Abstract
Canine histiocytic proliferative disorders include a wide spectrum of diseases characterized by different biologic behaviors. The etiology and pathogenesis of these diseases are largely unknown. The clinicopathologic, morphologic and immunophenotypic characteristics of canine localized and disseminated histiocytic sarcoma were examined in 39 dogs. Rottweilers, Bernese Mountain Dogs, and retrievers were most commonly affected (79%). Localized histiocytic sarcomas (19 dogs) arose from a single site, and metastatic lesions were observed in draining lymph nodes. Predilection sites were subcutis and underlying tissues on extremities, but tumors occurred in other locations, including spleen, lung, brain, nasal cavity, and bone marrow. Disseminated histiocytic sarcomas (20 dogs), a multisystem disease previously described as malignant histiocytosis, primarily affected spleen, lungs, bone marrow, liver, and lymph nodes. Both localized and disseminated canine histiocytic sarcomas were composed of pleomorphic tumor cell populations. CD1+, CD4-, CD11c+, CD11d-, MHC II+, ICAM-1+, Thy-1± tumor cells were identified in all snap-frozen samples (31 dogs). This phenotype is characteristic for myeloid dendritic antigen-presenting cell lineage. Hence, canine localized and disseminated histiocytic sarcomas are likely myeloid dendritic cell sarcomas. Dendritic antigen-presenting cells are a heterogeneous cell population with regards to their ontogeny, phenotype, function, and localization. The exact sublineage of the proliferating dendritic antigen-presenting cells involved in canine histiocytic sarcomas remains to be determined. Phenotypic analysis of formalin-fixed tissues from eight dogs was limited by available markers. Morphologic features and the phenotype CD18+, CD3-, and CD79a- were the most useful criteria to indicate likely histiocytic origin.
Keywords
Previous reports of canine proliferative histiocytic diseases have documented a range of disorders with marked differences in clinical behavior and pathologic features. These disorders include cutaneous histiocytoma, reactive histiocytosis (cutaneous or systemic form), and malignant histiocytosis. 1,22,26,30,32,33,39,47 Canine cutaneous histiocytoma is a mostly benign epitheliotropic neoplasm composed of intraepidermal dendritic antigen-presenting cells (APC) also called Langerhans cells. 15,22,33 Most canine cutaneous histiocytomas spontaneously regress. Their clinical and pathologic features resemble those of some forms of Langerhans cell histiocytosis (LCH) in humans. 11,13,45 Canine reactive histiocytosis is either limited to the skin (cutaneous histiocytosis) or simultaneously affects skin and other organ systems (systemic histiocytosis). 1,26,30,41,46 Skin lesions are characterized by an angiocentric pleocellular infiltrate composed of activated interstitial myeloid perivascular dendritic APC of the dermis, lymphocytes, and neutrophils. 1 The clinical course and response to immunosuppressive therapy are consistent with a reactive process, which likely arises in the context of immune system dysregulation. 1
Canine histiocytic neoplasia occurs as a localized tumor or a disseminated neoplastic process. 14,17,19,23,32,39 Localized histiocytic sarcomas develop from a single site. They are locally invasive and metastasize to draining lymph nodes. The majority arise in the subcutis, but other primary locations have been observed. 17 Disseminated canine histiocytic sarcoma was first described as malignant histiocytosis of Bernese Mountain Dogs. 32,37,39 However, this condition since has been documented in other breeds. 18,23,35,43,44,57,60 Disseminated histiocytic sarcoma is an aggressive multisystem disease characterized by presence of multiple tumor masses in several organ systems. Primary sites are spleen, lung, and bone marrow. Secondary lesions are observed in lymph nodes and liver, and subsequently other organs can be affected. The cellular origin of canine histiocytic sarcomas is largely unknown. Malignant histiocytic proliferations have been documented best in humans and include histiocytic sarcomas and Langerhans cell histiocytosis. 3,11,45 Histiocytic sarcomas in humans are of either macrophage origin or myeloid dendritic APC origin. 3,11,45
The goal of this study was to document morphologic and phenotypic characteristics of canine localized histiocytic sarcoma (19 dogs) and canine disseminated histiocytic sarcoma (20 dogs). Canine histiocytic sarcomas are composed of a pleomorphic proliferation of myeloid dendritic APC and hence resemble dendritic cell sarcomas described in humans. 11,45
Material and Methods
Tissue handling
Tissue samples from 39 dogs included needle biopsies, punch and excisional biopsies, bone marrow smears, or postmortem tissues. The samples were submitted by veterinary practitioners, the Veterinary Medicine Teaching Hospital at the University of California at Davis, or other veterinary schools throughout the United States. Fresh tissue samples (31/39) were bisected, and one half was fixed in 10% neutral buffered formalin and embedded in paraffin. The other half was embedded in OCT compound (Finetek, Sakura USA, Torrance, CA) and snap frozen in isopentane cooled to the point of freezing in liquid nitrogen. Normal canine control tissues for immunohistochemistry (skin, tongue, spleen, thymus) were handled identically. The remaining tissue samples (8/39) were submitted in formalin only.
Classification
Morphologic features were evaluated on 4–6-µm, hematoxylin and eosin (HE)-stained sections of formalin-fixed, paraffin-embedded tissue. The presumptive diagnosis of a histiocytic neoplasm was based on morphologic features previously described. 18,23,32 According to the criteria used for humans, the cases were categorized as localized or disseminated histiocytic sarcomas. 11,45
Antibodies
A panel of leukocyte-specific antibodies (Table 1), previously used for characterization of canine cutaneous histiocytomas and canine reactive histiocytosis, was used in this study. 1,33 The sources and reactivity of these antibodies were fully documented previously. 31 Cryosections (6 µm) from 31 dogs were evaluated with the entire panel of antibodies (Table 1). A limited number of antibodies recognize formalin-fixed epitopes (Table 1). These antibodies can detect expression of CD3∊, CD11d (αD), CD18, CD45RA, and CD79a in formalin-fixed, paraffin-embedded tissues. These reagents were used for evaluation of eight dogs where only formalin-fixed, paraffin-embedded tissues were available.
Monoclonal antibodies specific for canine leukocyte antigens. 31

∗ Available antibodies for formalin-fixed, paraffin-embedded tissue.
Immunohistochemistry
Cryosections were fixed in acetone (3 minutes), and endogenous peroxidase was quenched by immersing slides in hydrogen peroxide (0.3%), sodium azide (0.1%), and phosphate-buffered saline (PBS) for 10 minutes. Following a blocking step with 10% heat inactivated horse serum in PBS (20 minutes), the dog-specific monoclonal antibodies were applied to the sections for 30 minutes. Appropriate antibody dilutions, prepared in 10% horse serum in PBS, were determined by previous titration of each antibody on frozen sections of normal canine spleen. If available, skin samples from each dog were used as control tissue for the expression of the CD1a allotype by the intraepidermal Langerhans cells recognized by CA9.AG5 (Table 1). Omission of the primary antibody or application of an isotype-matched nonspecific antibody was used as a negative control in each run. Secondary biotinylated horse anti-mouse IgG (Vector, Burlingame, CA) was applied to each section for 30 minutes followed by streptavidin–horseradish–peroxidase (Zymed, South San Francisco, CA) for 20 minutes according to instructions of the manufacturers. Between each step, the sections were washed thoroughly in PBS. Amino-9-ethyl-carbazole (AEC; Sigma Chemical Co., St. Louis, MO) was used as the chromogen. The tissue sections were counterstained with hematoxylin (Gill's formula 3; Fischer, Pittsburgh, WA), air dried, and coverslipped.
Eight tissue samples were submitted in formalin only. Immunohistochemical phenotyping was performed on 6-µm paraffin-embedded sections. The sections were deparaffinized in xylene and hydrated through graded ethanol solutions. Several antigen retrieval procedures to unmask antigen epitopes were evaluated, including enzymatic digestion with trypsin (0.1% trypsin and 0.1% CaCl2) for 20 minutes at 37C) or microwaving (19 minutes) or steaming (30 minutes) of tissue sections immersed in 10 mM citrate, pH 6 (Dako, Carpinteria, CA). 48 The best results were achieved with steaming of the tissue sections in citrate buffer. Formalin-fixed tissue sections of all eight dogs were therefore steamed previous to the blocking step with 10% heat inactivated horse serum in PBS. The tissue sections were subsequently stained as described above.
Results
Clinical summary and distribution of lesions
Histiocytic sarcomas were observed in a variety of breeds (Table 2), but Rottweilers, Bernese Mountain Dogs, and retrievers were highly represented (79%). The ages of affected dogs ranged from 2 to 13 years, and a female:male ratio of 1.2:1 was observed. Localized histiocytic sarcoma was diagnosed in 19 dogs, and disseminated histiocytic sarcomas were seen in 20 dogs. Frequency of tissue involvement with localized histiocytic sarcoma and disseminated histiocytic sarcoma are listed in Table 3. The majority of localized histiocytic sarcomas were observed in the subcutis (13/19), whereas most disseminated histiocytic sarcomas presented with widespread lesions in several internal organ systems, and subcutaneous masses were seen in only seven dogs (Table 3).
Dog breeds affected with localized (LHS) and disseminated (DHS) histiocytic sarcoma.
Frequency of tissue involvement in 19 dogs with localized histiocytic sarcoma (LHS) and 20 dogs with disseminated histiocytic sarcoma (DHS).
∗ Metastatic lesions.
All except one of the subcutaneous localized histiocytic sarcomas were located on limbs. One dog presented with a mass on the chest. The tumor masses often invaded the deep dermis and underlying skeletal muscles and fascia. The remaining six localized histiocytic sarcomas (Table 3) affected the spleen, the tongue, a lung lobe, the brain stem, the nasal cavity, and a vertebral bone and epidural space (Fig. 1). For five dogs, samples from enlarged draining lymph nodes were submitted together with the primary tumor. Metastases were present in three lymph nodes, and two lymph node samples had no evidence of neoplasia. The localized histiocytic sarcoma in the spleen was associated with numerous liver metastases.

Vertebral column and spinal cord; sagittal section; Rottweiler, dog No. 19. A nonencapsulated, white homogeneous mass originates from the dorsal portion of the vertebral body. The mass protrudes into the vertebral canal and exerts compression on the spinal cord. Bar = 2 cm.
Disseminated histiocytic sarcomas were most often found in the spleen (Fig. 2), liver, lung, bone marrow, and lymph nodes (Table 3). Additional lesions were noted in many different organ systems (Table 3). The majority of dogs had a history of nonspecific initial clinical signs, which included anorexia, lethargy, weakness, and weight loss, and clinical evaluation revealed involvement of several organ systems at the time of presentation. Additional clinical signs varied. Dogs with pulmonary masses and neoplastic involvement of the hilar lymph nodes presented with dyspnea. Lameness was observed in dogs with extensive proliferative bone marrow lesions associated with destruction of surrounding bone. Intravertebral lesions were seen in only two Rottweilers; both dogs presented with ataxia and hind leg paresis and paralysis.
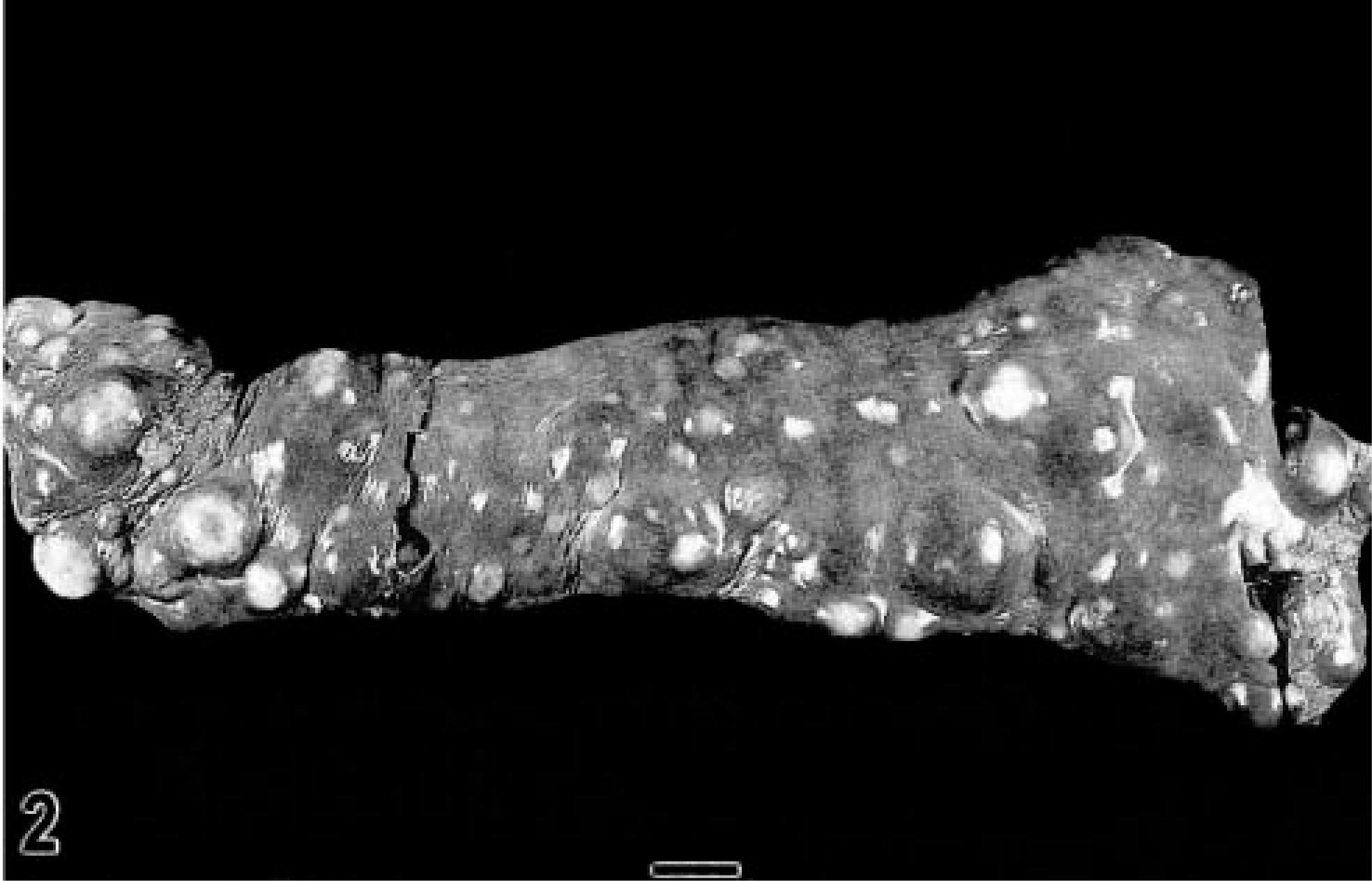
Spleen; Bernese Mountain Dog with disseminated histiocytic sarcoma, dog No. 29. The spleen is markedly enlarged and contains numerous white, nonencapsulated, and often coalescing nodules. The nodules range in size from a few millimeters to 2 cm in diameter. Bar = 2 cm.
Clinical follow-up revealed a more favorable outcome for dogs with localized histiocytic sarcomas than for dogs with disseminated lesions. Wide surgical excisions or amputation in dogs with large masses on extremities were performed in all 13 dogs with subcutaneous sarcomas. No postsurgical recurrence or metastasis was reported in 5/13 dogs. The remaining eight dogs were lost to further clinical follow-up. Radiotherapy was used successfully to treat a histiocytic sarcoma in the tongue, and no subsequent recurrence was reported. Of four dogs with internal localized histiocytic sarcomas, two were euthanatized shortly after the diagnosis was established and for two others submitted tissues had been collected upon necropsy. One dog was lost to clinical follow-up.
Clinical outcome in dogs with disseminated histiocytic sarcomas reflected the very aggressive behavior and poor prognosis of this disease. Fast deterioration and lack of response to chemotherapy was seen in seven dogs. One dog died spontaneously, and the other 6 dogs were euthanatized upon the owners' requests. Necropsies to evaluate the extent of the disease were performed on five of these dogs. Another seven dogs were in very poor general condition at the time of presentation. These dogs were euthanatized, and tissue samples for histology and immunophenotyping were collected upon necropsy. No clinical follow-up was available for six dogs.
Morphologic features
Localized and disseminated histiocytic sarcomas were characterized by identical morphologic features. Poorly demarcated, locally invasive tumor masses were composed of a dense pleomorphic cell proliferation, which effaced normal tissue architecture. There were multiple areas of necrosis. Morphology and arrangement of the tumor cell population varied among nodules and among different areas within a lesion (Figs. 3, 4). Some tumor nodules were composed of individualized large round cells with abundant amphophilic cytoplasm. Occasional cytoplasmic vacuolization was observed. A second pattern was characterized by variably dense proliferations of plump spindle cells with long cytoplasmic processes. Often both cell types were present within one lesion. Neoplastic multinucleated giant cells were common and presented as either large round cells or as bizarre stellate cells with long cytoplasmic processes (Fig. 3). There was marked anisokaryosis (Figs. 3). Large vesicular nuclei were round to oval or indented and twisted and contained one or several nucleoli; other nuclei were slightly smaller, round or elongated, and characterized by more condensed, coarse chromatin (Figs. 3, 4). The mitotic rate varied and ranged from one to seven mitosis per high power field (40× objective). Phagocytosis by the tumor cells was occasionally seen. The phagocytic vacuoles contained neutrophils or cellular fragments. The extent of a reactive leukocytic infiltrate varied among dogs and ranged from a few dispersed cells to a prominent reactive infiltrate. Neutrophils were consistently observed. The number of small lymphocytes ranged from occasional dispersed cells to aggregates.
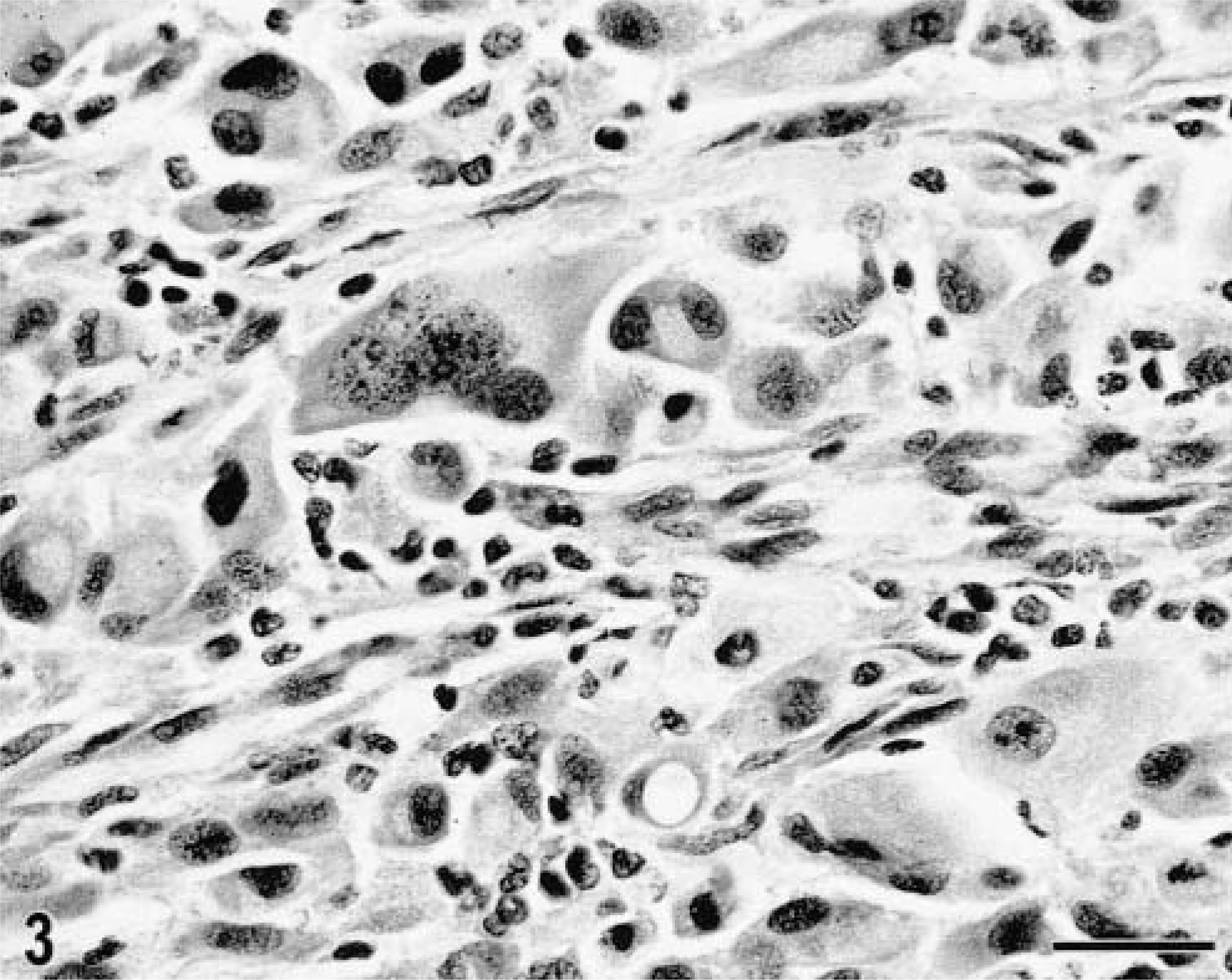
Lung, histiocytic sarcoma; Bernese Mountain Dog, dog No. 15. The pleomorphic tumor cell population consists of spindle cells, individualized round cells, and multinucleated giant cells. There is marked anisokaryosis. Admixed are scattered reactive small lymphocytic. HE. Bar = 30 µm.
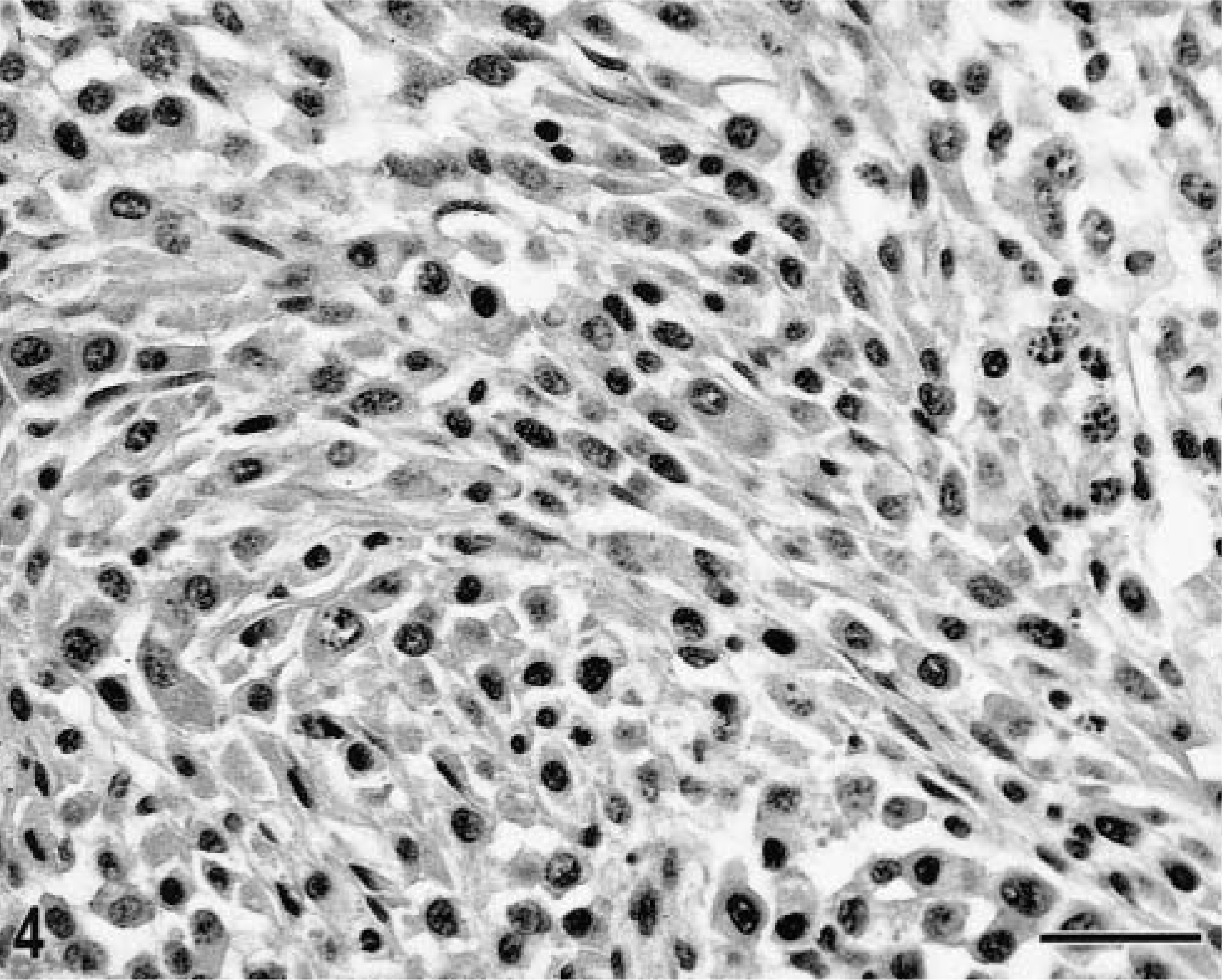
Subcutaneous tissue, histiocytic sarcoma; Golden Retriever, dog No. 11. The tumor is composed of plump spindle cells arranged in loose bundles. Admixed are occasional round tumor cells. The tumor cells have round to oval nuclei, mostly characterized by condensed chromatin. A few round vesicular nuclei with more prominent nucleoli are present; occasional karyorrhexis is also present. HE. Bar = 30µm.
Immunohistochemistry
Despite their variable morphology, the tumor cells expressed a consistent phenotype. Moreover, no differences in the phenotype was noted between localized and disseminated histiocytic sarcomas. Based on the expression of the leukocyte antigens CD45 and CD11a/CD18, all 39 sarcomas were of leukocytic origin (Table 4). Tumor cells in snap-frozen tissues of 31 dogs consistently expressed molecules involved in antigen presentation, including CD1b, CD1c (Fig. 5), and major histocompatibility complex (MHC) class II. In carriers of the CD1a allotype recognized by CA9.AG5, coexpression of CD1a was seen (Table 4). The tumor cell populations expressed the β2-integrin adhesion molecule CD11c (Fig. 6) and the intercellular adhesion molecule ICAM-1. With consistent expression of CD1, CD11c, MHC class II, and ICAM-1, the tumor cells therefore displayed a phenotype characteristic of myeloid dendritic APC origin. 5,29,33 The tumor cells were consistently negative for CD4, which is upregulated upon activation of dendritic APC, as seen in canine reactive histiocytosis. 1 Additional surface proteins were identified on these tumor cells; however, they did not contribute to further differentiation of the tumor cell population. These surface molecules are either broadly expressed by leukocytes, such as CD44, or they were inconsistently expressed by the tumor cells, such as CD45RA or the adhesion molecules CD11b, CD49d, and ICAM-3 (Table 4). The majority of tumor tissues lacked expression of CD90 (Thy-1); its expression was limited to a perivascular subpopulation of tumor cells in a few cases (Table 4). Reactive lymphocytic infiltrates consisted mainly of CD8+, TCRαβ+ T cells. CD21+ B lymphocytes were less common and occurred as small aggregates. CD4+ T cells and TCRγδ T cells were rare.
Phenotype of dendritic APC in localized and disseminated histiocytic sarcomas evaluated from frozen (n = 31) or formalin-fixed (n 8) canine tissues.

∗ CD1a allotype recognized by CA9.AG5.
† Antibodies not available.

Spleen; histiocytic sarcoma; Labrador Retriever, dog No. 20. The cytoplasmic membranes of the tumor cells stain positive for dog-specific CD1 antibody. Cryosection, immunhostochemistry with CA13.9H11, AEC. Bar = 30 µm.

Spleen; histiocytic sarcoma; Labrador Retriever, dog No. 20. The cytoplasmic membranes of the tumor cells also stain positive for dog-specific CD11c antibody. Cryosection, immunhostochemistry with CA11.6A1, AEC. Bar = 30 µm.
Limited conclusions could be drawn from immunohistochemical results for formalin-fixed, paraffin-embedded tissues from eight dogs. The histiocytes consistently expressed CD18, and those from two dogs expressed CD45RA. The tumor cell populations in all samples were negative for CD3∊ and CD79a, two surface molecules used to identify lymphocytes of T- or B-cell origin. A dispersed population of reactive small CD3∊+ lymphocytes was present in all cases; the number of reactive lymphocytes, however, varied among different tissue samples.
Discussion
In this investigation, we documented that canine histiocytic sarcomas are proliferations of myeloid dendritic APC displaying a CD1+, CD11c+, MHC class II+, ICAM-1+ phenotype. CD1 is consistently expressed by dendritic cells in the thymic medulla, the epidermis and mucosal epithelia, the dermis, and other tissues in humans and dogs. 5,12,29,31,33,36,49,59 Coexpression of CD1 and high levels of MHC class II antigens, both involved with antigen presentation, are characteristic for professional APC. 5,29,31,36 Adhesion molecules are variably expressed by different leukocyte subpopulations. The β2-integrin CD11c and the ICAM-1, which are consistently present on canine myeloid dendritic APC, 33 were strongly expressed by these tumor cells, whereas expression of other adhesion molecules such as CD11b, CD49d, and ICAM-3 was variable (Table 4). These observations confirm the myeloid dendritic cell origin of these tumor cells.
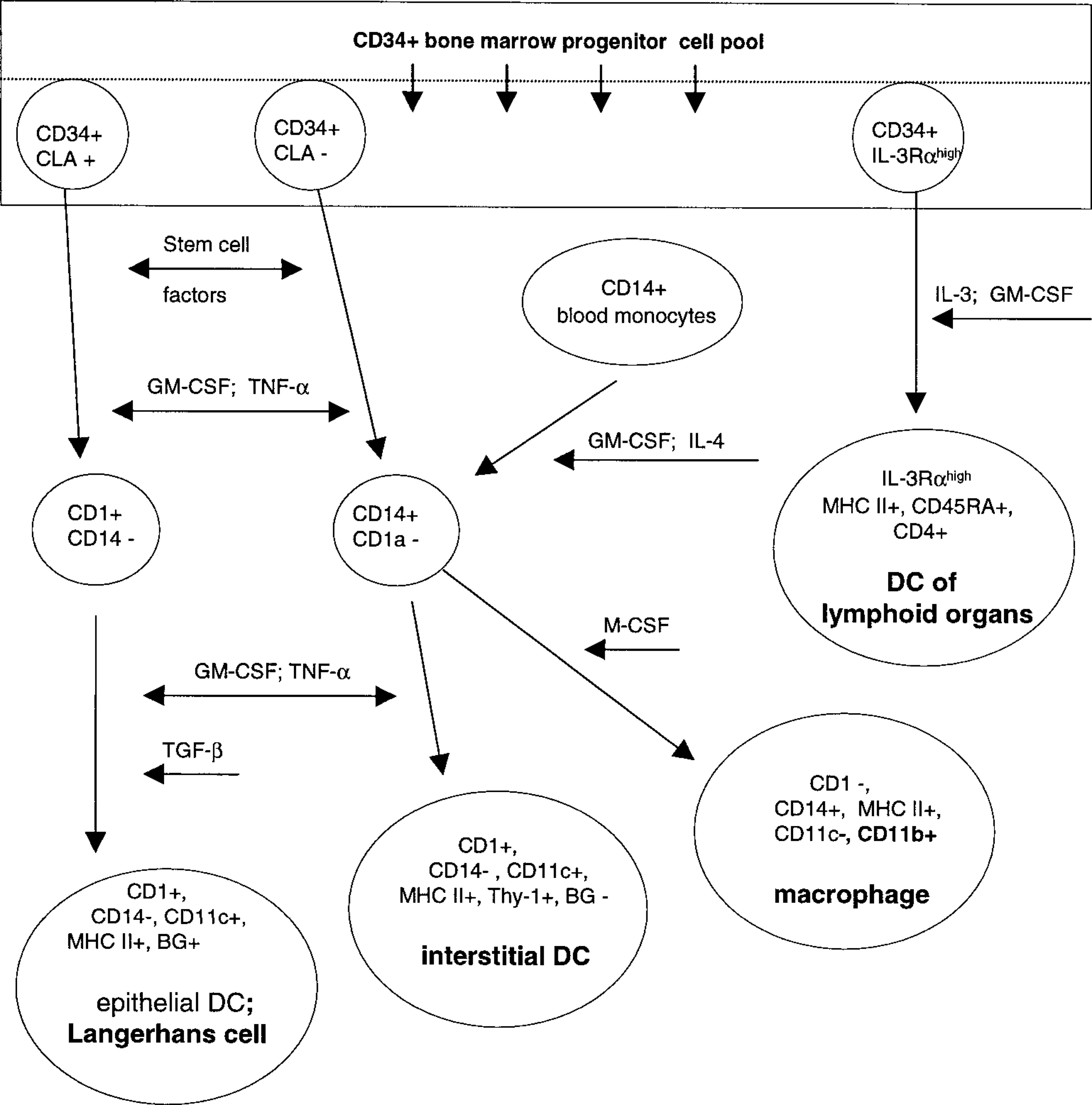
Myeloid dendritic APC themselves are a heterogeneous population of cells in terms of their ontogeny, phenotype, function, and localization 34,49 (Fig. 7). Together with macrophages, they evolve from a common CD34+ progenitor cell in the bone marrow (Fig. 7). Various stem cell factors and cytokines (Fig. 7) induce differentiation into either macrophages or myeloid dendritic cells. 6–8,40,49,54,55 Under the influence of granulocyte-macrophage colony-stimulating factor (GM-CSF), tumor necrosis factor α (TNF-α), and interleukin (IL)-4, CD34+ precursors differentiate into CLA+ (cutaneous lymphocyte-associated antigen) intraepithelial dendritic APC, called Langerhans cells or CLA− interstitial dendritic cells. Langerhans cells reside in the epidermis, follicular epithelium, and mucosal epithelia, and interstitial dendritic APC are present in various nonlymphoid tissues, e.g., as perivascular dermal dendritic APC. 5,9,29,33,34,36,49,59 As professional APC, myeloid dendritic cells are involved in the immune surveillance of their environment. 49 After antigen uptake, Langerhans cells and interstitial dendritic cells migrate to the lymphoid tissue. As interdigitating cells, they present processed antigens in context with MCH class I, MHC class II molecules or CD1 to lymphocytes and hence induce an immune response. 5,36,53 A third subset of myeloid dendritic APC has been documented in humans. 34,49 They develop from a CD34+ bone marrow precursor subset that expresses high levels of IL-3α receptors (IL-3Rα) (Fig. 7). At an undifferentiated stage and independently of inflammatory stimuli or previous uptake and processing of foreign antigens, IL-3Rαhigh, CD45RA+, CD4+ dendritic cells migrate from the bone marrow directly into T-cell dependent areas of lymphoid tissues. 34,49 In vitro studies have shown that upon maturation these dendritic cells become potent APC. 34
Proliferative diseases of dendritic APC have been well documented in humans. A large group of diseases with biologically variable behavior are categorized as LCH. 3,11 True malignant histiocytic proliferations are rare and may consist of macrophages or dendritic APC. 4,11,16 Disseminated forms of histiocytic sarcoma may mimic other malignancies such as anaplastic large cell lymphoma. 4,16,42 Localized histiocytic sarcomas often have been referred to as malignant fibrous histiocytomas. 21,28,50,58 Immunophenotyping is therefore essential to confirm the diagnosis. 4,11,16,42,61 S100, lysozyme, α1-antitrypsin, α1-antichymotrypsin, soya bean agglutinin, and peanut agglutinin have previously been used to diagnose the histiocytic origin of round cell and spindle cell sarcomas in humans. 10,13,38,42,51 However, CD1, CD11b, CD11c, CD14, CD68, and MHC class II are considered the most reliable markers for identifying histiocytic origin and differentiating between dendritic cell origin (CD1+, CD11c+) and macrophage origin (CD11b+, CD14+, CD68+). 10,20,24,42,51
Proliferative diseases of different dendritic APC subpopulations have recently been documented in dogs. Canine cutaneous histiocytoma, a mostly benign self-limiting cutaneous epitheliotropic neoplasm, is composed of a proliferation of CD1+, CD11c+, Thy-1−, CD4− canine Langerhans cells. 33 Canine cutaneous histiocytoma resembles some forms of human LCH. 3,11 Canine reactive histiocytosis, which includes cutaneous and systemic forms, has recently been documented as an angiocentric, pleocellular infiltration of CD1+, CD11c+, Thy-1+, CD4+ activated interstitial dendritic APC, CD8+, TCR αβ+ T cells, and neutrophils. 1 Clinical behavior and response to immunosuppressive therapy are consistent with an immunoregulatory disorder. 1
As in humans, localized and disseminated histiocytic sarcomas in dogs can mimic other spindle cell or round cell sarcomas. Accurate diagnosis therefore requires immunophenotypic evaluation of the lesions. Canine localized histiocytic sarcoma presented with a distribution, clinical behavior, and morphologic features that resembled those of previously documented canine malignant fibrous histiocytoma. 19,23 The diagnosis of canine malignant fibrous histiocytoma has always been controversial. More recent studies confirmed that this entity likely represents a collection of sarcomas of different cellular origin but with similar histologic features, e.g., fibrosarcomas, leiomyosarcomas, rhabdomyosarcomas, liposarcomas, synovial cell sarcomas, and histiocytic sarcomas. 23,56 In a recent study, fibrohistiocytic nodules in the spleen were described. 52 Immunhistochemistry was used to document the mesenchymal origin of these nodules. However, the large variation in morphologic features, immunophenotype and clinical behavior of these fibrohistiocytic nodules suggest that these proliferations were of various mesenchymal cell lineages, which likely included soft tissue sarcomas as well as dendritic cell sarcomas. However, a direct comparison of canine splenic dendritic cell sarcomas of this current study with the previously described fibrohistiocytic splenic nodules is not possible because those nodules were not evaluated for expression of leukocyte antigens. 52
Because of the diversity of the dendritic APC, the exact dendritic cell sublineage of histiocytic sarcomas has not been determined. Histiocytic sarcomas of nonlymphoid tissues most likely arise from peripheral interstitial dendritic APC, which have been documented in several organ systems. 25,27,29 Some dendritic cell sarcomas were characterized by coexpression of Thy-1 (Table 4), a molecule consistently expressed by normal perivascular dermal dendritic APC and by intralesional dendritic cells in canine reactive histiocytosis. 1,29,33 However, expression of Thy-1 by interstitial dendritic cells of nonlymphoid tissues often affected by histiocytic sarcoma, such as subcutis and lung, is unknown. Histiocytic sarcomas of lymphoid organs most likely arise from the interdigitating cells within the lymphoid tissue. Interdigitating cells in humans arise from three different subpopulations of dendritic APC. 2,34,49 Langerhans cells and interstitial dendritic cells initiate migration after antigen uptake and activation in peripheral tissues and enter the lymph nodes through afferent lymphatics. Human IL-3Rα-rich, CD45RA+, CD4+ dendritic APC enter the lymph organs as undifferentiated cells through the vascular system. Langerhans cells and interstitial dendritic cells have been identified in dogs, but the existence of canine IL-3Rα-rich, CD45RA+, CD4+ dendritic APC cannot be evaluated at this point because appropriate tools to identify canine IL-3Rα expression are not available. However, all tumor samples were consistently negative for CD4, and only a few canine histiocytic sarcomas coexpressed CD45RA.
The prognosis of canine disseminated and localized histiocytic sarcoma of internal organs was poor, but localized histiocytic sarcomas of the subcutis were less devastating. Despite the behavioral differences, no histologic or phenotypic differences were noticed. Early recognition of tumors in clinically easily accessible areas such as the subcutis facilitated timely surgical excision or radiotherapy, which might have decreased the chances of spreading. Disseminated histiocytic sarcomas predominantly affected internal organs, which could explain the delay in their recognition. Most dogs presented with widespread lesions at the time of diagnosis. It remains unknown whether disseminated sarcomas arise as a primary multicentric malignancy, which would justify the use of the term malignant histiocytosis. Alternatively, disseminated histiocytic sarcoma may represent a terminal stage of a localized histiocytic sarcoma of an internal organ with rapid proximal and distant spread.
The majority of canine localized and disseminated histiocytic sarcomas represent aggressive proliferations of myeloid dendritic APC. The broad range of morphologic features observed in these tumors requires immunophenotypic evaluation to confirm the myeloid dendritic cell origin. Given the complexity of dendritic cell differentiation, 34,49 additional studies are required to identify the precise origin(s) of the neoplastic myeloid dendritic APC population.
