Abstract
Immunohistochemistry is widely utilized in diagnostic laboratories to study neoplastic and nonneoplastic diseases. Knowledge of the immunohistochemical characteristics of normal tissue is essential for interpretation of immunoreactivity in pathologic conditions. In this study, immunohistochemistry was performed with a broad panel of diagnostically relevant antibodies on 4 normal canine globes—namely, vimentin, pan-cytokeratin (AE1/AE3), cytokeratin 7, cytokeratin 8/18, cytokeratin 20, α–smooth muscle actin, muscle specific actin, desmin, Melan-A, microphthalmia transcription factor, S-100, glial fibrillary acidic protein, triple neurofilaments, neuron-specific enolase, chromogranin A, synaptophysin, laminin and CD31. Results include cytokeratin immunoreactivity limited to the conjunctival epithelium, corneal epithelium, and retinal pigment epithelium; distinct patterns of immunopositivity of muscle markers; and widespread immunoreactivity for vimentin and most neural/neuroendocrine markers. These findings in normal eyes provide the basis for interpretation of ocular immunohistochemistry in dogs. Published immunophenotypes of primary ocular neoplasms are also reviewed.
Immunohistochemistry has been used in various forms since the early 1940s to identify cell types, structures, organisms, and materials secreted by cells. 13,38,57,74 In diagnostic settings, immunohistochemistry is most often used to determine the histogenesis of neoplasms, characterize neoplastic subtypes, or establish prognosis. 38,74 In one human study, immunohistochemistry was performed to establish a diagnosis in 64% of cases, determine prognosis in 19% of cases, and identify microorganisms or acellular structures in 17% of cases. 74
Antibodies that may be beneficial in diagnostic ocular pathology are similar to those of value in other organs. Antibodies directed against intermediate filaments (IFs), such as cytokeratins (CKs), vimentin, desmin, glial fibrillary acidic protein (GFAP), and neurofilaments, may be useful. CKs are classified as type I or II IFs and are typical of epithelial cells. 32,36,55 Antibodies can target individual CKs or groups of CKs, such as AE1/AE3. Vimentin, a type III IF, is the major IF of nonmyocyte mesenchymal cells. It is commonly coimmunoreactive with other IFs. 32,35,36,55 Desmin, a type III IF, is found mostly in striated, myocardial, and smooth muscle cells. 32,35,36,55 GFAP is immunoreactive in glial cells and can be coimmunopositive with CKs in some epithelial cells. GFAP is also a type III IF. 32,36,55 Neurofilaments (triple neurofilaments [TNF]) are type IV IFs and consist of a triplet of proteins in neural tissues and a subset of neuroendocrine cells. 32,35,36,55 Actin microfilaments can be identified using antibodies that recognize either all 4 muscle actins (HHF35/muscle-specific actin [MSA]) or only α–smooth muscle actin (SMA). 32,36
Melanocytic markers, such as Melan-A and microophthalmia transcription factor (MITF), may also be helpful in diagnostic immunohistochemistry. The Melan-A antibody targets an antigen that is recognized by autologous cytotoxic T cells and is mostly used in the diagnosis of melanoma, although Melan-A is also immunoreactive in nonmelanocytic cells.58,59 MITF is a protein essential for the synthesis of melanin pigment as well as the embryogenesis and postnatal survival of melanocytes. 26 Other helpful neural/neuroendocrine markers include S100, neuron-specific enolase (NSE), synaptophysin and chromogranin A. S100 is a calcium-binding protein that is utilized in the diagnosis of melanoma; however, S100 antibodies also stain a variety of normal tissues and nonmelanocytic neoplasms.36,59,67 NSE is a glycolytic isoenzyme present in neurons, neuroendocrine cells, and other cell types, including smooth muscle. 36,59,67 Synaptophysin is a membrane glycoprotein of presynaptic vesicles of neurons and neuroendocrine cells. 2,36 Chromogranins are the major proteins in neurosecretory granules of neuroendocrine cells and sympathetic nerves. 2,36 Laminin is a glycoprotein and a major component of basal lamina/basement membranes. 36 CD31, or PECAM-1, is normally immunoreactive in endothelium and some leukocytes. 36
Accurate interpretation of immunohistochemical reactions in neoplastic and nonneoplastic lesions requires the knowledge of normal tissue’s immunohistochemical properties. There has been an exponential increase in the number of scientific publications using immunohistochemistry over the last several decades. 74 Greater than 50% of all studies in the dog utilizing immunohistochemical techniques have been published since 2000, and 75% were published after 1990 (PubMed search performed January 18, 2011). Despite the ever-expanding utilization of immunohistochemistry, there has been little published on the immunohistochemical properties of normal eyes in animals. Only a few reports describe immunohistochemical findings of normal canine ocular structures. These reports examined specific diseases or structures and utilized a select number of antibodies (Supplemental Table 1, available online at http://vet.sagepub.com/supplemental). The purpose of this study was to characterize the immunohistochemical reactivity of antibodies routinely used in a diagnostic setting in normal canine globes.
Materials and Methods
Animals
Four histologically normal globes were obtained from dogs with no known history of previous ocular disease and were euthanized for reasons unrelated to the study. All globes were from adult dogs (4–10 years): 1 was an intact male; 3 were neutered males. Breeds included 1 Pit Bull Terrier, 1 Pit Bull Terrier cross, 1 Labrador Retriever cross, and 1 Weimaraner. The globes with retrieved within 1 hour of euthanasia, fixed in 10% neutral buffered formalin for 24 to 48 hours, and routinely processed for histology.
Immunohistochemistry
This study used antibodies against vimentin, pan-CK (AE1/AE3), CK7, CK8/18, CK20, SMA, MSA, desmin, Melan-A, MITF, S-100, GFAP, TNF, NSE, chromogranin A, synaptophysin, laminin, and CD31. A standard streptavidin–biotin horseradish peroxidase method was used. Antibodies, positive control, and antigen retrieval methods are listed in Supplemental Table 2. Briefly, tissues sections were deparaffinized with xylene and rehydrated using a graded ethanol series. Endogenous peroxidase activity was quenched by incubating sections in 0.3% H2O2 in methanol for 30 minutes. Antigen retrieval was performed as noted in Supplemental Table 2. Sections were incubated in 3% normal goat serum for 30 minutes at room temperature. Sections were incubated with the primary antibody for 60 minutes or 2 hours (CD31) or overnight at 4°C (Melan-A) in a moist chamber. Slides were incubated with a biotinylated anti-mouse link reagent (Biocare Medical, Concord, CA) for 10 minutes and then incubated with streptavidin horseradish peroxidase (Biocare Medical) for 10 minutes. Positive reaction was visualized with 3-amino-9-ethylcarbazole chromogen (Zymed Labs, San Francisco, CA), followed by several rinses in phosphate buffered saline. Slides were counterstained with Mayer’s hematoxylin (Sigma Chemical Co., St. Louis, MO). Negative controls were prepared by substituting the primary antibody with an irrelevant species-matched antibody (monoclonal antibodies) or phosphate buffered saline (polyclonal antibodies) (Supplemental Table 2).
Sixty structures were evaluated for each globe. In addition to those listed in Table 1, the following cells and structures were evaluated: collagen matrix (corneal, scleral, uveal), blood vessels and peripheral nerves of the uvea, lens zonules, nonvascular smooth muscle cells of the choroid, 40,56 and retinal muller cells. The ciliary epithelium of the pars plicata and pars plana were initially examined as separate sites but are combined in Table 1. Similarly, the pigmented and nonpigmented retinal pigment epithelium (RPE) was initially examined separately. The immunoreactivity was assessed independently by 2 observers (P.L., C.M.R.). The immunoreactivity was noted to be positive when coloration was distributed appropriately within the cell/structure for each antibody. Positive reaction was moderate or strong unless noted to be weak. Other findings, such as distribution pattern, were noted separately.
Immunohistochemical Reactivity in Normal Canine Eyesa
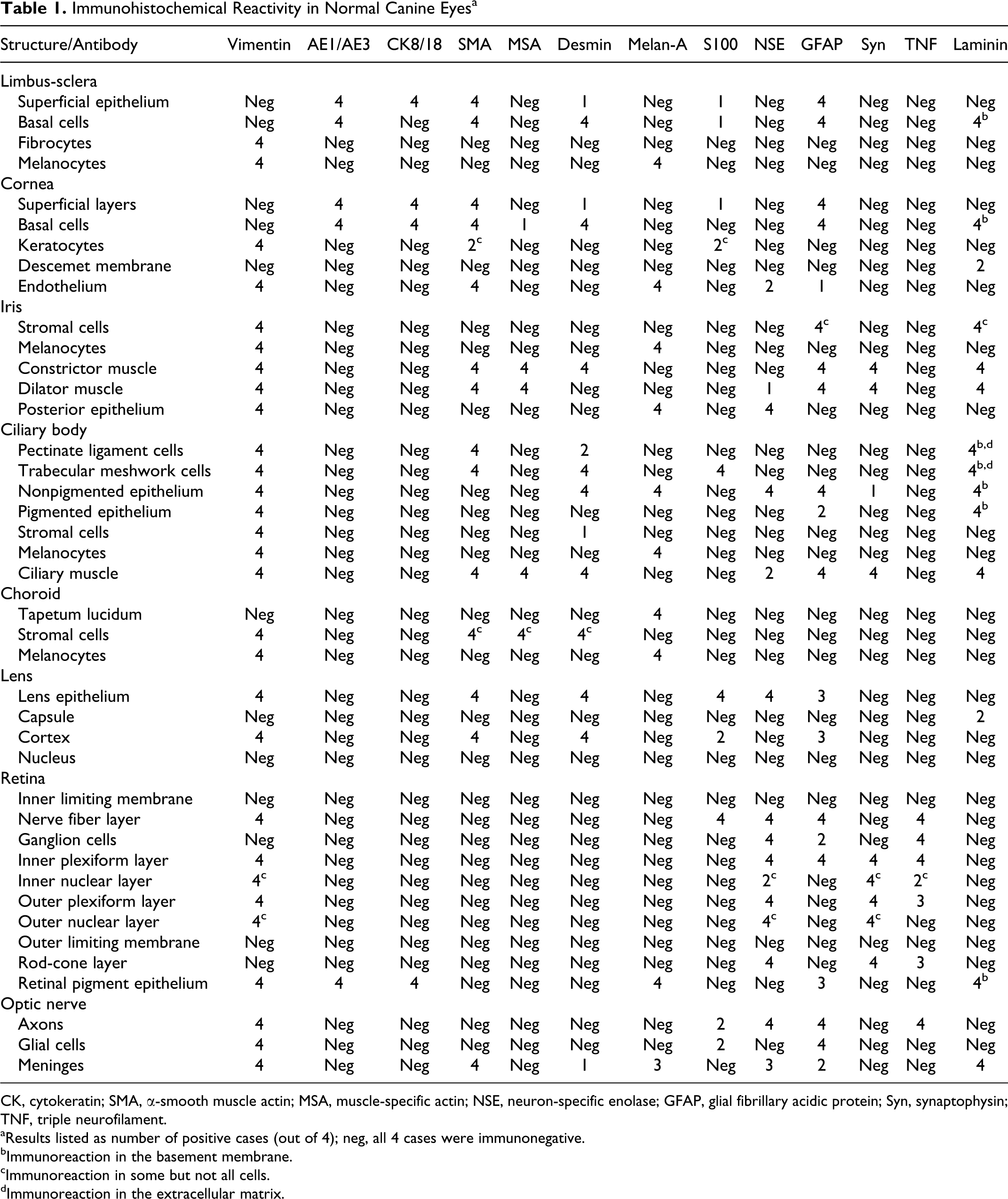
CK, cytokeratin; SMA, α-smooth muscle actin; MSA, muscle-specific actin; NSE, neuron-specific enolase; GFAP, glial fibrillary acidic protein; Syn, synaptophysin; TNF, triple neurofilament.
aResults listed as number of positive cases (out of 4); neg, all 4 cases were immunonegative.
bImmunoreaction in the basement membrane.
cImmunoreaction in some but not all cells.
dImmunoreaction in the extracellular matrix.
Results
Results are summarized in Table 1. The immunoreactivity of each stain was similar for the 4 globes in 97% of the cells/structures evaluated. There was no positive immunoreaction with CK7, CK20, or chromogranin A in any ocular structure. MITF immunoreactivity was present in melanocytes and RPE as expected, but there was also positive immunoreaction in almost all other structures evaluated, making conclusive interpretation impossible (results not shown).
Limbus
The conjunctival epithelium was immunopositive for AE1/AE3, SMA, and GFAP. In addition, the superficial cells were immunopositive for CK8/18 (basal cells were negative); there was weak immunoreactivity for desmin mostly in basal cells; and 1 case was weakly immunoreactive for S100 in the basal and superficial epithelium. In 2 cases, rare vimentin and Melan-A immunopositive cells were scattered in the epithelium (interpreted as melanocytes). The basement membrane was immunoreactive for laminin. In the substantia propria, the fibrocytes were vimentin immunopositive, and the melanocytes were vimentin and Melan-A immunoreactive.
Cornea
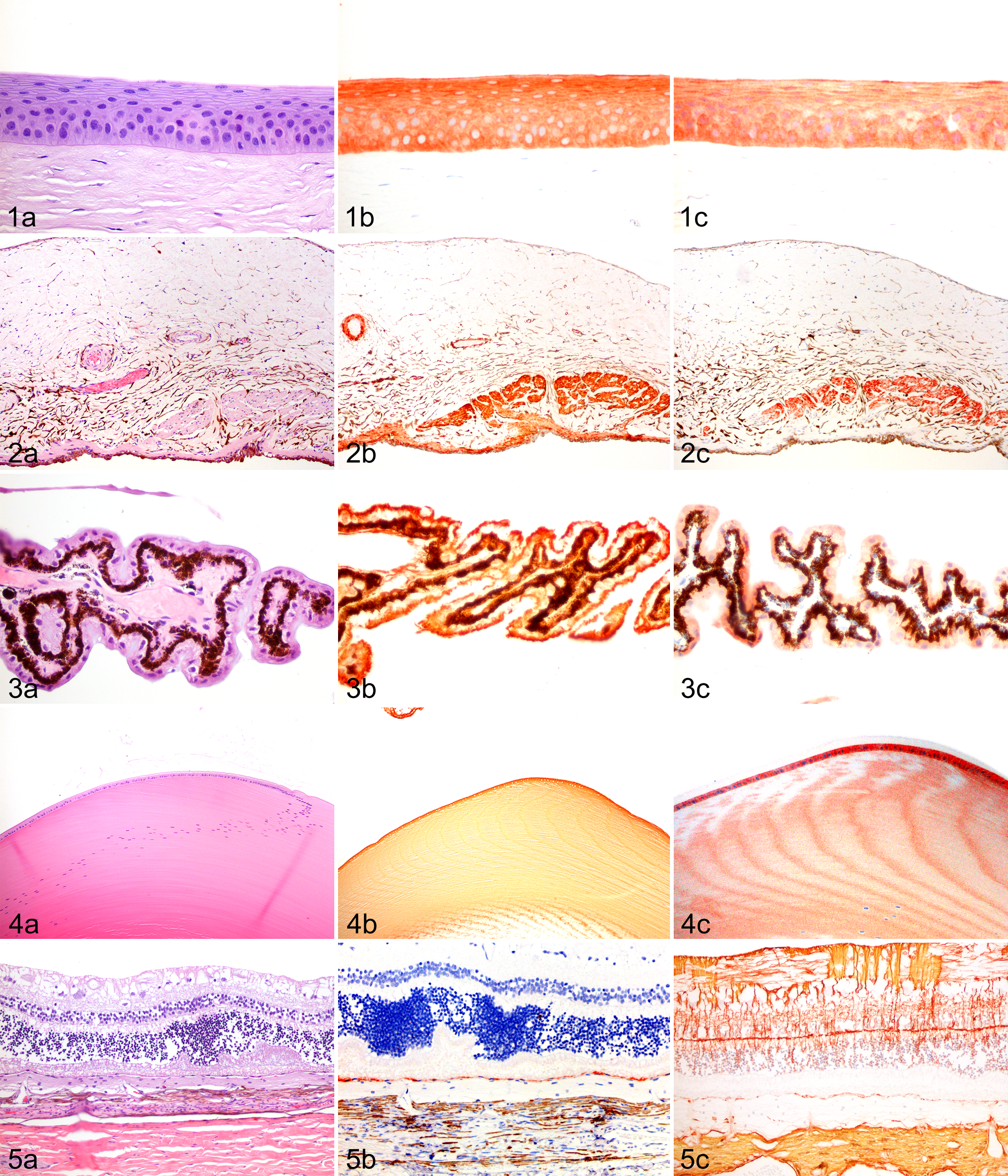
The basal cells and superficial layers (wing and superficial cells) of the epithelium were immunopositive for AE1/AE3, CK8/18, SMA, and GFAP (Fig. 1a–1c). There was weak immunoreaction for desmin and S100 (3 cases), mostly in basal cells, and 1 case was weakly immunoreactive for MSA in the basal cells. In 1 case, a small cluster of 10 to 12 basal cells was immunopositive for vimentin, but the epithelium was overall negative and listed as such in Table 1. The basement membrane was weakly immunoreactive for laminin. The stromal keratocytes were immunopositive for vimentin; 2 cases were immunoreactive for SMA and S100, mostly in the peripheral cornea. The stromal collagen was negative for all stains. Laminin was weakly immunoreactive on the stromal aspect of Descemet membrane in 2 cases. The corneal endothelium was immunoreactive for vimentin, SMA, and Melan-A, with 2 cases immunopositive for NSE and 1 case immunopositive for GFAP.
Iris
All stromal cells were immunoreactive for vimentin. Some stromal cells surrounding vessels were immunopositive for GFAP, and some diffusely scattered cells were immunopositive for laminin. The stromal melanocytes, including those forming the iris anterior border, were immunoreactive for vimentin and Melan-A. The constrictor muscle was immunoreactive for vimentin, laminin, and SMA; moderately reactive for MSA, desmin, and GFAP; and weakly positive for synaptophysin (Fig. 2a–2c). The dilator muscle was immunoreactive for vimentin, laminin, and SMA; moderately reactive for MSA and GFAP; and weakly positive for synaptophysin (Fig. 2a–2c). One case was positive for NSE. The iris posterior epithelium was immunoreactive for vimentin, NSE, and Melan-A.
Ciliary Body
The cells lining the pectinate ligament were immunoreactive for vimentin and SMA, and 2 cases were positive for desmin. The basement membrane and extracellular matrix were multifocally weakly immunopositive for laminin. The cells lining the trabecular meshwork were immunopositive for vimentin, SMA, and desmin and weakly positive for S100. The basement membrane and extracellular matrix were multifocally weakly immunopositive for laminin. The immunophenotype of the ciliary epithelium in the pars plicata and pars plana were identical in all cases. The nonpigmented ciliary epithelium was immunoreactive for vimentin, moderately positive for NSE, and weakly positive for GFAP, desmin, and Melan-A. One case was weakly positive for synaptophysin (Fig. 3a–3c). The internal limiting membrane / basement membrane was immunopositive for laminin. The pigmented ciliary epithelium was immunopositive for vimentin and moderately positive for GFAP in 2 cases (Fig. 3a–3c). The basement membrane was immunopositive for laminin. The ciliary stromal cells were immunoreactive for vimentin, and some stromal cells were positive for desmin in 1 case. The melanocytes were vimentin and Melan-A reactive. The ciliary muscle was immunoreactive for vimentin, laminin, SMA, and MSA and moderately reactive for desmin, GFAP, and synaptophysin; 2 cases were positive for NSE.
Choroid
All cells of the tapetum lucidum were immunoreactive for Melan-A. The stromal cells were vimentin immunopositive, and some of the stromal cells were reactive for SMA, MSA, and desmin. The melanocytes were vimentin and Melan-A reactive. The uveal (iris, ciliary body, and choroid) collagen/matrix was negative for all stains.
Lens
The lens capsule was immunopositive for laminin in 2 cases. The lens epithelium was immunopositive for vimentin, SMA, desmin, S100, NSE, and GFAP (3 cases) (Fig. 4a–4c). The cortical lens fibers were immunoreactive for vimentin, SMA, desmin, S100 (2 cases), and GFAP (3 cases). The lens nucleus and the lens zonules were negative for all stains.
Retina
The nerve layer was immunoreactive for vimentin, S100, NSE, GFAP, and TNF. The ganglion cells were immunopositive for NSE and TNF, and 2 cases were positive for GFAP. The inner plexiform layer was immunopositive for vimentin, NSE, GFAP, synaptophysin, and TNF. There was multifocal immunoreactivity in the inner nuclear layer with vimentin, synaptophysin, NSE (2 cases), and TNF (2 cases). The outer plexiform layer was immunopositive for vimentin, NSE, synaptophysin, and TNF (3 cases). There was mutifocal immunoreaction in the outer nuclear layer with vimentin, NSE, and synaptophysin. The rod-cone layer was immunoreactive for NSE, synaptophysin, and TNF (3 cases). The RPE was immunopositive for vimentin and AE1/AE3 and weakly immunoreactive for CK8/18, Melan-A, and GFAP (3 cases) (Fig. 5a–5c). The pigmented and nonpigmented RPE shared the same immunophenotype. The basement membrane was immunoreactive for laminin.
Optic Nerve
The axons were immunoreactive for vimentin, NSE, GFAP, TNF, and S100 (2 cases). The glial cells were immunopositive for vimentin, GFAP, and S100 (2 cases). The meninges were immunoreactive for vimentin, SMA, laminin, NSE (3 cases), GFAP (2 cases), and desmin (1 case).
Other Structures
Uveal and scleral peripheral nerves were immunoreactive for vimentin, S100, GFAP, TNF, and laminin, with SMA immunoreactivity limited to the sheath. The endothelium of blood vessels, including scleral veins, was immunopositive for vimentin and CD31 (immunoreactive in most vessels, weak reaction in veins). The walls of the blood vessels were immunoreactive for vimentin, SMA, desmin, MSA, and laminin. The scleral collagen was negative for all stains.
Discussion
The results of this study will serve as the basis for the future interpretation of neoplastic and nonneoplastic immunohistochemical studies performed on canine ocular diseases. In addition to selecting diagnostically relevant antibodies, we used a standard diagnostic protocol so that these results may be easily reproduced and so that results from other diagnostic laboratories may be more easily compared. As with any immunohistochemical study, the immunopositive reactions described here should be confirmed to be specific for each antibody by other methods. Our findings are compatible with the previous reports of positive immunohistochemical reaction in normal canine ocular structures (Supplemental Table 1). We did not observe SMA immunoreactivity in the ciliary epithelium as described by Zarfoss et al; 77 however, there was positive desmin immunoreaction in the nonpigmented ciliary epithelium.
The broad spectrum of antibodies used in this study revealed several interesting immunophenotypes. The corneal and limbal conjunctival epithelia share a similar immunophenotype, which includes immunoreactivity for CKs (AE1/AE3 and CK8/18), SMA, and GFAP with variable S-100 and desmin immunoreactivity (Fig. 1a–1c). One noteworthy difference between the corneal and conjunctival epithelia was the negative reactivity of the limbal basal cells for CK8/18, while the superficial limbal cells and all layers of the corneal epithelium were immunopositive. A similar distribution has been noted in humans with CK3/12, which supports the limbal localization of corneal stem cells. 42,55,65 The distribution of CK8/18 in dogs may also be related to the unique role of limbal basal cells. The immunoreactivity of CKs was expected and is diagnostically helpful. However, the immunopositivity for muscle markers and GFAP in the limbal and corneal epithelia is an unexpected immunophenotype, one that is similar to proliferating/neoplastic myoepithelial cells. 27,28 The functional significance of these findings is unclear and warrants further investigation.
The cells lining the pectinate ligament and trabecular meshwork share a similar immunophenotype. Vimentin and SMA immunoreactivity is likely to be the most helpful in the study of pectinate ligament and trabecular meshwork cells. There is also desmin immunopositivity, and the trabecular meshwork cells are immunoreactive for S100. There is no positive immunoreaction with CD31, indicating that the cells lining the pectinate ligament and trabecular meshwork are not vascular in origin despite being often termed
The distribution of the muscle markers in the dilator, constrictor (sphincter), and ciliary muscles is distinct. The constrictor and ciliary muscles are immunopositive for SMA, MSA, and desmin, while the iris dilator muscle is SMA and MSA immunopositive but desmin negative (Fig. 2a–2c). A similar distribution of muscle markers has been described in cats and was shown to be diagnostically helpful to determine the origin of an intraocular leiomyosarcoma. 34 The dilator, constrictor, and ciliary muscles were moderately immunoreactive for GFAP. This should be considered when making the diagnosis of uveal schwannomas / spindle cells tumors of blue-eyed dogs, which are GFAP immunopositive but negative for muscle markers (Table 2). 79 Numerous choroidal stromal cells are immunoreactive to all 3 muscle markers (SMA, MSA, desmin). It is possible that these cells are similar to the network of nonvascular smooth muscle cell of the choroid described in other species. The exact function is not known, but a role in the protection of choroidal architecture during accommodation has been proposed. 40,56
Previously Reported Diagnostic Immunohistochemistry of Primary Ocular Neoplasms in Dogs
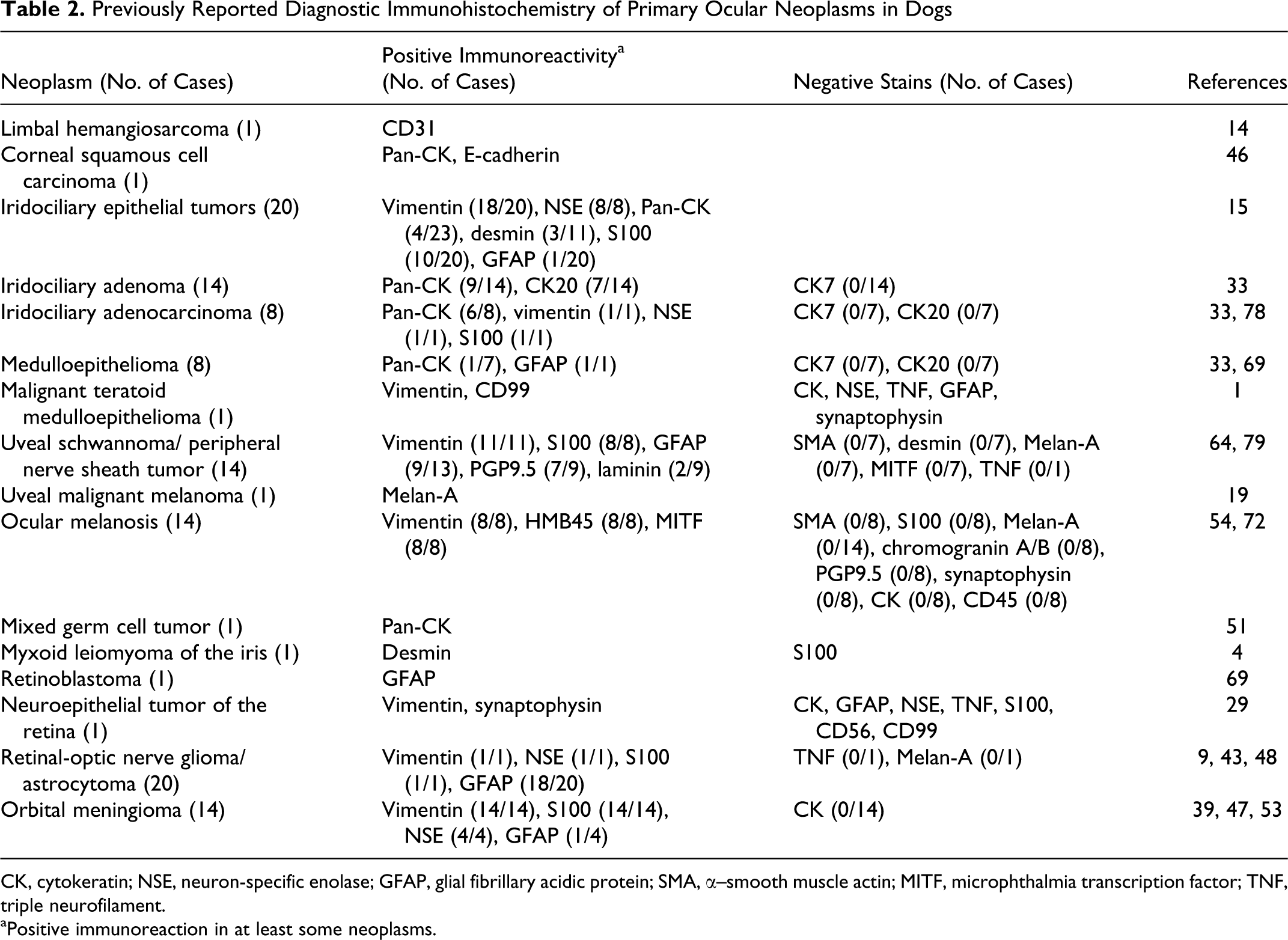
CK, cytokeratin; NSE, neuron-specific enolase; GFAP, glial fibrillary acidic protein; SMA, α–smooth muscle actin; MITF, microphthalmia transcription factor; TNF, triple neurofilament.
aPositive immunoreaction in at least some neoplasms.
The use of vimentin, NSE, and GFAP appears to be the best diagnostic combination to identify canine neuroepithelium of the iris and ciliary body (Fig. 3a–3c). The iris posterior epithelium is immunoreactive for vimentin, NSE, and Melan-A. The nonpigmented ciliary epithelium is immunoreactive for vimentin and NSE and weakly immunopositive for GFAP, desmin, Melan-A, and synaptophysin. The pigmented ciliary epithelium is immunopositive for vimentin and weakly immunoreactive for GFAP. These findings are consistent with previous published immunoreactivity in normal and neoplastic neuroepithelium (Supplemental Table 1, Table 2). 15,16 In humans, the ciliary epithelium is immunoreactive for vimentin as well as CK. 32,55
The normal canine lens epithelium is immunopositive for vimentin, SMA, desmin, S100, NSE, and GFAP (Fig. 4a–4c). The immunoreactions with vimentin and possibly SMA are likely to be the most helpful in the study of the lens epithelium. Vimentin, SMA, S100, NSE, and GFAP immunoreactivity has been noted in the lens epithelium of different species in various combinations and at different times in development, but desmin immunoreactivity appears unique to the canine lens epithelium. 6,20,44,45,61,68,80 The embryologic origin of the lens epithelium is the surface ectoderm, but CK immunoreactivity is lost in the early stages of lens development. 31,32 There is no CK immunopositivity in the normal adult lens epithelium in most species. 31,32 CK immunoreactivity has however been reported in the epithelium of lenses with cataracts. 11,32,70 The positive immunoreaction of SMA in normal canine lens epithelial cells is significant, since SMA immunoreactivity has been used as an indication of epithelial-mesenchymal transition / metaplasia in cataract formation. It has been recently discovered that normal human lens epithelium is also immunoreactive to SMA and that epithelial-mesenchymal transition may be dependent on increased immunoreactivity of SMA and other factors rather than simply the presence of SMA. 11,20,21,61
It is best to interpret the results in the neuroretina as a descriptive distribution of immunoreaction rather than an identification of specific cell types, since we did not perform double labeling or use antibodies specific for the various cells of the retina. The CK reaction of the AE1/AE3 antibody described in glial cells and neural tissue and interpreted in most cases to be a cross-reaction with GFAP was not present in this study. 17 The RPE is immunoreactive for both CKs (AE1/AE3 and weak reaction to CK8/18) and vimentin, and this is a diagnostic feature of the normal canine RPE (Fig. 5a–5c). There is also weak immunoreaction for Melan-A and GFAP. This immunophenotype is compatible with previous findings in dogs and other species. Dual immunoreactivity for vimentin and CK has been shown in cultured canine RPE cells. 41 Guinea pig RPE cells and a proportion of bovine RPE cells in situ also demonstrate both CK and vimentin IFs. 50 Normal human adult RPE cells are only immunopositive CKs, but coreactivity of vimentin with CKs has been documented during ocular development and in reactive, hyperplastic, and neoplastic RPE. 6,18,24,32,71 RPE of rhesus monkeys is immunopositive for vimentin and CK after retinal separation/detachment, but vimentin immunolabeling is not detected after long-term reattachment. 22 Triple immunoreactivity for CK–vimentin–GFAP has been documented in cells thought to be RPE in epiretinal membranes and in cultured RPE cells in humans. 8 Neoplastic human RPE can be immunoreactive for Melan-A. 24
The immunopositivity of Melan-A in all layers of the tapetum lucidum is consistent with the suspicion that the tapetal cells in tapeta cellulosa are modified melanocytes. 7,37,49 The lack of immunoreactivity to any other antibody tested, including vimentin, is puzzling. Vimentin filaments in melanocytes are thought to be involved in the movement of melanosomes. 10,30 The lack of vimentin may be a consequence of the specific structure and arrangement of melanosome-like rods in tapetal cells. The unique role of tapetal pigment in light reflection rather than absorption may also be a factor contributing to the unique immunophenotype of the tapetal cells. 7,37,49,63
Our findings are compatible with the published immunophenotypes of canine ocular neoplasms (Table 2). Melanocytic and iridociliary neoplasms are the 2 most common forms of primary ocular neoplasia. 15 Iridociliary tumors in dogs have a unique immunophenotype with immunopositivity for vimentin and NSE and variable immunoreaction with desmin and GFAP. 15 A subset of iridociliary tumors, more often in histologically malignant neoplasms, is also immunopositive for CK, 15,33,78 which was not observed in normal iris and ciliary epithelium. Melan-A immunoreactivity, noted in normal iris and nonpigmented ciliary epithelium, has not been investigated in canine iridociliary tumors. Melan-A immunoreactivity has however been reported in a human adenoma of the nonpigmented ciliary epithelium. 52 Although differentiation between iridociliary tumors and melanoma does not typically require immunohistochemistry, the immunoreactivity of Melan-A in the nonpigmented ciliary epithelium may pose a diagnostic challenge in rare cases of poorly differentiated neoplasms affecting the uveal tract. Investigation of the immunoreactivity of other melanocyte/melanoma markers, such as PNL2 and tyrosine-related proteins 1 and 2, 60,66 in normal and neoplastic ocular tissue might prove useful. The cells of ocular melanosis, considered by many to be a form of diffuse melanocytoma, have a unique immunophenotype that differs from normal melanocytes, with positive immunoreaction for vimentin and variable HMB-45 and MITF immunoreactivity but negative reaction with Melan-A. 54,72,75
In summary, this study provides a comprehensive description of the immunohistochemical features of normal canine ocular structures. Although interesting immunophenotypes were identified, this study reveals significant overlap in the immunoreaction patterns of various structures in the canine globe. Interpretation of immunohistochemistry performed on canine eyes should always be made in context of histopathologic findings and with knowledge that adjacent ocular structures may share immunohistochemical characteristics. Further research is needed to validate these findings and provide functional significance to the immunophenotypes identified.
Footnotes
Acknowledgements
We thank Dr Scott Moroff and Antech Diagnostics for providing funding for this study, as well as Marc Hardman for assistance with figures.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
