Abstract
A 9-y-5-mo-old, spayed female, mixed-breed dog with buphthalmia and elevated intraocular pressure in the left eye, consistent with glaucoma, was evaluated. Black-pigmented, slightly elevated tissue with irregular margins was noted on the dorsolateral aspect of the left globe. Ultrasonography detected a mass, later identified as lacrimal gland, adjacent to the globe and the thickened uvea. The surgically removed lacrimal gland was effaced by dense sheets of melanin-laden cells. Within the enucleated globe, numerous melanin-laden cells infiltrated and expanded the rostral two-thirds thickness of the cornea, the entire anterior uvea (iris and ciliary body), and a rostral portion of the choroid. Melanin-laden cells in the left lacrimal gland and globe showed no nuclear atypia or mitotic figures, and reacted to anti-S100 and anti–melan A antibodies by immunohistochemistry. Our final diagnosis was concurrent lacrimal gland melanocytoma and ocular melanocytosis. The trabecular meshwork of the eye was obliterated by melanin-laden cells, which was the likely cause of glaucoma in this patient. To our knowledge, melanocytoma affecting the lacrimal gland has not been reported previously in a non-human mammalian species. Veterinary clinicians are encouraged to include melanocytoma in the differential list when examining an enlarged lacrimal gland.
Melanocytoma is a benign neoplastic proliferation of melanocytes. Most melanocytomas of animals affect the skin and globe.5,8 Although intra-ocular and epibulbar melanocytomas have been described in dogs,4,5 such tumors affecting the lacrimal gland have not been reported to our knowledge. In humans, there is a single reported case of lacrimal gland melanoma. 15 We describe here the histopathologic features of concurrent lacrimal gland melanocytoma and ocular melanocytosis in a dog.
A 9-y-5-mo-old, spayed female, 13.5 kg, mixed-breed dog was evaluated because of glaucoma of the left eye, which did not respond to timolol maleate (Timolol XE; Nitto Medic) prescribed by the receiving veterinarian. Ophthalmologic examination by the referring veterinarian (S. Nishimura) revealed buphthalmia and markedly elevated intraocular pressure (IOP: 40 mm Hg, RI: 10–20 mm Hg) in the left eye; the IOP of the right eye was 19 mm Hg. The findings reported hereafter were confined to the left eye. Corneal edema and anterior chamber aqueous flare were present. We detected black-pigmented, slightly elevated tissue with irregular margins spreading on the dorsolateral aspect of the globe and blurring the limbus (Fig. 1). Ocular ultrasonography detected a mass adjacent to the globe and the thickened uvea (Fig. 2). Thoracic radiography did not reveal any tumor metastasis. No abnormality was detected by radiographic and ultrasonographic examinations of the liver, although by ultrasonography the gallbladder had scant viscous contents adhering to the inner surface of its wall.
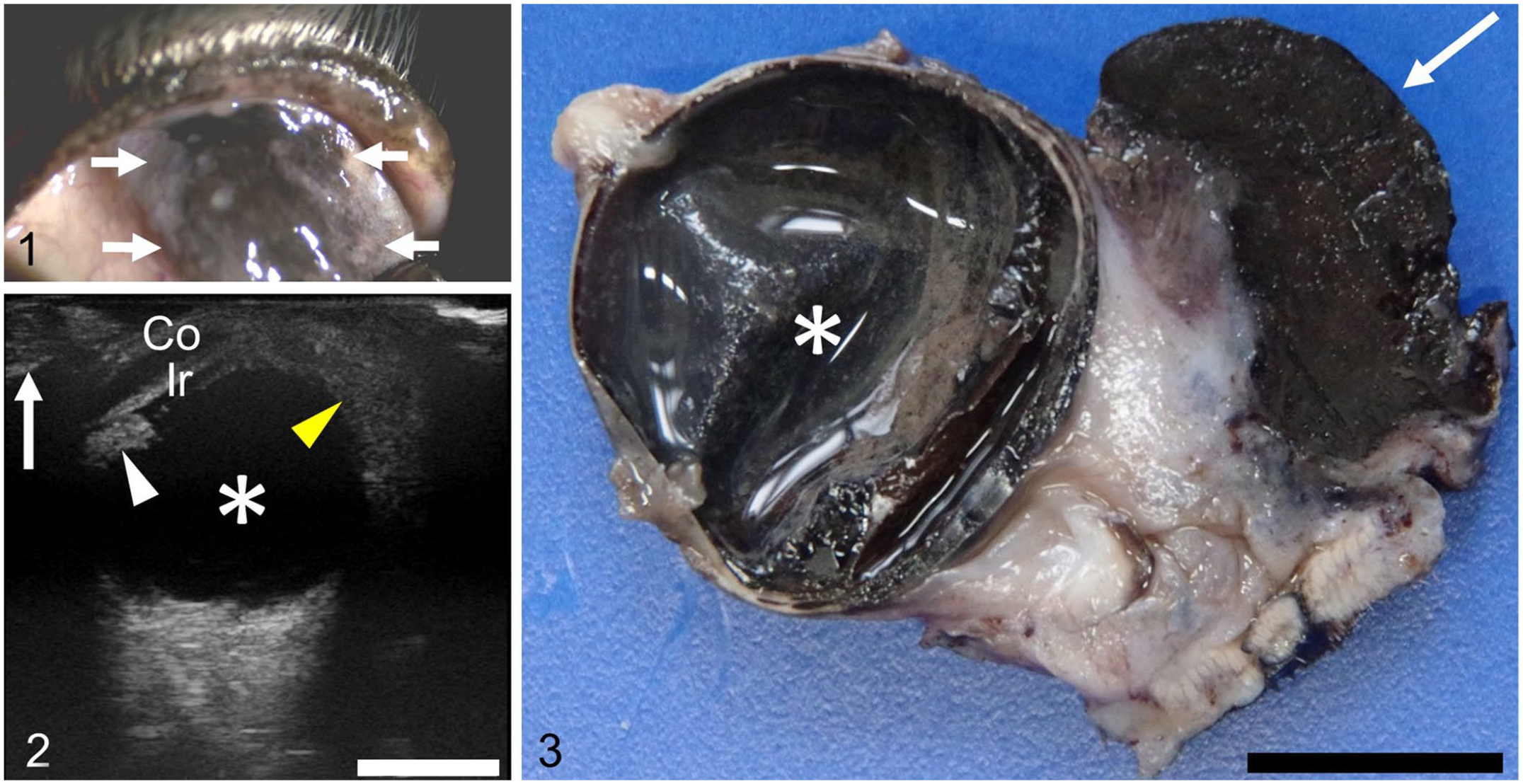
Concurrent lacrimal gland melanocytoma and ocular melanocytosis in a dog.
Difluprednate (Sterop; Senju Pharmaceutical) did not improve the ophthalmologic problem. Surgical removal of the mass and glaucomatous globe was performed after informed consent had been obtained from the dog’s owner. Presurgical complete blood count and blood chemistry revealed slight leukocytosis (leukocytes 20 × 109/L, RI: 6–17 × 109/L); elevated activities of alanine aminotransferase (3.17 μkat/L, RI: 0.28–1.30 μkat/L), aspartate aminotransferase (1.24 μkat/L, RI: 0.28–0.73 μkat/L), and alkaline phosphatase (5.54 μkat/L, RI: 0.78–4.24 μkat/L); slightly elevated total cholesterol (9.51 mmol/L, RI: 2.98–8.73 mmol/L); slightly elevated glucose (7.88 mmol/L, RI: 4.16–7.10 mmol/L); and significantly elevated total bile acid (45.6 μmol/L, RI: 0–7.9 μmol/L) concentration. Platelet count was decreased mildly (158 × 109/L, RI: 200–500 × 109/L). Cytologic evaluation of the regional lymph node via fine-needle aspiration was not performed. Surgery and post-anesthetic recovery were uneventful. The excised left globe and a 2.8 × 1.0 × 1.0-cm mass were fixed in 10% neutral-buffered formalin for 2 d, processed routinely, and 4-µm thick sections were prepared and stained with H&E for histologic examination by an American College of Veterinary Pathologists/Japanese College of Veterinary Pathologists board-certified veterinary anatomic pathologist (I. Mitsui).
The mass and the left globe lacked macroscopically or microscopically evident continuity (Figs. 3, 4). The mass consisted of dense sheets of melanin-laden cells with few remaining lacrimal glands (Fig. 5). On a bleached specimen, melanin-laden cells were pleomorphic, including round, epithelioid, and spindle shapes (Fig. 6). These cells had distinct cell boundaries, abundant cytoplasm, slightly anisokaryotic, round-to-ovoid euchromatic nuclei, and indistinct nucleoli (Fig. 6). No mitotic figures were observed in 10 hpfs (2.37 mm2). Vascular invasion by these cells was not observed.
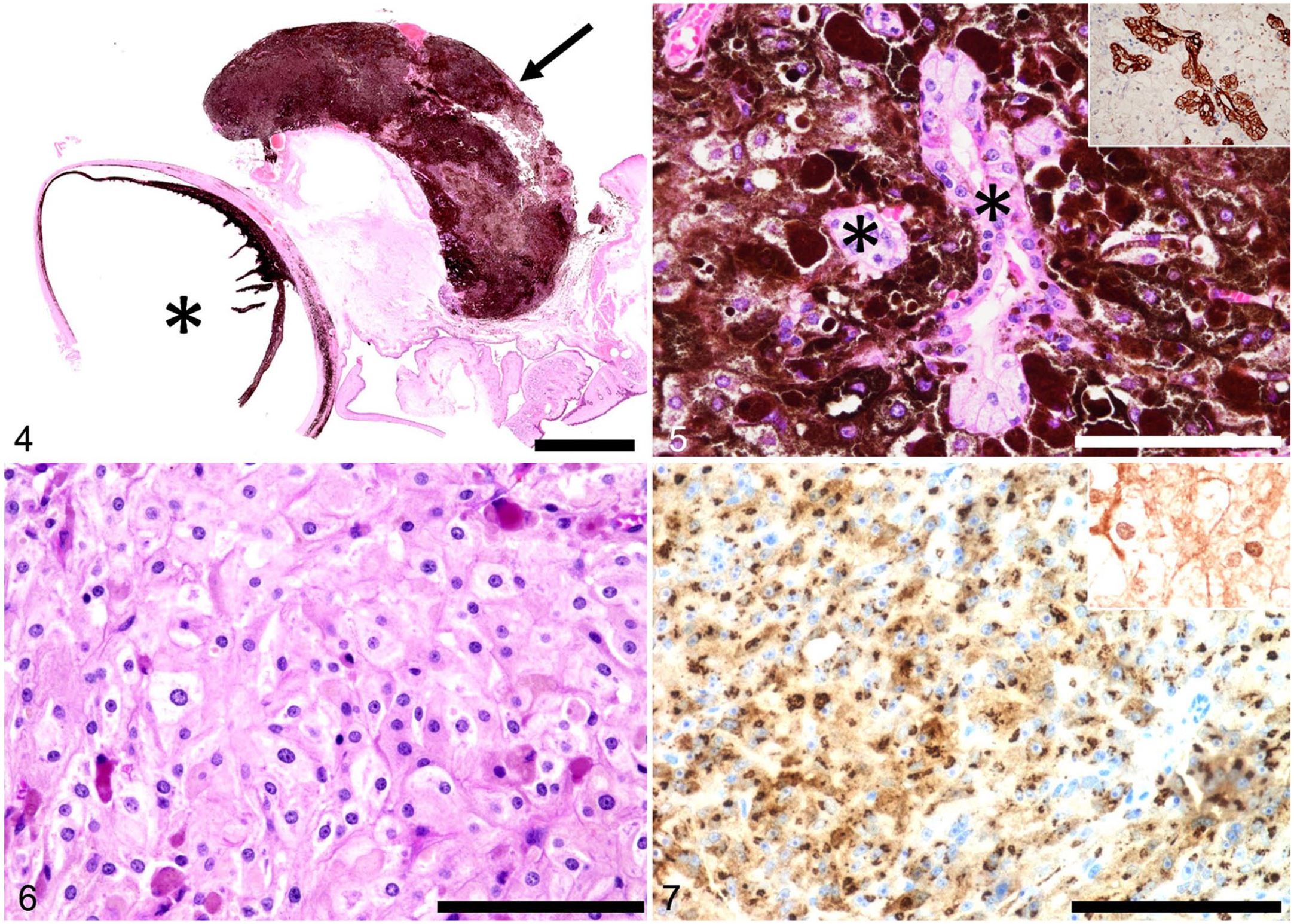
Concurrent lacrimal gland melanocytoma and ocular melanocytosis in a dog.
Immunohistochemistry (IHC) was performed using 4-μm thick formalin-fixed, paraffin-embedded tissues (Table 1). Lacrimal glandular epithelial cells were positive for cytokeratin AE1/AE3 (Fig. 5). Neoplastic melanin-laden cells in the lacrimal gland (Fig. 7) and globe were positive for melan A and S100 in bleached specimens. IHC results supported a final diagnosis of concurrent lacrimal gland melanocytoma and ocular melanocytosis.
Primary antibodies used for immunohistochemistry in a canine case of lacrimal gland melanocytoma and ocular melanocytosis.
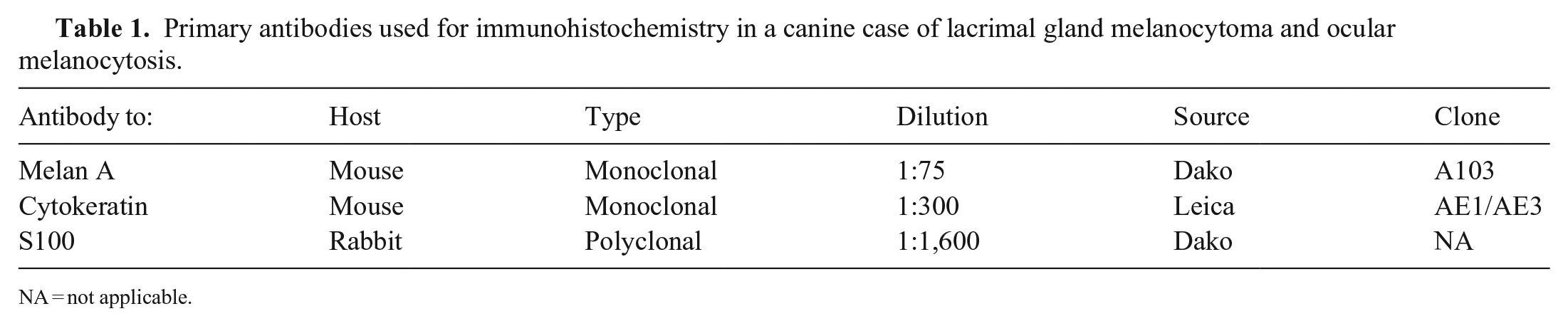
NA = not applicable.
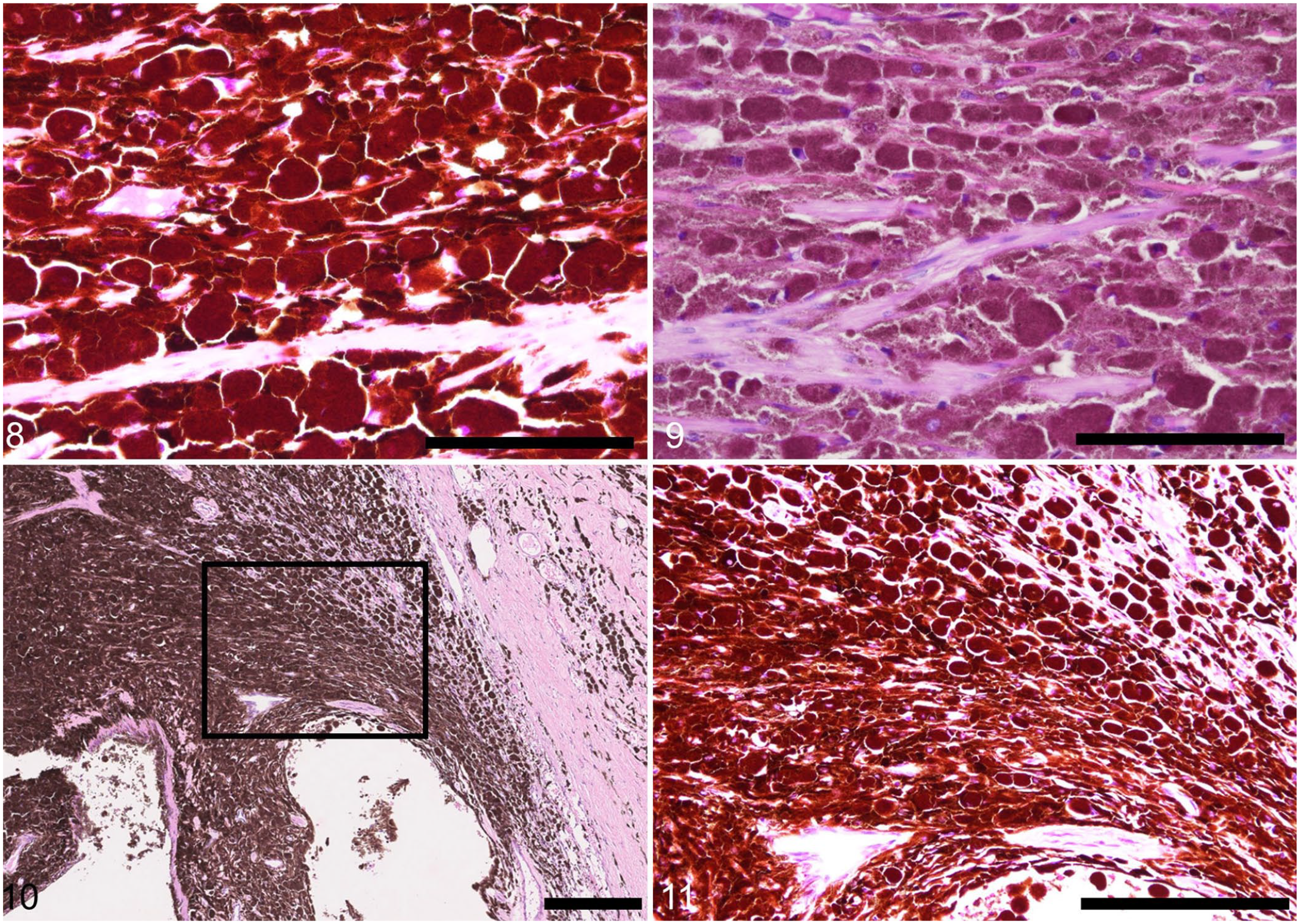
Within the globe, numerous melanin-laden cells infiltrated and expanded the rostral two-thirds thickness of the cornea, the entire anterior uvea (iris and ciliary body), and a rostral portion of the choroid. Most cells were round and contained more abundant cytoplasmic melanin pigments than those in the lacrimal gland (Figs. 8, 9). The trabecular meshwork was obliterated by these cells (Figs. 10, 11), which is consistent with the clinically diagnosed glaucoma in this dog. The retina, which was otherwise unremarkable, was detached from the retinal pigment epithelium (RPE). Given that no hypertrophy of the RPE cells was identified, the retinal detachment was judged an artifact. The sclera, lens, and palpebrae were histologically unremarkable. The tumor-free margins around the neoplastic tissue were <30 µm for the lacrimal gland lesion and ~70 µm for the ocular lesion.
Concurrent lacrimal gland melanocytoma and ocular melanocytosis in a dog.
In a telephone follow-up 7 mo after hospital discharge, the dog had no complications according to the owner. The dog’s owner did not wish to pursue a liver biopsy, and we could not determine the cause of the abnormal blood chemistry liver values.
Melanocytoma is a benign neoplastic proliferation of melanocytes. It is a common skin neoplasm in domestic animal species, including dogs, horses, and pigs. 8 In dogs, the haired skin of the eyelid is a common site for this neoplasm. 8 Intra-ocular tumors of melanocyte origin are also common in dogs; melanocytoma of the anterior uveal tract is the most common. 4 According to some literature, the nomenclature describing a proliferation of bland melanin-laden cells in the canine uvea as either melanocytoma or melanocytosis is acceptable given that there is no solid scientific support for one diagnosis versus the other. 16 In other literature, although both melanocytoma and melanocytosis or melanosis of canine globes are regarded as benign neoplastic proliferation, these lesions can be distinguished by detailed histologic examination.2,5,9 Namely, although melanocytomas appear as a nodule or mass and have a mixed population of round, ovoid, and fusiform cells, melanocytosis or melanosis does not form a large mass and lacks a fusiform cell component. 2 Invasion by pigmented cells of the cornea, sclera, or epibulbar or peribulbar tissue can be detected in both of these disease categories; 5 hence, corneal invasion by melanin-laden cells in our report does not support a diagnosis of melanocytoma.
The unusual topography of the melanocytoma described here is distinct from past reports of melanocytomas. Dogs have 2 lacrimal glands proximate to each globe, namely, the lacrimal gland in the dorsolateral portion of the globe and the accessory lacrimal gland embedded in the third eyelid (colloquially known as nictitating gland or third eyelid gland). Based on its anatomic location (a mass dorsolateral to the left globe), we believe that the lacrimal gland melanocytoma of the dog in our report arose within and effaced the non–third-eyelid lacrimal gland. We found no previous reports in dogs or humans of melanocytic tumors affecting the lacrimal gland regardless of malignancy (benign vs. malignant), location (orbital vs. palpebral or third eyelid), or whether primary or metastatic, except for a single case of primary malignant melanoma of the palpebral lacrimal gland in a 50-y-old woman. 15 In that human case, the left lacrimal gland was swollen without infiltration of the adjacent eyelid, conjunctiva, orbit, or globe. 15 The lacrimal gland was replaced by tumor cells that were morphologically consistent with malignant melanoma and positive for HMB-45 and S100 using IHC. 15 The tumor in the human patient was successfully treated by a combination of surgery and adjuvant radiotherapy. 15 Nevus of Ota, which is oculodermal melanocytosis in the area innervated by the ophthalmic and maxillary branches of the trigeminal nerve, was ruled out based on lack of relevant gross findings.7,11
The cell of origin of the lacrimal gland melanocytoma in the dog in our report is unknown. The lacrimal gland of animals is a serous gland, except in pigs, in which the lacrimal gland is a mucous gland. We could not find any published reports that identified melanocytes or melanin-laden resident cells in the lacrimal gland in either humans or animals. Based on experiments that detected melanocyte-melanoma precursors in eccrine sweat glands of murine volar skin, 13 it is possible that secretory glands such as lacrimal glands can be a nidus for melanocytic neoplasia.
Lacrimal fluid (tears), secreted by the lacrimal gland, enters the upper and lower lacrimal puncta at the medial canthus and are drained through the lacrimal canaliculus. The lacrimal sac, a dilated portion of the lacrimal canaliculus, then passes lacrimal fluid to the nasolacrimal duct, which is continuous with the nares. Although a melanocytic tumor of the lacrimal gland is extremely rare in humans, 15 melanocytic tumors arising from or affecting lacrimal drainage are not rare.12,14 When such tumors occur, a lacrimal drainage neoplasm can be difficult to treat given its location whereby complete surgical excision is challenging. In animals, neoplasms affecting lacrimal drainage have not been reported.
Non-melanocytic neoplasms of the human lacrimal gland vary, including pleomorphic adenoma, cystadenoma, hemangioma, mucoepidermoid carcinoma, sebaceous carcinoma, follicular lymphoma, and synovial sarcoma, to name a few. 1 The reported canine non-melanocytic lacrimal gland tumors, if the nictitating gland is included, comprise pleomorphic adenoma, adenoma, adenocarcinoma, and squamous cell carcinoma, with adenocarcinoma overwhelmingly predominant.3,10 One should be aware that previous reports of nictitating gland tumors in animals may include those of the surface structures (lymphoid tissues and conjunctiva) or the stroma of the substantia propria, which are not essentially components of the lacrimal gland. The reasons why human lacrimal gland tumors are more prevalent and diverse than those of animals are unknown.
Lacrimal gland melanocytoma of the dog in our report was not suspected at the time of surgery. The dog did not have a history of melanodacryorrhea (black tears) as seen in a human patient suffering from uveal melanoma. 6 The mass (later proved to be the lacrimal gland) was removed surgically because that is the standard surgical protocol when ophthalmectomy is performed for lesions with a suspicion of neoplasm, which in our case was anterior uveal melanocytic tumor. The glaucoma in our patient was most likely caused by obliteration of the trabecular meshwork by numerous melanin-laden cells, resulting in sluggish flow of aqueous humor. There was no histologic evidence of invasion or metastasis of proliferating cells from either the lacrimal gland or the globe to the other. We hypothesize that these lesions occurred coincidentally within a narrow timeframe. The discovery and reporting of similar cases may provide deeper insight into the tumorigenesis of this rare neoplasm of the lacrimal gland.
Footnotes
Acknowledgements
Proofreading and critical review of the manuscript by Dr. Mary Durbin is highly appreciated. We thank Shikoku Cytopathological Laboratory for their help in histopathology, histochemistry, and immunohistochemistry.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
