Abstract
A 10-year-old, neutered, male, domestic short-haired cat had numerous, small, firm, round, red, nonpruritic, nonpainful, dermal nodules 5–16 mm in diameter that ruptured within 48 hours of their appearance and subsequently crusted over. The masses were located in all regions of the body. One mass was excised from the dorsal right carpus and examined histologically, and 2 masses from the interscapular region were cultured for bacteria. The excised dermal mass from the carpus effaced normal dermal architecture, pressed tightly against the epidermis, and was composed of tightly packed round to polyhedral cells that extended to the deep margins of the sections. The overlying epidermis was extensively ulcerated and vesiculated with intraepidermal nests of cells identical to those in the dermis. There was marked anisokaryosis in the deeper regions of the mass with numerous multinucleated cells and cells with giant bizarre nuclei. The histological appearance and CD18 immunocytochemical staining of this mass are consistent with a diagnosis of feline progressive epitheliotropic dendritic cell histiocytosis.
History
A firm, ulcerated, dermal mass tightly adherent to the overlying epidermis and measuring 5 × 16 mm was surgically removed from the dorsal right carpus of a 10-year-old, neutered, male, domestic short-haired cat. There was a 2 week history of multiple dermal nodules throughout all regions of the dermis. The nodules varied from 5 to 16 mm in diameter and ruptured within 48 hours of their appearance and then crusted over. They were nonpruritic and nonpainful. Formalin-fixed, hematoxylin and eosin and Giemsa stained 5-μm-thick sections were prepared using standard method. Standard culture methods were used in an attempt to identify pathogenic bacteria within 2 similar lesions from the interscapular area.
Microscopic Findings
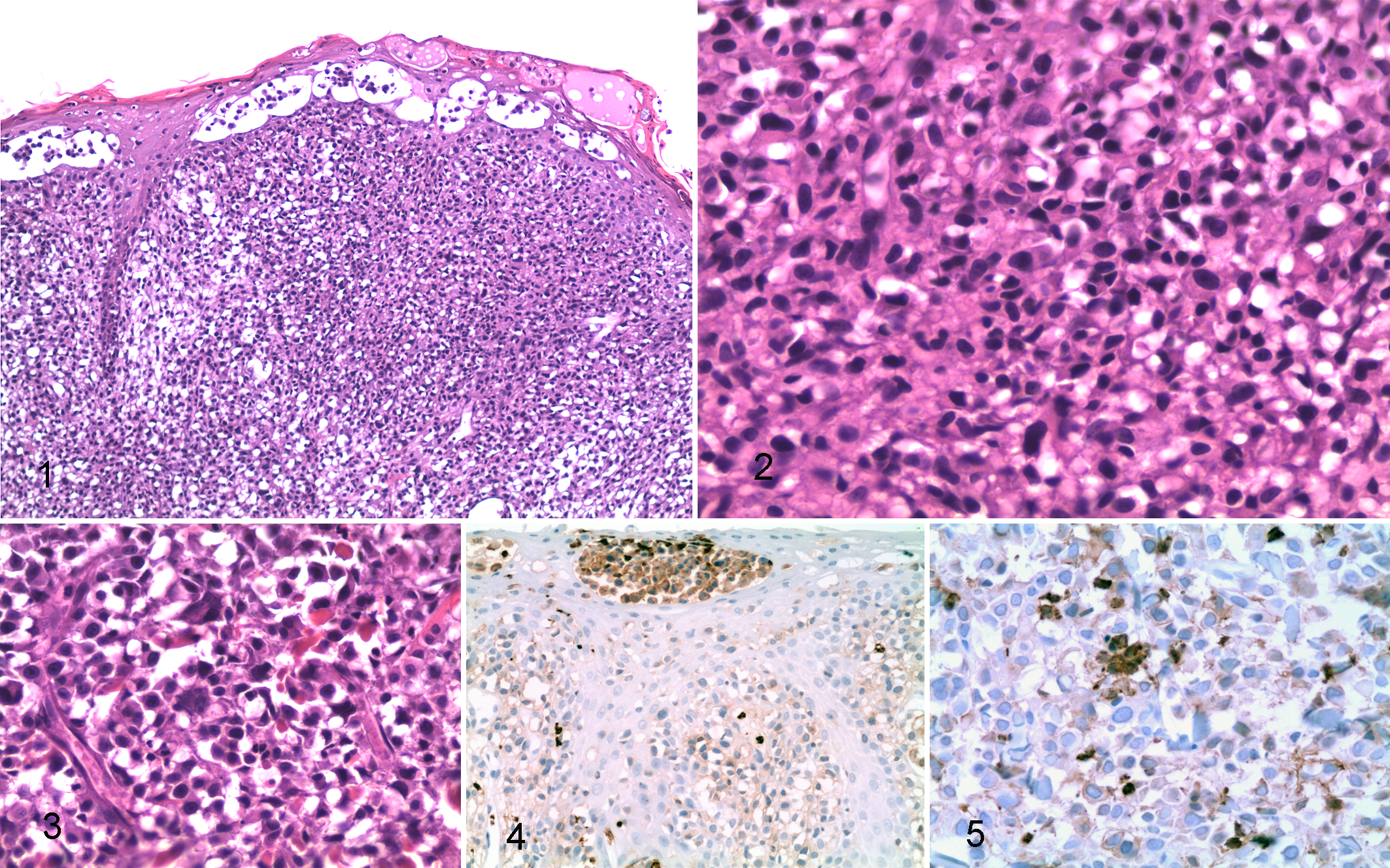
The sections were lined by ulcerated, haired skin containing superficial vesicles/pustules. Pressing tightly against the epidermis, effacing dermal architecture, and extending to the deep margins of the sections was an infiltrative, densely cellular mass organized as loose to pavemented sheets and incomplete lobules (Fig. 1). The cells comprising the mass were round to polyhedral and obliterated adnexal structures. Dense to areolar nests of similar cells were present within the epithelium. The neoplastic cells had distinct cell borders; abundant, pale, eosinophilic cytoplasm; and occasional circumnuclear clearing. Nuclei were centrally located within the cells, round to oval to reniform and hyperchromatic (Fig. 2). Multinucleated cells and cells with giant bizarre nuclei were especially numerous at the deep margins of the sections where acute necrosis was also present (Fig. 3). Small numbers of neutrophils were present in small areas of necrosis, and there was 1 mitotic figure per 10, 400× fields. Giemsa staining for metachromatic cytoplasmic granulation was negative, and no bacteria were isolated by bacterial culture or visualized in HE sections.
Differential Diagnoses
Round cell tumors of feline skin include trichoblastomas, melanomas, mastocytomas, plasmacytomas, Merkel cell tumors, xanthomas, lymphomas, and histiocytomas/histiocytosis. Of these, epidermal invasion may occur in melanomas, epitheliotropic lymphomas, and the epitheliotropic form of feline progressive dendritic cell histiocytosis.
Immunohistochemistry
Unstained, paraffin-embedded tissue was sectioned at 3–5 μm and placed on positively charged glass slides. Immunohistochemical staining was performed using an automated staining device (Dako Autostainer, Carpinteria, CA) with Nova Red (Vector Labs, Burlingame, CA) as the chromogen and a hematoxylin counterstain. The negative control for all tissues and antibodies was the universal negative control provided by Dako. Antibodies tested were CD3, CD18, CD20, and Melan A (Table 1). The tumor cells, including the intraepidermal component and multinucleate forms, were strongly positive for feline CD18 (Figs. 4, 5). Lymphocytes did not bind CD18 stain in the feline lymph node control tissue.
Immunohistochemistry Results.

LN, lymph node. aDako Cytomation, Carpinteria, CA.
bLeukocyte Antigen Biology Laboratory, Davis, CA.
cThermo Fisher Scientific, Fremont, CA.
Diagnosis
The diagnosis was feline progressive epitheliotropic dendritic cell histiocytosis.
Discussion
Feline progressive epitheliotropic dendritic cell histiocytosis is an infrequently reported, nonpruritic, nonpainful, indolent, dermal neoplasm of older cats apparently first described as multiple cutaneous histiocytomas in 2000. 3 The neoplastic dendritic cells are of unknown origin but are not of the Langerhans type. 4 Lesions may be solitary, multiple or coalescent, dermal or epitheliotropic. Although long-term clinical follow-up is limited, it is believed that the neoplasia waxes and wanes without spontaneous resolution but with spread to draining lymph nodes. 1 Surgical removal and anti-inflammatory and antineoplastic therapies are ineffective. 1 The cutaneous lesions may progress to histiocytic sarcoma, which may also affect internal organs. Feline malignant histiocytosis is similar to its canine counterpart and is differentiated from feline progressive histiocytosis by the historical absence of cutaneous lesions. 4 Case reports are insufficient to determine whether there is a breed, age, or sex predisposition. 1
CD18 recognizes a B2 integrin subunit on all leukocytes. In general, macrophages and granulocytes have a 10-fold greater affinity for CD18 than do lymphocytes. Additionally, lymphocytes in formalin-fixed tissue whose processing includes steps for antigen retrieval may be negative for CD18. In the case presented here, neoplastic cells were also negative for CD3 and CD20, greatly reducing the possibility that the neoplasm was of lymphoid origin.
Round cell tumors of feline skin include those composed of basal cells, melanocytes, mast cells, plasma cells, Merkel cells, xanthoma cells, lymphoid cells, and histiocytes. Of these, those with epidermal invasion include melanomas, epitheliotropic lymphomas, and the epitheliotropic form of dendritic cell histiocytosis. Nodular packaging of cells occurs in plasmacytomas, melanomas, and relatively early cases of histiocytosis. In the case presented herein, lymphoma was ruled out by a lack of CD3 and CD20 immunostaining. Melanoma was eliminated by a lack of CD18 and Melan-A reactivity. CD18 is specific for dendritic cells when lymphocytes do not stain, such as in the current case. 5 Lesions of progressive histiocytosis that are older than the one presented here may contain cytoplasmic vacuoles that may appear similar to xanthocytes, which are also positive for CD18. 4 Biopsy of newer lesions may be necessary to separate xanthomas from nonepitheliotropic progressive histiocytosis. 4
Footnotes
Acknowledgements
I thank Kay Fischer and the personnel of the histopathology section of the Veterinary Diagnostic Laboratory for their excellent technical assistance.
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
