Abstract
Histiocytic disorders of dogs include histiocytoma, localized histiocytic sarcoma (HS), disseminated HS (malignant histocytosis), and the reactive histiocytoses: cutaneous and systemic. A common element to these diseases is proliferation of dendritic cells (DC) of either Langerhans cell (epithelial DC) or interstitial DC lineage. In this report, 17 dogs with hemophagocytic HS are described. Breeds affected included Bernese Mountain Dog (6), Golden Retriever (4), Rottweiler (3), Labrador Retriever (2), a mixed-breed dog, and a Schnauzer, which were from 2.5 to 13 years old. The dogs presented with Coombs negative responsive anemia in 16/17 dogs (94%), thrombocytopenia in 15/17 dogs (88%), hypoalbuminemia in 16/17 dogs (94%), and hypocholesterolemia in 11/16 dogs (69%). All dogs died or were euthanized. The clinical course ranged from 2 to 32 weeks (mean 7.1 weeks). Diffuse splenomegaly with ill-defined masses was consistently present. Microscopic lesions were prevalent in spleen, liver, lung, and bone marrow. Metastasis occurred by insidious intravascular invasion with minimal mass formation. Histiocytes were markedly erythrophagocytic and accompanied by foci of extramedullary hemopoiesis. Cytologically, the histiocytes varied from well differentiated to atypical, with atypia more prevalent in spleen than bone marrow. These tumors arose from splenic red pulp and bone marrow macrophages, which expressed major histocompatibility complex class II and the β2 integrin, CD11d. They had low and/or inconsistent expression of CD1 and CD11c, which are dominantly expressed by canine nonhemophagocytic HS of DC origin. Canine histiocytic proliferative diseases now encompass proliferation of all members of the myeloid histiocytic lineage: Langerhans cells, interstitial DC, and macrophages.
Histiocytic proliferative disorders are prevalent in dogs, especially so within a narrow range of breeds. Reactive and neoplastic histiocytic disorders have been described. These include histiocytoma, localized histiocytic sarcoma (HS), disseminated HS (equivalent to malignant histocytosis), and the reactive histiocytoses: cutaneous histiocytosis and systemic histiocytosis. 1, 2, 28, 33– 35 A common element to these diseases is proliferation of dendritic cells (DC) of either Langerhans cell (epithelial DC) or interstitial DC lineage. 1, 2, 27, 35
Canine cutaneous histiocytoma is a common localized tumor of epidermal Langerhans cells. 27, 35 Disseminated histiocytomas and Langerhans cell histiocytosis occur uncommonly in dogs; these diseases closely resemble some forms of human Langerhans cell histiocytosis. 35, 37 Reactive histiocytoses are complex disorders likely arising in the context of disordered immune regulation and manifest with infiltration or proliferation of lymphocytes and perivascular interstitial DC of the dermis and subcutis. 1 Histiocytic sarcomas (localized and disseminated) likely arise from interstitial DC, which are prevalent in almost all tissues and organs, with the most notable exception being the brain. 2 Within lymphoid tissues, interstitial DC are concentrated in or adjacent to organized lymphoid domains. Lymphoid sinuses and splenic red pulp, by contrast, are enriched for macrophages. It is thought that in most instances, HSs arise from interstitial DC, and hence emanate from lymphoid domains when they arise from lymphoid organs, such as spleen and lymph node. 2
Macrophages and myeloid DC arise in bone marrow and differentiate along divergent lines from a common multilineage hemopoietic stem cell precursor. Blood monocytes retain some developmental plasticity and can be induced to differentiate into either interstitial DC or macrophages, depending on the applied cytokine and growth factor milieu. 41, 42 Dendritic cells are specialized antigen-presenting cells that interact with and lead to T cell activation following contact with antigen. DC constitutively or inducibly express many of the cell surface molecules important in antigen presentation and costimulation, such as major histocompatibility complex (MHC) class I and II, CD1, CD80, and CD86. 3, 21 Macrophages are more specialized in phagocytic/degradative pursuits; with appropriate stimulation they can be induced to present antigen to T cells, although at a lower efficiency than DC. This major functional difference is reflected in the cell surface antigen expression pattern of macrophages. Constitutively, they express low levels of MHC class II and the costimulatory molecules CD80 and CD86. Also, CD1 is not constitutively expressed on macrophages. 30, 31 CD1 molecules, which are highly expressed on DC, are important antigen-presenting molecules for lipid and glycolipid moieties often derived from bacterial cell walls. 11
In this report, the clinical and pathologic features of HS, which manifested prominent hemophagocytosis and Coombs negative responsive anemia, are described. These tumors arose from splenic red pulp and bone marrow macrophages, which expressed the β2 integrin, CD11d, and had low and/or inconsistent expression of CD1 and CD11c, which are dominantly expressed by canine nonhemophagocytic HS of DC origin. 2
Materials and Methods
Case selection and tissue handling
Tissue samples were sourced from the Veterinary Medical Teaching Hospital, University of California, Davis, and from private veterinary practitioners. Seventeen dogs with a diagnosis of hemophagocytic HS were selected from a pool of submissions spanning 1996 to 2005 (n > 40). The cases were selected for inclusion based on completeness of clinical records and/or presence of fresh tissue samples to enable more comprehensive immunophenotyping. Tissue samples submitted included unfixed fresh tissue and formalin-fixed tissue acquired as surgical biopsy specimens or at necropsy. In some instances, fine needle aspirate (FNA) smears of spleen and bone marrow smears were submitted. Fresh tissue samples were bisected, and one half was fixed in 10% neutral buffered formalin and embedded in paraffin. The other half was embedded in optimal cutting temperature compound (Finetek, Sakura USA, Torrance, CA) and snap frozen in isopentane cooled to the point of freezing in liquid nitrogen. Normal canine control tissues for immunohistochemistry (spleen, liver, thymus, skin, and bone marrow) were handled identically. The clinical records of a cohort of dogs (n = 26) diagnosed with nonhemophagocytic HS during the same time period were analyzed for the comparison of key clinicopathologic findings.
Statistical analysis
Differences in key clinicopathologic data between hemophagocytic and nonhemophagocytic HS were analyzed using unpaired 2-tailed Student's t-test. A P value <.05 was considered significant.
Antibodies
A panel of monoclonal antibodies (MAb) specific for canine leukocyte antigens was used in this study (Table 1). These MAb have been used extensively for characterization of canine histiocytic disorders. 1, 2, 32, 35 All MAb were sourced from the Leukocyte Antigen Biology Laboratory, University of California, Davis, with the exception of anti-CD79a (clone HM57, Dako, Carpinteria, CA). Cryosections were evaluated with the entire panel of antibodies (Table 1). A limited number of antibodies recognized formalin-denatured epitopes following heat-based antigen retrieval (Table 1). Hence, expression of CD3∊, CD11d, CD18, CD45, CD45RA, and CD79a was also assessed in formalin-fixed, paraffin-embedded tissues.
Monoclonal antibodies specific for canine leukocyte antigens.

∗ Reactive in formalin-fixed paraffin embedded tissue sections after heat-based antigen retrieval.
† V. K. Affolter and P. F. Moore, manuscript in preparation.
Immunohistochemistry
Cryosections (6 μm) and cytology smears were fixed in acetone (3 minutes), and endogenous peroxidase was quenched by immersing slides in hydrogen peroxide (0.3% w/v), sodium azide (0.1% w/v) diluted in phosphate-buffered saline (PBS) for 10 minutes. Following a blocking step with 10% heat-inactivated horse serum in PBS (20 minutes), the dog-specific monoclonal antibodies were applied to the sections for 30 minutes. Appropriate antibody dilutions, prepared in 10% horse serum in PBS, were determined by previous titration of each antibody on frozen sections of normal canine spleen. Omission of the primary antibody or application of an isotype-matched nonspecific antibody was used as a negative control in each run. Secondary biotinylated horse anti-mouse IgG (Vector, Burlingame, CA) was applied to each section for 30 minutes followed by streptavidin–horseradish–peroxidase (Zymed, South San Francisco, CA) for 20 minutes according to the manufacturers' instructions. Between each step, the sections were washed thoroughly in PBS. Amino-9-ethyl-carbazole (AEC; Sigma Chemical Co., St. Louis, MO) was used as the chromogen. The tissue sections were counterstained with hematoxylin (Gill's formula 3; Fischer, Pittsburgh, WA), air dried, and coverslipped. Paraffin-embedded sections (6 μm) were deparaffinized in xylene and hydrated through graded ethanol solutions. An antigen retrieval procedure was conducted to unmask antigen epitopes. This consisted of steaming tissue sections immersed in 10-mM citrate buffer (pH 6.6) for 30 minutes. The tissue sections were allowed to cool for 20 minutes and subsequently stained as described above, with the omission of the acetone fixation step.
Results
Clinical features
The demographics of 17 dogs with hemophagocytic HS are listed in Table 2. Breeds affected included the Bernese Mountain Dog (6), Golden Retriever (4), Rottweiler (3), Labrador Retriever (2), a mixed breed dog, and a Schnauzer. Dogs ranged in age from 2.5 to 13 years. No sex predilection was noted, since 8 female and 9 male dogs were affected. Abnormalities in the clinical history included lethargy, inappetance, weight loss, and pale mucous membranes. Jaundice was only evident in dog No. 5 and dog No. 8. Abdominal palpation and radiographic and/or ultrasound evaluation were performed in 14 dogs and revealed splenomegaly (14 dogs) and hepatomegaly (7 dogs). All dogs died or were euthanized. The clinical course from initial onset of clinical signs to death or euthanasia ranged from 2 to 32 weeks (mean 7.1 ± 7.2 weeks; n = 17).
Demographics, organ involvement, cytologic score, and survival of dogs with hemophagocytic HS.∗
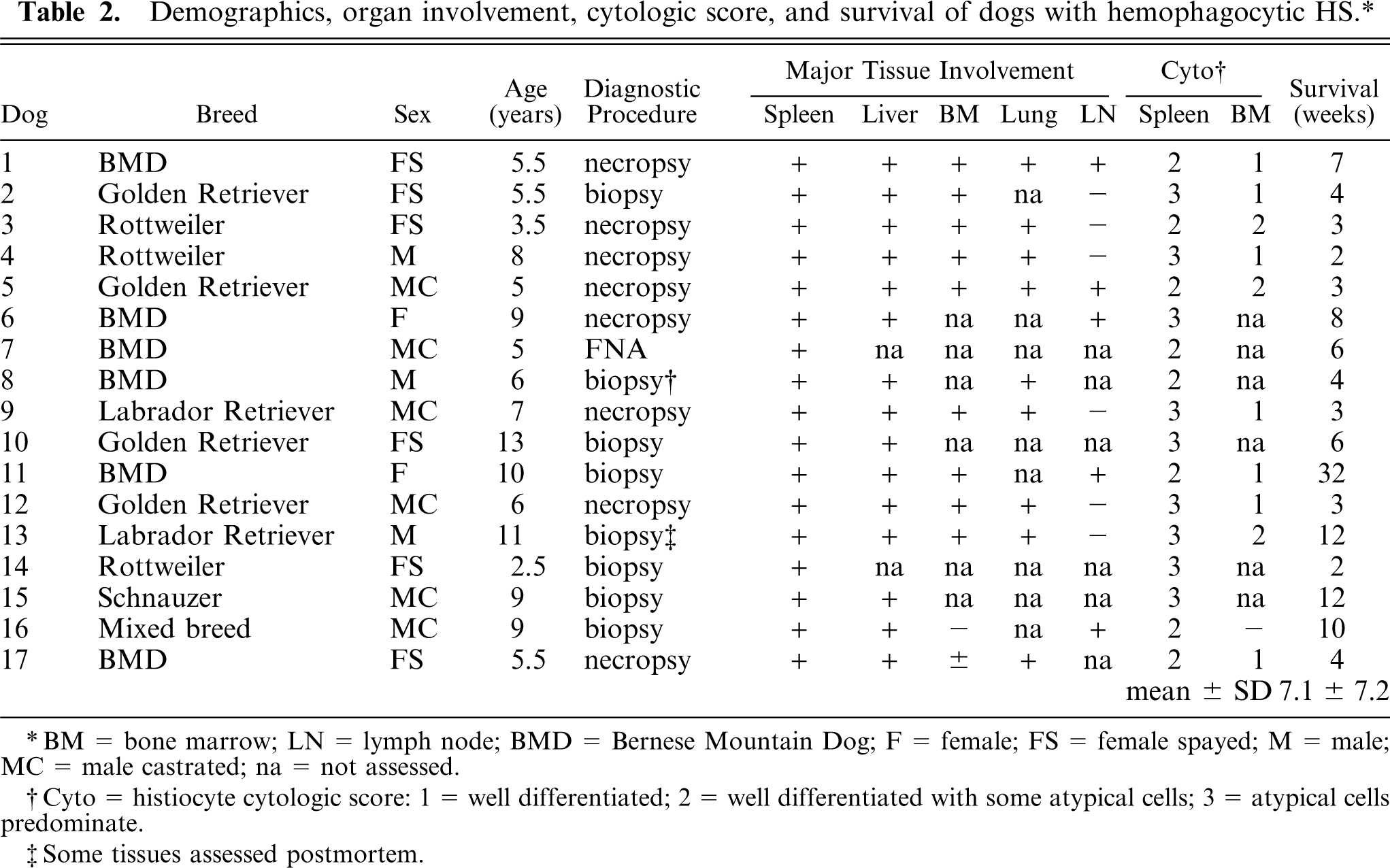
∗BM = bone marrow; LN = lymph node; BMD = Bernese Mountain Dog; F = female; FS = female spayed; M = male; MC = male castrated; na = not assessed.
†Cyto = histiocyte cytologic score: 1 = well differentiated; 2 = well differentiated with some atypical cells; 3 = atypical cells predominate.
‡Some tissues assessed postmortem.
Clinicopathologic features
Many dogs with hemophagocytic HS were initially evaluated for immune-mediated hemolytic anemia. A responsive anemia (mean hematocrit = 23.5%, mean reticulocyte count 232,911/μl) was initially present in 94% (16/17) of dogs and eventually developed in all dogs. Thrombocytopenia (mean platelet count 116,200/μl) was also common, occurring in 88% (15/17) of dogs (Table 3). Coagulation abnormalities were prevalent, with mild prolongation of partial thromboplastin time (PTT; mean 14.9 seconds; reference range 9–12 seconds) occurring in 70% (7/10) of dogs. Other common laboratory abnormalities included hypoproteinemia (mean 4.8 g/dl) in 82% (14/17) of dogs, hypoalbuminemia (mean 2.2 g/dl) in 94% (16/17) of dogs, and hypocholesterolemia (mean 123 mg/dl) in 69% (11/16) of dogs. Hyperbilirubinemia was present in 44% (7/16) of dogs and was usually mild (mean 0.6 mg/dl) (Table 3). Coombs tests were done in 8 dogs, and all were negative.
Clinicopathologic findings in blood in hemophagocytic HS∗.
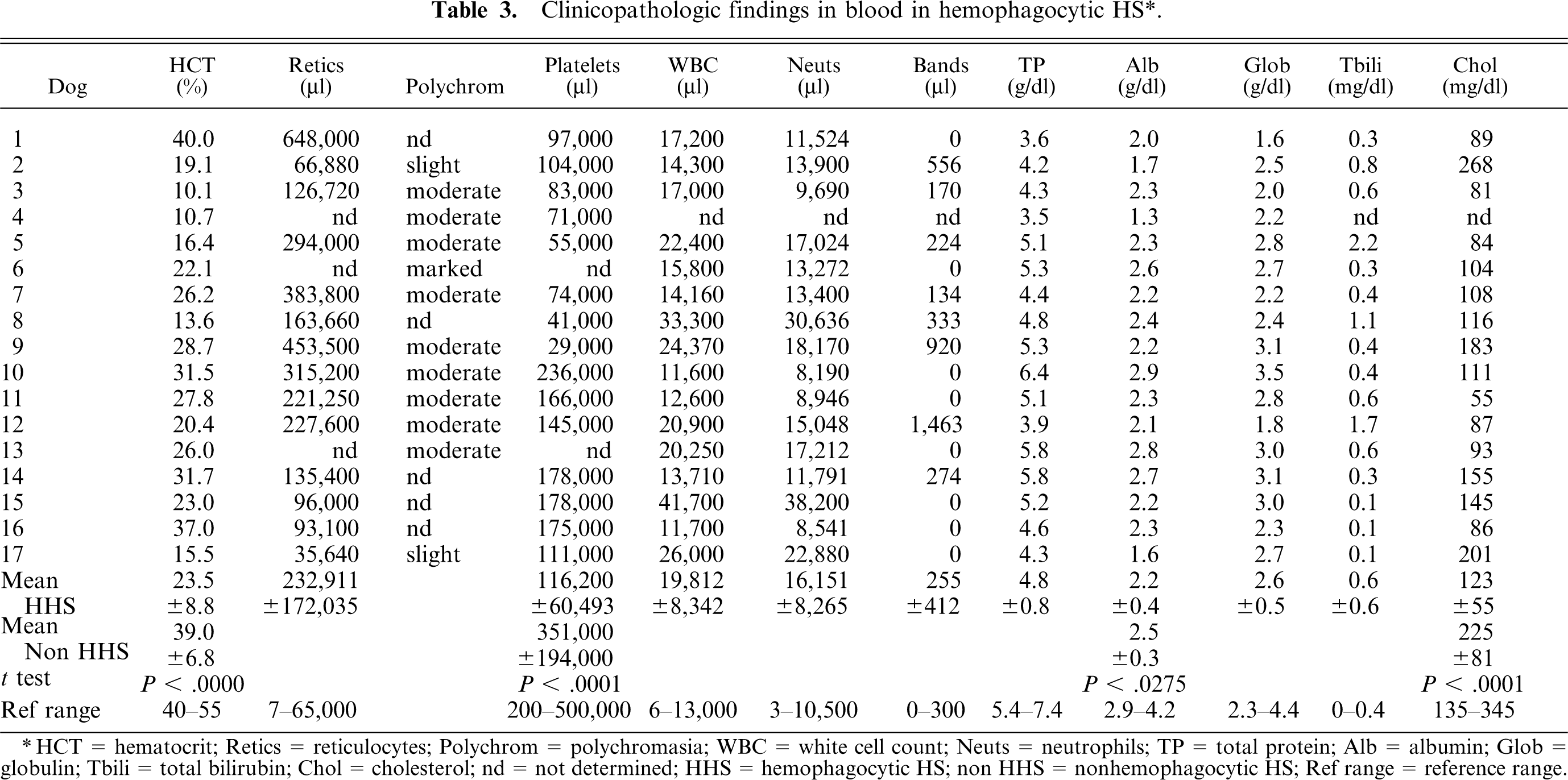
∗HCT = hematocrit; Retics = reticulocytes; Polychrom = polychromasia; WBC = white cell count; Neuts = neutrophils; TP = total protein; Alb = albumin; Glob = globulin; Tbili = total bilirubin; Chol = cholesterol; nd = not determined; HHS = hemophagocytic HS; non HHS = nonhemophagocytic HS; Ref range = reference range.
Clinicopathologic abnormalities were also encountered in the nonhemophagocytic HS cohort (Table 3). Mild anemia (usually nonresponsive) was observed in 54% (14/26) of dogs, and hypoalbuminemia was observed in 88% (23/26) of dogs, although the mean serum albumin concentration (2.5 mg/dl) was significantly higher than in hemophagocytic HS. Interestingly, hypocholesterolemia was uncommon in nonhemophagocytic HS, and occurred in only 8% (2/26) of dogs, and the mean serum cholesterol (225 mg/dl) was significantly higher than that of dogs with hemophagocytic HS.
Macroscopic lesions
Macroscopic lesions were most prevalent in the spleen and liver. The spleen was assessed grossly in 14 dogs and was consistently enlarged. Splenomegaly was diffuse with few to many, ill-defined, tan to red nodular masses (3–5 cm diameter) also discernible (Figs. 1, 2). Discolored green-tan to white splenic infarcts were often visible through the splenic capsule (Figs. 1, 2). Hepatomegaly (7 dogs) was characterized by diffuse hepatic enlargement, a textured capsular surface, a light tan to reddish discoloration, and a prominent reticular pattern. Distinct mass formation was not observed. Pulmonary lesions were grossly visible in only 2 dogs (dog Nos. 1 and 3); they consisted of randomly distributed, innumerable, white nodules up to 2-mm diameter. Extensive necropsies were performed in 6 dogs (dog No. 1, dog Nos. 3–5, dog No. 9, and dog No. 12). In 2 of these dogs, the kidneys contained numerous, white, 1–5-mm-diameter cortical nodules (dog No. 1) or 2 white, 10–15-mm-diameter cortical nodules (dog No. 9).
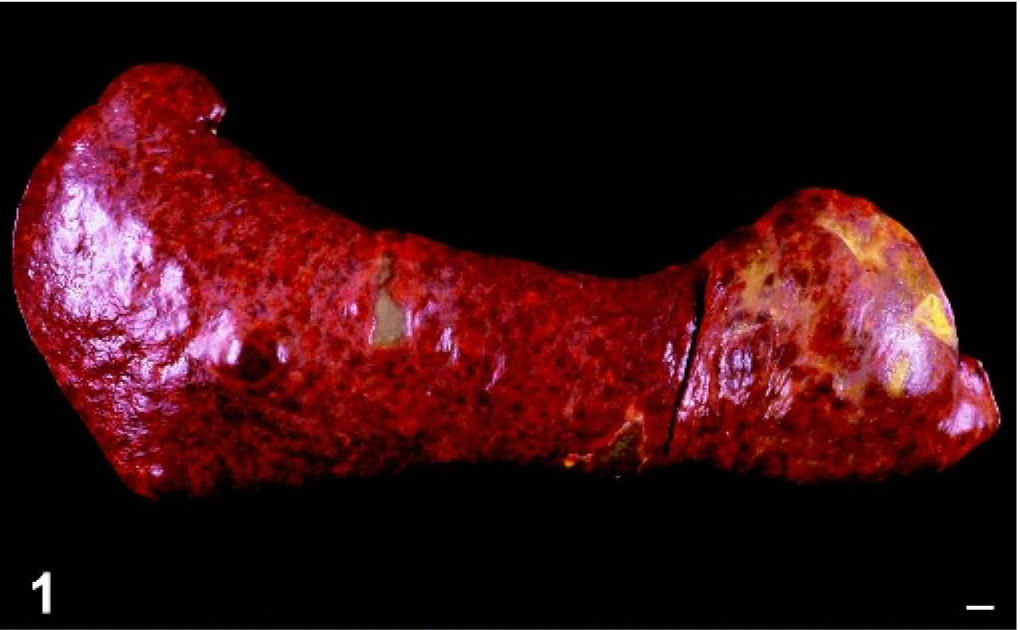
Spleen; dog No. 5. Diffuse splenomegaly with small discrete nodules is evident. Infarcts are visible through the capsule as irregular shaped grey-green regions. Bar = 1 cm.
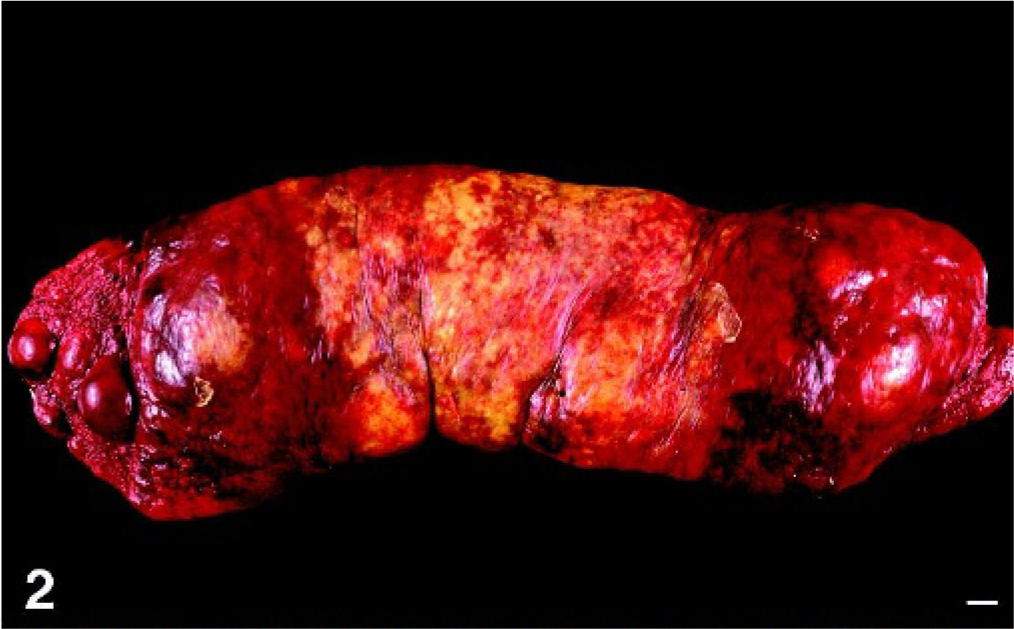
Spleen; dog No. 2. Diffuse splenomegaly with additional masses up to 5 cm. Infarcts are visible through the capsule as irregular yellow-tan coalescing foci in the mid-portion of the spleen. Bar = 1 cm.
Microscopic lesions
The most consistently involved organs in dogs with hemophagocytic HS were spleen, liver, bone marrow, and lung (Table 2). The spleen was infiltrated in all dogs. The splenic red pulp was diffusely expanded by histiocytes, which occupied the red pulp cords, invaded the red pulp sinuses, and frequently obliterated the adjacent white pulp (Fig. 3). Foci of fibrin deposition were present in the red pulp sinuses and cords (Fig. 3). Extensive areas of red pulp infarction were found in association with red pulp thrombosis. Thrombi were also found in splenic trabecular veins. Histiocytes were markedly erythrophagocytic in some and less so in other regions of the spleen (Fig. 4). Histiocytes frequently contained hemosiderin deposits reflecting previous erythrophagocytosis. Histiocytes also engulfed red cell precursors and granulocytes, although the latter was less frequent. Extensive foci of extramedullary hemopoiesis (EMH) were often intimately associated with histiocytic infiltrates (Fig. 4). Clusters of plasma cells and individual lymphocytes were identifiable within the histiocytic expansion. These lymphoid infiltrates were numerically insignificant in the majority of dogs.
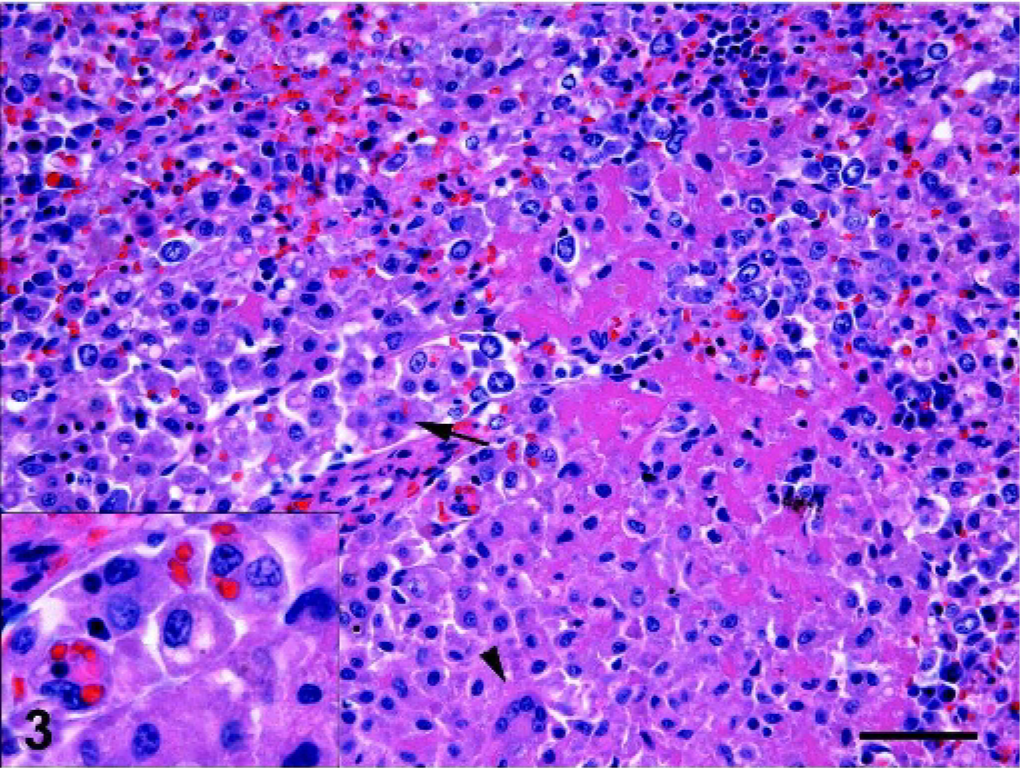
Spleen; dog No. 5. Red pulp cords and sinus (arrow) infiltration by cytologically well differentiated histiocytes. Multinucleated giant cell (arrowhead). Fibrin deposition in red pulp cords. Inset: erythrophagocytic histiocytes, some with moderate cytologic atypia characterized by hyperchromatic nuclei and large irregular nucleoli. HE; bar = 50 microns.
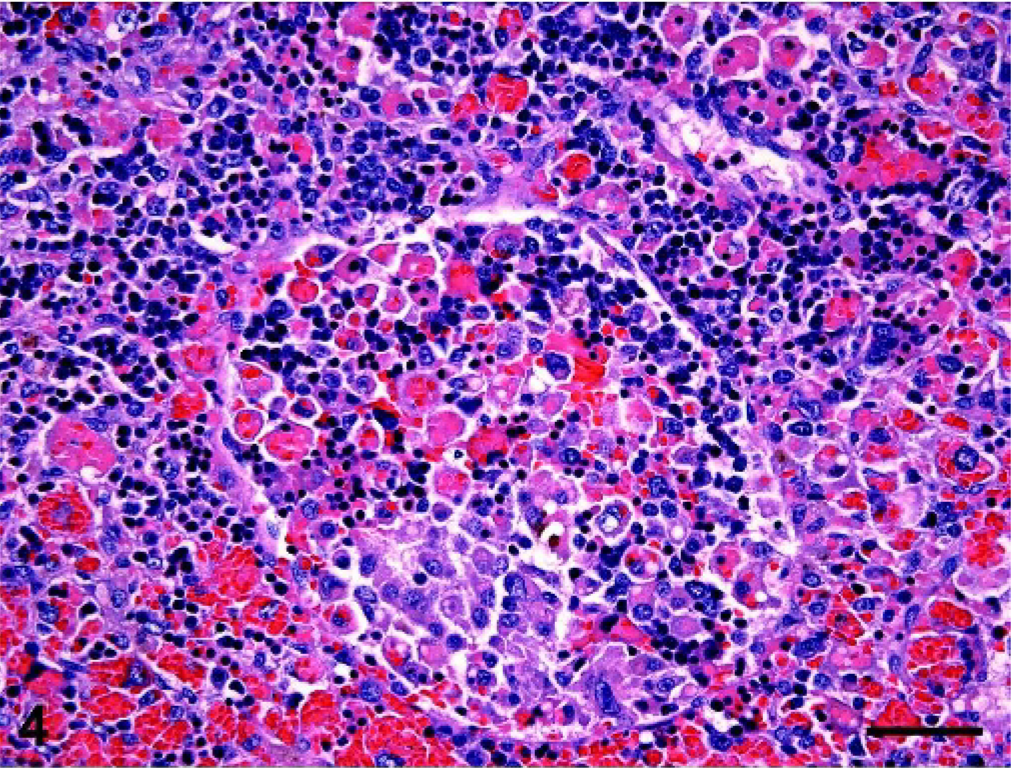
Spleen; dog No. 14. Histiocytic infiltration with marked EMH in splenic red pulp and sinus (center). Histiocytes exhibiting moderate cytologic atypia have engulfed red cells, red cell precursors, and occasional granulocytes. HE; bar = 50 microns.
There was marked variation in the cytologic characteristics of splenic histiocytes (Table 2). In some instances they resembled normal red pulp macrophages, although occasional anisocytosis, anisokaryosis, and hyperchromatic nuclei were discernible. Sporadic multinucleated giant cells intermingled with these mononuclear, cytologically well differentiated histiocytes (Fig. 3). The cytoplasm of histiocytes was eosinophilic, abundant, and often coarsely vacuolated. The mitotic rate was 0–1 per 40× field. In other instances, histiocytes were more frequently cytologically atypical, evidenced by hyperchromatic nuclei, anisocytosis, anisokaryosis, and karyomegaly (Figs. 4, 5). Multinucleated cells with similar atypical cytologic properties were more frequently encountered in the latter lesions. Nuclei were most frequently ovoid to indented; only occasionally were bizarre forms observed (Fig. 5). The mitotic rate was 1–3 per 40× field.
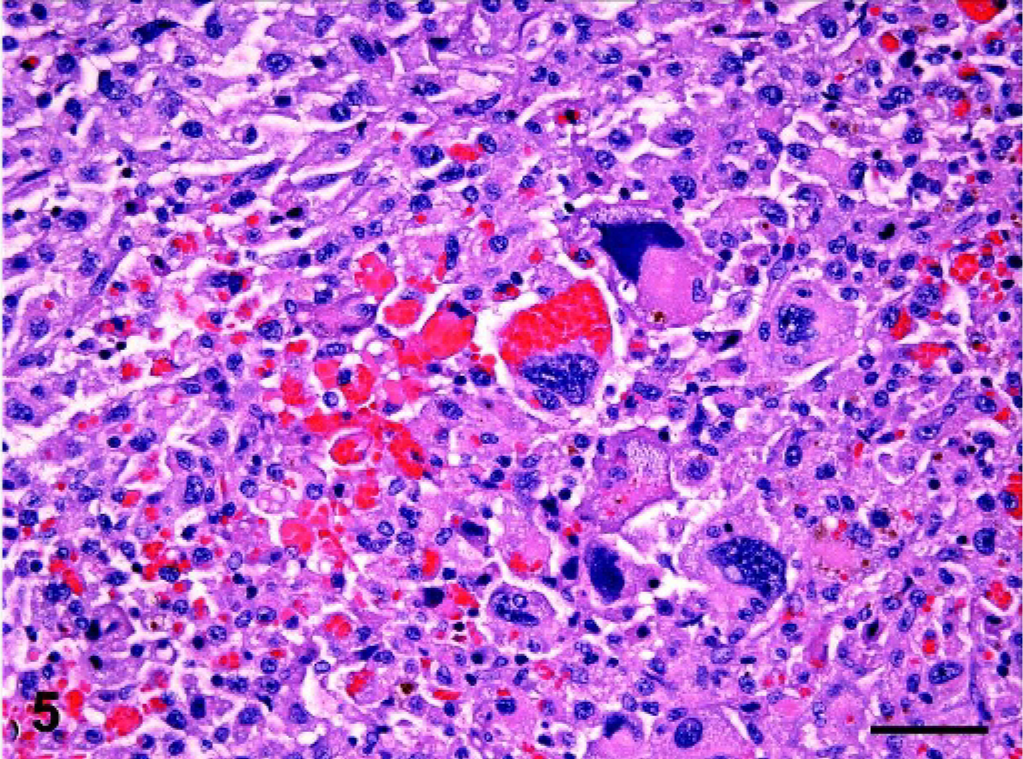
Spleen; dog No. 14. Erythrophagocytic histiocytes with marked cytologic atypia characterized by ← anisocytosis, anisokaryosis, hyperchromatic nuclei, and multiple nucleoli have invaded the splenic red pulp. HE; bar = 50 microns.
The liver was infiltrated by hemophagocytic HS in all dogs assessed (n = 15). Liver invasion occurred via demonstrable intravascular spread within the portal vein. Portal vein branches within the liver usually contained rafts of cytologically atypical histiocytes, many of which exhibited erythrophagocytosis (Fig. 6). Extension into the hepatic parenchyma occurred via hepatic sinusoids. In most instances, histiocytes were confined to the sinusoids and uncommonly breached the hepatic cords to form coalescing histiocytic foci. Foci of EMH were often associated with the invading histiocytes within hepatic sinusoids. In some dogs, the foci of EMH telegraphed the presence of intrasinusoidal histiocytes, which were otherwise largely unapparent without the use of immunostains (CD11d, CD18, see immunohistochemistry section) to disclose their presence (Figs. 7, 8). The hepatic sinusoids often contained fibrin thrombi, which were peripheral to foci of periacinar hepatocellular necrosis with dissociation of hepatic cords. Examination of hepatic cords in dogs with hyperbilirubinemia also revealed canalicular plugging with bile (Fig. 6).
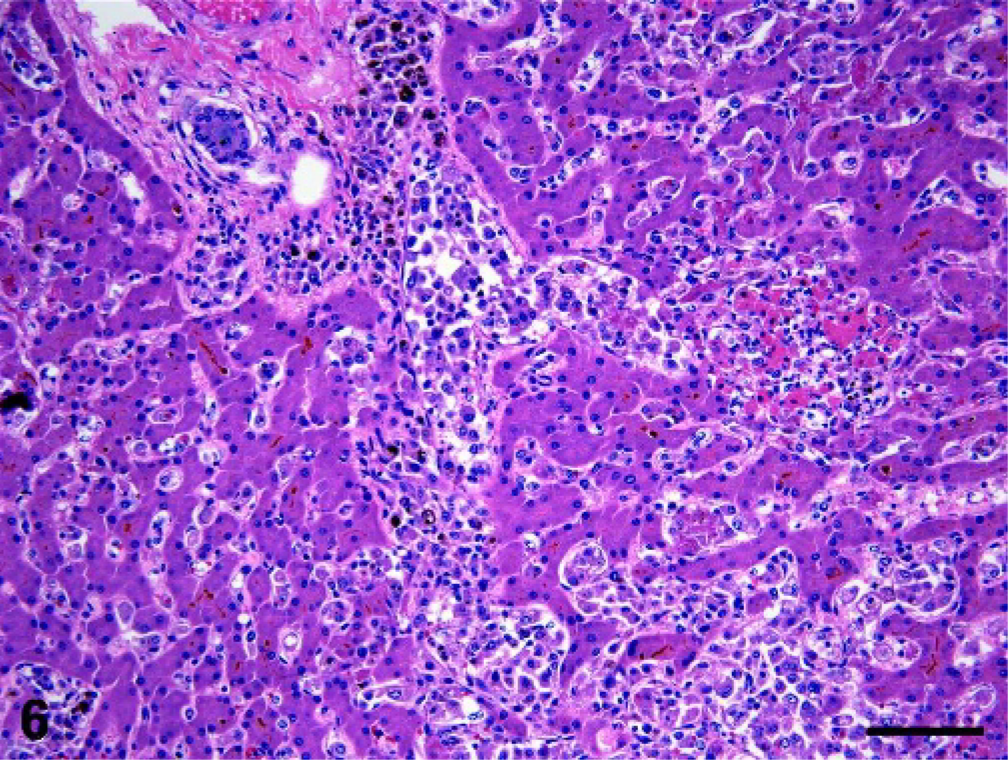
Liver; dog No. 5. Histiocytes invade the liver via the portal vein and sinusoids. Bile canaliculi are distended with bile and hemosiderin-laden histiocytes are evident. A focus of hepatocellular necrosis is present to the right of center. HE; bar = 100 microns.
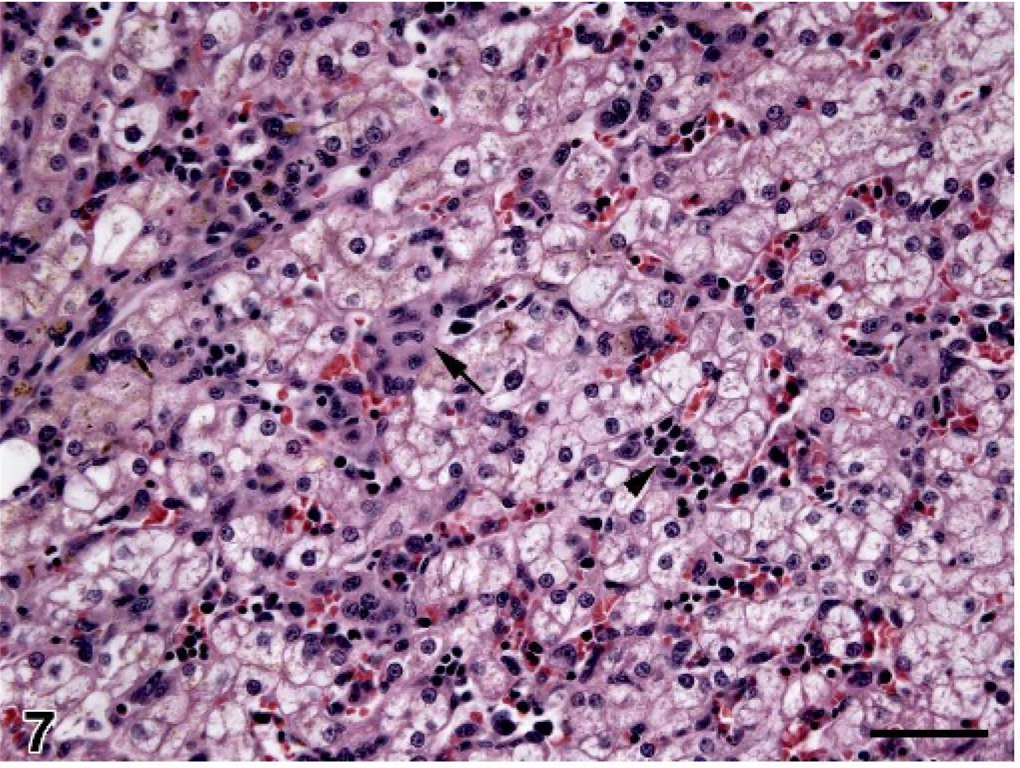
Liver; dog No. 15. Scant histiocyte infiltration within hepatic sinusoids (example at the arrow). Scattered foci of EMH (arrowhead) are also visible adjacent to swollen, vacuolated hepatocytes. HE; bar = 50 microns.
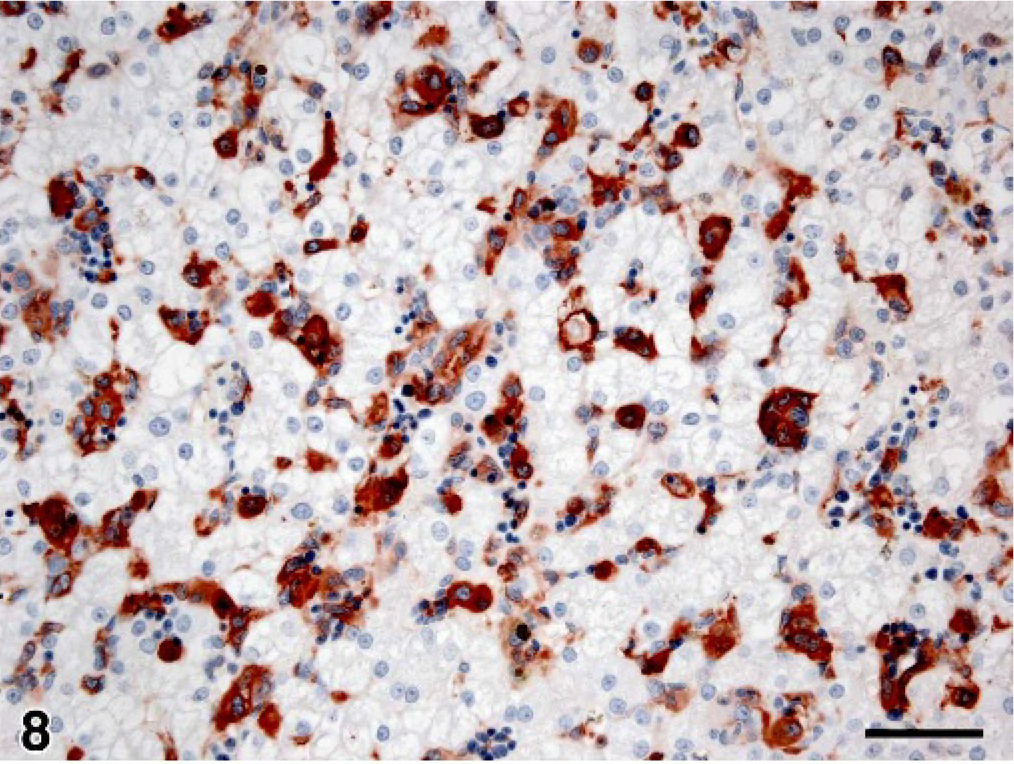
Liver; dog No. 15. Serial section to Fig. 7 illustrating CD11d+ histiocytes invading the hepatic sinusoids mostly as single cells. Immunoperoxidase stain for CD11d; AEC chromogen; hematoxylin counterstain; bar = 50 microns.
The bone marrow was infiltrated by hemophagocytic HS in 10 of 11 dogs. Histiocytes infiltrated between foci of normal hemopoietic precursors of erythroid and myeloid lineages (Fig. 9). Cytologically, histiocytes varied from an almost normal well differentiated appearance to cells with atypia characterized by anisocytosis, anisokaryosis, and karyomegaly (Table 2). The cytoplasm of the histiocytes was eosinophilic, abundant, and often coarsely vacuolated. Many histiocytes contained engulfed red cells and red cell precursors and occasional granulocytes; others contained abundant, finely granular or clumped hemosiderin deposits (Fig. 9). Multinucleated giant cells were infrequently encountered in bone marrow.
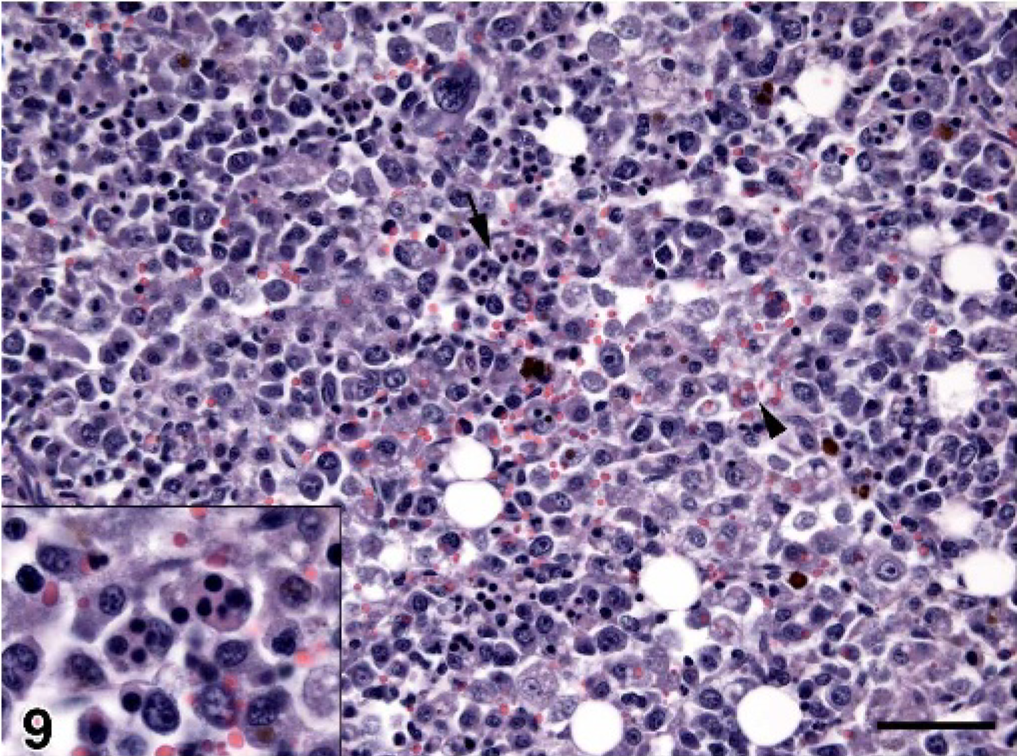
Bone marrow; dog No. 5. Marked infiltration by histiocytes, which have engulfed red cell precursors (arrow and inset) and red cells (arrowhead). HE; bar = 50 microns.
The lung was infiltrated by hemophagocytic HS in all dogs assessed (n = 9). Invasion of the lung was often limited to cytologically atypical, intravascular histiocytes within alveolar septa with limited spread to the alveoli. Immunostains (CD11d, CD18) were particularly effective in detecting tumor histiocytes. In other instances, tumor emboli were readily visible within pulmonary arterial vessels; alveolar septal infiltration was marked in these dogs. Intravascular histiocytes were erythrophagocytic and were accompanied by EMH. Fibrin thrombi were frequently encountered in small arteries and alveolar septal capillaries.
Lymph nodes were less frequently infiltrated by hemophagocytic HS; only 5 of 11 dogs exhibited lymph node involvement. Lymph nodes infiltrated included tracheobronchial, mediastinal, sternal, and abdominal lymph nodes (not exactly specified). In most instances the drainage field of these lymph nodes included the organs most often infiltrated by hemophagocytic HS (spleen, liver, and lung). In lymph nodes, cytologically atypical histiocytes expanded the sinuses of the cortex and medulla. The medullary cords were often spared, whereas the paracortical lymphoid domains were invaded by histiocytes and were partially to totally effaced.
Microscopic infiltration of organs, other than the renal cortex in dog No. 1 and dog. No 9, was not observed in the 6 dogs subjected to extensive necropsy examination, which included examination of tissues from all major organ systems including the central nervous system.
Immunohistochemistry
Identification of histiocytes in normal and diseased tissues was achieved through the use of monoclonal antibodies specific for molecules essential for the function of macrophages and dendritic antigen-presenting cells (i.e., DC). These include molecules involved in antigen presentation such as CD1, MHC class II molecules, CD80 and CD86, and the β2 integrins CD11/CD18, which are adhesion molecules expressed exclusively by leukocytes. In hemophagocytic HS, the β2 integrin staining pattern was particularly valuable in classifying the lineage of the histiocytes and the tissue compartments from which they arose.
The β2 integrin expression by histiocytes was highly ordered in normal canine spleen. CD11c was expressed by interdigitating DC of the white pulp and by marginal zone histiocytes (Fig. 10). CD11c expression in the red pulp was largely limited to circulating granulocytes. CD11d was exclusively expressed by red pulp macrophages beyond the marginal zone (Fig. 11). In the bone marrow, resident macrophages expressed CD11d almost exclusively. CD11b and CD11c were expressed by myeloid precursors, mature granulocytes, and monocytes in hemopoietic foci. In normal liver, CD11d was not expressed by resident Küpffer cells, which expressed CD11b instead. 9, 10
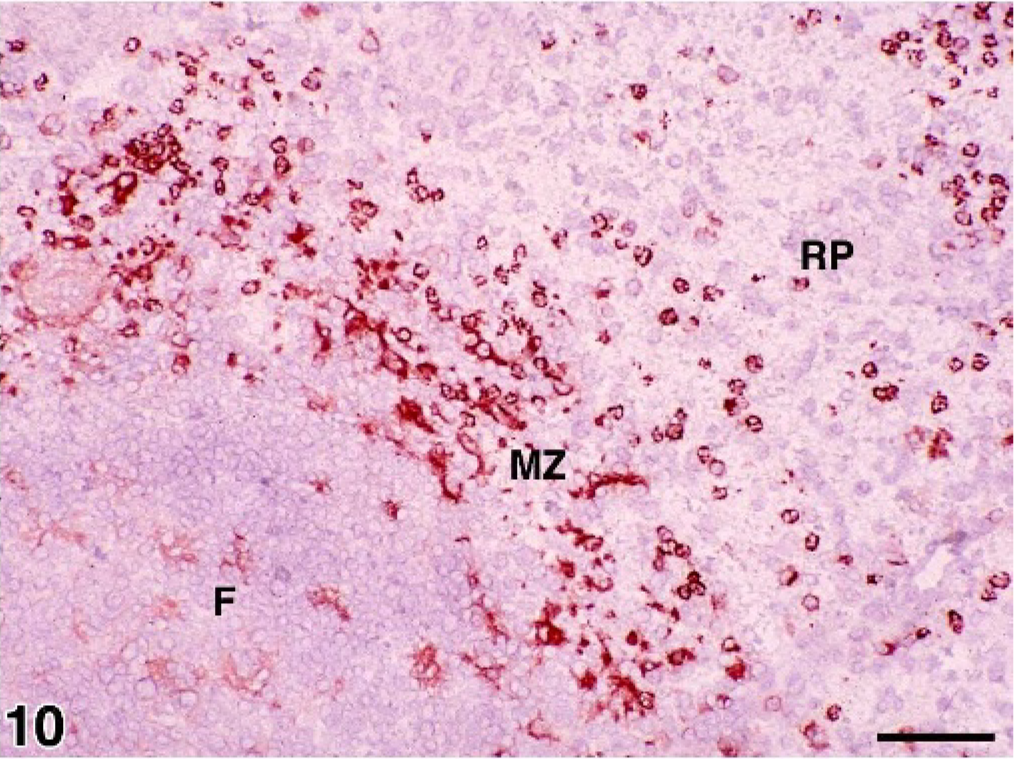
Spleen; normal control dog. Frozen section illustrating CD11c+ histiocytes in a lymphoid follicle (F) ← and the marginal zone (MZ). Only CD11c+ granulocytes are visible in the red pulp (RP). Immunoperoxidase stain for CD11c; AEC chromogen; hematoxylin counterstain; bar = 50 microns.
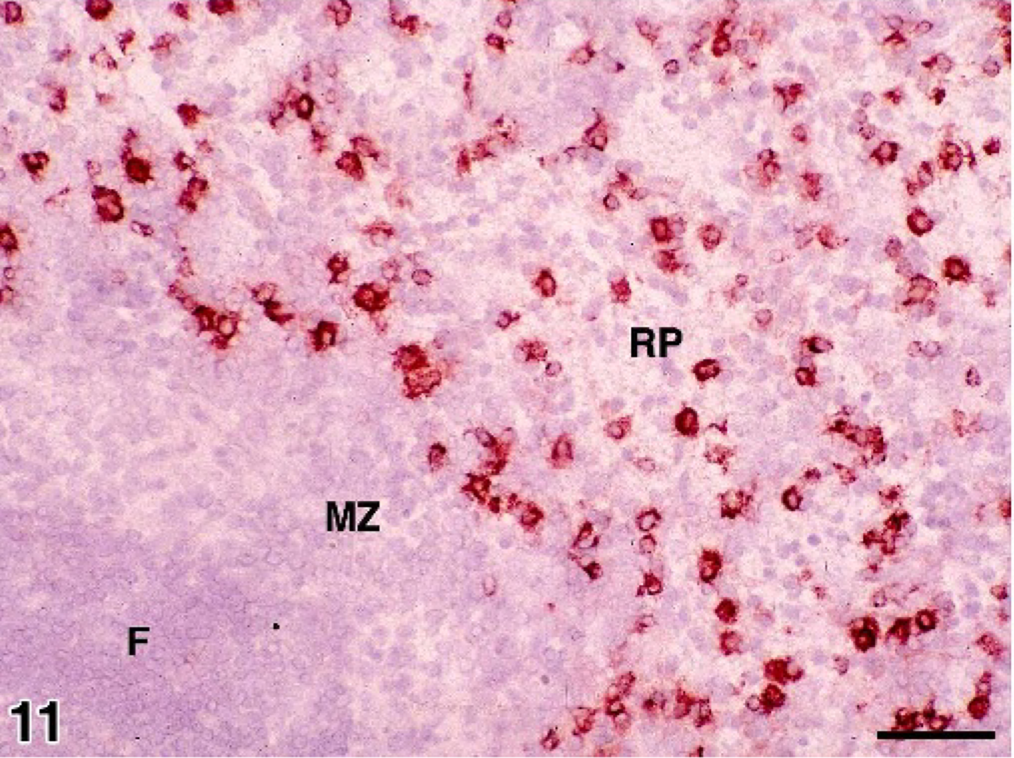
Spleen; normal control dog. Frozen section illustrating CD11d+ histiocytes in the red pulp (RP) peripheral to the marginal zone (MZ). Immunoperoxidase stain for CD11d; AEC chromogen; hematoxylin counterstain; bar = 50 microns.
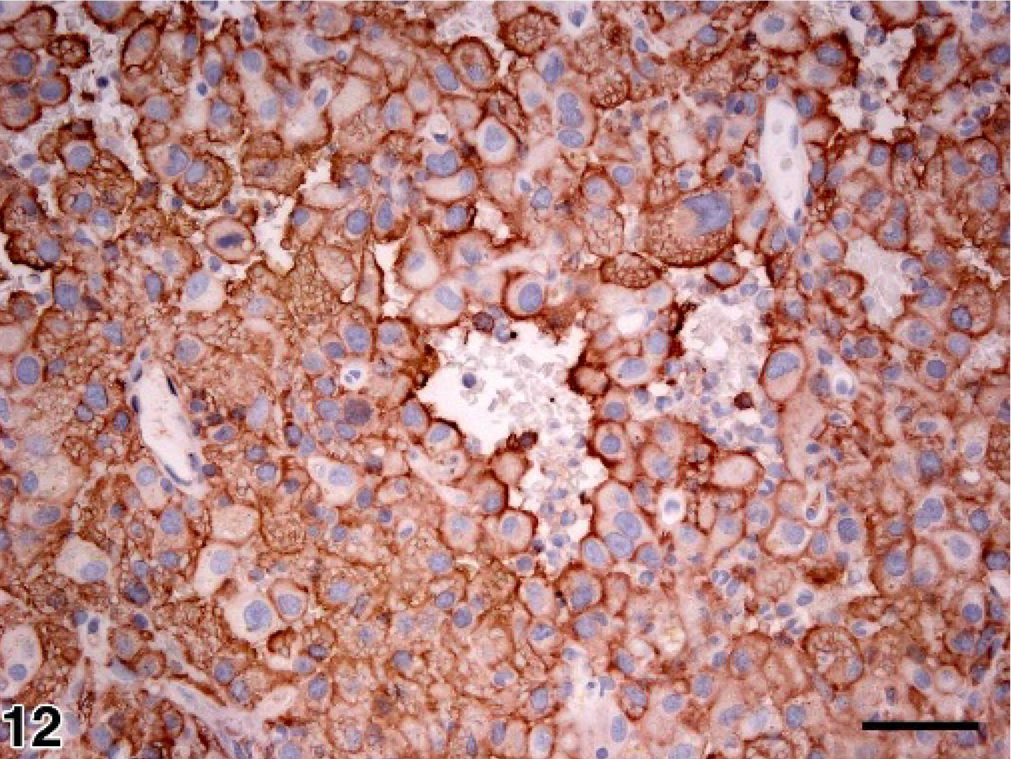
Spleen; dog No. 14. Diffuse expression of CD11d by neoplastic splenic red pulp histiocytes in hemphagocytic HS. Immunoperoxidase stain for CD11d; AEC chromogen; hematoxylin counterstain; bar = 50 microns.
In hemophagocytic HS, neoplastic histiocytes in formalin-fixed tissue sections of spleen from all dogs dominantly expressed CD11d, CD18, and CD45 (Fig. 12). Staining of tissue sections for CD11d expression proved to be a sensitive method to reveal early invasion of neoplastic histiocytes into liver and lung. This was particularly so when examination of HE-stained sections revealed minimal, easily overlooked intravascular invasion (Figs. 7, 8). In 7 dogs frozen tissue sections or FNA smears of spleen were assessed for a broader range of leukocyte antigens (Table 4). Expression of CD11b and CD11c was far less prevalent than CD11d. Neoplastic histiocytes also diffusely expressed MHC class II and CD45. Expression of CD1c, CD80, CD86, and CD90 was more variable and generally not prevalent (Table 4). In no instance was CD45RA or CD4, an activation marker of macrophages and DC, expressed by neoplastic histiocytes.
Expression of leukocyte antigens by histiocytes in hemophagocytic HS∗.

∗Score: 0 = 0% cells; 1 = 1–5% cells; 2 = 5–25% cells; 3 = 25–50% cells; 4 = 50–75% cells; 5 = 75–100% cells. na = not assessed. Frozen sections of spleen: dog Nos. 3, 5, 6, 8, 13, and 17; splenic FNA: dog No. 7.
Scant to moderate numbers of infiltrating lymphocytes were present within lesions of hemophagocytic HS. An evaluation of lymphoid antigen expression revealed mixed populations of T cells, which expressed CD3, TCR αβ more than TCR γδ, and CD4 or CD8. B cells were present in lower numbers and were identified by expression of CD21 and CD79a. Plasma cells were often present in clusters recognizable by expression of CD79a.
Discussion
Hemophagocytic HS was a distinctive clinical and pathologic entity, marked by an aggressive clinical course dominated by splenomegaly, regenerative anemia, thrombocytopenia, hypoalbuminemia, and hypocholesterolemia. Hemophagocytic HS was prevalent in the same breeds affected by localized and disseminated HS. Hemophagocytic HS initially involved spleen and bone marrow simultaneously and later spread to liver and lungs often via insidious intravascular invasion with minimal mass formation. The splenic lesions consisted of diffuse splenomegaly often with additional, ill-defined masses and infarction. Neoplastic histiocytes dominantly expressed MHC class II and the leuko-integrin CD11d/CD18; expression of CD11c/CD18 and CD1c were far less prevalent. The leuko-integrin expression profile was that expected of macrophages resident in splenic red pulp and bone marrow. In contrast, localized and disseminated HS as previously reported dominantly expressed CD1c, CD11c, and MHC class II and lacked expression of CD11d, which supported their origin from interstitial DC. 2
The clinical presentation of hemophagocytic HS was often confused with immune-mediated hemolytic anemia (IMHA) or more specifically with Evan's syndrome, because thrombocytopenia usually occurred concurrently. However, the direct anti-globulin (Coombs) test was negative in all dogs tested (n = 8), and only 2 dogs were clinically icteric. The mean serum total bilirubin concentration in dogs with hemophagocytic HS was 0.6 mg/dl, with a range of 0.1–2.2 mg/dl. In contrast, dogs with IMHA typically have higher serum bilirubin concentrations, with mean values between 6.5 and 8.2 mg/dl. 7, 39 Additionally, hypoalbuminemia and hypocholesterolemia were common in dogs with hemophagocytic HS, which also helped to differentiate it from IMHA. 7 Splenic and bone marrow aspirates revealed expanded numbers of erythrophagocytic histiocytes, many with a deceptively well differentiated cytologic appearance, in about half of the afflicted dogs, which no doubt obfuscated the diagnosis of hemophagocytic HS. In those instances, histiocytes manifesting cytologic atypia were more prevalent in splenic aspirates than in bone marrow aspirates. However, the sheer number of macrophages, often present as aggregates, in bone marrow aspirates was frequently overwhelming, a feature that has been noted previously. 50 This was another useful distinguishing feature versus IMHA which, in our experience, lacks the same degree of marrow histiocytic expansion.
Responsive anemia and thrombocytopenia were the most common hematologic abnormalities. Marked erythrophagocytosis by neoplastic macrophages in the spleen, liver, bone marrow, and lung was the most likely cause of the anemia, because macrophage expansion in the bone marrow did not produce myelophthisis. Additionally, there was prominent EMH in both the spleen and liver, and the anemia was therefore almost always responsive. The pathogenesis of the thrombocytopenia was probably multifactorial. Although not easy to observe, phagocytosis of platelets by neoplastic macrophages was likely. Additionally, thrombosis was prominent in affected organs. Platelet consumption was therefore likely an additional factor, and the frequent occurrence of PTT elevations was consistent with the presence of a consumptive coagulopathy. The consistent presence of a responsive anemia and thrombocytopenia helped to clinically distinguish hemophagocytic HS from the more common nonhemophagocytic HS of DC type. The mean hematocrit and platelet counts of the latter were significantly higher than those of hemophagocytic HS (Table 3).
Hypoalbuminemia (mean 2.2 g/dl) was the most consistent biochemical abnormality, followed by hypocholesterolemia (mean 123 mg/dl) and mild hyperbilirubinemia. The hypoalbuminemia and hypocholesterolemia did not appear to be caused by hepatic insufficiency, as serum urea and glucose concentrations were usually within reference limits. In humans, hypoalbuminemia and hypocholesterolemia can occur concurrently with underlying inflammation, as both albumin and cholesterol are negative acute phase reactants. 4, 5, 13 Elevated levels of the proinflammatory cytokines interleukin 6 (IL-6) and tumor necrosis factor α (TNF-α) have been most implicated as the cause of decreased hepatic synthesis of albumin and cholesterol. In dogs with hemophagocytic HS, production of elevated levels of both IL-6 and TNF-α may play a role in the pathogenesis of hypoalbuminemia and hypocholesterolemia, as macrophages are a rich source of these cytokines. Increased catabolism of albumin and cholesterol may also be involved, but has not been evaluated. Although hypoalbuminemia (mean 2.5 g/dl) was also common in nonhemophagocytic HS (i.e., HS of DC type), the mean serum albumin concentration was significantly higher. Curiously, hypocholesterolemia was uncommon in nonhemophagocytic HS and may be useful in the clinical distinction of these 2 histiocytic malignancies.
The unique pattern of organ infiltration evident in canine histiocytic sarcomas with prominent hemophagocytosis compared with other histiocytic sarcomas has not been emphasized in most previous reports. 6, 22, 34, 40, 51 Splenic lesions similar to those presented in this article have been described previously under the designation myeloid metaplasia (i.e., EMH) with histiocytosis and hypersplenism. 43 In that report, lesions from dogs with IMHA (Coombs positive) and histiocytic sarcoma/malignant histiocytosis were described as a continuum underpinned by the presence of EMH. The presence of giant cells was the most significant and discriminatory predictor of a negative outcome, and presumably some of these dogs had hemophagocytic HS based on evidence of metastatic disease in 8 of 15 dogs necropsied. Interestingly, 5 of 7 dogs without gross evidence of extrasplenic disease had bone marrow histiocytosis. Other than this, the pattern of organ infiltration beyond the spleen was not emphasized. Furthermore, consistent clinicopathologic abnormalities, save for a poorly responsive normochromic normocytic anemia, were not described.
Hemophagocytic HS arose in the splenic red pulp, which was essentially diffusely infiltrated. Grossly, this manifested as diffuse splenomegaly, although mass formation often occurred as a coincidental lesion. Simultaneously, there was usually an expansion of the bone marrow histiocyte population, which also exhibited hemophagocytosis. Cytologic atypia manifested by the splenic histiocytes was often more prominent than that of bone marrow histiocytes. Histiocytes invaded the splenic red pulp sinuses and traveled in the splenic and portal veins to involve the liver. The pattern of liver involvement was sinusoidal permeation, which was occasionally occult and easily overlooked. The liver could be massively infiltrated without noticeable mass formation. A similar pattern of intravascular invasion with mimimal mass formation occurred in the lung. Immunohistochemical staining to demonstrate CD11d expression was a valuable adjunct to morphologic assessment, particularly in early liver and lung infiltration.
Hemophagocytic HS is a proliferative disease of splenic red pulp and bone marrow macrophages; it is not a proliferative disorder of interstitial DC, as are most other histiocytic sarcomas in the dog. 2 Histiocytic sarcomas of interstitial DC type diffusely express CD1c and CD11c, but not CD11d. Hemophagocytic HS diffusely expressed CD11d, but inconsistently expressed CD1c and CD11c. CD1 molecules are highly expressed by DC, but are not constitutively expressed by macrophages. 30, 31 CD1 molecules are functionally important for DC, because they are involved in the presentation of lipid and glycolipid antigens to T cells. 3, 11, 21
The original premise for the development of MAb specific for canine β2 integrins (CD11/CD18), was their potential for characterizing lineages of cells in canine hemopoietic neoplasia. 9 The unique expression pattern of CD11d in spleen facilitated both its discovery and characterization as a novel β2 integrin and led directly to the discovery of the human analogue. 10, 46 The expression of the β2 integrins, CD11c and CD11d, in canine spleen appears highly regulated. CD11c is dominantly expressed by histiocytes in the white pulp and marginal zone. In contrast, CD11d is expressed only in splenic red pulp by resident macrophages and T cells, the majority of which were CD4–CD8–. 10 We later found that CD11d+ γδ T cells were 5-fold enriched in splenic red pulp compared to peripheral blood (S. M. McDonough and P. F. Moore, unpublished observations). Canine T cell chronic lymphocytic leukemia of granular lymphocyte type and hepatosplenic γδ T cell lymphoma are lympho-proliferative diseases that show prominent expression of CD11d; both originate in splenic red pulp. 20, 29, 47 Hemophagocytic HS completes the picture, since it should now be recognized as the first canine histiocytic proliferative disease of macrophage lineage, which originates in splenic red pulp and bone marrow and retains the β2 integrin expression pattern expected of macrophages in those compartments—diffuse expression of CD11d/CD18. CD11d and CD18 are clearly not lineage-specific molecules. Hence, expression of CD11d/CD18 should be interpreted in the context of clinical and pathologic findings as well as the expression profile of other leukocyte antigens.
Hemophagocytic HS of dogs shares clinicopathologic and morphologic features with a rare subtype of malignant histiocytosis (i.e., disseminated HS) of humans, also referred to as histiocytic medullary reticulosis. 49 In histiocytic medullary reticulosis, anemia, thrombocytopenia, and hyperbilirubinemia occur in association with diffuse hepatosplenomegaly and bone marrow infiltration. Cytologically atypical, hemophagocytic histiocytes expand the splenic red pulp and invade the hepatic sinsusoids without significant mass formation. 49
Hemophagocytic syndromes in humans also occur in the context of infection and malignancy and as familial disorders, which affect cytotoxic T cell and NK cell function. 16– 19, 23– 25, 44 Hemophagocytic syndromes associated with infection, malignancy, or unknown cause have also been reported in dogs. 45, 48, 50 Immunologic deficiencies leading to hemophagocytic syndrome have been best documented in human familial hemophagocytic lympho-histiocytosis (FHLH). 17, 18, 44 Defective expression of the cytotoxic granule protein, perforin, or defective transportation of cytotoxic granules to the cell surface because of mutations in proteins involved in the secretory cytotoxic pathway are involved in the pathogenesis of FHLH. 12, 17, 18, 44, 52 A role for the secretory cytotoxic pathway in immune system regulation and lymphocyte homeostasis has emerged from these studies. 8, 12, 14, 26, 36 Although the pathogenesis remains elusive, FHLH and secondary HLH are associated with infiltration of tissues by activated CD8+ T cells, which are associated with activated hemophagocytic macrophages. Massive cytokine release, particularly of IL-1, IL-6, TNF-α and interferon γ, underlies the macrophage activation. 12, 19 Viral infection, usually Epstein-Barr virus or Cytomegalovirus, may trigger the onset of the hemophagocytic syndrome in FHLH, as well as in nonfamilial or secondary HLH. 19, 23 It is believed that defective cytotoxic T cell and NK cell function in FHLH allows the persistence of activated T cells, which infiltrate tissues and incite the hemophagocytic syndrome by promoting inflammatory cytokine release. 12, 26 Important distinguishing features of FHLH are that disease onset mostly occurs in early childhood and the hemophagocytic macrophages are activated, well differentiated normal cells, which are accompanied by substantial numbers of activated T cells. Tissues infiltrated by lymphocytes and macrophages include central nervous system, liver, lymph nodes, spleen, and bone marrow, in decreasing order of frequency. The histologic pattern of liver infiltration is most like chronic persistent hepatitis with marked involvement of the portal tracts. 15, 16, 38 The pattern of tissue involvement with clear intravascular invasion by cytologically atypical histiocytes (in tissues other than bone marrow) and the sparse T cell infiltration accompanying the histiocytes are important features of canine hemophagocytic HS that distinguish it from the familial and secondary hemophagocytic syndromes of humans and the secondary hemophagocytic syndromes of dogs.
In conclusion, we have described a macrophage variant within the canine histiocytic sarcoma disease complex, which previously featured diseases derived entirely from interstitial DC. The unique pattern of organ infiltration evident in canine HS with prominent hemophagocytosis differs markedly from HS of DC origin. It should also be mentioned for completeness that mixed histiocytic tumors have been observed rarely, i.e., dogs that have hemophagocytic HS occurring in the context of HS of DC origin (dogs not included in this report). The tissue infiltration patterns remain discrete by HS type. These complex situations are further resolvable with immunostains for an array of leukocyte antigens; CD1, CD11c, CD11d, and CD18 are particularly relevant. Canine histiocytic proliferative diseases now encompass proliferation of all known members of the myeloid histiocytic lineage: Langerhans cells, interstitial DC, and macrophages. It is currently unknown if proliferative diseases of lymphoid type DC, known as plasmacytoid DC, occur in dogs because the identification of canine plasmacytoid DC has been hampered by lack of specific markers for these cells.
Footnotes
Acknowledgements
We thank the numerous clinicians and pathologists who contributed case material for this report. We also thank Diane Naydan for immunohistochemistry assistance, and the Center for Companion Animal Health, University of California, Davis, for support for monoclonal antibody development.
