Abstract
A 17-year-old, gelded Quarter Horse cross was found to have a large, intra-abdominal mass. Clinical signs included infrequent mild colic, weight loss, and chronic anemia. Surgery revealed a very large, discrete, hemorrhagic, multilobular mass with vascular attachments to the transverse colon, mesocolon, jejunal mesentery, and omentum; the site of origin was the transverse colon. Histologic examination demonstrated dense sheets, fascicles, palisades, and interconnecting streams of neoplastic spindle cells with lesser numbers of admixed multinucleated giant cells. Based on morphology alone, this neoplasm might have been misdiagnosed as a peripheral nerve sheath tumor because many of the morphologic features were suggestive of neural differentiation. Neoplastic cells expressed cluster of differentiation (CD)117 (c-kit), vimentin, desmin, smooth muscle actin, neuron-specific enolase, and S-100 protein and did not express cytokeratin. Based predominantly on the immunohistochemical profile, especially the CD117 positivity, this neoplasm was diagnosed as a gastrointestinal stromal tumor with both myogenic and neurogenic differentiation. The morphology and immunohistochemical profile of this neoplasm were different from published cases of equine gastrointestinal stromal tumors. Unusual aspects included the large size of this neoplasm, the neuroid rather than myxomatous morphology, the presence of multinucleated giant cells, and the expression of desmin.
Historically, nonlymphoid mesenchymal tumors of the gastrointestinal tract were diagnosed based on strictly morphologic features as either leiomyomas or their malignant counterpart, leiomyosarcomas. 2,3,13 Ultrastructural and immunohistochemical investigation of many gastrointestinal tumors morphologically resembling smooth muscle has revealed that they are variably composed of undifferentiated cells as well as differentiated cells with neural and/or smooth muscle features. 3,13,14 Because the histogenesis of these neoplasms remains unclear, the term gastrointestinal stromal tumor (GIST) is often applied. 2,3,6,11,12,14 The majority of human GISTs express cluster of differentiation (CD)117, and a slightly smaller number are also positive for CD34. 6–8,11,12 Because the interstitial cells of Cajal (ICCs), which differentiate into the pacemaker cells of the intestinal tract, have a similar immunophenotype, ICCs are hypothesized by many to be the progenitor cells of GISTs. 2,3,6–8,11 Neoplasms with many analogous features have been reported in nonhuman primates, 1,15 dogs, 4,9,10 horses, 3,6 and most recently in a Fischer 344 rat. 5 The current case report describes an atypical equine GIST with several unique features.
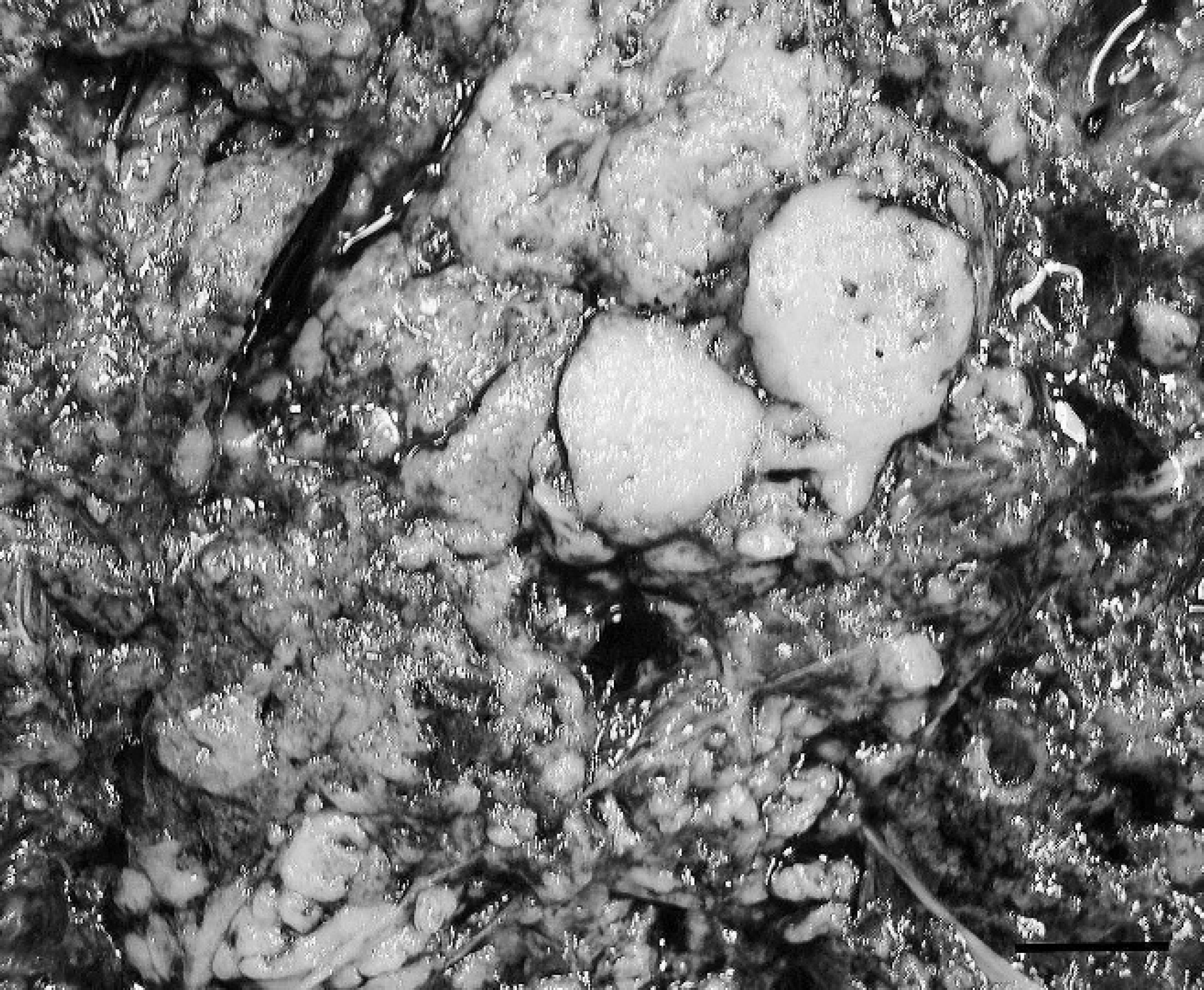
Intra-abdominal mass, gastrointestinal stromal tumor cut section; horse. The parenchyma is mottled and contains many variably sized cribrous areas. Bar = 3.5 cm.
A 17-year-old Quarter Horse cross gelding presented with infrequent mild colic, weight loss, and chronic anemia. Abdominal ultrasound revealed a large intra-abdominal mass, which extended along the ventral body wall from the 7–16th intercostal space, and a mild peritoneal effusion. When the horse returned 5 months later for surgical excision of the mass, ultrasound demonstrated mild peritoneal and pleural effusions and revealed that the mass now occupied the majority of the ventral abdomen. It extended from the xyphoid cartilage to the prepuce and slightly dorsal to the costochondral junctions and was not obviously associated with any internal organs. At surgery, an extremely large, discrete, hemorrhagic, multilobular mass was observed. It contained 3 large, independent vascular attachments to the transverse colon, the base of the mesocolon, and the distal jejunal mesentery, with many, smaller vascular attachments to the omentum. The site of origin of this solitary mass was considered to be the transverse colon because a portion of it had to be resected with the mass. The mass was removed in toto and submitted for gross, histologic, and immunohistochemical examination.
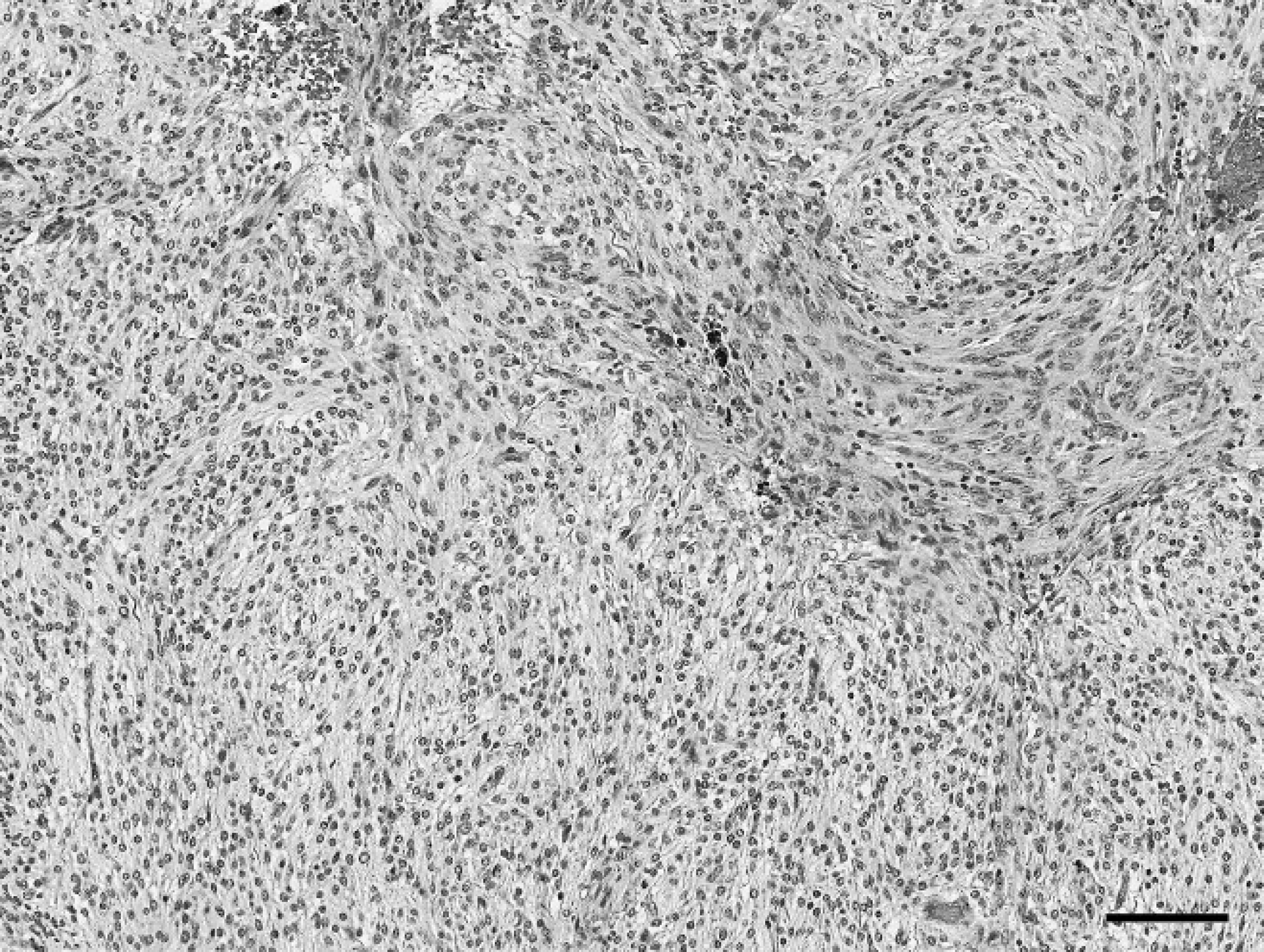
Intra-abdominal mass, gastrointestinal stromal tumor; horse. The highly cellular neoplastic proliferation forms dense sheets, fascicles, and interconnecting streams. Hematoxylin and eosin. Bar = 300 μm.
The mass measured 1 m × 40 cm × 40 cm and weighed 45.5 kg. It was moderately soft, friable, and tan with intervening red cribrous areas. On cut section, it was surrounded by a thin connective tissue capsule, and the parenchyma contained many variably sized, blood-filled cavities (Fig. 1). For histologic examination, multiple sections of the mass were preserved in 10% buffered formalin, processed routinely, and stained with hematoxylin and eosin. Histopathology revealed a highly cellular, well-vascularized, neoplastic spindle-cell proliferation. The neoplastic cells were arranged mainly in sheets but also were in fascicles, palisades, and interconnecting streams (Fig. 2). They were variably and incompletely separated by a scant amount of fibrovascular stroma. The majority of the neoplastic cells were monomorphic, with indistinct cell borders and moderate to abundant amounts of fibrillar to vacuolated eosinophilic cytoplasm. Nuclei were ovoid to elongate with coarsely clumped to cleared chromatin and sometimes had an amphophilic to basophilic nucleolus. Mitotic figures were rare; there was 1 per 10 40× fields. Occasionally, among the neoplastic cells, there were variable numbers (0–3 per 40× field) of large (up to 60 μm at their widest point), polygonal multinucleated giant cells (MNGCs) with abundant amounts of amphophilic cytoplasm and up to 15 bland nuclei (Fig. 3). There was scattered hemorrhage and hemosiderophages as well as small numbers of scattered lymphocytes and plasma cells. Based on morphology, differential diagnoses that were considered included GIST, including the more neuroid variant, which is sometimes termed a gastrointestinal autonomic nerve tumor, 2,6,11 or a benign peripheral nerve sheath tumor (PNST).
Indirect immunohistochemical staining was performed using a commercial-labeled streptavidin-biotin kit a and a commercial highly sensitive 2-step visualization system. a Antibodies to the following antigens were used: vimentin (VIM; V9; monoclonal, a dilution 1:30), S-100 protein (S-100; polyclonal, a 1:800), desmin (DES; monoclonal, a 1:40), smooth muscle actin (SMA; monoclonal, a 1:25), and neuron-specific enolase (NSE; monoclonal, a 1:1,500), CD117 (polyclonal, a 1:50), and cytokeratin (CK; AE1/AE3; monoclonal, b 1:500). Sections used for CD117 immunostaining were pretreated with a citrated (pH 6) buffer c by heating in a microwave oven (boil 7 min, then cool 20 min). Additional pretreatments included trypsin and proteinase K for S-100– and CK-treated sections, respectively. Equine tissue sections known to express the relevant antigen served as positive controls, and negative serum controls also were utilized. There was strong, diffuse, cytoplasmic staining of neoplastic cells for CD117, VIM, DES, SMA, and NSE as well as diffuse, strong, intracytoplasmic and intranuclear staining for S-100. The neoplastic cells did not express CK, and stromal fibroblastic cells of the neoplasm expressed only VIM. The immunohistochemical profile, especially the CD117 reactivity, is most compatible with a GIST, and without it, this neoplasm very likely would have been misdiagnosed as a PNST.
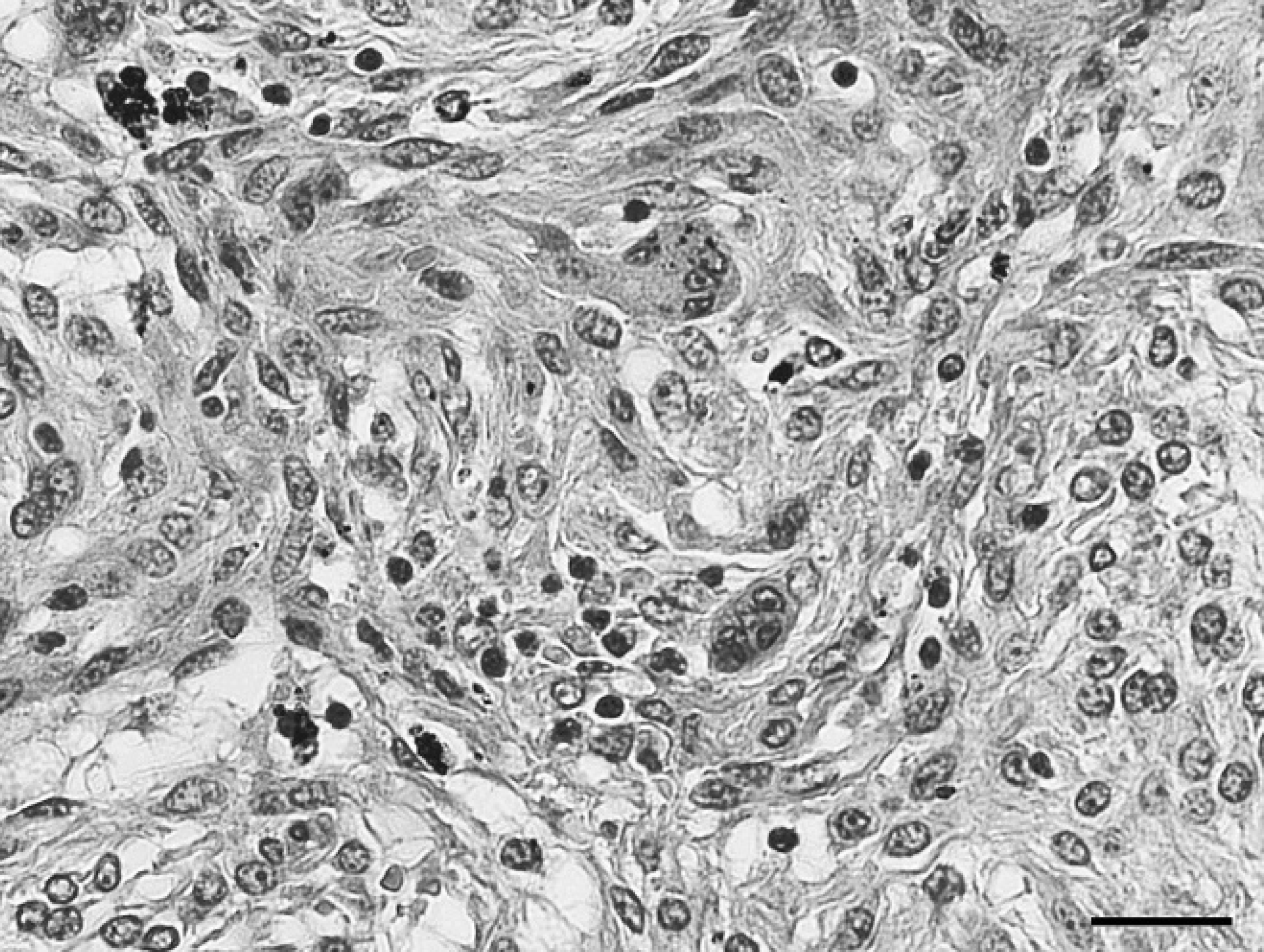
Intra-abdominal mass, gastrointestinal stromal tumor; horse. There are interconnecting streams of spindle cells with intervening multinucleated cells. Hematoxylin and eosin. Bar 5 90 μm.
Akin to the majority of other equine cases, the mass in the current study occurred as a solitary, well-demarcated, moderately soft, tan, cribrous mass 3,6 in a mature horse. 3 The mass was attached to the transverse colon by a vascular peduncle, which is somewhat similar to a previously reported equine case. 3 Although the cecum and ileum 3,6 are the most commonly reported sites for this neoplasm, equine GISTs also have rarely been reported in the large colon, 3 as in the current case. In this horse, as in a small number of other cases, there were associated clinical signs. 3 The clinical signs in this particular animal were likely a result of the extremely large size of the intraabdominal mass; the mass was large when the horse was initially examined, and its size increased significantly during the interval between detection and surgery. Most importantly, despite its large size and prolonged time course, this neoplasm did not metastasize as has been reported in many other species, including humans, 8,12 nonhuman primates, 1 dogs, 4 and the single reported case in a rat. 5 This provides further evidence supporting the benign nature of this neoplasm in horses. 3,6
The tumor was much larger than the largest previously reported equine GIST, which at its widest point measured only 13 cm in diameter. 3 Microscopically, the neoplasm was composed of often densely packed spindle cells arranged in a more neuroidal rather than myxomatous pattern as is most commonly described. 3 It also contained admixed MNGCs, which have infrequently been reported in human GISTs 8,12,14 and canine GISTs. 9,10 In human GISTs, both tumor giant cells 12,14 as well as reactive osteoclast-like giant cells 8,12,14 have been described. Because of the bland morphology of the MNGCs in the reported neoplasm, the authors suspected that the MNGCs were reactive rather than neoplastic.
In the present case, the neoplastic cells expressed CD117 and various mesenchymal markers, including VIM, as well as several more or less specific myogenic (DES, SMA) and neurogenic (S-100, NSE) markers. In previous reports of equine GISTs that were subjected to immunohistochemistry, the CD117 and vimentin markers were consistently positive; NSE, S100a, and SMA were variably reactive; and in no previous case was positive DES staining reported. 3,6 The reported neoplasm was unusual in that it stained with desmin. Coexpression of both myoid and neural markers has been seen in human 14 and canine 10 cases as well as a few other previously reported equine GISTs. 3
In conclusion, based on morphologic features and immunohistochemical staining, the neoplasm in the current case was diagnosed as an atypical, benign, equine GIST with both myogenic and neurogenic differentiation as well as MNGCs. The presence of MNGCs and this neuroidal pattern have not previously been documented in equine GISTs and, therefore, broaden the spectrum of morphologic features associated with this neoplasm in horses. Although the morphology of this neoplasm is similar to a PNST, the immunohistochemical profile is distinctive because these neoplasms are comprised of different cell types. The origin of both the MNGCs and the neoplasm itself are unclear, as is the relationship of this neoplasm with the ICC. Further immunohistochemical investigation might help clarify if the MNGCs are in fact reactive, as is suspected. As for the origin of GISTs and their relationship to the ICC, investigation in the human medical field is ongoing and still controversial.
Acknowledgements. The authors are grateful to Drs. Pamela Wilkins and John C. Higgins-Biddle for critical review of the manuscript and to Mrs. Jaqueline Ferracone for assistance with immunohistochemistry.
Footnotes
a.
EnVision™+ System, Dako North America Inc., Carpinteria, CA.
b.
Boehringer Mannheim Biochemica, Mannheim, Germany.
c.
Antigen Retrieval Citra Solution, BioGenex Laboratories Inc., San Ramon, CA.
