Abstract
A 3-year-old female neutered Staffordshire Bull Terrier presented with a mixed germ cell tumor involving the base of the iris and the ciliary body of the right eye. The tumor mass was composed primarily of packeted vacuolated, polygonal (hepatoid) cells and small round cells; epithelial cells lining tubuloacinar structures were a less prominent component. The hepatoid and round cells stained positively for alpha-fetoprotein and cytokeratin. The epithelial cells stained positively for cytokeratin only, and some contained cytoplasmic mucin droplets. The polygonal cells were interpreted as a hepatoid variant of yolk sac tumor, and the epithelial cells were considered a teratomatous component. Trabeculae of bone were observed within the mass and may have been metaplastic or a teratomatous element. Extragonadal germ cell tumors are rare in dogs and have previously been reported only in the suprasellar region. This is the first report of this tumor type in the eye of a nonhuman species.
Germ cells give rise to spermatogonia in the testis and oogonia in the ovary. Gonadal germ cell tumors reported in animals include seminomas, teratomas, embryonal carcinomas, and mixed germ cell–sex cord stromal tumors. 5 Germ cell tumors in dogs most commonly occur in the ovaries and testes; 8 however, small numbers of extragonadal tumors have been reported in the intracranial suprasellar region. 4,10,11 Here, we describe a case of mixed germ cell tumor (yolk sac tumor and teratoma) in the eye of an adult Staffordshire Bull Terrier.
This 3-year-old female neutered Staffordshire Bull Terrier presented with a 4-mm-diameter mass of unknown duration in the lateral aspect of the anterior chamber of the right eye. The globe was removed, and the eye was immersion fixed in 10% neutral buffered formalin, routinely processed, and embedded in paraffin. Five-micrometer sections were mounted on glass slides and stained with hematoxylin and eosin (HE). Other sections were immunohistochemically stained for cytokeratin AE1/AE3, vimentin, and alpha-fetoprotein by the avidin–biotin–peroxidase complex method. Sections were also stained under identical conditions with the normal mouse and rabbit sera as negative controls. Immunostained sections were counterstained with Mayer's haematoxylin.
Microscopically, the base of the iris, the ciliary body and processes, and the anterior choroid were expanded by a poorly demarcated, densely cellular neoplastic mass (Fig. 1). Tumor cells extended along the anterior margin of the iris and formed short papillary projections into the anterior chamber of the eye; they also extended anteriorly from the base of the iris, with obliteration of the filtration angle and extension along the peripheral, posterior aspect of Descemet's membrane. In the posterior chamber, loose clusters of tumor cells coated the pigmented epithelial cell layers of the posterior surface of the iris. Most tumor cells were arranged in packets, with a fine fibrovascular stroma. Multifocally, tumor cells surrounded accumulations of vacuolated eosinophilic extracellular material. The majority of the tumor cells were round or polygonal with round to oval nuclei, finely stippled chromatin, one to three small, central nucleoli, and a moderate amount of pale-staining, delicately vacuolated eosinophilic cytoplasm that often contained a single large, clear vacuole (hepatoid cells; Fig. 2). Elsewhere, tumor cells were round, with a small amount of homogeneous, deeply eosinophilic cytoplasm; small numbers of these round cells contained a small cytoplasmic vacuole (Fig. 2). Nuclei were central and round to ovoid with clumped chromatin and one to three small central, deeply eosinophilic nucleoli. There was moderate anisokaryosis with scattered large nuclei and up to five mitotic figures per 400× field. Some of the mitotic figures were bizarre. A third, less prominent component was comprised of cuboidal to columnar epithelial tumor cells forming short branching cords or lining tubuloacinar structures consistent with a mature teratoma (Fig. 3). These tumor cells had a larger amount of more brightly eosinophilic, granular to vacuolated cytoplasm and basal or central nuclei with sparse, often marginated chromatin and one or two prominent central, deeply eosinophilic nucleoli; some of the cells contained apical mucus and had flattened basal nuclei, and there was focal squamous differentiation. Bony trabeculae with appositional bone formation were found within the tumor mass in the base of the iris, filtration angle, and peripheral anterior chamber (Fig. 1). The hepatoid and round cells showed moderate to strong cytoplasmic staining for alpha-fetoprotein (Fig. 3) and moderate positive staining for cytokeratin (Fig. 4). Epithelial tumor cells were strongly positive for cytokeratin (Fig. 4).
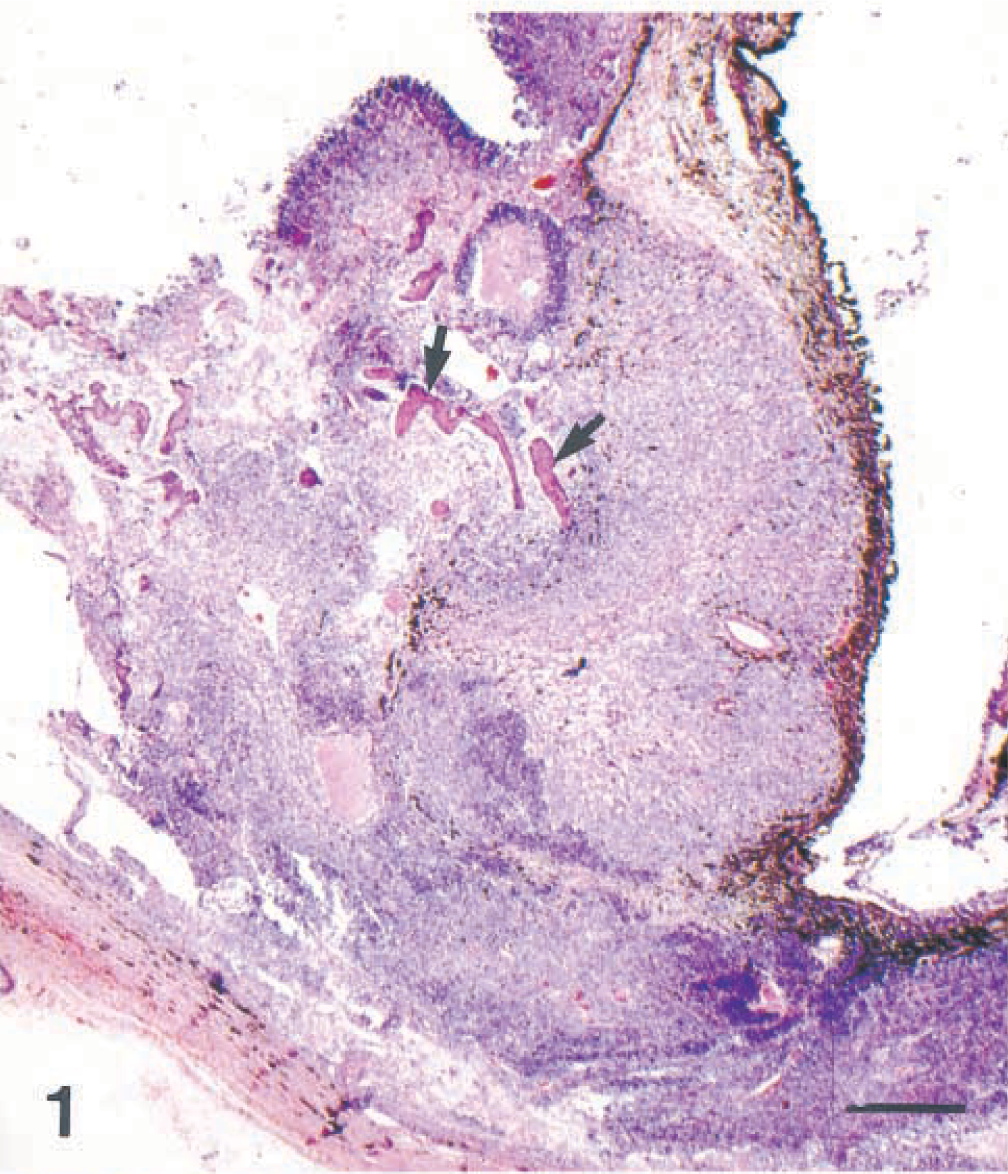
Eye; dog. Mixed germ cell tumor involving the iris and ciliary body. Bone trabeculae (arrows) are present. HE. Bar = 500 µm.
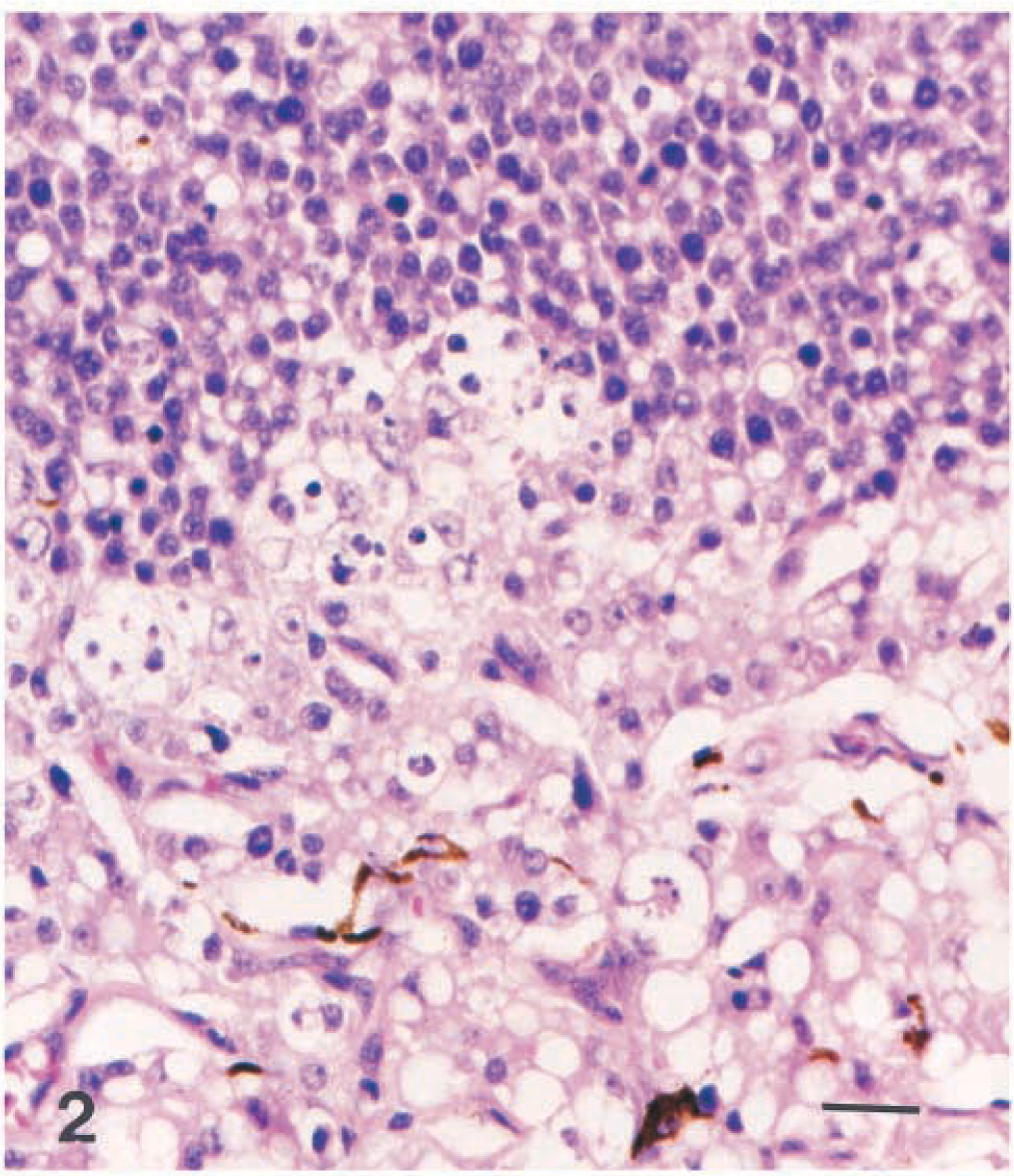
Eye; dog. Mixed germ cell tumor with polygonal cells that often contain a single large vacuole (hepatoid) at the bottom and small round cells that occasionally contain smaller vacuoles at the top. HE. Bar = 25 µm.
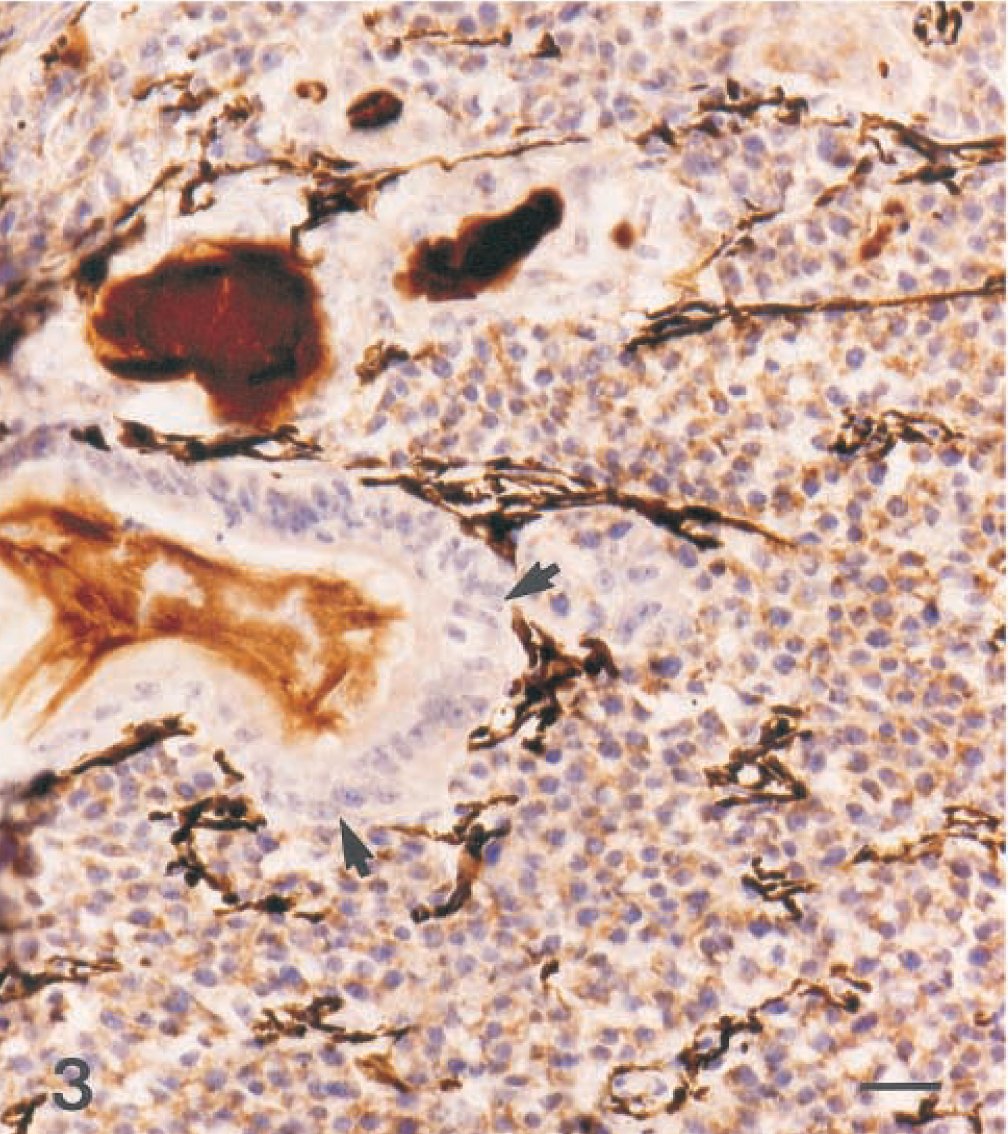
Eye; dog. Mixed germ cell tumor. Neoplastic cells are positive for alpha-fetoprotein, with the exception of the epithelial component (arrows). Hematoxylin and avidin–biotin immunoperoxidase. Bar = 25 µm.
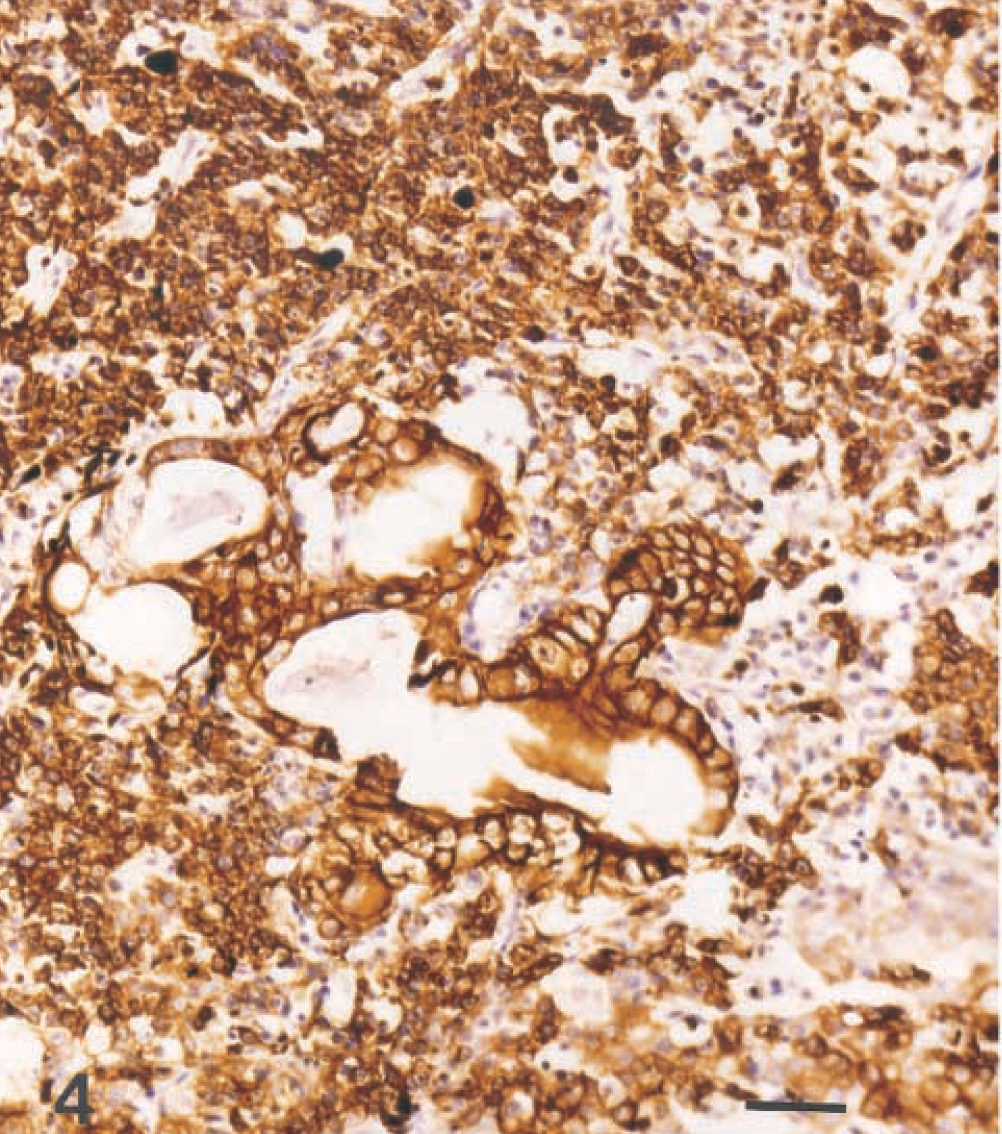
Eye; dog. Mixed germ cell tumor. The epithelial and hepatoid components stain strongly for cytokeratin, but the round cells are less strongly positive. Hematoxylin and avidin–biotin immunoperoxidase. Bar = 50 µm.
Recent studies on mouse embryos indicate that germ cells originate in the epiblast, which is the upper cell layer of the blastocyst and contributes to formation of the ectodermal, mesodermal, and endodermal layers. 3 The germ cells migrate to the caudal yolk sac endoderm and adjacent splanchnic mesoderm of the embryo and reach the gonadal ridge on the medial side of the mesonephros via the dorsal mesentery of the hindgut. 9 Extragonadal germ cell tumors have been proposed to arise by neoplastic transformation of cells that have failed to reach the gonadal ridge by aberrant migration, failure of initiation of migration, or entrapment by and migration with mesodermal cells to the developing neural axis during the formation of the primitive streak. 3 Many normal embryos contain extragonadal germ cells, and in chick embryos the optic cup is one of the sites in which they have been observed. 7 Neoplastic transformation of these cells is rare, however, and it has been suggested that these cells do not normally persist.
Most extragonadal germ cell tumors in humans arise intracranially in midline diencephalic structures, i.e., the pineal region or the suprasellar–hypothalamohypophyseal axis. 3 They have also been reported in the suprasellar region in a small number of young adult dogs. 4,10,11 There are a few reports of retrobulbar germ cell tumors in children 6 and one report of an intraocular yolk sac tumor in a 4-year-old girl. 2 In this dog, the germ cell tumor involved the ciliary body and iris. The stroma of these structures originates from the midbrain neural crest, and the epithelium originates from the anterior aspect of the neuroectodermal optic cup. 1
Teratomas are tumors that arise from totipotential germ cells that have undergone somatic differentiation, 5 whereas seminomas and their ovarian equivalents, dysgerminomas, arise from germ cells that have not undergone differentiation. Other germ cell tumors arise from cells that have undergone only early stages of differentiation, and in humans these tumors include yolk sac tumors (endodermal sinus tumors), embryonal carcinomas, choriocarcinomas, and mixed forms. 5 Such tumors in animals are rare. Suprasellar germ cell tumors described in dogs are comprised of small round cells interpreted as seminomatous or germinomatous cells, vacuolated hepatoid cells, and glandular structures similar to this ocular tumor; this appearance was previously proposed to be unique to the canine suprasellar germ cell tumors. 11 Alpha-fetoprotein is a positive marker for germ cell tumors in humans and dogs, 11 and is produced by yolk sac tumors, enteric elements of teratomas, and some embryonal carcinomas focally. Glandular cells in this case were cytokeratin positive, indicating epithelial differentiation.
Bony trabeculae in this tumor mass may have been metaplastic, but it is more likely to have been another manifestation of teratomatous tissue, as previously proposed 10 for similar trabeculae in a suprasellar mixed germ cell tumor. Metaplastic bone in the eye has been associated with proliferative retinal lesions in humans. 12
This is the first report of an intraocular mixed germ cell tumor in a nonhuman species. The histologic appearance of this mass was similar to that of previously described suprasellar germ cell tumors in dogs.
Footnotes
Acknowledgements
We thank Ms. R.A.V. Ferris, Mr. K. Vrtacnik, and Mr. D. Landry for photographic assistance.
