Abstract
Histiocytic proliferative diseases include reactive and neoplastic proliferations of dendritic cells (DC) or macrophages. Various forms of DC proliferations have been documented in humans and dogs; their etiology is largely unknown. With the exception of a few case reports, histiocytic proliferations have not been characterized in cats. This study summarizes clinical, morphologic, and immunophenotypic features of a feline progressive histiocytosis (FPH) in 30 cats. There was no breed or age predilection. Females were more often affected than males. Solitary or multiple nonpruritic firm papules, nodules, and plaques had a predilection for feet, legs, and face. Lesions consisted of poorly circumscribed epitheliotropic (13/30) and nonepitheliotropic (17/30) histiocytic infiltrates of the superficial and deep dermis, with variable extension into the subcutis. The histiocytic population was relatively monomorphous early in the clinical course. With disease progression, cellular pleomorphism was more frequently encountered. Histiocytes expressed CD1a, CD1c, CD18, and major histocompatibility complex class II molecules. This immunophenotype suggests a DC origin of these lesions. Coexpression of E-cadherin, a feature of cutaneous Langerhans cells, was only observed in 3 cats. FPH followed a progressive clinical course; the lesions, however, were limited to the skin for an extended period of time. Terminal involvement of internal organs was documented in 7 cases. Treatment with chemotherapeutics or immunosuppressive and immunomodulatory drugs was not successful. The etiology of FPH remains unknown. FPH is best considered an initially indolent cutaneous neoplasm, which is mostly slowly progressive and may spread beyond the skin in the terminal stage.
Keywords
Histiocytic proliferative diseases include proliferations of dendritic cells (DC) and macrophages. 38 The latter are most efficient in phagocytosis of pathogens, whereas DC are primarily involved in antigen presentation. Different DC sublineages home to specific tissues. 2, 8, 21, 29, 33, 38 Langerhans cells populate the epidermis, the epithelia of mucous membranes, and the follicular epithelium, and a few cells may be seen in the perivascular dermis. Interstitial DC are encountered in many organs; in the skin (dermal DC) they mainly live in the perivascular dermis. The plasmacytoid DC constitute a major DC population of the peripheral lymphoid organs in humans. 29, 33 The latter DC population has not been identified in companion animals to date.
Histiocytic proliferative diseases have been well documented in humans and dogs. 1, 3, 5, 24, 27, 28, 31 Their etiology and pathogenesis are largely unknown. Canine cutaneous histiocytomas occur as solitary or multiple skin nodules and are composed of Langerhans cells. 28 Histiocytomas are mostly self-limiting and regress spontaneously. 28 Canine reactive histiocytosis presents with a cutaneous form and a systemic form. 1 The lesions are characterized by a nodular, pleocellular infiltrate composed of activated dermal DC, lymphocytes, and fewer neutrophils. 1 The morphologic features and the clinical behavior of canine reactive histiocytosis as well as its response to therapy with immune-suppressive drugs suggest a dysregulation of the immune system. 1 The canine solitary or disseminated histiocytic sarcomas, also referred to as malignant histiocytosis, are malignant tumors and have a poor prognosis. 3 The majority of cases are of DC origin. However, the exact sublineage of DC involved in these malignant tumors is mostly unknown. Some canine histiocytic sarcomas are of macrophage origin and primarily arise in the splenic red pulp or the bone marrow. 26
Histiocytic proliferative diseases are uncommon in cats, and their documentation is limited to a few case reports. 10, 12– 14, 18 The goal of this study was to characterize a histiocytic proliferative disease observed in 30 cats. Feline progressive histiocytosis (FPH) presents with solitary or multiple skin nodules. Although initially limited to the skin, the lesions have the potential to evolve into a malignant histiocytic neoplasia with metastases to lymph nodes and various internal organs.
Material and Methods
Animals and tissue samples
Tissue samples from 30 cats (Table 1) were submitted by veterinary practitioners, the Veterinary Medicine Teaching Hospital at the University of California at Davis, or other veterinary schools throughout the United States and Canada to the Leukocyte Antigen Biology Laboratory at the University of California for a second opinion and immunohistochemical stains to determine cell lineage of the lesions. As such, the true incidence of this disease is difficult to estimate, but is likely to be very low. The samples submitted included punch biopsies or excisional biopsies of skin nodules. In 7 cats samples were collected at different times throughout the disease process and submitted for histology and immunohistochemistry. Clinical history and follow-up were received upon submission of the biopsies and by contacting referring veterinarians or owners. Seven cats were necropsied, and internal organ samples were evaluated.
Feline progressive histiocytosis in 30 cats.∗
∗ u = unknown; DSH = Domestic Shorthair cat; Mix = mixed breed cat; DLH = Domestic Longhair cat; Siam = Siamese; TS = Tortoise Shell cat; m = male; mc = male castrated; f = female; fs = female spayed.
Tissue handling
Fresh skin samples obtained from 16 cats were bisected, and one-half of each sample was fixed in 10% neutral buffered formalin and embedded in paraffin. The other half was embedded in OCT-compound (Finetek, Sakura USA, Torrance, CA) and snap frozen in methylbutane cooled to the point of freezing with liquid nitrogen. Formalin-fixed, paraffin-embedded tissue blocks containing one or more skin samples were submitted from 9 cats. Morphologic features were evaluated on 4–6-μm hematoxylin and eosin (HE)–stained paraffin sections. Immunohistochemistry was performed on 6-μm cryosections (16 cats) or on 6-μm paraffin sections (9 cats). Postmortem samples of internal organs (7 cats) were handled as described above. For the remaining 5 cats, only HE-stained paraffin sections were available.
Antibodies
A panel of monoclonal antibodies specific for leukocyte cell surface molecules (Table 2) was used to evaluate tissues from 25/30 cats. Most of these antibodies are specific for feline leukocyte antigens (Table 2) and have been fully characterized by the Leukocyte Antigen Biology Laboratory at the University of California. 25 A few antibodies are specific for canine leukocyte antigens, but cross-reactive for feline antigens. 25 Anti-E-cadherin, anti-CD3∊, and anti-CD79a are antibodies specific for conserved cytoplasmic domains of the respective human proteins. These antibodies have been characterized as cross-reactive and have been used extensively in veterinary immunohistochemistry. 17, 22
Monoclonal antibodies (MABs) used for immunophenotyping of 25 cats with feline progressive histiocytosis: fresh, snap-frozen tissue from 16 cats, formalin-fixed tissues from 9 cats.∗

∗ LABL = Leukocyte Antigen Biology Laboratory, Peter F. Moore, University of California, Davis; DC = dendritic cells; SE = Serotec, Oxford, UK; BD-PH = BD Biosciences Pharmingen, San Diego, CA; MHC = major histocompatibility complex; TL = Transduction Laboratories, Lexington, KY.
†Cross-reactive with feline tissue.
Immunohistochemistry
Immunohistochemistry was performed as previously described. 1 Cryosections were fixed in acetone, and endogenous peroxidase was quenched with hydrogen peroxide (0.3%) and sodium azide (0.1%) in phosphate-buffered saline (PBS). Blocking of nonspecific binding was achieved with 10% heat-inactivated horse serum in PBS (20 minutes at room temperature). Most primary antibodies (Table 2) were applied as a 1°10 diluted tissue culture fluid in PBS containing 10% inactivated horse serum (30 minutes at room temperature). Anti-CD3∊ and anti-CD79a were used as previously described. 17, 20 Omission of the primary antibody or application of an isotype-matched, non–cross-reactive antibody was used as a negative control in each run. Secondary biotinylated horse anti-mouse IgG (Vector, Burlingame, CA) was applied on each section, and subsequently the sections were stained by the biotin-streptavidin-horseradish-peroxidase method according to the manufacturer's instructions (Zymed, South San Francisco, CA) and as previously described. 11 Amino-9-ethyl-carbazole (Sigma Chemicals Co, St. Louis, MO) was used as chromogen. Subsequently the sections were counterstained with hematoxylin (Gill's formulation 2; Fischer, Fair Lawn, NJ) and cover-slipped with an aqueous mounting media (Faramount; DAKO, Carpinteria, CA).
Paraffin sections were deparaffinized in xylene and hydrated through graded ethanol solutions. Hydrated sections were steamed in 10-mM Citrate buffer, pH6 (DAKO, Carpinteria, CA) at 95–98°C for 20 minutes. Samples were subsequently stained as described above.
Double-labeling immunofluorescence
Lesions from 4 cats were also evaluated by double-label immunofluorescence to evaluate coexpression of CD1 and CD5 by the lesional histiocytes and coexpression of CD3∊ and CD5 by admixed lymphocytes.
Cryosections were fixed in acetone and washed in PBS. Blocking of nonspecific binding was achieved with 10% heat-inactivated horse serum in PBS (20 minutes at room temperature). All antibodies were diluted in PBS containing 10% inactivated horse serum. Anti-feline CD5 or anti- CD3∊, respectively, was applied as a 1°10 dilution of tissue culture fluid for 30 minutes at room temperature. After a wash in PBS, the secondary antibody Texas red anti-mouse IgG (Vector) was applied as a 1°100 dilution for 30 minutes. A subsequent wash in PBS was followed by an additional blocking with an irrelevant, non–cross-reactive, canine-specific, anti-CD3 antibody (Ca17.2A12; ascites; dilution at 1°100). Without an additional wash, biotinylated anti-feline CD1a was applied for 30 minutes. After washing with PBS, fluorescein anti-biotin antibody (Vector) was applied for 30 minutes at a 1°50 dilution. Subsequent to the terminal wash the slides were air-dried and cover-slipped with aqueous mounting medium (Faramount, Dako).
Results
Clinical features and management
The 30 cats included 18 Domestic Short Hair, 3 Domestic Long Hair, 1 Siamese and 1 Siamese-cross, 1 Tortoise Shell, and 1 mixed breed cat. The breed of 5 cats was not known. The age was known for 27/30 cats and ranged from 2 to 17 years with a mean age of 8.79 years; 14 cats were older than 10 years of age, 5 cats were between 7 and 10 years old, and 8 cats were younger than 5 years of age. Sixteen (61%) cats were female or female spayed, and 10 (39%) cats were male or male castrated. In 4 cats the sex was not indicated.
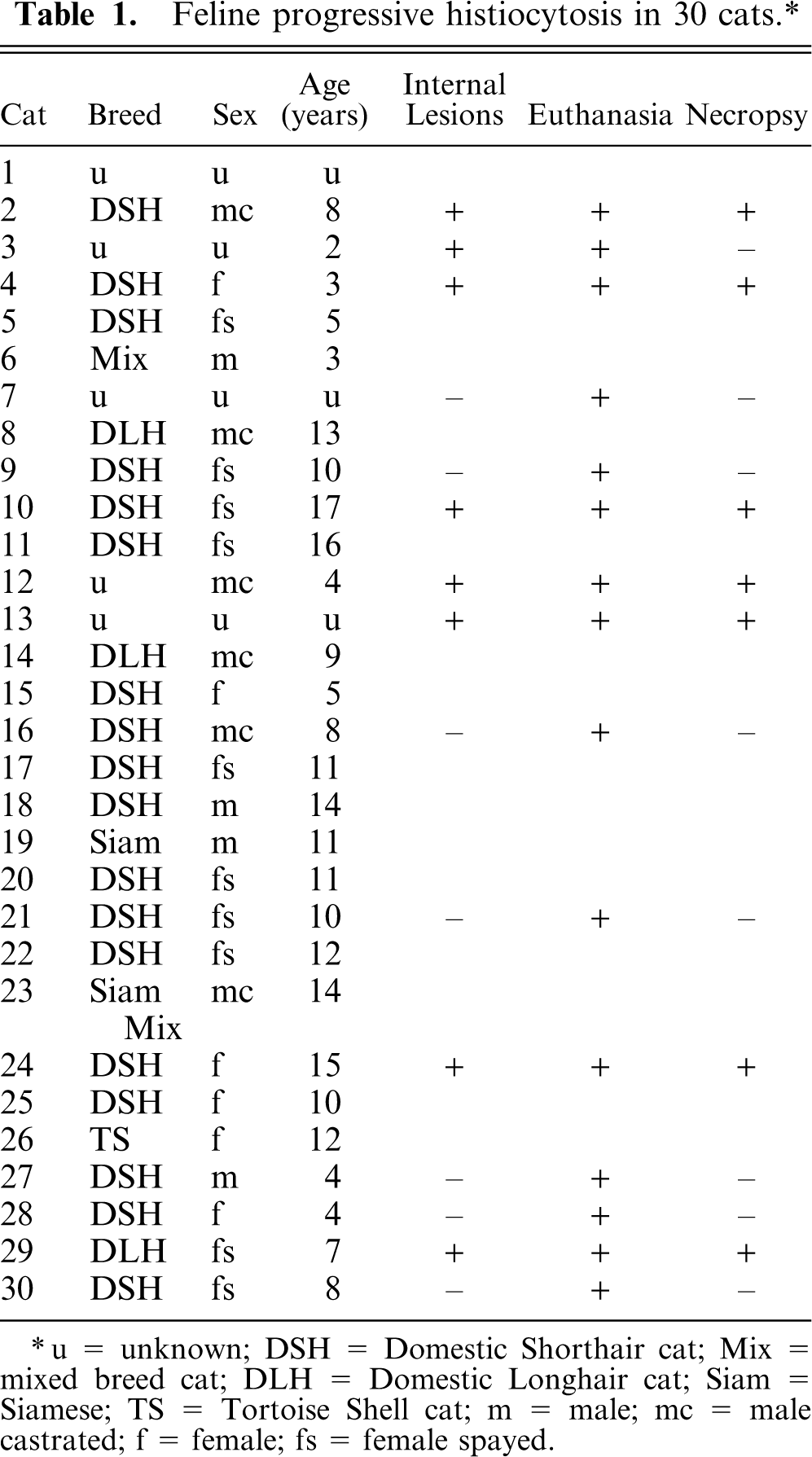
All 30 cats presented with skin lesions. Occasionally solitary, but mostly multiple, intradermal firm papules and nodules were observed. They measured up to 1.5 cm in diameter. The nodules did wax and wane in size, but complete spontaneous regression did not occur. The nodules progressed in size over time, and some coalesced to larger plaques. Early lesions had an intact skin surface, which was either normally haired or partially or completely alopecic. Pruritus was not observed. Initially the lesions were not painful. Over time some lesions became eroded, ulcerated, and painful. The locations of the skin lesions are listed in Table 3. Predilection sites for primary lesions were the feet, legs, and head (Fig. 1), but lesions were also described less frequently in other topographic locations (Table 3). In 8/30 cats the exact locations of the primary lesions were not indicated.
Skin, histiocytic nodules, cat No. 16. Multiple, coalescing, partially alopecic and eroded skin nodules involving the forehead and dorsal nasal planum and dorsum of the nose.
Location of skin lesions of feline progressive histiocytosis in 22 cats.

At the time of diagnosis, 18/30 cats were bright and alert and had no clinical evidence of internal disease. The lesions remained limited to the skin for a prolonged period of time. At the terminal stage, 4 of these cats presented with clinical evidence of internal involvement. The clinical signs included anorexia, weight loss, lymphadenopathy, respiratory signs, and anemia. An additional 4 cats presented with skin masses and concurrent clinical signs of internal involvement at the time of diagnosis. For the remaining 8 cats no clinical history of their general health was provided. The exact duration of the disease process after diagnosis was monitored in 10/30 cats and ranged from 1 month to 3 years with a mean of 13.4 months.
Information about clinical management was available in 15/30 cats. If number and location permitted, the lesions were removed surgically (8/15). Postsurgical local recurrence was seen in 4 of these cats; all 4 cats had infiltrative lesions on extremities. However, all 8 cats developed additional lesions distant from the surgery site. Corticosteroids were administered to 11 cats with multiple lesions, including some cats with previous surgery. Other therapies, including antibiotics (3/15), interferon-γ (2/15), retinoids (1/15), vincristine and vinblastine (2/15), cyclosporine A (1/15), nitrogen mustard (2/15), leflunomide (1/15), and L-asparaginase (1/15) were administered in an attempt to manage this disease. In all 15 cats the skin lesions progressed over time despite these various therapies.
Fifteen cats were euthanized (Table 1). In 7/15 cats the owners elected euthanasia because of marked progression of the skin lesions. There was no clinical evidence of internal disease, and necropsies were not performed. Necropsy confirmed the presence of internal disease in 7/15 cats. Lesions were found in lymph nodes (7/7), lungs (4/7), kidneys (3/7), pancreas (3/7), spleen (2/7), liver (2/7), heart (2/7), adrenals 1/7), and the mediastinum (1/7). Necropsy was not performed in 1 cat with clinical signs of systemic involvement.
Histopathologic features
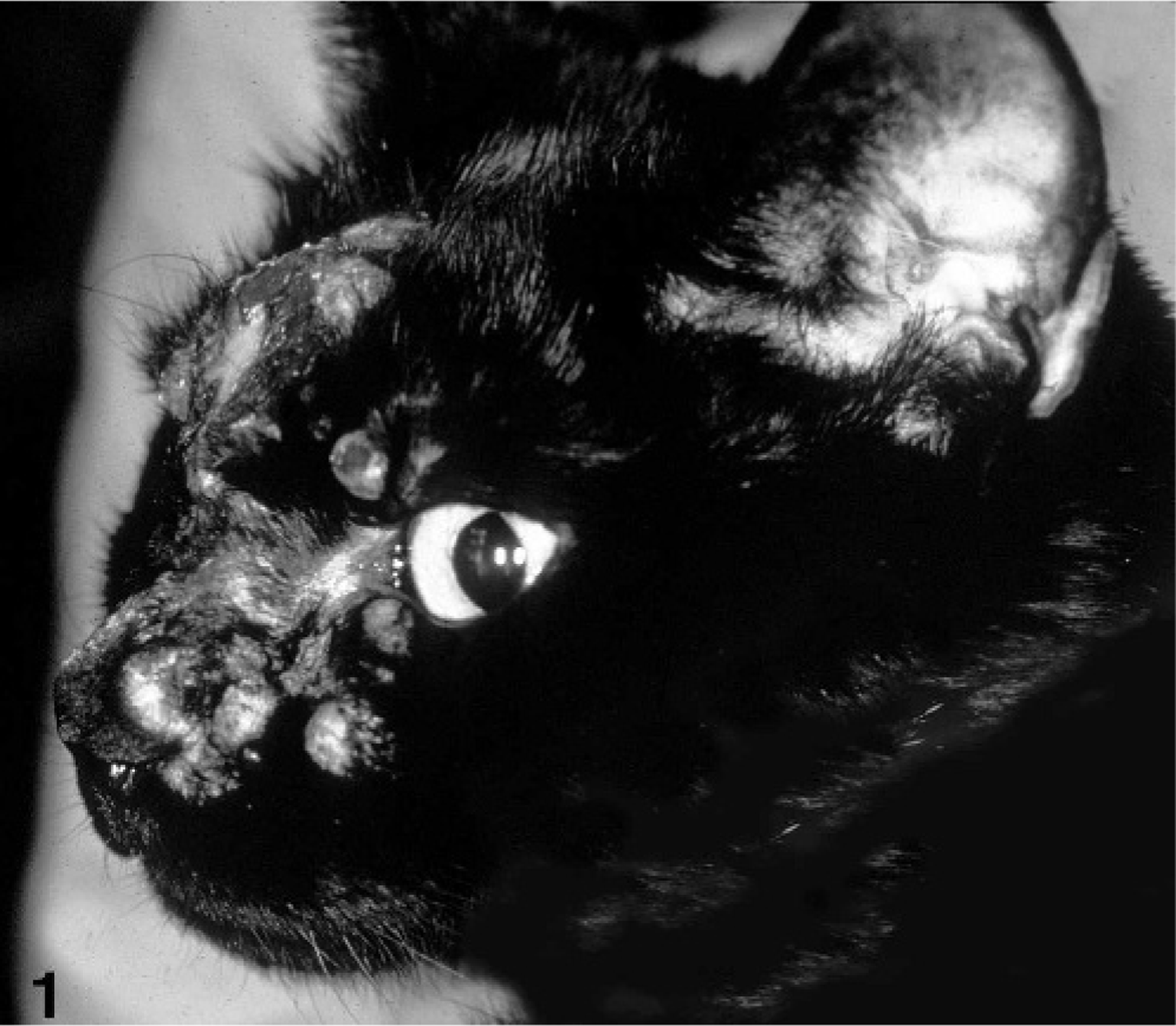
Characteristically, poorly circumscribed, nonencapsulated dermal nodules or masses with a top-heavy configuration were noted (Fig. 2). The cellular infiltrate extended from the superficial dermis into the deep dermis and occasionally into the subcutis. Biopsies from newly developing skin nodules revealed superficial and deep multinodular aggregates of large round cells. With progression, the nodules coalesced and formed a poorly demarcated mass.
Skin, histiocytic nodule, cat No. 10. The poorly circumscribed, nonencapsulated dermal and subcutaneous nodule has a slightly base-narrow and top-heavy configuration and is composed of a dense cell proliferation. HE. Bar = 1 mm.
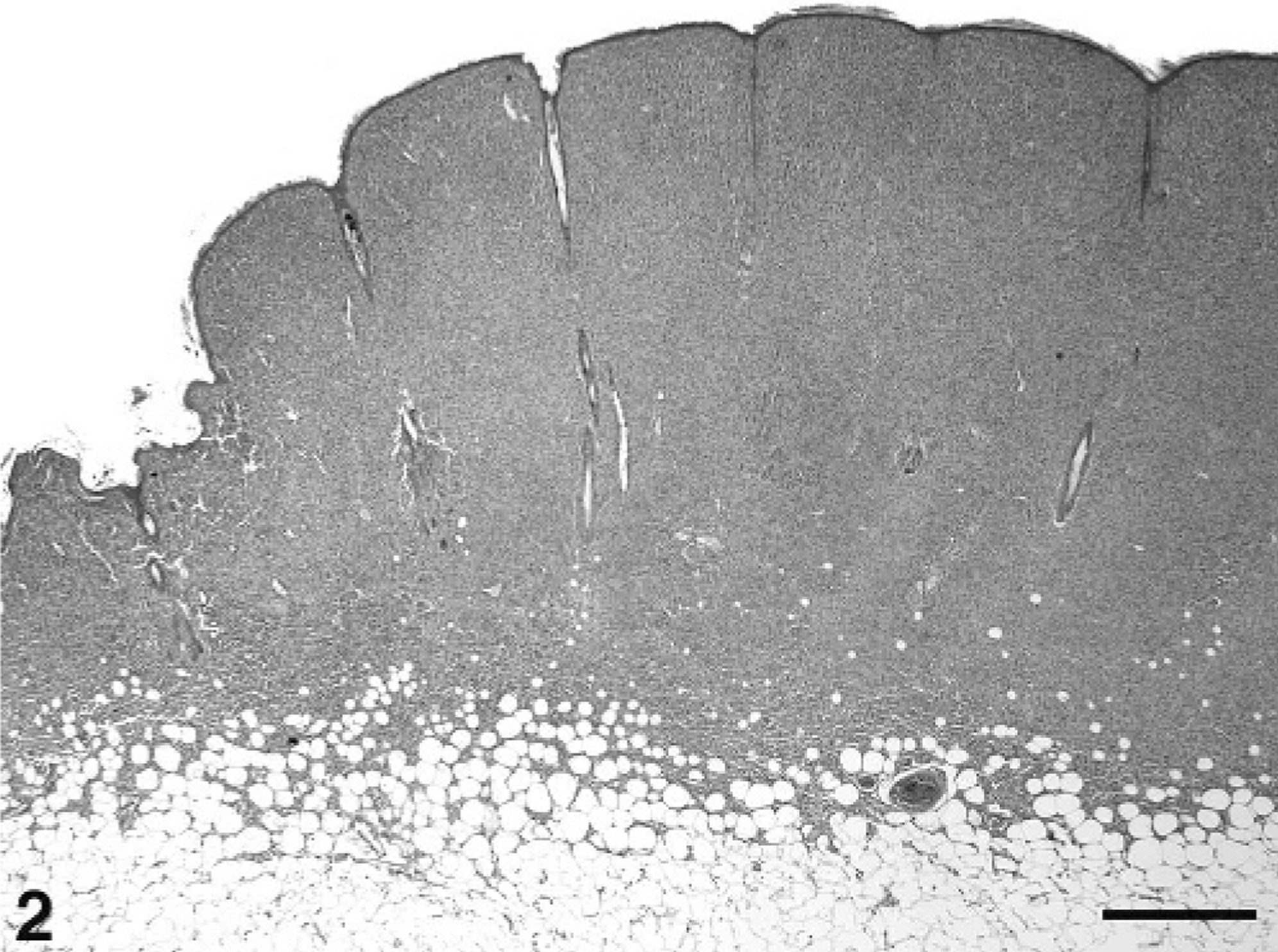
Typically, the nodules were composed of a dense population of large round-to-polygonal cells with discrete cell borders (Fig. 3). The cells had centrally located, large vesicular round-to-oval or indented nuclei with marginated, finely clumped chromatin and often inconspicuous or single and rarely multiple nucleoli. A moderate-to-abundant amount of lightly eosinophilic cytoplasm was observed (Fig. 3). Occasionally, the cytoplasm was vacuolated, resulting in a foamy, xanthomatous appearance of the lesion. No cytoplasmic granules were noted. These morphologic features are consistent with histiocytes. A mild-to-moderate degree of anisocytosis and anisokaryosis were noted, and 0–1 mitotic figures were observed per high-power field (40×) throughout most of the lesions. On rare occasions, 2–3 mitotic figures were seen per high-power field. A small-to-moderate number of multinucleated giant cells were present. Few to numerous small reactive lymphocytes and neutrophils were dispersed throughout the dermal nodules (Fig. 3).
Skin, histiocytic nodule, cat No. 10. The mass is composed of a dense cell proliferation of large round-to-polygonal cells with discrete cell borders. The large vesicular round-to-oval nuclei have marginated or fine granular chromatin and inconspicuous or single nucleoli. The cells have a moderate-to-large amount of pale eosinophilic cytoplasm. Note the mild-to-moderate degree of anisocytosis and anisokaryosis. A small number of reactive lymphocytes and neutrophils are dispersed throughout the lesion. HE. Bar = 50 μm.
The overlying epidermis was either intact or eroded and ulcerated. In most samples the cellular infiltrate abutted the basement membrane zone (Fig. 4). In 17/30 cats the lesions lacked epidermal involvement (Fig. 4). Occasionally, the dermal cellular infiltrate was separated from the overlying epidermis by a small zone of normal dermis (Grenz zone). In the remaining 13/30 cats intraepidermal infiltrates were noted (Fig. 5). Intraepithelial involvement was characterized by the presence of single cells or small aggregates of cells within the overlying epidermis. On rare occasions larger aggregates were noted (Fig. 5). The cytologic features of these intraepidermal aggregates resembled those of the dermal nodules. Involvement of follicular epithelium was not evident. In some skin lesions intralymphatic cell aggregates were noted.
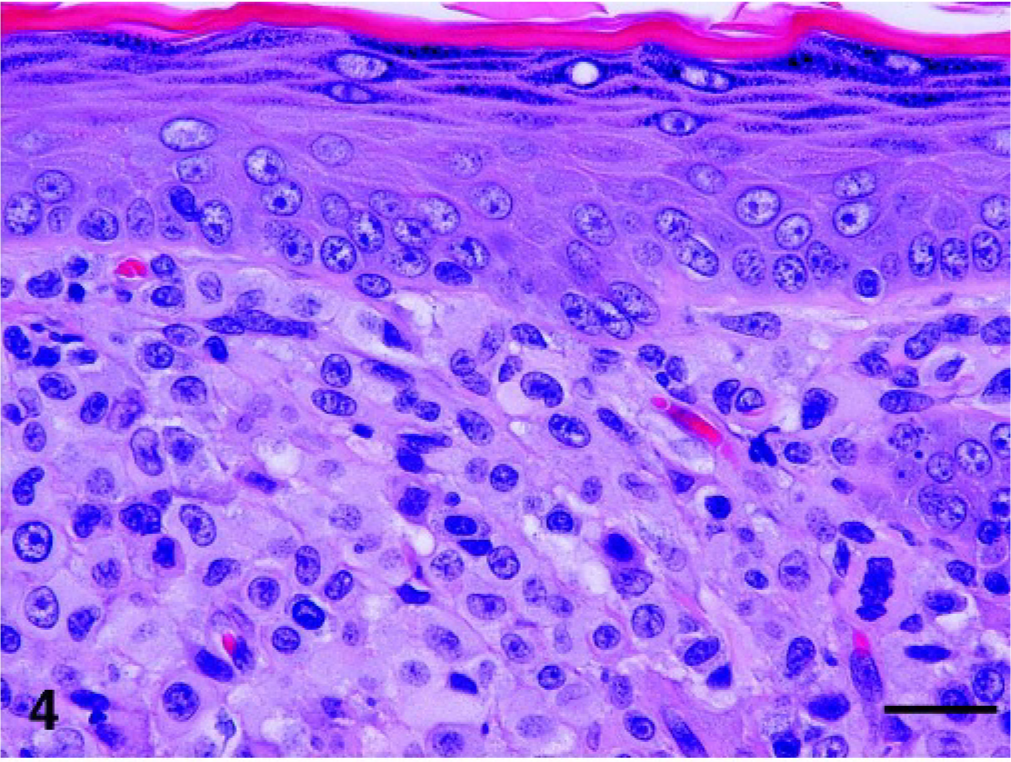
Skin, histiocytic nodule, cat No. 9. Nonepitheliotropic form: histiocytes abut the basement membrane zone, but there is no evidence of epidermal invasion. HE. Bar = 50 μm.
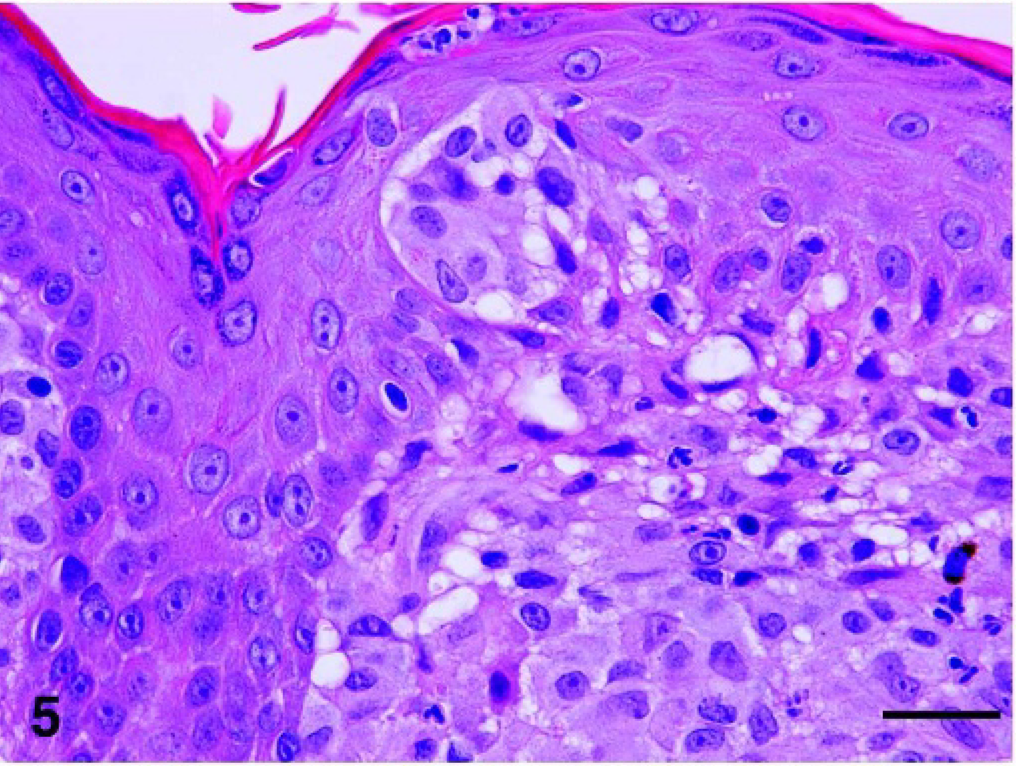
Skin, histiocytic nodule, cat No. 10. Epitheliotropic form: histiocytes have penetrated the basement membrane zone and form an intraepidermal aggregate. HE. Bar = 50 μm.←
In samples collected during the later phase of the disease and in skin lesions collected at necropsy, the cellular infiltrate often extended deep into the subcutis. Increased cellular pleomorphism was noted with marked anosicytosis and anisokaryosis (Fig. 6). Multinucleated giant cells were more numerous. A higher mitotic index (>4 mitotic figures per high-power field) and rare bizarre mitotic figures were seen. Intralymphatic cell aggregates were often noted.
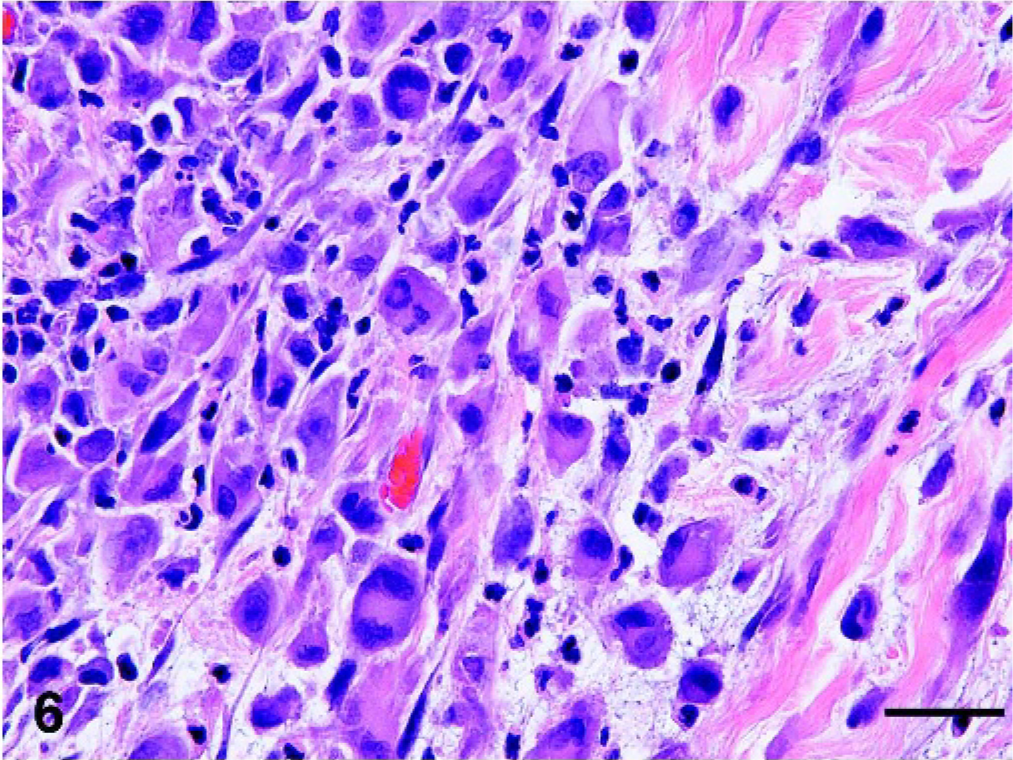
Skin, histiocytic sarcoma, cat No. 24, sample collected upon necropsy. The infiltrative mass is composed of a pleomorphic population of individualized large round and polygonal cells embedded in a loose myxomatous stroma. There is marked anisocytosis and anisokaryosis, and binucleated cells are noted. The nuclei have granular or dense chromatin and are often in paracentral location. A small number of reactive lymphocytes and neutrophils are dispersed throughout the lesion. HE. Bar = 50 μm.
Internal lesions (lymph nodes, lungs, kidneys, pancreas, spleen, liver, heart, adrenals, and mediastinum) were evaluated in 7 cats. Invasive and expansile masses effaced and replaced normal parenchymal tissues (Fig. 7). The masses comprised dense proliferation of large, moderately to markedly pleomorphic, round cells. Cytologic features, degree of anisocytosis and anisokaryosis, and the mitotic rate were most comparable with late-stage lesions in the skin.
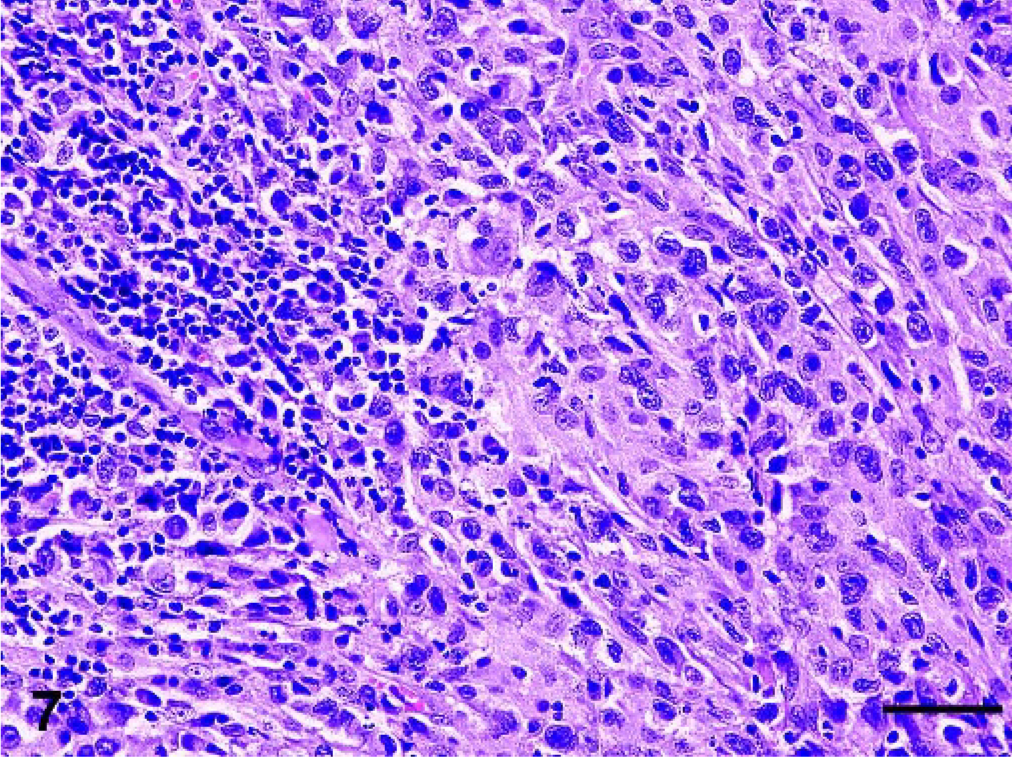
Lymph node, metastasis, cat No. 29. The majority of the lymph node tissue is replaced by a dense infiltrate of pleomorphic large histiocytes. Small remnants of cortical lymphoid tissue are still present in upper left corner. HE. Bar = 100 μm.
Immunohistochemistry and double-label immunofluorescence
Fresh, snap-frozen tissues from 16 cats were evaluated with the entire antibody panel (Table 2). A limited antibody panel (Table 2) was applied on formalin-fixed, paraffin-embedded tissues of 9 cats.
The lesional, large, round cell population consistently expressed CD18, the leukointegrin β subunit, and variably expressed the α subunit, CD11b. They lacked expression of CD3∊ and CD79a. This immunophenotype supports the presence of histiocytes. The strong and consistent coexpression of the antigen-presenting molecules CD1a, CD1c (Fig. 8), and major histocompatibility complex (MHC) II supports DC origin of these histiocytes. Furthermore, it rules out the differential diagnosis of amelanotic melanoma, because melanocytes do not express leukocyte markers, and poorly granulated mast cell tumors, which lack expression of CD1 and variably express CD18. The admixed reactive lymphoid infiltrate was mostly composed of CD3∊+, CD8+, and CD5+ cytotoxic T cells.
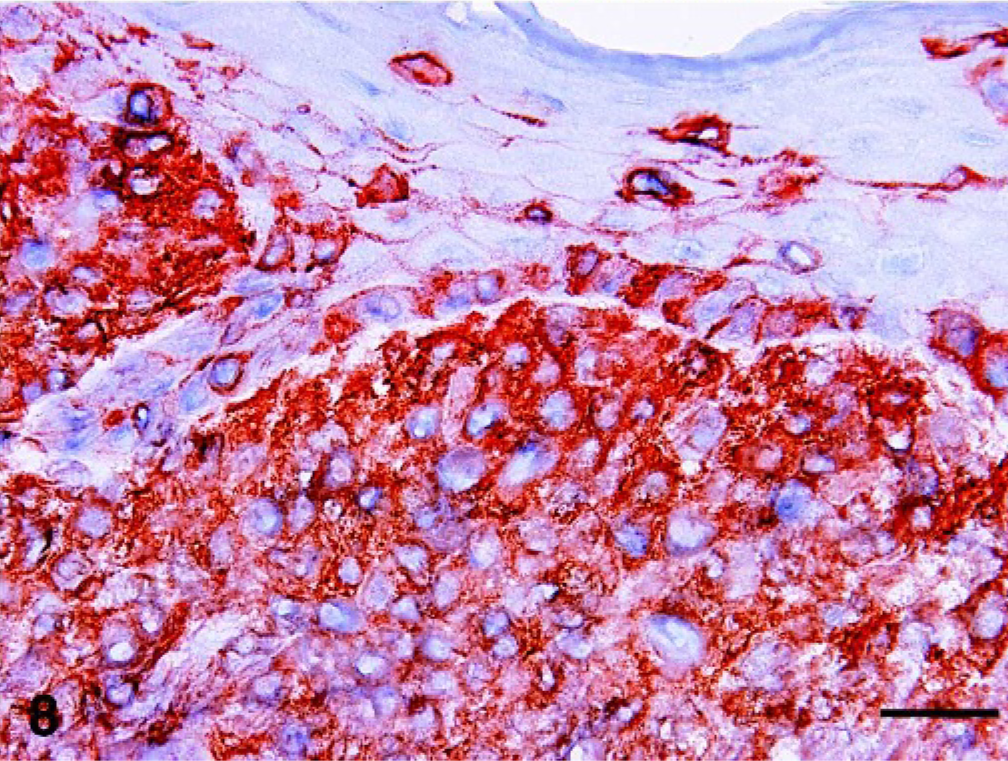
Skin, histiocytic nodule, cat No. 10. The lesional dermal histiocytic infiltrate expresses CD1c. The stained intraepidermal cells may represent resident Langerhans cells or individual lesional histiocytes. Immunohistochemistry; chromogen: amino-9-ethyl-carbazole, hematoxylin counterstain. Bar = 50 μm.
The majority of cases, including samples from epitheliotropic and nonepitheliotropic lesions, lacked expression of E-cadherin, a molecule usually expressed by Langerhans cells. 6, 15, 36 Expression of E-cadherin was detected in 3/25 cats only. Two cats had epitheliotropic lesions; the third had nonepitheliotropic lesions. E-cadherin expression was seen in a subpopulation of the lesional histiocytes and was usually weak.
In 7/16 cats diffuse CD5 expression was observed throughout the skin lesions. CD5 is a molecule usually expressed by lymphocytes. 19 Double-label immunofluorescence in 4 cats confirmed the coexpression of CD1 and CD5 by a subpopulation of the lesional histiocytes, as well as coexpression of CD3∊ and CD5 by reactive T cells in 3/4 cats tested. In the remaining cat the CD5+ cells coexpressed CD3∊ only.
Discussion
This investigation documents FPH in 30 cats, a disease process characterized by a progressive histiocytosis of the skin with potential for involvement of internal organs. The cutaneous masses are composed of histiocytes with a dendritic cell immunophenotype.
DC are a complex cell population, which include several different subpopulations. 2, 8, 16, 33 Their ontogeny, characteristics, and immunophenotype vary markedly between species. 2, 16, 30, 33 Regardless of their differences, DC are primarily involved in antigen presentation. Three major populations of DC have been documented in humans. A common CD34+ precursor cell in the bone marrow gives origin to the myeloid DC, which include Langerhans cells and interstitial DC. 8, 38 Langerhans cells populate the epidermis and epithelia of mucous membranes. They express CD1, CD11c, MHC II, langerin, and E-cadherin. 2, 6, 8, 15, 33 In some species, including the cat, the Langerhans cells also possess Birbeck's granules, a cytoplasmic structure induced by transforming growth factor-β. 8, 15, 30 The interstitial DC have been documented in many organs and, in the skin, include dermal DC. Dermal DC express CD1, CD11c, MHC II, and CD90, but lack expression of E-cadherin; they reside mainly adjacent to the dermal microvasculature. 2, 8, 21, 23, 32, 38 The evaluation of CD11c and CD90 expression in feline DC is not possible because of the lack of feline-specific or cross-reactive reagents. The third DC population in humans and rodents are the plasmacytoid DC. 29, 33 Their expression of some lymphoid markers is indicative of a lymphoid origin. 16 Plasmacytoid DC have not been identified in dogs and cats.
Various histiocytic proliferations of either DC or macrophage origin have been documented in humans and dogs. 1, 3, 5, 24, 27, 28, 31 Canine cutaneous histiocytoma, a mostly benign neoplasm of Langerhans cells, is predominantly seen in young dogs. 28 It tends to regress spontaneously. 9, 28, 35 Canine reactive histiocytosis occurs as a cutaneous or systemic form; 1, 24 both forms are characterized by a pleocellular, angiocentric process with numerous dermal DC. Reactive histiocytosis, a proliferation of activated dermal DC, is likely induced by dysregulation of the immune system. 1 Canine solitary and disseminated histiocytic sarcomas of DC origin—the latter has been referred to as malignant histiocytosis—develop as subcutaneous, periarticular, or internal tumor masses. 3, 27 Histiocytic sarcomas originate from interstitial DC based on tumor location and immunophenotype. Some canine histiocytic sarcomas arise from the splenic red pulp and the bone marrow; their immunophenotype and marked hemophagocytic activity indicates a macrophage origin. 26
Histiocytic proliferative diseases are uncommon in cats and their documentation is limited to a few case reports. Most cases were not associated with cutaneous lesions, but presented with internal disease and prominent weight loss. 10, 14, 18, 37 The cats had a low packed cell volume (19–20%), normochromic or hypochromic, regenerative or nonregenerative anemia, hypoproteinemia, hyperbilirubinemia, thrombocytopenia, and prolonged thromboplastin time. 10, 14, 18, 37 Hepatosplenomegaly with ascites and mediastinal and pulmonary masses with hydrothorax were observed. Histologic evaluations of the tumor masses revealed a proliferation of pleomorphic histiocytes, which usually displayed marked hemophagocytosis. The diagnosis of malignant histiocytosis was based on the morphology of the tumor cells, the hemophagocytic activity, and the variably positive immunohistochemical stains for lysozyme and Mac387 14, 18, 37 or positive reactions with α-1 antitrypsin, acid phosphatase, or nonspecific esterase. 10, 18 These findings are most consistent with disseminated hemophagocytic histiocytic sarcomas of macrophage origin rather than histiocytic sarcomas of DC origin. One case report describes a progressive multinodular cutaneous round cell proliferation similar to the cases described in this study. 12 Based on cytologic features and a MHC II+, CD79a–, and CD3- immunophenotype, the diagnosis of multiple histiocytomas was made. 12 More extensive immunophenotyping of the tumors was not performed on this cat. Hence, it was not possible to confirm a macrophage or DC origin.
The presence of multiple cutaneous nodules, the initially mostly benign cytologic features of the lesional histiocytes, and the slowly progressive clinical course of the disease separates FPH from primary histiocytic sarcoma. Clinical follow-up in 10 cats revealed that FPH may remain limited to the skin for an extended period of time, in some cats as long as 3 years. The fairly monomorphic tumor cells display mild anisocytosis and anisokaryosis, a low mitotic index, and occasional multinucleated cells. It is only at a late stage of the disease that pleomorphism, anisocytosis, increased mitotic index, and more numerous multinucleated giant cells were seen. Then histologic differentiation from a primary histiocytic sarcoma was no longer possible. Histiocytic sarcomas are characterized by a markedly aggressive behavior with fast progression and metastasis to draining lymph nodes and subsequent development of distant metastasis. Histiocytic sarcomas have been well described in dogs. 3 The authors have encountered 5 cats with solitary histiocytic sarcoma, which were essentially similar to the canine counterpart (V. K. Affolter and P. F. Moore, unpublished data). Therefore, differentiation between late-stage FPH and primary histiocytic sarcoma should be based on the clinical history with an emphasis on the course.
The DC sublineage involved in FPH is unknown. Epitheliotropism of the tumor cells was observed in 13 cats, suggesting that, similar to canine histiocytomas, FPH may originate from Langerhans cells or their precursors. 28 However, only 2/13 cats with epitheliotropic lesions expressed E-cadherin, a marker of Langerhans cell differentiation. 6, 34 For the remaining 11/13 cats Langerhans cell origin was unlikely, as they consistently lacked expression of E-cadherin. After activation, Langerhans cells downregulate their expression of E-cadherin as they migrate to the dermis. 6, 34 Hence, Langerhans cell origin cannot entirely be ruled out, because lesional histiocytes may have downregulated their expression of E-cadherin. This issue could be resolved by electron microscopic evaluation of future lesions to detect the presence of Birbeck's granules, a cytoplasmic organelle characteristic of feline Langerhans cells. 30 Wet tissue suitable for ultrastructural evaluation is not available from cats in the current series. Alternatively, FPH may arise from dermal DC. Unfortunately, the lesions cannot be evaluated for coexpression of CD1 and CD90, a molecule expressed by canine dermal DC and not by Langerhans cells, because feline-specific anti-CD90 antibodies are not available. 1 Furthermore, the coexpression of CD5 by the CD1+ lesional DC in 3 cats was unexpected. CD5 is a molecule that is usually only expressed by lymphocytes. 19 Evaluation of frozen tissue sections in 16 cats revealed that the reactive CD3+ T cell population was invariably positive for CD5 as expected. However, in 3 cats the coexpression of CD5 by a subpopulation of the lesional CD1+ DC was confirmed. CD5 expression has been observed on plasmacytoid dendritic cells in rats, which indicates that CD5 expression is not limited to lymphoid cells in some species. 16 Because of a lack of appropriate cell differentiation markers, we are not able to identify plasmacytoid DC in the cat. Plasmacytoid DC in humans and rats lack expression of CD1 when they first enter lymphoid tissue. However, they can upregulate this molecule upon activation. 29 Alternatively, expression of CD5 by lesional histiocytes in some cats with FPH may be ectopic and indicative of disordered cell regulation.
The etiology of FPH is not known. Chronic antigen stimulation was considered as a possible triggering factor. However, a reactive process, similar to reactive histiocytosis in dogs, 1 is unlikely as all cats failed to respond to immunomodulatory drugs. Moreover, the presence of a monomorphic cell infiltrate is more indicative of neoplasia than a pleocellular, inflammatory process. FPH likely represents a primary low-grade neoplasia of cutaneous DC with initial indolent behavior. Over time, accumulated mutations in a subpopulation of the lesional histiocytes may lead to the evolution of histiocytic sarcoma.
Neoplasia of histiocytic origin tends to respond poorly to chemotherapy. 3– 5, 7, 27 Follow-up data regarding attempted therapy for FPH was available in 8 cats. The lack of long-term response to high doses of corticosteroids, vincristine, vinblastine, nitrogen mustard, and L-asparaginase in these cats is consistent with observation in humans and dogs with histiocytic neoplasia. 3– 5, 7, 27 Surgical excision was performed in 8 cats with solitary masses or a small number of skin lesions. Local recurrence occurred in 4 cats. All surgically treated cats developed additional lesions in other locations.
In summary, we have described a histiocytic proliferative disorder of unknown etiology, which originated in the skin of 30 cats. We have chosen to call it “feline progressive histiocytosis” to highlight the clinical behavior, which although initially indolent, eventually progresses to involve extracutaneous sites. In the terminal phase, FPH is histologically indistinguishable from histiocytic sarcoma.
Footnotes
Acknowledgements
We thank all clinicians from private practice and veterinary schools as well as pathologists throughout the United States and Canada who provided some of the case material. We appreciate Dianne Naydan's assistance in handling the immunohistochemical stains of these cases.
