coupled with a reduced oxygen tension. Two tubular segments in particular—the late proximal tubules (straight or S3 segment) and the medullary thick ascending limbs—are sensitive to ischemic injury. This corresponds with another study in feline kidneys with natural and experimental AKI where kidney injury molecule 1 was shown to be most highly expressed in the S3 segment of the proximal tubule. 7 In the cats of the present study, the CMJ had significantly more mononuclear inflammatory cells, necrotic tubules, and mitotic figures during regeneration. This is further emphasized by the significantly greater amount of SMA and IC staining in the CMJ compared with the cortex and medulla. Although the most severe tubular necrosis occurred in the CMJ, streaks of atrophic tubular profiles and fibrosis were also present in the cortex. The relative sparing of proximal convoluted tubules within the cortical labyrinth on either side of these linear fibrotic foci is consistent with localization of degenerative changes to the medullary rays. Medullary rays extend from the cortex to the medulla (hence the name medullary rays) between the cortical labyrinths. Initial injury to the late proximal tubule (straight or S3 segment) and the thick ascending limb (which extends from the thin ascending limb up to the distal tubule at the level of the macula densa) within the CMJ likely encompassed the portions of these nephron segments within the cortex. There was evidence of tubular rupture in the CMJ, suggesting that the ischemia caused tubular basement membrane breaks in addition to epithelial necrosis, perhaps contributing to a loss of atrophic tubular profiles, interstitial inflammation, and renal fibrosis.
Evidence of epithelial necrosis and regeneration was present at the first sampled period (day 3 postischemia) and persisted through day 21. In studies of renal ischemia in mice, injured tubular epithelial cells were replaced within 2 days by proliferation of surviving epithelial cells. 24 A similarly rapid regenerative phase in the cats of this study was confirmed by the prominence of tubular cell mitoses on day 3 and epithelial cell hyperplasia at days 3 and 6. However, by day 42 irreversible changes of tubular atrophy and interstitial fibrosis predominated, and these chronic changes were still present at day 70.
Renal tubular injury was accompanied by inflammation at all sampled periods, with the ischemic kidney containing significantly more inflammatory cells of all types than the contralateral control kidney. An initial moderate to marked infiltrate of eosinophils was present in the superficial cortex of all cats at day 6. At later periods, eosinophils were only rarely present. In people, high numbers of eosinophils are known to be present early in acute tubulointerstitial nephritis, especially after drug-induced AKI. 9,34,36 In feline kidneys postischemia, most inflammatory cells were present as focal granulomas throughout the CMJ. These foci of inflammation were first apparent at day 6 and were present at all later time points. Some of these foci contained free and/or phagocytized lipid, which was presumed to originate from lipid-laden tubular epithelium or free lipid within atrophic/ischemic tubules. Free lipid in areas of granulomatous inflammation also occurs in cats with naturally occurring CKD. 31 The presence of fewer atrophic tubules at day 70 was attributed to previous tubulorrhexis, tubular dropout, and/or expansion of the interstitium by fibrous connective tissue and inflammatory cells.
Inflammation was frequently observed surrounding subcapsular veins, particularly at early time points. Other studies in cats have identified a similar distribution in association with renal injury. In one study evaluating feline renal allografts, 48% of grafts had mild to severe lymphocytic inflammation surrounding the subcapsular or interlobar and arcuate veins. 17 Similarly, in cats suffering from melamine and cyanuric acid toxicity, 12 of 13 cats exhibited subcapsular perivascular inflammation, characterized as vessel wall disruption by neutrophils, eosinophils, and macrophages. 15 The significance of subcapsular venous inflammation is unknown, but this finding appears to be associated with acute or ongoing renal injury in cats.
In this study, macrophages/monocytes were a predominant inflammatory cell following ischemia. Macrophages have a complex role in augmenting tubular injury, promoting tubular repair, and promoting interstitial fibrosis following AKI. 1,23 Monocytes/macrophages were initially scattered throughout the interstitium in areas of tubular injury, occurring as nodular foci in the cortex and CMJ at later time points. However, definitive differentiation among macrophages, endothelial cells, and epithelial cells originating from ruptured tubules could not be certain via the techniques of the present study.
We demonstrated that SMA and IC are more prevalent in ischemic kidneys compared with contralateral controls. At the final measurement on day 70, the percentage of positive staining tissue, especially in the CMJ, was reduced compared with previous time points. This reduction in SMA and IC staining could be due to a reparative process with a parallel reduction in fibrosis. 4 This seems unlikely because other histologic features of the kidneys on day 70 suggest ongoing chronic injury. For example, there were increased numbers of inflammatory cells, particularly monocytes, during these later time points. Subjectively, the presence of these cells made histomorphometry difficult as they expanded the interstitium and made the SMA and IC less easily quantifiable. It is also possible that increased expression of matrix metalloproteinases, collagenases, or other factors associated with the injury response may have altered SMA or IC content. 4,14 Thus, is it reasonable to speculate that the changes observed on day 70 reflect a yet-to-be characterized process or a sampling error. Regardless, evaluation of this model to later time points is warranted to provide long-term characterization of this process in cats.
Systemic hypertension is believed to be both a cause of renal injury and an effect of renal disease. 10 In this study, Doppler methodology was used to measure blood pressure in a small number of cats up to day 6 after ischemia, and differences were not observed. This may reflect a low number of cats evaluated, significant inter- and intraindividual variability, pronounced “white coat effect” in this population, 5 and failure to evaluate at chronic time points. Histologically, lesions associated with severe hypertension (hyperplasia or hyaline arteriosclerosis) were not observed in any samples; however, the prevalence of hypertension in this model remains poorly characterized. Further work is needed to evaluate systemic blood pressure in this model in a larger number of animals over time.
In cats, unilateral renal ischemia appears to be a viable model for investigation of AKI and the relation among AKI, ischemia, and CKD. Unilateral renal ischemia resulted in a significant reduction in GFR associated with 2 distinct patterns of structural changes. During the first 3 weeks postischemia, tubular cell necrosis and regeneration predominated. Significant, repeatable lesions of AKI were gradually replaced with interstitial inflammation, tubular atrophy, and fibrosis—changes similar to those observed in naturally occurring feline CKD. 11,31 The small number of animals studied precludes definitive conclusions, but in addition to characterizing the structural changes in ischemic AKI in cats, these studies are consistent with the hypothesis that transient ischemia and/or AKI is a contributory factor to the genesis or progression of CKD in cats. Longer-term studies (>70 days) will be required to further characterize chronic changes following transient renal ischemia in cats.
Footnotes
Authors' Note
Presented in part at the American College of Veterinary Surgeons Surgery Summit (San Antonio, TX, October 2013; San Diego, CA, October 2014) and the American College of Veterinary Pathology Annual Meeting (Atlanta, GA, November 2014).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was funded by intramural funds made available through the Veterinary Medical Experiment Station of the College of Veterinary Medicine of the University of Georgia.
