Abstract
Chronic kidney disease (CKD) is prevalent in elderly cats. Frequently, a diagnosis is made in later stages of disease, by which time many renal lesions are irreversible. As such, little headway has been made in identifying an etiology and preventing this common disease. The aim of this study was to evaluate the presence and severity of both reversible and irreversible histopathologic changes in the kidneys of cats at each stage of CKD and, in addition, to determine if lesion prevalence and character were different between stages. A total of 46 cats with CKD were classified according to the International Renal Interest Society (IRIS) as stage I (3 cats), stage II (16 cats), stage III (14 cats), and stage IV (13 cats). Eleven young, nonazotemic and 10 geriatric, nonazotemic cats were included as controls. The severity of tubular degeneration, interstitial inflammation, fibrosis, and glomerulosclerosis was significantly greater in later stages of CKD compared with early stages of disease. Proteinuria was associated with increased severity of tubular degeneration, inflammation, fibrosis, tubular epithelial single-cell necrosis, and decreased normal parenchyma. Presence of hyperplastic arteriolosclerosis, fibrointimal hyperplasia, or other vascular lesions were not found to be significantly different between hypertensive and normotensive cats. The greater prevalence and severity of irreversible lesions in stage III and IV CKD implies that therapeutic interventions should be targeted at earlier stages of disease.
Keywords
Chronic kidney disease (CKD) affects as many as 50% of elderly cats, and prevalence increases with age. 24,25 The etiology of CKD in cats is unknown. In humans, the leading cause for end-stage renal failure is type 2 diabetes mellitus and hypertension. 4 However, diabetic nephropathy has not been identified in cats, and renal lesions in diabetic cats have been no different from those in nondiabetic cats. 44 The most frequent morphologic diagnosis in cats with CKD is chronic tubulointerstitial nephritis and fibrosis, which are relatively nonspecific lesions. 10,12,20 Therefore, feline CKD is currently considered the consequence of a variety of etiologies that lead to a final common pathway of irreversible, progressive kidney damage. 13 This damage culminates in reduction of the glomerular filtration rate, which, when severe, results in retention of metabolic by-products such as creatinine and serum urea nitrogen. Eventually, uremia may develop and manifest as gastrointestinal, cardiovascular, pulmonary, neuromuscular, or hematologic diseases. 13,36
CKD is categorized into disease stages established by the International Renal Interest Society (IRIS) based on serum creatinine measurements. 33,37 Clinical progression, or stage, is associated with decreased survival. 6 Clinicopathologic data are useful for diagnosing, staging, and prognosticating but give few clues as to the distribution and pattern of injury within the kidney. Therefore, it is difficult to determine which stages are characterized by irreversible lesions and at which stage interventional therapies should be targeted. In a previous study, renal fibrosis in cats with CKD from the United Kingdom correlated with clinicopathologic derangements such as azotemia, hyperphosphatemia, and anemia. 10 Renal scarring encompasses interstitial fibrosis, which is an increase in extracellular matrix, as well as glomerulosclerosis and tubular atrophy. 14,16,17,32 Collectively, these changes imply a loss of function and are considered, at least to date, irreversible. 16,32 However, not all injury leads to irreversible damage. Replication and repair can lead to a return of normal function. Inflammation, edema, and tubular epithelial damage have the potential to resolve (ie, reversible). Interest in these types of morphologic responses guided the design of this study. The aim of the current study was to further explore the relationship between clinical parameters and renal pathology by characterizing and quantifying a wide variety of interstitial, tubular, glomerular, and vascular lesions (both reversible and irreversible) in the kidney of cats with CKD from the United States and assessing the correlation between these histopathologic changes and clinical IRIS stage and substage.
Materials and Methods
Case Selection
Cats autopsied at Colorado State University Diagnostic Medical Center between 2000 and 2013 with a history of CKD, appropriate clinicopathologic data necessary for IRIS staging, and adequate tissue available for histologic review were included in this study. Exclusion criteria included renal neoplasia, ureteral or urethral obstruction, or pyelonephritis. Nonazotemic geriatric cats were humanely euthanized with client consent for health problems unrelated to renal disease. Young nonazotemic cats were euthanized at a local humane society, according to humane society guidelines and protocols. Study samples were obtained from these cats after euthanasia, and no cats were euthanized for the purpose of this study. Age was estimated by surrender history and/or dental assessment.
Clinicopathologic Data
Using the IRIS staging scheme, patients (designated CKD cats) were assigned a stage of I to IV based on clinical evaluation of the cat’s medical history by a board-certified internist. Clinical evaluation for IRIS stage determination took into account 2 or more serum creatinine levels no less than 48 hours apart, clinical history, fluid therapy, body condition score, and fluctuation in weight and muscle mass over the course of the disease. Patients assigned to stage I had a urine-specific gravity (USG) <1.035, serum creatinine <1.6 mg/dl, and other evidence of renal abnormalities such as abnormal imaging (radiograph and/or ultrasound) and/or proteinuria. Stages II, III, and IV were defined by creatinine measuring 1.6 to 2.8 mg/dl, 2.9 to 5.0 mg/dl, and >5.0 mg/dl, respectively. Nonazotemic control cats had no clinical evidence or history of renal disease, USG >1.035, and creatinine ≤1.6 mg/dl. Control cats were divided into either nonazotemic geriatric controls (GCs) if they were ≥7 years of age or nonazotemic young controls (YCs) if they were <4 years of age.
Substaging according to proteinuria and elevation of systemic blood pressure was determined based on data availability. Urine samples were collected via cystocentesis during routine examination and submitted to the Clinical Pathology Laboratory at Colorado State University Diagnostic Medical Center. Quantification of urine protein was determined by urine protein to creatinine ratio (UPC) measurement, and only CKD cats in which UPC data were available were included in this portion of the analysis. GC cats were confirmed to have no urinary protein by the sulfosalicylic acid precipitation test. Hypertension was determined by repeated mean systolic blood pressure measurements of >160 mm Hg, prior clinical diagnosis of hypertension, or evidence of hypertensive therapy (eg, amlodipine) in the medical record; only cats with blood pressure data available were included in this portion of the analysis.
Histology
Tissues were preserved in 10% neutral buffered formalin, routinely processed, and embedded in paraffin. Sections of kidney were cut at 3 μm and stained with hematoxylin and eosin and periodic acid–Schiff–hematoxylin to assess basement membranes of tubules and blood vessels, as well as Masson’s trichrome for assessment of fibrosis. Tissues were evaluated for a range of histologic lesions by 2 pathologists. The scoring schematic for dichotomous and semiquantitative histologic variables is outlined in Supplemental Table S1. In addition, continuous variables included global glomerulosclerosis and frequency of tubular epithelial single-cell necrosis. Global glomerulosclerosis was determined by examining 50 randomly distributed glomeruli per kidney to determine the proportion in which >75% of the capillary tuft was effaced by extracellular matrix. When present, specific glomerular lesions (eg, glomerulonephritis or focal segmental glomerulosclerosis) were noted. As renal lesions can be unilateral, if both kidneys were available, each was scored separately for the following variables: percent normal parenchyma, interstitial cortical scarring, medullary scarring, interstitial inflammation, tubular epithelial single-cell necrosis, and globally sclerotic glomeruli. Scarring was defined as a percentage of cortex or medulla occupied by trichrome-confirmed collagenous matrix, in conjunction with fibroplasia, parenchymal collapse due to tubular atrophy and loss, and glomerulosclerosis (Fig. 4). 14,17
Vasculature was assessed for degenerative changes, specifically arteriolosclerosis. Evaluation of arteriolosclerotic lesions included concentric medial smooth muscle hyperplasia (hyperplastic arteriolosclerosis) or segmental or circumferential intimal proliferation with disruption of the internal elastic lamina (fibrointimal hyperplasia; Fig 6). Other vascular lesions (eg, fibrinoid vascular necrosis, arteritis) were recorded if present.
Statistical Analysis
A univariate approach was used to determine if histologic variables differed between nonazotemic cats and those with CKD. Serum creatinine, IRIS stage, and proteinuria status were designated as independent variables. For semiquantitative outcome variables (eg, pattern of interstitial scarring), medians were reported and data were converted into “ranks” for linear regression analysis. For variables with multiple measurements from the same individual (ie, both kidneys available for review), repeated measures were taken into account for regression analyses. Similarly, for continuous variables, differences in means among groups were determined by linear regression after assumptions for normality were confirmed. Fisher exact test was used for dichotomous variables, and odds ratios were calculated with 95% confidence intervals. Statistical significance for all analyses was set at P ≤ .05 and P ≤ .0125 for pairwise comparison to account for multiple comparisons. Statistical calculations were performed by SAS v9.3 (SAS Institute, Cary, NC).
Results
Signalment
Of 331 CKD cats autopsied between 2000 and 2013, 46 had sufficient clinical data and tissue for evaluation. Age and cause of death are outlined in Table 1. There was no statistically significant difference among groups with regard to sex or breed. Of the CKD cats in this study, 2 died of natural causes and the others were euthanized. Those euthanized for poor quality of life or acute decompensation of CKD (21/46) were typically in later stages of the disease (stage II, n = 2; stage III, n = 7; and stage IV, n = 12). Twenty-five cats were euthanized for reasons not directly related to kidney disease, including neoplasia (n = 9); gastrointestinal disease, liver disease, or pancreatitis (n = 5); cardiopulmonary disease (n = 4), including congestive heart failure, thromboembolism, or pulmonary disease; and other metabolic or neurologic illnesses (n = 7). The 10 GC cats were euthanized for nonrenal neoplasia (n = 5), cardiopulmonary disease (n = 4), and pancreatitis (n = 1).
Signalment, Cause of Death, and Clinicopathologic Data for Controls and CKD Cats by Stage.
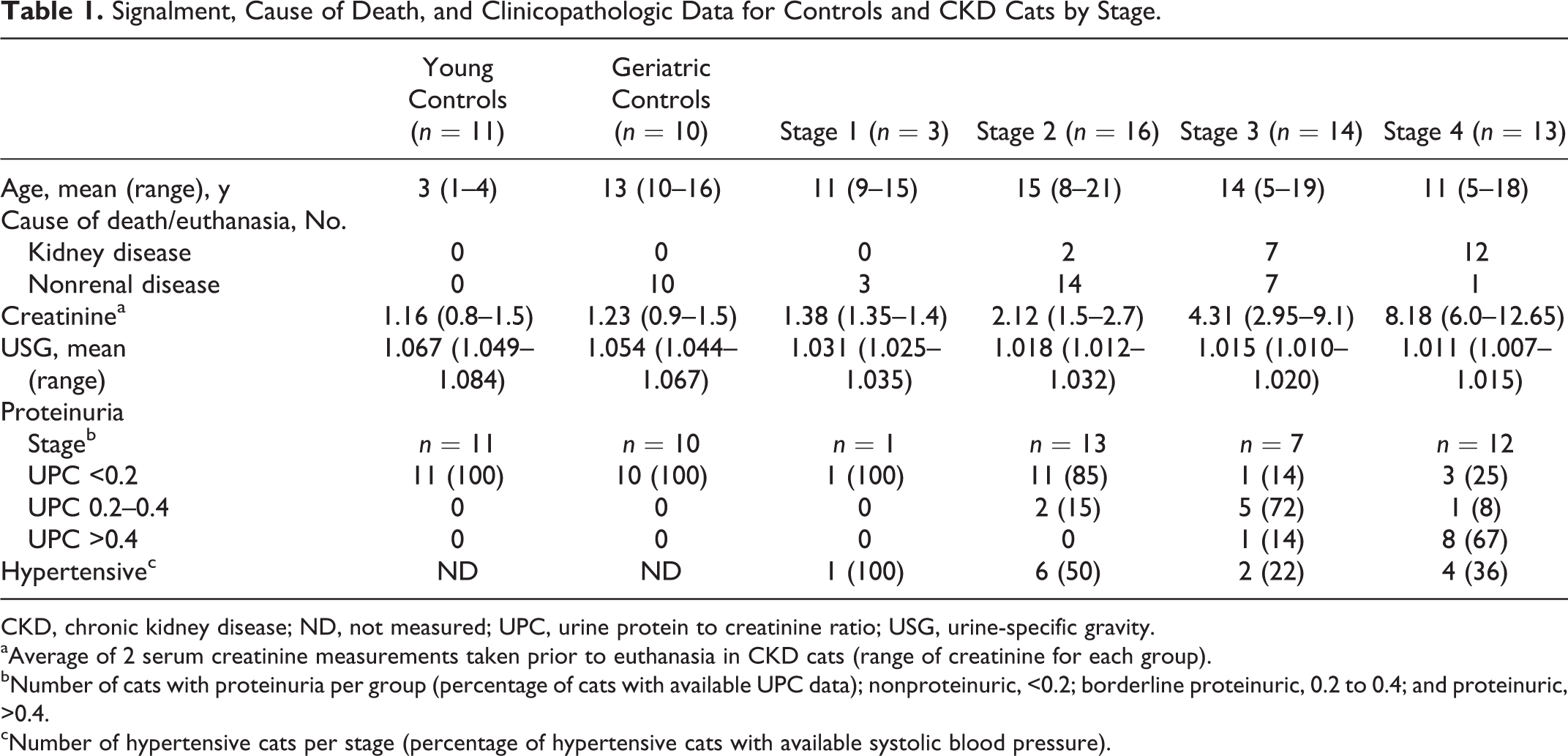
CKD, chronic kidney disease; ND, not measured; UPC, urine protein to creatinine ratio; USG, urine-specific gravity.
aAverage of 2 serum creatinine measurements taken prior to euthanasia in CKD cats (range of creatinine for each group).
bNumber of cats with proteinuria per group (percentage of cats with available UPC data); nonproteinuric, <0.2; borderline proteinuric, 0.2 to 0.4; and proteinuric, >0.4.
cNumber of hypertensive cats per stage (percentage of hypertensive cats with available systolic blood pressure).
Histopathology
Detailed histopathologic findings are present in Figures 1 –3, Tables 2 and 3 and in the Supplemental Materials. The following summarizes the salient lesions and their progression.
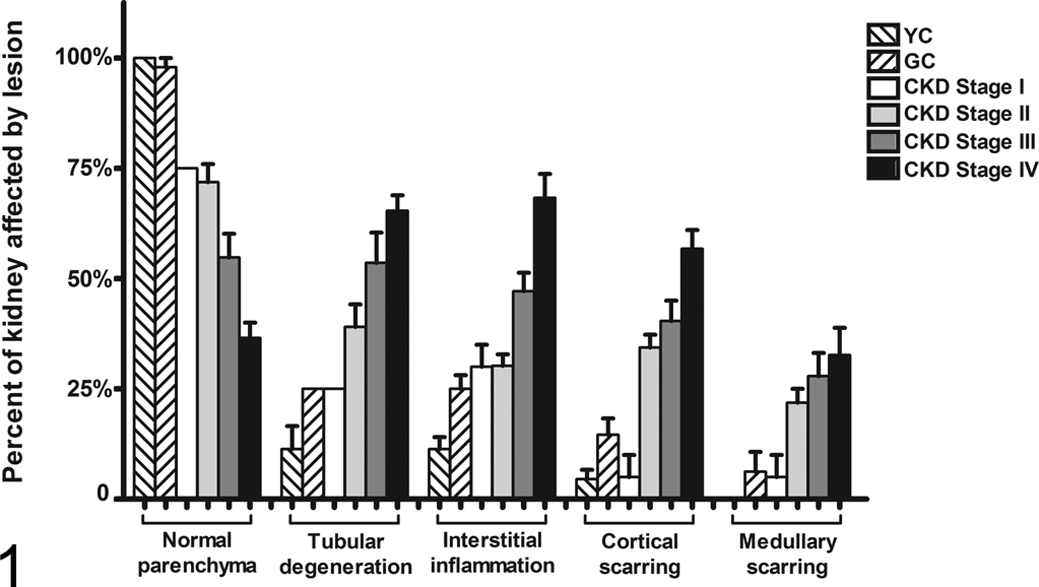
Categorical histologic variables by group (GC, geriatric controls; YC, young controls) or chronic kidney disease (CKD) stage. Columns represent mean percentage of tissue affected by histologic lesion while bars represent the standard error of the mean.
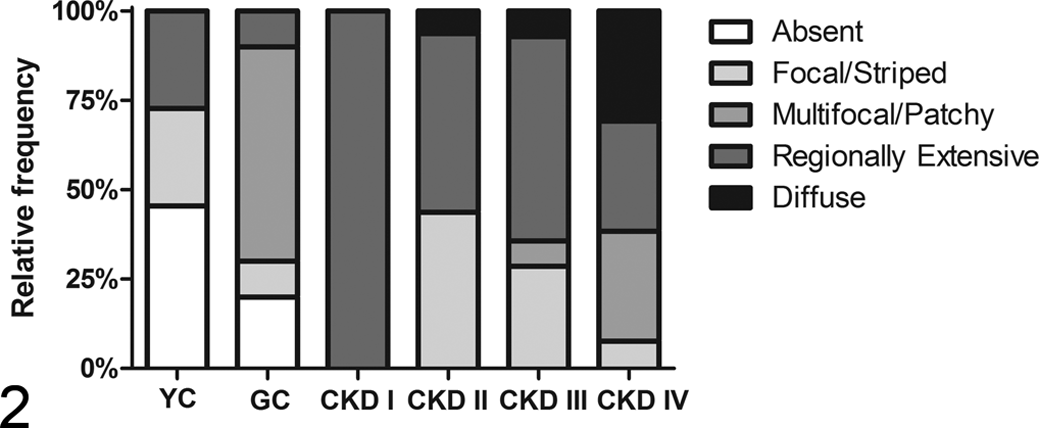
Relative frequencies of 4 different patterns of interstitial inflammation for each chronic kidney disease (CKD) stages I to IV or control group (GC, geriatric controls; YC, young controls).
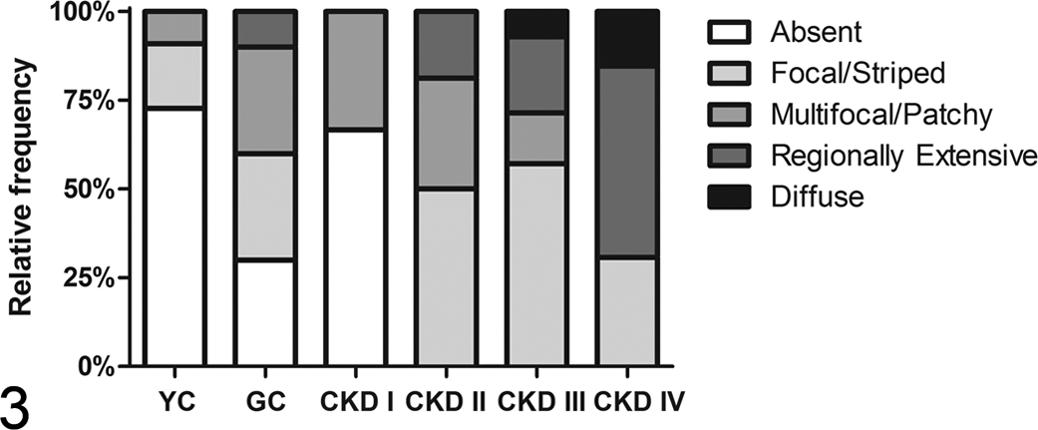
Relative frequencies of 4 different patterns of cortical scarring for each chronic kidney disease (CKD) stages I to IV or control group (GC, geriatric controls; YC, young controls).
Dichotomous and Continuous Histologic Variables With Statistical Significance.
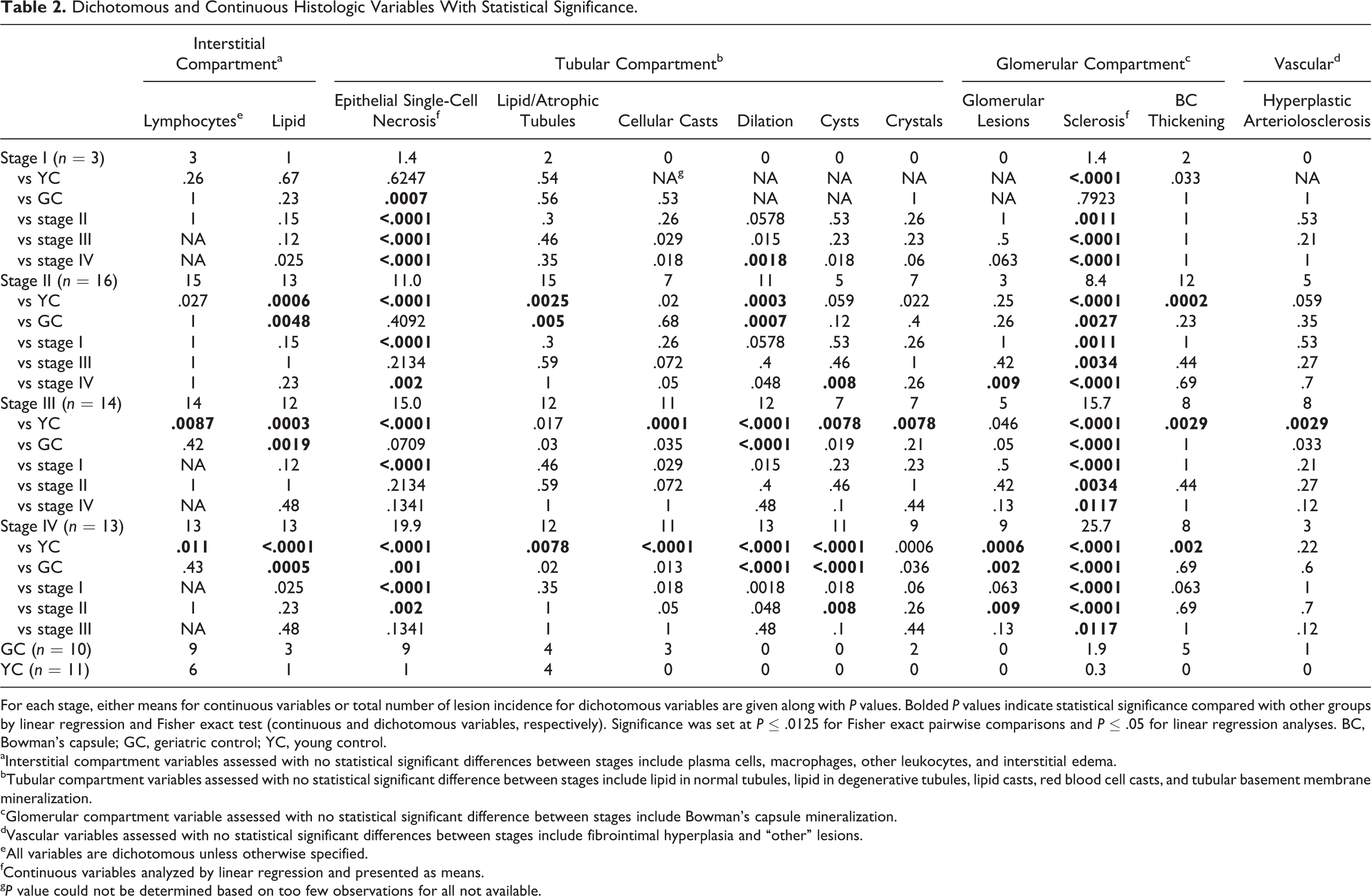
For each stage, either means for continuous variables or total number of lesion incidence for dichotomous variables are given along with P values. Bolded P values indicate statistical significance compared with other groups by linear regression and Fisher exact test (continuous and dichotomous variables, respectively). Significance was set at P ≤ .0125 for Fisher exact pairwise comparisons and P ≤ .05 for linear regression analyses. BC, Bowman’s capsule; GC, geriatric control; YC, young control.
aInterstitial compartment variables assessed with no statistical significant differences between stages include plasma cells, macrophages, other leukocytes, and interstitial edema.
bTubular compartment variables assessed with no statistical significant difference between stages include lipid in normal tubules, lipid in degenerative tubules, lipid casts, red blood cell casts, and tubular basement membrane mineralization.
cGlomerular compartment variable assessed with no statistical significant difference between stages include Bowman’s capsule mineralization.
dVascular variables assessed with no statistical significant differences between stages include fibrointimal hyperplasia and “other” lesions.
eAll variables are dichotomous unless otherwise specified.
fContinuous variables analyzed by linear regression and presented as means.
g P value could not be determined based on too few observations for all not available.
Categorical Histologic Variables With Statistical Significance.
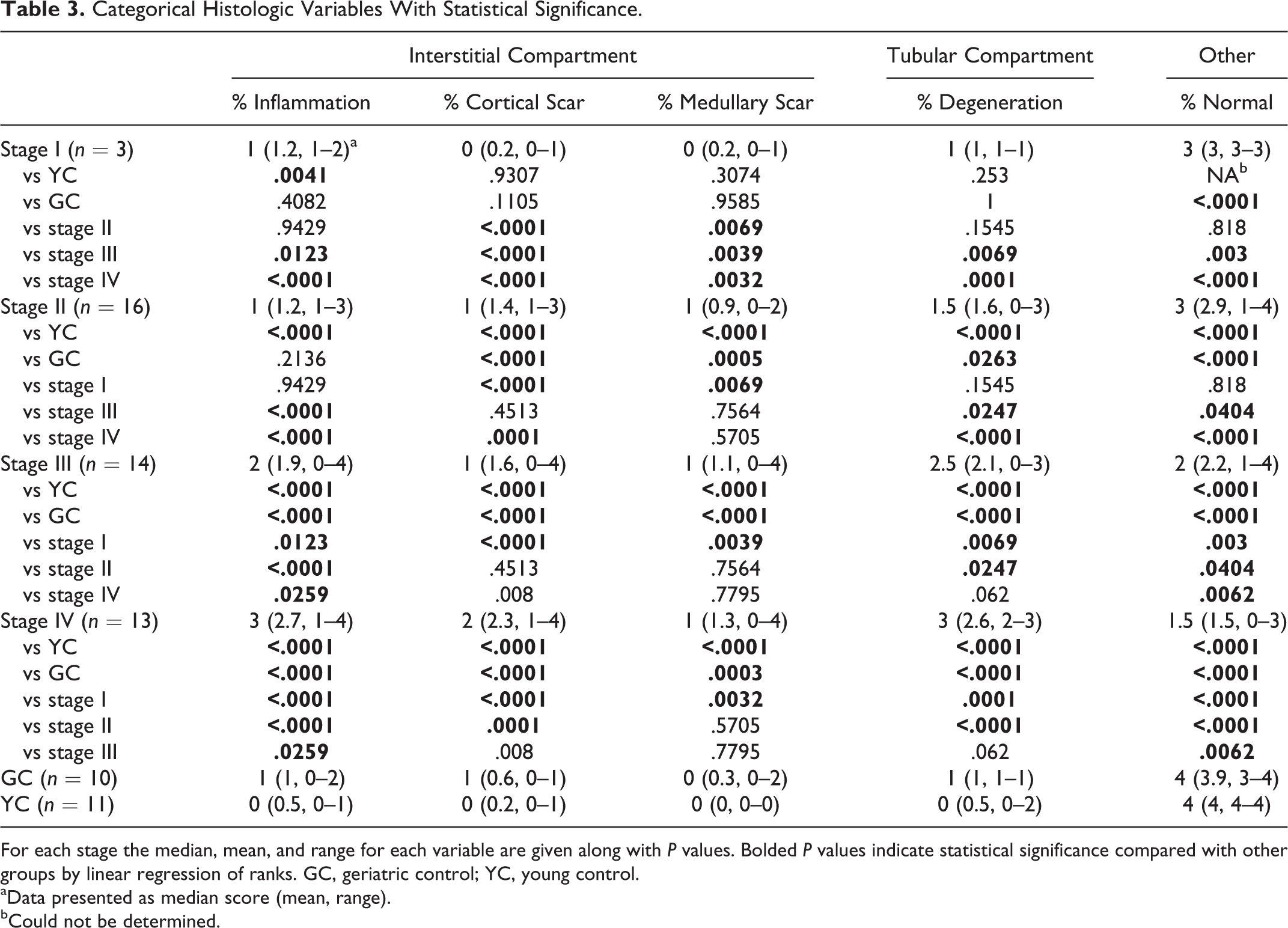
For each stage the median, mean, and range for each variable are given along with P values. Bolded P values indicate statistical significance compared with other groups by linear regression of ranks. GC, geriatric control; YC, young control.
aData presented as median score (mean, range).
bCould not be determined.
Stage I CKD Cats
Remaining normal renal parenchyma (51%–75%) was significantly less than in geriatric controls. Interstitial inflammation consisted exclusively of lymphocytes in a regionally extensive distribution. This distribution was similar for all stages, but the severity of inflammation in stage I (<25%) was greater than in YCs and less than later CKD stages. Cortical and medullary scarring (the presence of collagenous matrix visible with trichrome stain) was nearly absent (<25% in a single cat) at this stage and significantly less than all other stages (Fig 4). Tubular degeneration was mild, focal to scattered, which was significantly less severe than later stages (stages III and IV). Single-cell necrosis of tubular epithelial cells—characterized by loss of basement membrane adhesion, pyknotic to karyorrhectic nuclei, and shrunken, hypereosinophilic cytoplasm—was infrequent and significantly less than that observed in geriatric controls and other CKD stages. Global glomerulosclerosis—in which >75% of the capillary tuft was effaced by extracellular matrix—was observed less frequently in stage I than all other CKD stages but was greater than that observed in YCs. Other glomerular lesions and vascular lesions were absent.
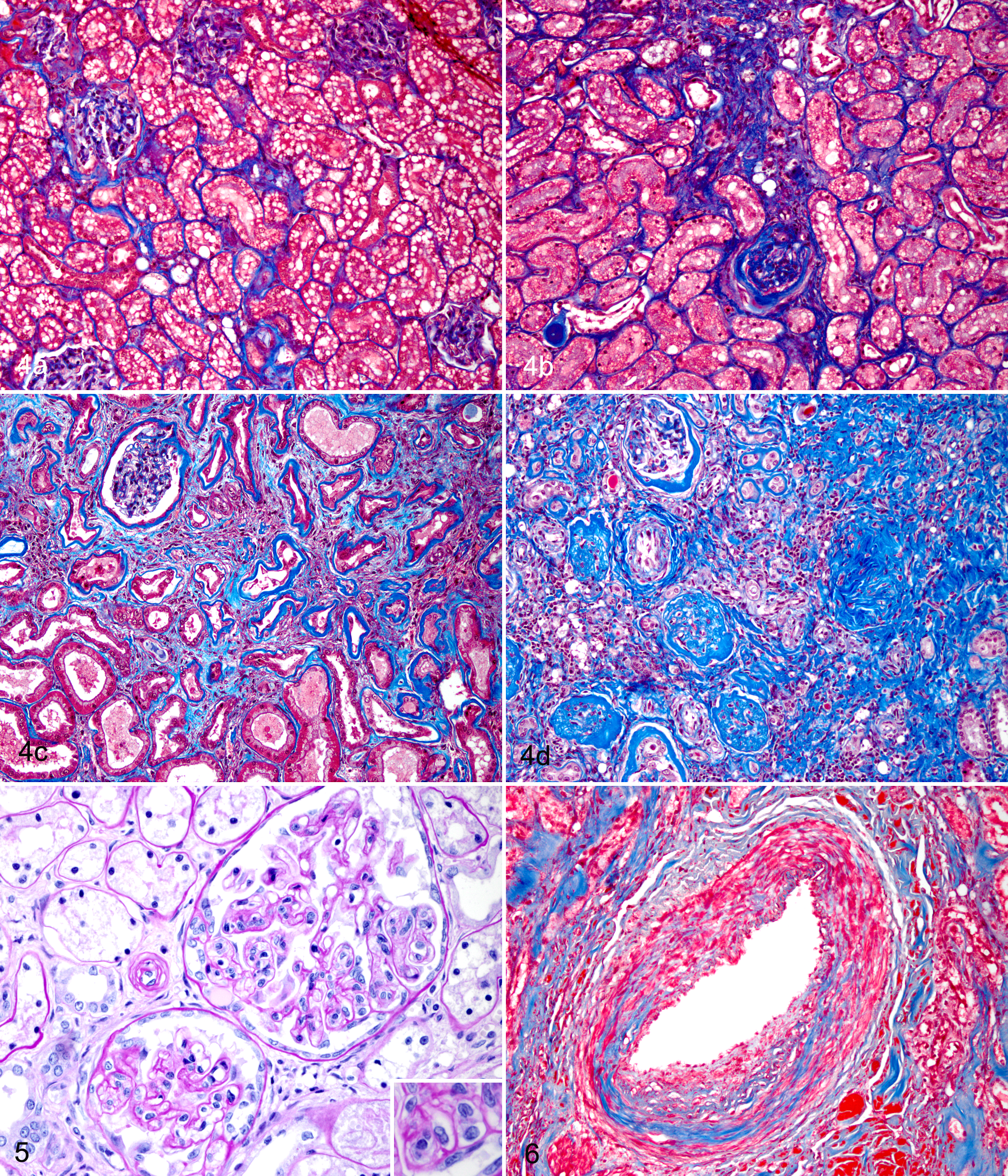
Scarring in chronic kidney disease, kidney, cat. (a) Cortical scarring score of 1 with minimal tubular atrophy and interstitial expansion by increased matrix. Masson’s trichrome stain. (b) Scarring score of 2 with increased interstitial fibrosis, periglomerular fibrosis, and tubular atrophy. Masson’s trichrome stain. (c) Score of 3 with at least 50% of the tissue affected by interstitial fibrosis, tubular atrophy, and glomerulosclerosis (not present in image). Masson’s trichrome stain. (d) Score of 4 with a majority of the cortical parenchyma replaced by fibrosis, tubular atrophy and loss, and glomerulosclerosis. Masson’s trichrome stain.
Stage II CKD Cats
The amount of remaining normal parenchyma (51%–75%) and severity of inflammation (<25%) were similar to that in stage I cats. Interstitial inflammation consisted of primarily lymphocytes and plasma cells and less frequently macrophages and granulocytes. Cortical and medullary scarring was significantly greater than in controls or CKD stage I cats but not different from stage II. Interstitial lipid was present in most cats and was more frequent than in controls. Mild to moderate tubular degeneration was observed and was greater than in both controls and less than that in stages III and IV. Epithelial single-cell necrosis was greater than in YCs or stage I cats but similar to GC cats. Global glomerulosclerosis was significantly greater than in controls and stage I cats but significantly less than in later stages.
Other glomerular lesions were identified in 3 cats: 1 each with membranoproliferative glomerulonephritis (MPGN) and focal segmental glomerulosclerosis (FSGS), cystic glomerular atrophy, or mesangial expansion. Bowman’s capsule thickening with or without parietal cell hypertrophy was present in most cats. Vascular lesions included fibrointimal hyperplasia, hyperplastic arteriolosclerosis, hyalinosis, and torturous vessels in regions of scarring; their prevalence was not statistically different among groups.
Stage III CKD Cats
Significantly less normal parenchyma remained at this stage (25%–50%) compared with stages I and II. Interstitial inflammation (25%–50%) was greater than earlier CKD stages and less than in stage IV cats and appeared as regionally extensive infiltrates of lymphocytes accompanied in half of cases by plasma cells, macrophages, and granulocytes. Severity of renal scarring (<25%) was similar between stage II and stage III cats; cortical scarring was significantly less compared with stage IV. Tubular degeneration was moderate to severe and significantly greater than in controls and stages I and II. Global glomerulosclerosis was greater than in controls and earlier stages but less than in stage IV cats. Other glomerular lesions were observed in 5 cats and included 1 or more of the following: FSGS, thrombotic microangiopathy (Fig 5), MPGN pattern, basement membrane remodeling, mesangial expansion, mesangiolysis, and/or glomerular hypertrophy. Thickening of Bowman’s capsule and parietal cell hypertrophy was present in more than half of stage III cats. Hyperplastic arteriolosclerosis was observed more frequently than in YCs but was not different from other groups.
Stage IV CKD Cats
Significantly less normal parenchyma remained than any other group. Inflammation affecting 51% to 75% of the tissue section was significantly greater than in all other groups and consisted of lymphocytes and plasma cells in most cats. Cortical scarring (25%–50%) was typically greater than medullary scarring (<25%); cortical scarring was significantly greater in stage IV than all other groups, while medullary scarring was significantly different only from controls and stage I cats. Regionally extensive scarring was most frequently encountered and was significantly different from controls and stages I and II. Interstitial lipid was present in all cats. Tubular degeneration, affecting entire nephrons, and single-cell necrosis of tubular epithelial cells were significantly more severe than in controls and earlier stages but similar to stage III cats. Tubular dilation and cysts were more prevalent than in stage I or Stage II, respectively. Global glomerulosclerosis was the most severe at this stage compared with all other groups. Most cats had at least one other glomerular lesion, including FSGS, glomerular hypertrophy, mesangial expansion, endothelial hypertrophy, MPGN pattern, and cystic glomerular hypertrophy; these were significantly more prevalent than in controls and stage II cats. Kidneys frequently contained fibrointimal hyperplasia but infrequently were affected by hyperplastic arteriolosclerosis. The prevalence of vascular lesions was not significantly different from controls or other stages.
Clinicopathologic Data
Clinicopathologic data for each group are summarized in Table 2. Analysis of the relationship between histologic lesions and serum creatinine did not reveal any major differences from what was observed in the comparison of histologic variables and individual IRIS stages (data not shown). Of the 33 CKD cats for which UPC measurements were available, 9 were proteinuric (UPC >0.4; stage III: n = 1, stage IV: n = 8), 8 were borderline proteinuric (UPC 0.2–0.4; stage II: n = 2, stage III: n = 5, stage IV: n = 1), and 16 were nonproteinuric (stage I: n = 1, stage II: n = 11, stage III: n = 1, stage IV: n = 3). All control cats (YCs and GCs) were nonproteinuric. Significant histologic findings in cats with or without proteinuria are outlined in Supplemental Table S2. When cats with proteinuria were compared with nonproteinuric cats (reference variable), statistical significance was found with the following lesions: lower percentage of normal parenchyma (P < .0001), higher percentage of interstitial inflammation (P < .0001) and cortical scarring (P < .0001), more tubular degeneration (P < .0001), cellular casts (P = .0162), lipid casts (P = .05), tubular crystals (P = .0046), tubular cysts (P = .0029), tubular epithelial single-cell necrosis (P = .0081), glomerular lesions (P = .0004), and glomerulosclerosis (P = .007). Comparing borderline proteinuric cats with nonproteinuric cats (reference variable), significantly less normal parenchyma (P = .0014) was present with a greater percentage of interstitial inflammation (P = .04), cortical and medullary scarring (P = .0052 and .0006, respectively), more tubular degeneration (P = .0003), cellular and lipid casts (P = .028), tubular dilation and cysts (P = .017 and .0299, respectively), tubular crystals (P = .031), tubular epithelial single-cell necrosis (P = .0009), and Bowman’s capsule mineralization (P < .0001). Glomerular lesions were the only histologic variable significantly different when comparing borderline proteinuric with proteinuric cats (P = .018). Populations of inflammatory cells and the occurrence of vascular lesions were not significantly different among groups. Patterns of inflammation (diffuse vs multifocal) and fibrosis (diffuse vs focal) were only significantly different when comparing proteinuric with nonproteinuric cats (P = .0046 and .0139, respectively).
A total of 33 of 46 CKD cats and 10 of 10 GC cats had blood pressure data available. Hypertension was present in 1 of 1 stage I, 6 of 12 stage II, 2 of 9 stage III, and 4 of 11 stage IV cats. All 10 GC cats were normotensive. Blood pressure data were not available for YC cats. Hypertension was more likely to occur in cats with CKD than in GC cats (P = .03). Presence of hyperplastic arteriolosclerosis, fibrointimal hyperplasia, or other vascular lesions was not significantly different between hypertensive and normotensive cats.
Discussion
While progress has been made in better categorizing stages of feline CKD and the associated prognosis and treatment recommendations, a great deal is still unknown about the etiopathogenesis of such a common disease. 41 Clinicopathologic data are useful for diagnosing, staging, and prognosticating but give few clues about the distribution and pattern of injury within the kidney. Therefore, it is difficult to determine which stages are characterized by irreversible lesions and at which stage therapeutic interventions should be targeted. In an attempt to bridge clinical parameters with renal pathology, reversible and irreversible histologic lesions affecting all renal compartments were identified for each stage of CKD. This study showed that reversible lesions were present throughout the stages of CKD while irreversible lesions were more prevalent in later stages than in early stages of CKD.
Histologic variables that were significantly different among stages affected the interstitial, tubular, and glomerular compartments. Overall, earlier stages of CKD (ie, stages I and II) retained a greater proportion of normal parenchyma in comparison to later stages (ie, stages III and IV). Within the interstitial compartment, the severity of inflammation was similar in earlier stages and significantly less than later stages. Lymphocytes were the most common constituent of inflammation at any stage, and a regionally extensive pattern was most frequent. Lipid within the interstitium was frequently associated with interstitial inflammation and scarring while edema was rare. Cortical scarring was prevalent in the final stages of CKD (stage IV) but was mild in cats in stages II and III. Tubular degeneration was significantly greater in later stages compared with earlier stages, although the degree of degeneration was similar between individual later stages (ie, stages III and IV). Tubular lipid was a frequent finding in normal and atrophic tubules. Single epithelial cell necrosis was significantly less in the earliest stage of disease (stage I) than in all other stages, but coagulative necrosis was rare. Glomerulosclerosis progressively worsened with CKD stage while other glomerular lesions were uncommon. Vascular lesions did not differ among IRIS stages.
Normal parenchyma unaffected by degeneration, atrophy, inflammation, or fibrosis was significantly less in later stages of CKD (ie, stages III and IV) compared with earlier stages (stages I and II) but similar between stages I and II. Interestingly, as little as 25% to 50% of the parenchyma was affected in the earlier stages of CKD, implying that even a mild degree of lesions could have functional significance. This is in contrast to the dogma that at least 75% of functional mass must be lost before clinical evidence of renal disease is evident. 8,9,21,30 The determination of the functional consequence of histologic lesions involving only ≤50% of the renal tissue was based on serum creatinine levels as an estimate of renal function, as is commonly used in clinical practice. Other functional tests such as glomerular filtration rates, which have been demonstrated to decline with 75% or greater surgical reduction in renal mass in cats, were not measured in this study. 1,7,8,38 Reasons for this contradiction may include uncertainty of the relationship between histologic assessment of normal parenchyma and functional renal mass and potential variation in whether the most or least severe renal lesions were sampled for histopathology in the 11 of 46 cases that were collected retrospectively in a unilateral manner.
Percent tubular degeneration was significantly greater in later stages than in earlier stages of disease but was not different between controls and stage I. This could be a result of small sample size for stage I. In fact, the only histologic lesions significantly different between geriatric controls and stage I CKD cats were percent normal parenchyma and tubular epithelial single-cell necrosis, with the latter being greater in geriatric controls. When comparing all CKD stages, there was an obvious upward trend of tubular epithelial single-cell necrosis with each stage, although statistical significance was not reached between stages. Last, the authors acknowledge that the methods used would not differentiate single-cell necrosis and include apoptosis.
Interstitial fibrosis and scarring, confirmed by Masson’s trichrome stain, was statistically greater in stage IV compared with all other stages. Cats in stage IV were most likely to have 25% to 50% of their kidneys affected by scarring in comparison to ≤25% scarring in other stages. Interstitial fibrosis did not increase significantly between cats in IRIS stage II and IIIs. This is in contrast to a previous study (which did not evaluate tissues stained with trichrome) in which interstitial fibrosis was the lesion that best correlated with severity of azotemia. 10 This suggests that additional pathologic processes other than fibrosis are involved in disease progression and implies that initiation of any potential antifibrotic therapies in CKD cats should occur prior to stage IV, when irreversible fibrosis is most severe.
While mean percentage of tissue affected by interstitial inflammation increased between stages II and IV, histologic scores for inflammation were typically greater than scarring scores for each stage. This would be compatible with inflammation preceding and inducing fibrosis. 16 However, patterns of scarring did not parallel that of interstitial inflammation, and a significant progression in scarring patterns from focal to regional to diffuse with increasing IRIS stage was not found. This suggests that instigators of fibrosis other than inflammation may be players in the progression of CKD and should be identified and evaluated as potential therapeutic targets.
Glomerulonephropathies, with the exception of FSGS, were infrequently diagnosed by light microscopy (4/46). Due to the retrospective nature of this study, tissues were not available for ultrastructural evaluation. However, our results are similar to previously published work on feline renal disease in which glomerular disease was uncommon, with a reported prevalence of 8% to 15%. 12,20,24,29 In the present study, the most common glomerular changes were global glomerulosclerosis and FSGS. Global glomerulosclerosis was the only histologic variable in this study that precipitously increased with IRIS stage progression. Interestingly, glomerulosclerosis was more severe in nonazotemic geriatric cats than in stage I CKD cats. This suggests that although global glomerulosclerosis appears to be a feature of aging, it can be considered pathologic in certain scenarios. 43 Additional studies on nonazotemic geriatric cats are needed to better characterize normal renal aging changes.
Focal segmental glomerulosclerosis is a poorly characterized entity in veterinary medicine. 3,42 Histologic features are one or more glomeruli with segmental consolidation of capillary tufts with replacement by increased extracellular matrix, cellularity, or both. 11,40 Clinically, proteinuria is a hallmark of FSGS and typically is accompanied by variable degrees of hypoalbuminemia, hypercholesterolemia, and edema (nephrotic syndrome) in human patients. 11 Etiologies for FSGS are classified as either primary (idiopathic or genetic) or secondary (adaptive); the latter encompasses drug-induced or viral-associated causes. 11,40 In humans, distinct histologic variants of FSGS have been identified, based on location and character of the lesion within the glomerular tuft, and have been found to relate to specific therapeutic responses and prognostic outcomes. For example, the variant perihilar FSGS is defined by sclerosis at the vascular pole and often seen in secondary, adaptive FSGS. Individuals with the perihilar variant of FSGS typically have nephrotic syndrome less frequently with a milder degree of proteinuria than other variants. 40 Aging, advanced renal disease, and obesity are 3 examples of conditions that are associated with adaptive FSGS. 11,40
In the current study, 8 cats with CKD had histologic evidence of FSGS. Urinalysis and UPC measurements were available for 6 of the 8 cats. Although none of the cats had nephrotic syndrome, 5 of 6 were proteinuric, with only 2 of 6 within nephrotic range (UPC >2.0). A single cat was hypoalbuminemic, but proteinuric status was not available for this cat. However, hypoalbuminemia in this cat was most likely due to pleural effusion from severe mediastinitis secondary to complications from an esophageal feeding tube rather than glomerular disease. While Leishmania infection in dogs has been associated with chronic glomerulosclerosis, other primary or secondary etiologies for FSGS in veterinary medicine have not been reported. 2 Viral infections (eg, parvovirus or feline immunodeficiency virus) were not suspected clinically. None of the cats in the current study was treated with any FSGS-associated drugs. While these 8 cats may have had primary FSGS, that is typically a diagnosis of exclusion. A more plausible explanation would be that these lesions were secondary to adaptive changes due to the loss of functional renal mass in these cats. Loss of functional glomeruli leads to hyperfiltration of the remaining glomeruli, which can result in podocyte injury and eventually sclerosis. In support of this theory, a perihilar pattern of segmental sclerosis was identified in 4 of the 8 cats with FSGS in this study. Based on histopathology and clinicopathologic data, it seems most likely that FSGS in these 8 cats were likely secondary, or adaptive, to loss of renal mass.
In the present study, proteinuria was an uncommon finding and typically present in later stages of disease. Despite the infrequency of proteinuria in CKD cats, an association with increased severity of tubular degeneration, inflammation, fibrosis, tubular epithelial single-cell necrosis, and decreased normal parenchyma was detected. Similarly, positive correlations between severity of proteinuria and tubular epithelial injury, interstitial inflammation, and fibrosis have been found in humans. 18 Protein exposure to tubular epithelial cells perpetuates renal injury by direct damage to these cells or indirectly through production of proinflammatory and profibrotic cytokines. 5,18,35
Cats with glomerular lesions had an average UPC greater (1.3 ± 1.8) than those cats without glomerular lesions (0.2 ± 0.1), but a UPC >2 was present in only 3 cats. These were late stage (stage III: n = 1, stage IV: n = 2), and glomerular lesions were marked (eg, MPGN pattern and thrombotic microangiopathy [TMA]). A UPC >2 typically indicates glomerular disease, while tubular reabsorption defects are suspected if <2. 15 These data suggest that proteinuria in study cats is unlikely a result of primary glomerular disease in most cases and is more likely tubular in origin. In this study, proteinuria was more common in late-stage cats, implying that it is a consequence rather than an initiator of disease.
Mineralization of Bowman’s capsule and tubular basement membranes was more frequent in cats with more severe renal disease (ie, stages III and IV) than those with milder disease (stages I and II). While calcium-phosphorus products (CPPs) were not evaluated in the current study, mineralization may be the result of an increased CPP secondary to late-stage renal disease. 22,28
A novel glomerular lesion identified in this report was TMA. This cat presented for acute onset of ataxia and blindness. Systolic blood pressure was 190 mm Hg at presentation with severe proteinuria (UPC 4.8) and a creatinine of 3.2 mg/dl. Histologically, scattered glomeruli had endothelial swelling with occlusion of capillary lumina, which is a characteristic feature of the TMA variant glomerular endotheliosis. 31,39 Endotheliosis diminishes filtration and thus decreases overall glomerular filtration rate (GFR) in affected individuals. 39 Endotheliosis is often seen in women due to preeclampsia with clinical hypertension and proteinuria. Although unrelated to pregnancy in the cat reported here, confirmed hypertension may have resulted in endothelial injury with endotheliosis. The relationship of this to feline CKD is unknown.
Vascular lesions other than fibrointimal hyperplasia and hyperplastic arteriolosclerosis were uncommon. Fibrointimal hyperplasia appeared as segmental to circumferential thickening of the tunica intima accompanied by a discontinuous internal elastic lamina. It was present in over half of all CKD cats as well as in young normal cats and did not differ among stages of CKD. Fibrointimal hyperplasia has been associated with hypertension but is indistinguishable from similar lesions in aging, normotensive human patients. 19,26 Ultimately, vascular lesions associated with hypertension may lead to glomerular damage, sclerosis, tubular atrophy, and interstitial fibrosis. 27 In the present study, however, hypertensive cats were not more likely to be affected by fibrointimal hyperplasia than were normotensive cats. In a study evaluating lesions in the aging feline kidney, vascular lesions were infrequent (6/600 cats), although lesion detection may have been limited in that study by the absence of histochemical stains. 20 In a recent study assessing renal histologic lesions in diabetic cats, vascular lesions were infrequent, with only 4 cats affected by arterial hypertrophy and a single cat with vascular thickening and splitting. 44 Lesions were not significantly different between those cats with or without diabetes mellitus. 44 Last, in the present study, fibrointimal hyperplasia was prevalent in young, nonazotemic cats (6/11). From these data, the relationship of this vascular lesion to feline kidney disease is uncertain.
Hyperplastic arteriolosclerosis, a feature of systemic hypertension thought to be due to microvascular injury, was found in 48% of normotensive cats with CKD. This is in contrast to a previous report of 3% in normotensive CKD cats. 10 The reason for this discrepancy is not clear but may indicate that systemic blood pressure does not adequately represent local renal hemodynamics in cats. For example, in a previous study, cats with CKD had significantly higher renal vascular resistance than cats without CKD, despite no difference in systemic blood pressure between groups, as well as no correlation between systemic blood pressure and renal arterial resistance indices. 34 In contrast, in humans, indices of vascular resistance (RI) are associated with the severity and progression of CKD and RI correlates with systemic blood pressure. The reason for the difference between species is unclear, but increased RI may be a more accurate measurement of local renal hemodynamics in cats.
Irregularly sized lipid vacuoles were frequent within the tubular epithelium and free within the interstitium of CKD cats in this study. Interstitial lipid is thought to originate from tubular lipid after rupture of basement membranes and cell lysis. 23 The role of tubular lipidosis in feline CKD has not been determined. Given that many different lipids have identical histologic appearances, it is unclear whether the larger lipid vacuoles and lipid casts are simply due to coalescence of smaller vacuoles from normal feline tubules or if they are composed of different types of lipids.
Conclusions
This study documents differences in histologic lesions and patterns among stages of CKD. The severity of tubular degeneration, interstitial inflammation, fibrosis, and glomerulosclerosis was significantly greater in later stages of CKD compared with early stages of disease, although glomerulosclerosis was the only variable that increased significantly with each disease stage. Proteinuria was associated with increased severity of tubular degeneration, inflammation, fibrosis, tubular epithelial single-cell necrosis, and decreased normal parenchyma. These findings will help pinpoint therapeutic targets and the stage of disease at which they should be initiated.
Footnotes
Acknowledgements
We thank Todd Bass in the histopathology laboratory at the Colorado State University Diagnostic Medical Center, Sangeeta Rao for statistical assistance, and William Tyler Brock for contributions to the completion of this project.
Author Contribution
Conception or design: SM, RC, CD, JQ Data acquisition, analysis, or interpretation: SM, RC, CD, JQ Drafting the manuscript: SM All authors participated in critically revising the manuscript, gave final approval, and agree to be accountable for all aspects of work to ensure integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support was provided by Frankie’s Fund for Feline Stem Cell Therapy.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
