Abstract
Although synovial cell sarcoma is reported to be the most common neoplasm of the canine synovium, this retrospective study of 35 canine synovial tumors found that the majority were of histiocytic origin. Five (14.3%) synovial cell sarcomas were identified by positive immunohistochemical staining with antibodies to cytokeratin. Eighteen (51.4%) histiocytic sarcomas were identified by cell morphology and immunohistochemical staining with antibodies to CD18. Six (17.1%) synovial myxomas were identified by histologic pattern. The remaining six (17.1%) synovial tumors represented a variety of sarcomas, including two malignant fibrous histiocytomas (actin positive), one fibrosarcoma, one chondrosarcoma, and two undifferentiated sarcomas. Rottweilers were overrepresented in the histiocytic sarcoma category and Doberman Pinschers were overrepresented in the synovial myxoma category. The average survival time was 31.8 months for dogs with synovial cell sarcoma, 5.3 months for dogs with histiocytic sarcoma, 30.7 months for dogs with synovial myxoma, and 3.5 months for dogs with other sarcomas. Among the dogs with follow-up information available, metastatic disease was detected in 25% of dogs with synovial cell sarcoma, in 91% of dogs with histiocytic sarcoma, in none of the dogs with synovial myxoma, and in 100% of dogs with other sarcomas. Immunohistochemical staining for cytokeratin, CD18, and smooth muscle actin is recommended to make the diagnosis and thereby predict the behavior of synovial tumors in dogs.
Keywords
Synovial cell sarcoma (SCS) is reported to be the most common joint tumor of dogs. 20,22,27 Excellent reviews of the radiographic, histologic, and behavioral features of this tumor in dogs have been published. 12,15,20,24 These tumors typically occur in the large joints of the extremities (especially the stifle) in middle-aged, large-breed dogs. 12,14 The histologic appearance is described as monophasic (spindle) or biphasic with both spindle cell and epithelioid components. 12,20 Positive cytokeratin staining of the epithelioid component has been associated with a good prognosis in dogs. 24
Normal synovium consists of a single or double layer of synoviocytes above a loose connective tissue layer containing adipocytes, fibroblasts, histiocytes, and blood vessels. The synoviocytes have been classically divided into type A (phagocytic function) and type B (synovial fluid synthesis function). However, immunohistochemistry has shown three distinct populations: type 1, phagocytic macrophages; type 2, antigen-presenting dendritic cells; and type 3, fibroblast-like mesenchymal cells that produce glyosaminoglycans (formerly type B cells). 25 Theoretically, a neoplasm could arise from any of these cell types.
In humans, the diagnosis of SCS is based on histopathology, immunohistochemistry, and cytogenetics. The monophasic variant is the most common form and immunohistochemistry for cytokeratin is used to differentiate these from other spindle cell sarcomas. 13,23 In addition, 90% of human SCSs have a specific translocation involving chromosomes X and 18. 2,6,25 This molecular marker is especially useful in identifying the 5–15% of human SCSs that are not associated with joints or tendon sheaths. These are thought to arise from primitive mesenchymal cells similar to type 3 synovial cells. 2,6
Other joint tumors reported in dogs include fibrosarcoma, rhabdomyosarcoma, osteosarcoma, malignant fibrous histiocytoma (MFH), liposarcoma, hemangiosarcoma, myxoma, malignant giant cell tumor of soft tissue, and undifferentiated sarcoma. 20,27 Recently, histiocytic sarcomas have been reported in the periarticular tissue of large appendicular joints. 17
Materials and Methods
The records of the University of Pennsylvania School of Veterinary Medicine surgical biopsy service were searched for synovial tumors diagnosed between 1993 and 2000. Only those cases seen by the Veterinary Hospital of the University of Pennsylvania and two referral hospitals were included to simplify medical record retrieval. Medical records were reviewed for breed, sex, age, weight, radiographic findings, surgical biopsy site, treatment, and outcome. The presence or absence of metastatic disease was determined by radiography, cytology, ultrasound, necropsy, or a combination of these. However, the cases in which metastatic disease was detected by radiographs or ultrasound were not all confirmed by histopathology as synovial tumor metastases.
All tissues were identified as intra-articular at surgery or necropsy. Tissues were submitted fixed in 10% neutral buffered formalin, embedded in paraffin, and 5-µm sections were stained with hematoxylin and eosin (HE). Tumors identified as myxomatous on HE were also stained with Alcian blue (1% Alcian blue in 3% acetic acid, pH 2.5).
Immunohistochemistry for cytokeratin (AE1/AE3 and CAM 5.2), vimentin, smooth muscle actin, CD3, CD18, and MAC387 was performed on formalin-fixed, paraffin-embedded 5-µm sections using a Autostainer Immunostaining System (DAKO, Carpinteria, CA). The slides were placed in a 60 C oven for 60 minutes then deparaffinized. Some of the antibodies required pretreatment with Proteinase K for 5 minutes (AE1/AE3, CAM 5.2, CD3, and MAC387). The CD18 slides were pretreated for 5 minutes with a protein block (CSA, DAKO). The slides were incubated for 30 minutes with the monoclonal antibodies AE1/AE3 (Boehringer Mannheim, Indianapolis, IN) at 1:500, CAM 5.2 (Becton Dickinson, San Jose, CA) at 1:20, vimentin (DAKO) at 1:30, actin (DAKO) at 1:25, CD18 (Dr. P. F. Moore, University of California, Davis) at 1:25, and MAC387 (DAKO) at 1:1,600, and for 10 minutes with the polyclonal antibody CD3 (DAKO) at 1:100. A two-step non–avidin–biotin detection method (ENVISION+, DAKO) was used for the vimentin, CAM 5.2, and AE1/AE3 antibodies. A labeled streptavidin–peroxidase method (LSAB+, DAKO), was used for the CD3, actin, and MAC 387 antibodies. A peroxidase-catalyzed signal amplification method (CSA, DAKO) was used for the CD18 antibody.
The Kaplan-Meier product limit method was used to estimate the portion of dogs that were alive or that had died in each histologic grouping (Stata 7.0 for Windows, College Station, TX). Statistical differences in survival among histologic groups were assessed by the log rank test. Additionally, analysis of variance was used to compare differences in age and weight among the histologic groups and the chi-square test was used to compare metastasis. Fisher's exact test was used to compare breed differences of synovial tumors to the surgical biopsy population. Statistical significance was defined as P < 0.05. These analyses were performed using SAS statistical software (SAS Institute Inc., Version 8.0, Cary, NC).
Results
Synovial cell sarcoma
Five (14.3%) of the synovial tumors were identified as SCSs by positive staining with the cytokeratin antibody AE1/AE3. The SCS cells were regularly spaced with oval nuclei, smooth chromatin, and central nucleoli. The cells were spindle shaped with indistinct cell borders and variable amounts of eosinophilic fibrillar cytoplasm and stroma (Fig. 1). They occasionally formed concentric rings around medium-sized blood vessels. The cytokeratin staining was limited to small clusters of cells constituting less than 10% of the neoplastic cells (Fig. 2). These small clusters had the same spindle shape as the cells that did not stain with cytokeratin. No epithelioid or glandular structures were identified by HE staining or cytokeratin staining. The overlying synovial membrane cells did not stain for cytokeratin. The SCSs also stained diffusely positive for vimentin, but were negative for all other antibodies tested, including CAM 5.2.
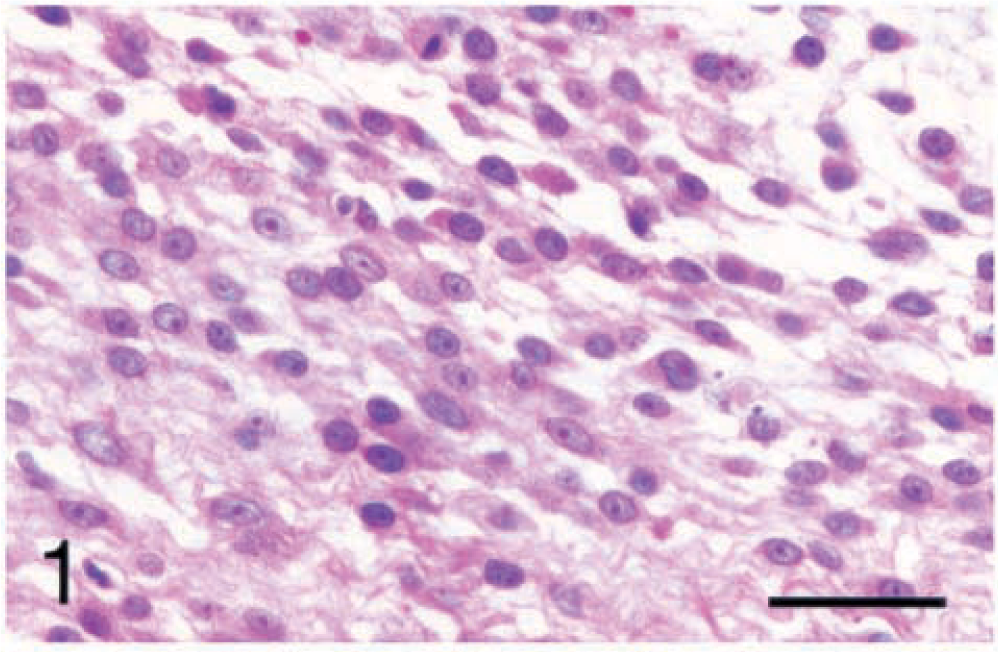
Carpus; synovial cell sarcoma; dog No. 1. The cells are spindle shaped with indistinct cell borders and variable amounts of eosinophilic fibrillar stroma and oval nuclei with smooth chromatin and central nucleoli. HE. Bar = 40 µm.
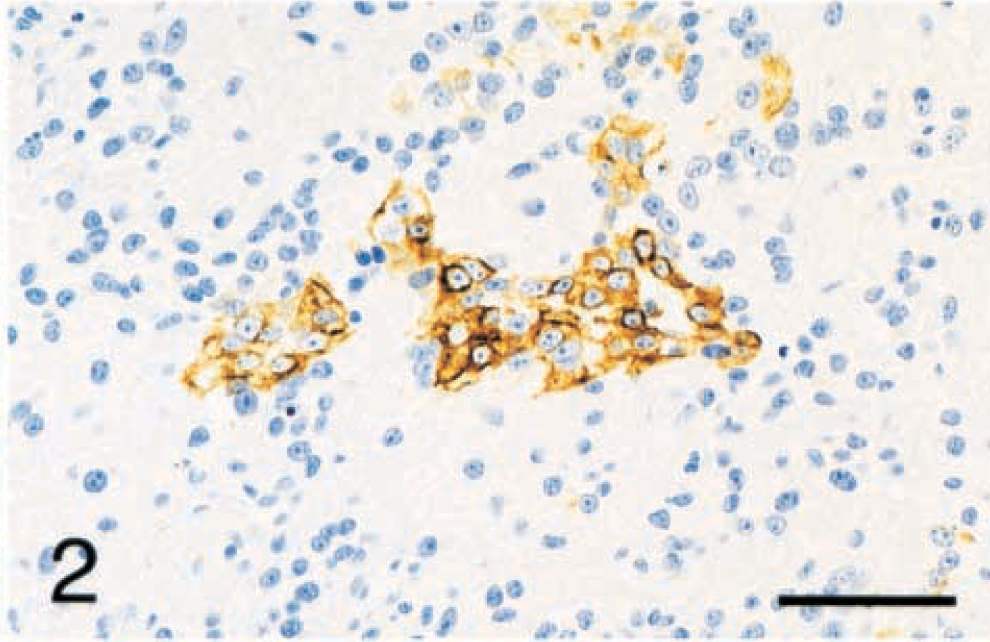
Tarsus; synovial cell sarcoma; dog No. 3. The cytoplasmic cytokeratin staining is limited to small clusters of cells that are otherwise indistinguishable from the surrounding cells. Immunohistochemistry for cytokeratin (AE1/AE3) with hematoxylin counterstain. Bar = 48 µm.
The site, breed, treatment, metastatic behavior, and survival of the dogs with SCSs are listed in Table 1. The average weight of affected dogs was 33.8 kg with a range of 8–60 kg. Three of the dogs were castrated males, one was an intact male, and one was a spayed female. The average age at time of diagnosis was 9.3 years, with a range of 7–12 years.
The breed, site, treatment, and outcome of five dogs with synovial cell sarcomas.

The average time from diagnosis to death or euthanasia of the dogs with SCSs was 31.8 months with a range of 0–74 months. Dog No. 1 was euthanatized at the time of diagnosis and a complete necropsy did not detect metastasis. Lung metastases were detected by radiographs at the time of euthanasia (4 months after diagnosis) in dog No. 2. Dog No. 3 died at home of unknown causes 15 months after diagnosis. Dog Nos. 4 and 5 were euthanatized 74 and 66 months after diagnosis, respectively, for reasons unrelated to the primary neoplasia or metastatic disease. Of the four dogs with SCS with definitive follow-up information, only one had evidence of metastatic disease.
Histiocytic sarcoma
Eighteen (51.4%) of the synovial tumors were identified as histiocytic sarcomas by cell morphology and positive staining with CD18 antibody. Most of the histiocytic sarcomas contained areas of spindle-shaped cells as well as round cells. The cell morphology was very anaplastic in the histiocytic sarcomas, with large pleomorphic nuclei and coarse chromatin. The cytoplasm was abundant and varied from smooth and brightly eosinophilic to fibrillar to vacuolated (Fig. 3). The immunohistochemical staining for CD18 was sparsely punctate to densely punctate and coalescing along cell borders (Fig. 4). The staining was strongest in areas where the neoplastic cells were round and multinucleated. The neoplastic cells were positive for vimentin, but negative for all other antibodies tested, including MAC387.
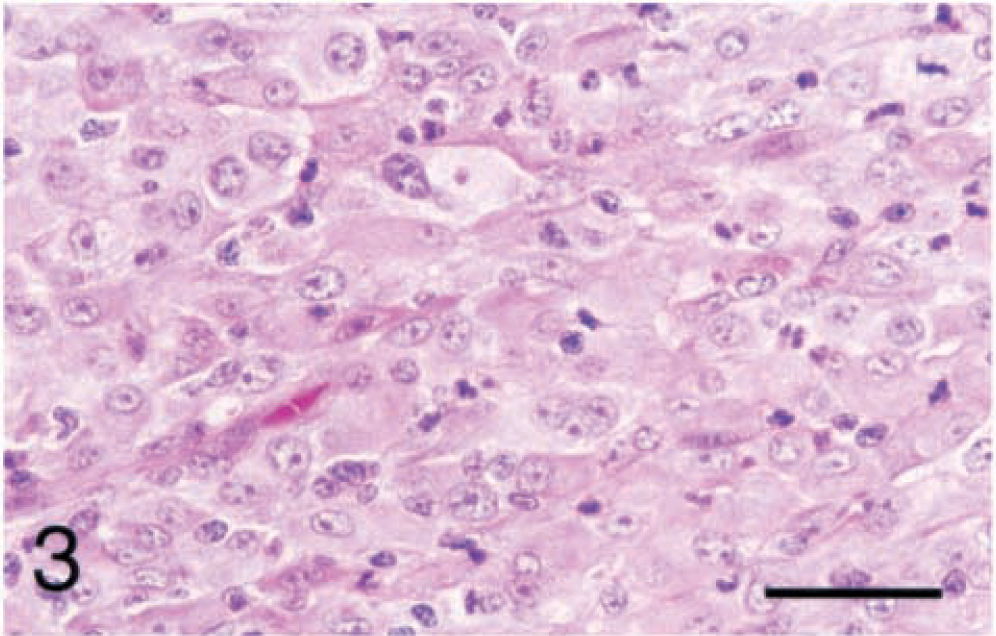
Stifle; histiocytic sarcoma; dog No. 11. The neoplastic cells are large with abundant eosinophilic cytoplasm and pleomorphic nuclei with coarse chromatin. Notice the neutrophils and lymphocytes between the neoplastic cells. HE. Bar = 40 µm.
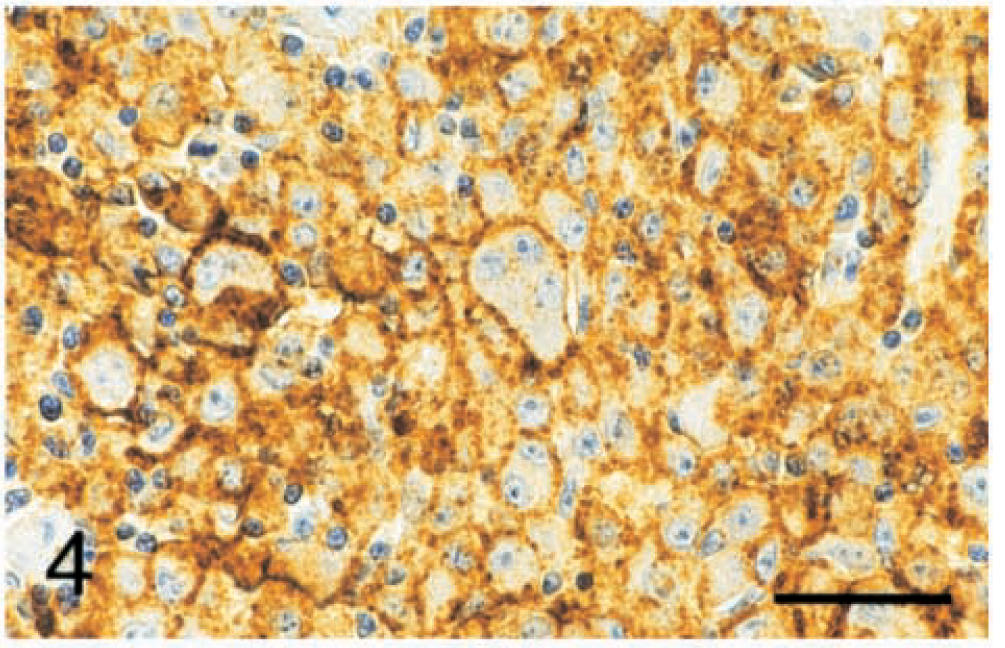
Stifle; histiocytic sarcoma; dog No. 11. The CD18 staining is punctate to coalescing along cell borders. Immunohistochemistry for CD18 with hematoxylin counterstain. Bar = 40 µm.
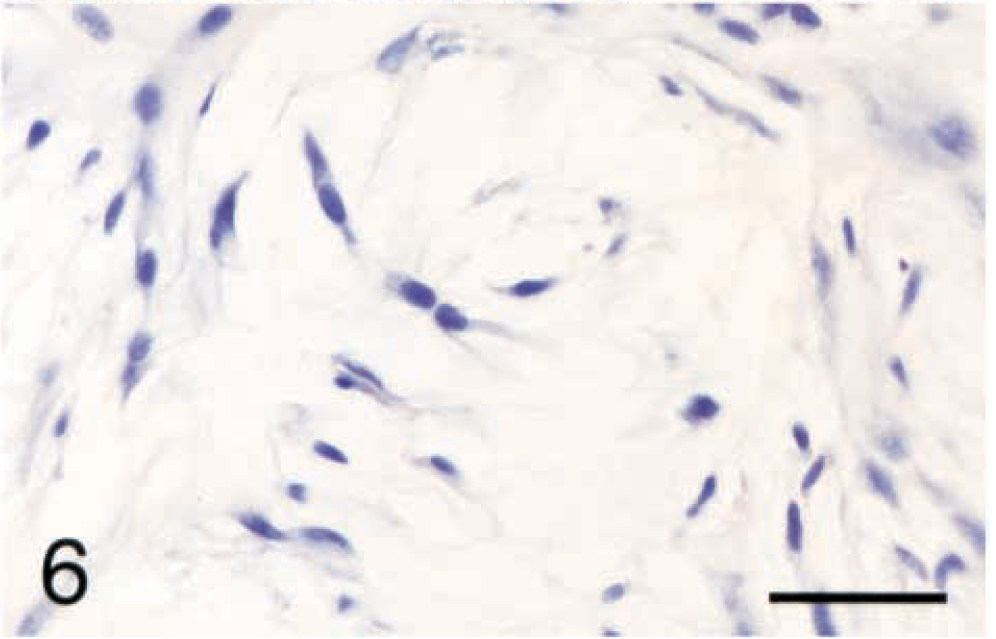
The site, breed, treatment, metastatic behavior, and survival of the dogs with histiocytic sarcomas are listed in Table 2. Eleven (61%) of the dogs with histiocytic sarcomas were Rottweilers. This is significantly more than expected (P < 0.01). The average weight of affected dogs was 43.1 kg with a range of 11.2–65.4 kg. The average age at time of diagnosis was 7.5 years with a range of 3–13 years. No sex predilection was found for histiocytic sarcomas (six castrated males, four intact males, five spayed females, and one intact female).
The breed, site, treatment, and outcome of 18 dogs with histiocytic sarcomas of the synovium.
Numerous inflammatory cells including CD3-positive T lymphocytes and MAC387-positive neutrophils and macrophages were distributed throughout the histiocytic sarcomas. The adjacent synovium also was often severely inflamed and contained infiltrates of lymphocytes, plasma cells, and macrophages. Eleven (61%) of the histiocytic sarcomas were in the stifle (eight Rottweilers, one Bernese Mountain Dog, and two mixed-breed dogs). Six (54.5%) of the dogs with stifle histiocytic sarcomas had a history of cranial cruciate ligament rupture in the same joint, including five Rottweilers (dog Nos. 10, 12, 13, 14, and 16) and one Labrador Retriever cross (dog No. 18). One of the Rottweilers (dog No. 16) had evidence of bilateral surgical repair of cranial cruciate ligament rupture at necropsy.
The average time from diagnosis to death or euthanasia was 5.3 months with a range of 0–16 months. Eleven of the 18 dogs with histiocytic sarcoma had follow-up information available regarding metastatic disease in the form of radiographs, lymph node cytology, ultrasound, or necropsy. Ten of those 11 (91%) had evidence of metastatic spread. The lungs, lymph nodes, and liver were the most common metastatic sites. The remaining dog (dog No. 20) was euthanatized for local spread of the primary tumor. Therefore, all of the dogs with histiocytic sarcoma for which follow-up information was available died or were euthanatized for reasons related to that neoplasm.
Synovial myxoma
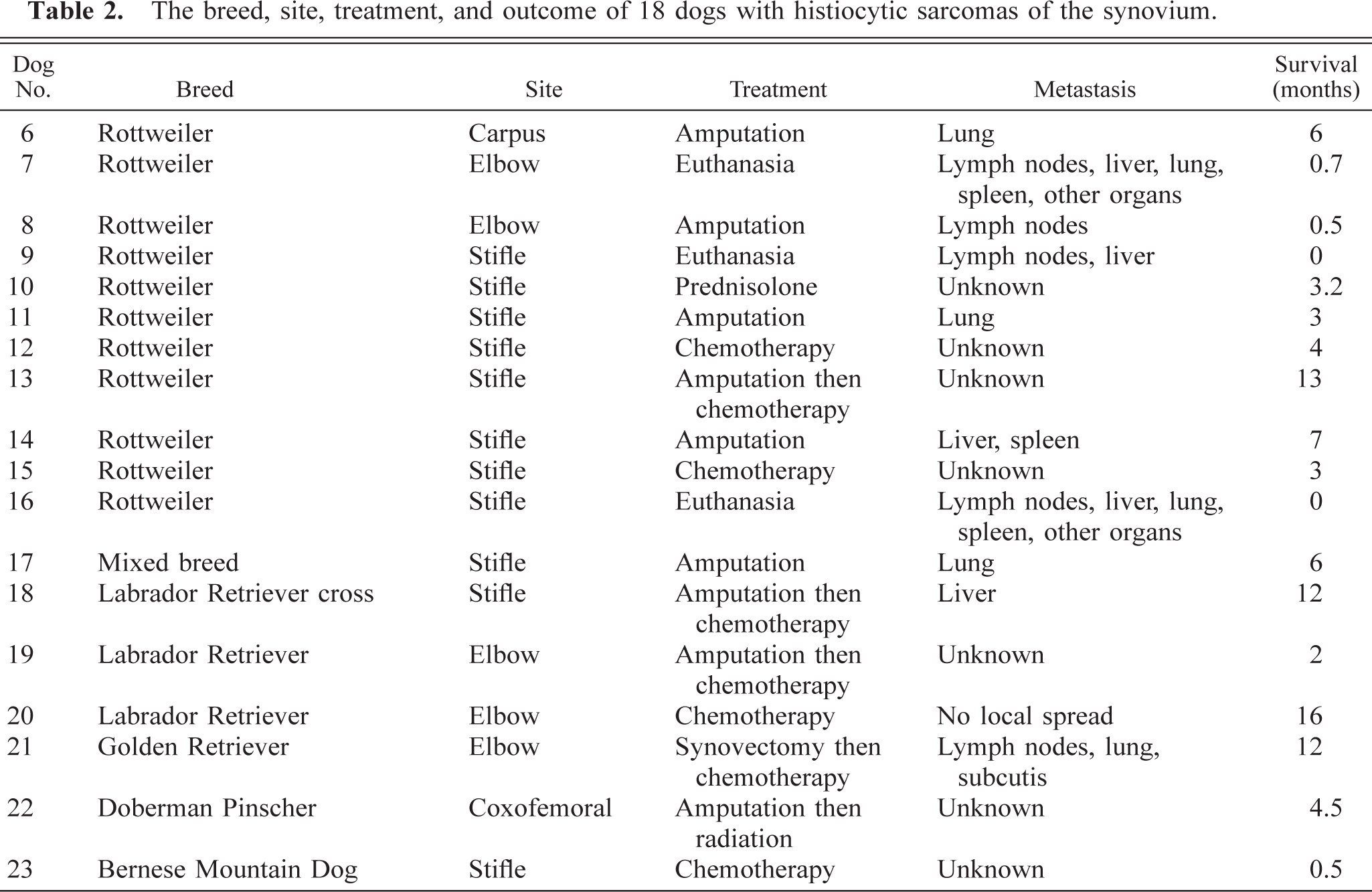
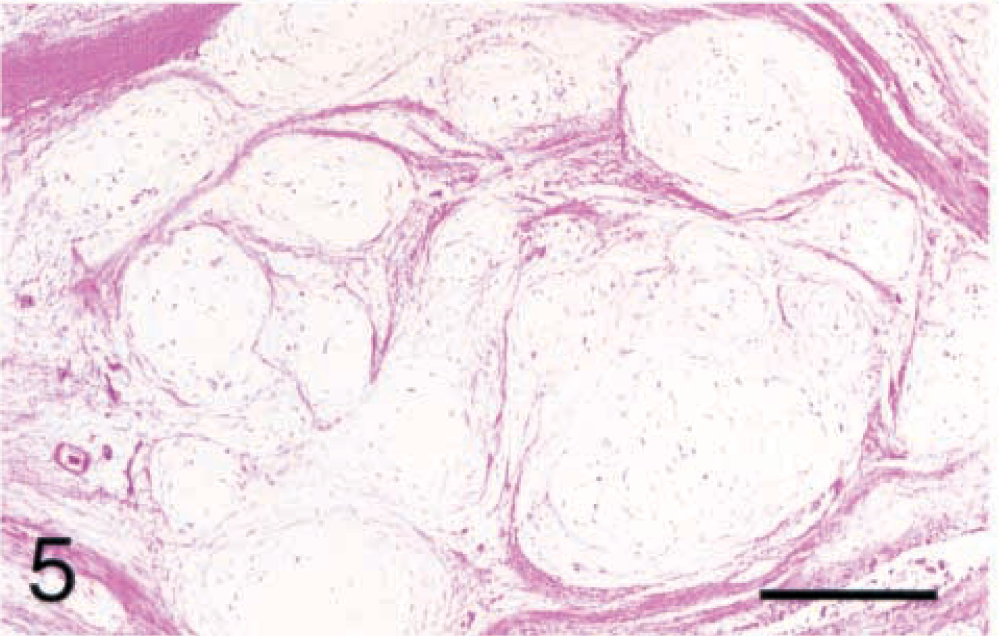
Six (17.1%) of the synovial tumors were identified as myxomas by their unique histologic appearance. These tumors consisted of multiple myxomatous islands (Fig. 5) containing widely separated stellate cells (Fig. 6). The overlying synovial membrane was often hyperplastic. The myxomatous islands extended into the bone on either side of the joint, resulting in areas of bony lysis radiographically and histologically. The myxomatous nature of the tumors was confirmed by positive staining with Alcian blue. These tumors also stained with vimentin, but were negative for all other antibodies tested.
Stifle; synovial myxoma; dog No. 28. The tumor consists of multiple myxomatous islands of variable sizes separated by collagenous septa. HE. Bar = 387 µm.
Stifle; synovial myxoma; dog No. 29. The stellate cells are widely separated in a mucinous stroma. HE. Bar = 40 µm.
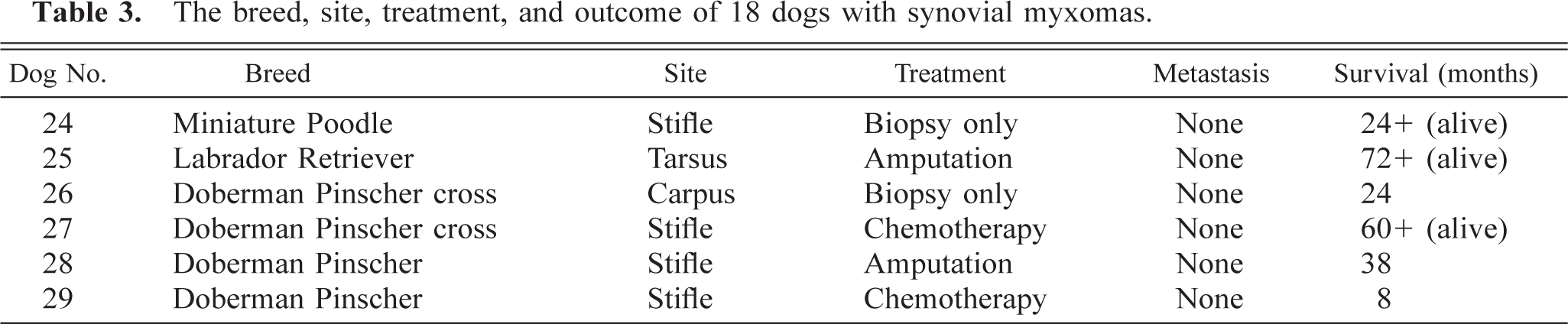
The site, breed, treatment, metastatic behavior, and survival of the dogs with synovial myxomas are listed in Table 3. Four (66.7%) of the dogs with synovial myxomas were Dobermans or Doberman crosses. This is significantly more than expected (P < 0.0001). The most common site for the synovial myxoma was the stifle (four of six; 66.7%), with one tumor each in the tarsus and carpus. The average weight of affected dogs was 31.8 kg with a range of 5–46.3 kg. The average age at time of diagnosis was 9.2 years with a range of 6–13 years. No sex predilection was found for synovial myxomas (one castrated male, two intact males, one spayed female, and two intact females).
The breed, site, treatment, and outcome of 18 dogs with synovial myxomas.
The average time from diagnosis to death or euthanasia of the dogs with synovial myxomas was 30.7 months with a range of 8–72 months. Three of the dogs are still alive 2, 5, and 6 years after diagnosis. The dog with the shortest survival (8 months) was a Doberman Pinscher (dog No. 29) that was treated with five cycles doxorubicin and died at home at the age of 11 years of unknown causes. No evidence was found that the synovial myxoma was the cause of death or euthanasia in any of these dogs.
Other sarcomas
In two dogs (dog Nos. 30 and 31), the neoplastic cells stained only with antibodies to vimentin and actin. These tumors were made up of large polygonal to spindle-shaped cells arranged in a storiform pattern. These were diagnosed as MFH. The remaining four synovial tumors stained only with antibodies to vimentin. One dog (dog No. 32) had a distinct herringbone pattern of spindle cells that was consistent with fibrosarcoma. Another dog (dog No. 33) had areas of myxomatous and chondromatous differentiation that were consistent with chondrosarcoma. The remaining two sarcomas consisted of small plump spindle cells with scant cytoplasm. These were diagnosed as undifferentiated sarcomas.
The site, breed, treatment, metastatic behavior, and survival of dogs with other sarcomas are listed in Table 4. Four tumors were in the tarsal joint and two were in the stifle. The average weight of affected dogs was 36.7 kg with a range of 24–54 kg. The average age at time of diagnosis was 7.8 years with a range of 4–11 years. Four of the dogs were castrated males, one was an intact male, and one was a spayed female.
The diagnosis, breed, site treatment, and outcome of six dogs with other sarcomas of the synovium.
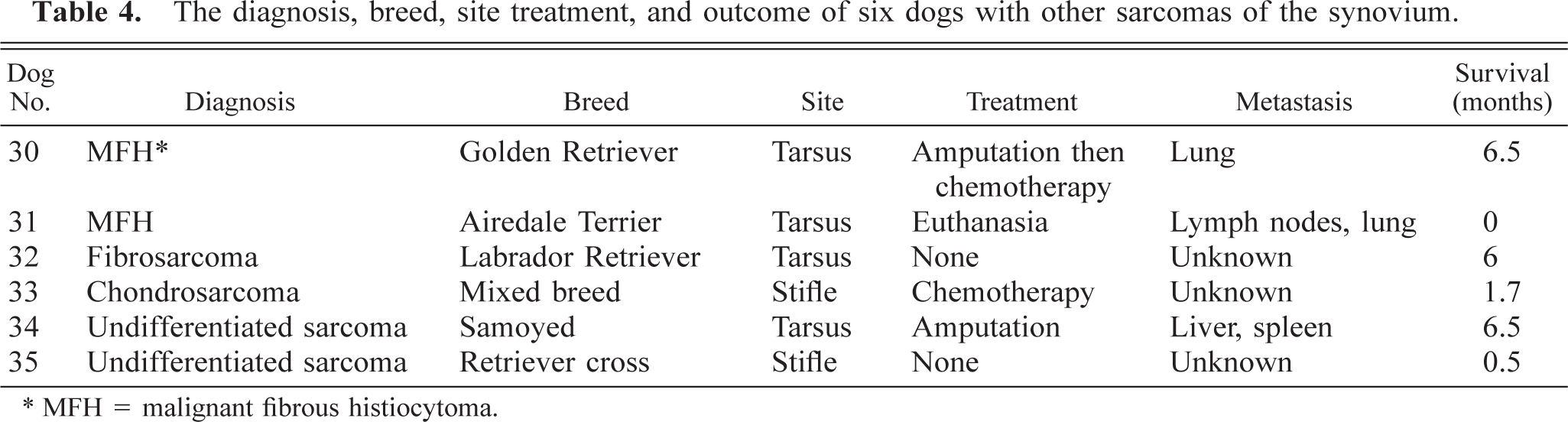
∗ MFH = malignant fibrous histiocytoma.
The average time from diagnosis to death or euthanasia was 3.5 months with a range of 0–6.5 months. Of the three dogs with follow-up information, all three had metastatic disease (both dogs with MFHs and one of the dogs with undifferentiated sarcoma).
Group comparisons
The low-magnification patterns of the SCS, histiocytic sarcoma, malignant fibrous histiocytoma, and other sarcomas were remarkably similar. They all formed multiple variably sized lobules of spindle cells covered by intact or ulcerated synovial membrane. Only the synovial myxoma had a distinctive low-magnification morphology consisting of myxomatous islands. They all showed a similar degree of invasion into surrounding soft tissue and bone.
A significant difference (P < 0.0015) was found in average survival times between the dogs with histiocytic sarcoma (5.3 months) or undifferentiated sarcoma (3.5 months) and those with SCS (31.8 months) or synovial myxoma (30.7 months). The Kaplan-Meier survival data of the three histologic groups is shown in Fig. 7. The group with undifferentiated sarcomas was not included in the graph because undifferentiated sarcomas represent a variety of tumor types.
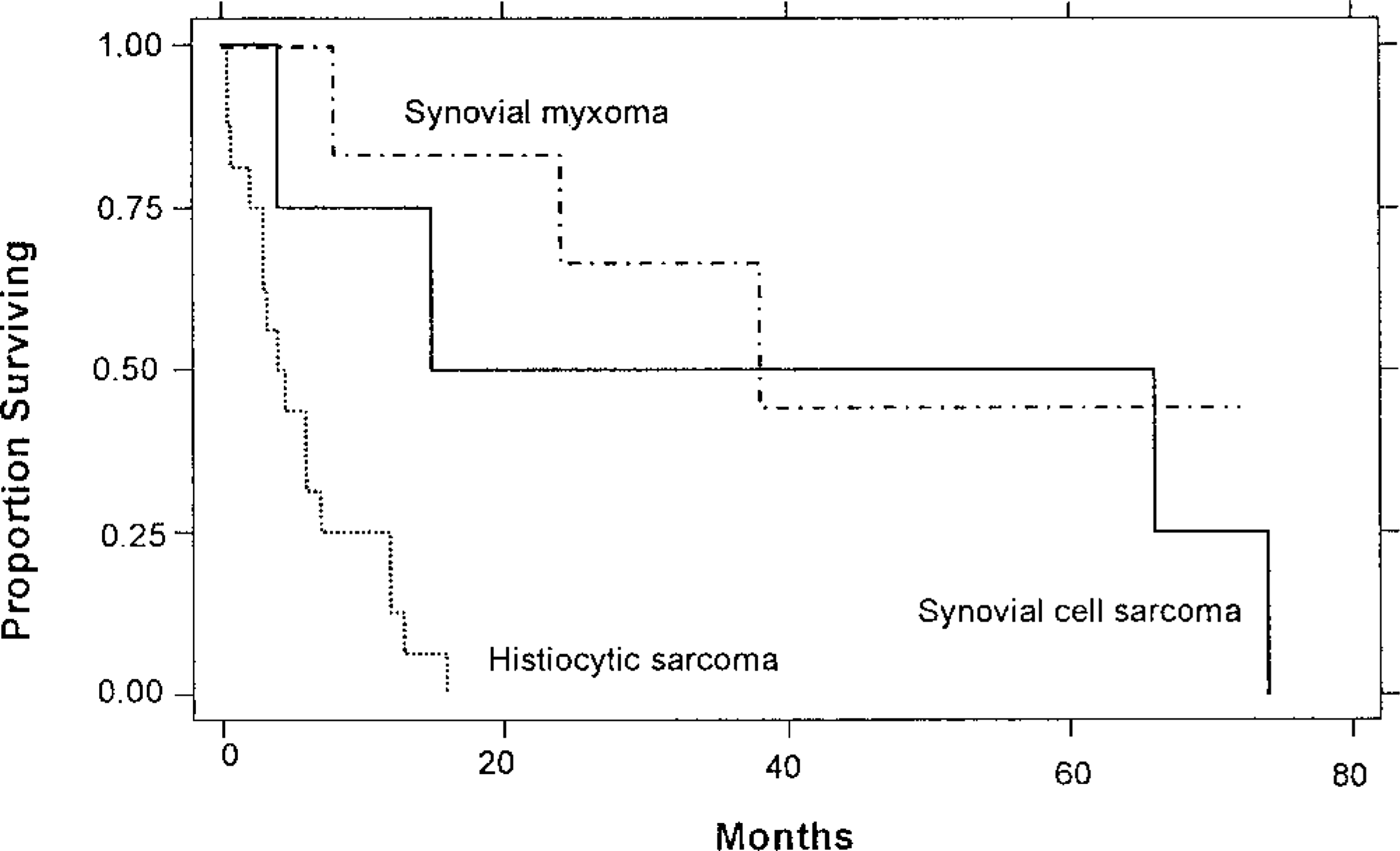
Kaplan-Meier survival graph of dogs with synovial myxoma, synovial cell sarcoma, or histiocytic sarcoma of the synovium.
The average age at time of diagnosis for Rottweilers (6.8 years) was significantly lower (P < 0.05) compared to the average age at time of diagnosis for all other breeds (8.7 years). However, no significant difference was found in age, weight, or sex distribution among the histologic groups.
A significant difference (P < 0.0005) also was found in the percentage of dogs with metastases between dogs with histiocytic sarcoma (91%) or undifferentiated sarcoma (100%) and those with SCS (25%) or synovial myxoma (0%).
Discussion
Only five (14.3%) of the synovial tumors in this study were identified as SCSs. Although the two undifferentiated sarcomas could represent monophasic SCSs, they did not contain any cytokeratin-positive cells. Because we could recognize no distinguishing histopathologic features of the SCS except cytokeratin staining, tumors negative for cytokeratin were excluded from the SCS category. Cytokeratin staining is helpful in making the diagnosis of SCS, but the absence of cytokeratin staining does not rule out SCS. This percentage of SCS in this study is similar to that reported in a recent review of canine joint tumors where only 8 of 30 (26.7%) were identified as SCSs. 27
The prognosis of dogs with SCS was variable. Two dogs treated only with amputation lived for more than 5 years, whereas another dog treated with chemotherapy and amputation was euthanatized for pulmonary metastatic disease only 4 months after the initial biopsy. The average survival (31.8 months) was significantly better than that of dogs with histiocytic sarcoma or undifferentiated sarcoma of the synovium.
The largest proportion of synovial tumors (51.4%) were identified as histiocytic sarcomas. This is a relatively new classification that includes tumors of macrophage and dendritic cell origin. 17 Primary histiocytic sarcomas are typically diagnosed in the spleen, lymph nodes, lung, bone marrow, skin, subcutis, brain, or periarticular tissue, but metastasis to multiple organs follow rapidly. In contrast, malignant histiocytosis, another tumor of macrophage and dendritic cell origin, presents with simultaneous involvement of multiple organs. Both malignant histiocytosis and histiocytic sarcoma occur most commonly in Bernese Mountain Dogs, Rottweilers, and Golden Retrievers. 11,17,18,21 Unfortunately, most of the surface markers of histiocytic tumors are destroyed by formalin fixation. However, CD18 is a formalin-resistant β2 integrin found on the surface of all bone marrow–derived leukocytes and dendritic cells. 1,17,19,28 Therefore, mononuclear neoplastic cells with abundant cytoplasm that stain with antibodies to CD18, but do not stain with antibodies to lymphocytes, are of histiocytic origin.
The preponderance of Rottweilers with synovial histiocytic sarcomas is an interesting finding. Although the Rottweiler is a common breed and is predisposed to this type of tumor, so is the Golden Retriever, 11,17 which was only represented by one dog. Many of the Rottweilers in this study had a history of cranial cruciate ligament rupture, which has been associated with an ongoing chronic synovitis, even after surgical repair. 7 Increased numbers of dendritic cells have been detected in the inflamed synovium after cranial cruciate ligament rupture. 10 We hypothesize that the overrepresentation of Rottweilers in this study is due to their predisposition to both cranial cruciate ligament rupture 5,26 and histiocytic sarcoma, 11,17 and that these neoplasms arise from the dendritic cells found within the inflamed synovium associated with degenerative joint disease.
The prognosis of dogs with synovial histiocytic sarcoma was very poor. All of the dogs for which follow-up information was available died or were euthanatized as a direct result of their primary or metastatic disease. The average survival was only 5.3 months after diagnosis. The average age at the time of diagnosis was significantly younger for Rottweilers (6.8 years) than for other dogs in this study (8.7 years). In another study, Rottweilers were found to have a younger age at death, regardless of cause, than three other large breeds. 3
Six (17.1%) of the synovial tumors were myxomas. Three cases of synovial myxoma were described previously by Pool. 20 Two of those were in Doberman Pinschers and synovectomy was considered to be curative. 20 Nevertheless, all of the myxomas in this study had been diagnosed previously as malignancies, five as myxosarcomas and one as a mucinous synovial cell sarcoma. It is important to recognize that despite their aggressive radiographic and histopathologic appearance, myxosarcomas have a good prognosis. Two dogs in this study (dog Nos. 24 and 26) had no evidence of recurrence or metastasis 2 years after diagnosis; these dogs received no treatment except nonsteroidal anti-inflammatory drugs. However, a recent case report of a synovial myxoma demonstrated extension into the surrounding tissue that necessitated amputation. At the time of follow-up, 18 months after surgery, that dog remained disease-free. 8
Of the remaining synovial tumors, one was a fibrosarcoma, one was a chondrosarcoma, two were undifferentiated sarcomas, and two were diagnosed as MFH by cell morphology and actin staining. Human MFH is a morphologically heterogenous group of sarcomas; however, in dogs, MFHs are typically composed of a mixture of spindle cells in a storiform pattern, large pleomorphic round cells, and a variable nonneoplastic inflammatory cell component. 9 Both canine and human MFHs stain most often with smooth muscle actin. 9,16 Recent molecular analysis has confirmed a close relationship between MFHs and leiomyosarcomas. 4 Despite this difference in histogenesis, the histopathologic appearance of the histiocytic sarcomas and MFHs are very similar. 11 In this study, immunohistochemical staining with smooth muscle actin and CD18 was used to differentiate the two. Three of the histiocytic sarcomas had been previously diagnosed as MFHs.
Of the 18 histiocytic sarcomas, 12 had been misdiagnosed. The diagnoses included liposarcoma (three), MFH (three), SCS (three), undifferentiated sarcoma (two), and fibrosarcoma (one). The prominent round vacuoles in the cytoplasm of some of the histiocytic sarcomas are similar to those seen in liposarcomas.
According to the results of this study, the prognosis varies greatly between histologic types of synovial tumors. Therefore, a panel of immunohistochemical stains for synovial tumors should include cytokeratin, CD18, and smooth muscle actin. Synovial cell sarcomas are spindle cell tumors with a small number of cells that are strongly cytokeratin (AE1/AE3, but not CAM 5.2) positive. MFHs stain diffusely positive with smooth muscle actin and have a storiform pattern. Histiocytic sarcomas are more anaplastic with stippled cytoplasmic CD18 staining and may contain inflammatory cells. The inflammatory cells stain positively for CD3 and MAC387, but these antibodies were not useful for tumor identification. Synovial myxomas have a distinct pattern of myxomatous islands. All of the synovial tumors stain uniformly positively with vimentin.
Footnotes
Acknowledgements
We thank Dr. R. J. Orsher of Veterinary Specialty and Emergency Center, Langhorne, Pennsylvania, and Dr. R. M. Fred of Redbank Veterinary Hospital, Redbank, New Jersey, for providing case material and medical records; Mr. J. E. Hayden and Dr. R. M. Walton for assistance with the illustrations; and Drs. K. J. Drobatz and F. S. Shofer for statistical analysis.
