Abstract
Immunohistochemical detection of inhibin-α, -βA and -βB chains and 3β-hydroxysteroid dehydrogenase (HSD) was carried out on primary testicular tumors from 15 dogs and normal testes from three adult dogs. Histopathologically, the tumors were composed of three types: Leydig cell tumors in five dogs, Sertoli cell tumors in five dogs, and seminoma in five dogs. In normal testes, immunostaining against inhibin-α, -βA, and -βB chains and 3β-HSD revealed positive reactivity in the cytoplasm of Leydig cells. In testicular tumors, immunoreactive cells against inhibin-α, -βA, and -βB chains and 3β-HSD were localized in all Leydig cell tumors but not in any Sertoli cell tumors or seminomas. The results of radioimmunoassay for plasma inhibin in dogs with Leydig cell tumors showed higher concentrations than those in dogs with Sertoli cell tumors and seminomas and those in normal dogs. The concentration of inhibin in the plasma was markedly decreased by the surgical removal of the Leydig cell tumor in one dog. Our findings suggest that inhibin is synthesized by normal and neoplastic Leydig cells in the canine testis, and the secreted inhibin may be inhibin A and inhibin B.
Inhibin is a gonadal hormone that selectively inhibits the secretion of follicle-stimulating hormone (FSH) from the pituitary gland. Inhibin consists of an α-subunit linked by a disulfide bridge to one of two highly homologous β-subunits to form either inhibin A (α-βA) or inhibin B (α-βB). Although inhibin and activin are closely related peptides, activin is composed of homodimers or heterodimers of the same β-subunits, resulting in activin-A (βA-βA), activin-AB (βA-βB), or activin-B (βB-βB). 1,7 Inhibin is secreted from the granulosa cells of ovarian follicles, corpus luteum, and placenta in females 2 and presumably from the Sertoli cells in males of humans and rats 11,16,17 but not from the Leydig cells in horses. 13,14 3β-Hydroxysteroid dehydrogenase (3β-HSD), a steroidogenic enzyme, in the normal equine testis is localized only in the Leydig cells and not in the Sertoli cells and germ cells. 13 However, the secreting cells of inhibin and 3β-HSD in canine testes remain unidentified.
Primary testicular tumors are very common in dogs. The three main types are derived from three specialized testicular elements: the interstitial cells of Leydig, the sustentacular cells of Sertoli, and the spermatic germinal epithelium. 15 The tumors occur mostly in mature and old dogs, and the incidence of Leydig cell tumors in particular increases with age. 15 Previous studies in humans and animals have produced conflicting data on the pattern of expression of the inhibin subunits in normal testes 13,14,17–19 and have yielded little information on expression of the inhibin subunits in primary testicular tumors. 3,4,6,10 The purpose of the present study was to investigate the secreting cells of inhibin in primary testicular tumors and normal adult testes in dogs.
Materials and Methods
Tissue preparation
Tissue samples were collected from 15 testicular tumors, including five Leydig cell tumors, five Sertoli cell tumors, and five seminomas, and from three normal testes for control (Table 1). All testicular tumors and normal testes were in the bursula testium and were surgically excised for treatment and castration. Tumor and normal tissues were fixed in 4% phosphate-buffered paraformaldehyde solution, embedded in paraffin, and cut at 4 µm for histopathologic and immunohistochemical examinations. Sections for histopathologic examination were stained with hematoxylin and eosin (HE).
Results of immunohistochemical examinations of canine tissues for inhibin types and 3β-HSD.
Immunohistochemistry
After deparaffinization, sections were autoclaved for 15 minutes at 121 C in sodium citrate buffer (0.01 M, pH 6.0). The sections were incubated in 3% H2O2-methanol for 30 minutes to quench endogenous peroxidase activity and then washed three times for 5 minutes each in 0.05 M Tris HCl (pH 7.6, with 0.15 M NaCl). Sections were incubated with Block Ace (Dainippon Pharmaceutical Co., Osaka, Japan) at 37 C in a moisture chamber for 30 minutes to prevent nonspecific reactions. The specific antisera used in the present study were 1:500 anti-porcine-[Tyr30]-inhibin-α (1–30) chain (Dr. N. Ling, Neuroendocrine Inc., San Diego, CA), 1:200 anti-porcine-cyclic inhibin-βA (81113) (305-24-D) and 1:500 anti-human-cyclic inhibin-βB (80112) (305-25-D) chains (Dr. W. Vale, Salk Institute for Biological Studies, La Jolla, CA), and 1:500 anti-human-3β-HSD (Dr. J. I. Mason, Cecil H. and Ida Green Center for Reproductive Science, University of Texas, Southern Medical Center, Dallas, TX) rabbit polyclonal antibodies. Sections were incubated with the primary antibodies at 4 C in a moisture chamber for 6 hours. After washing with Tris HCl buffer, sections were further incubated with 0.5% biotinylated goat anti-rabbit secondary antibody (ABC-peroxidase staining kit Elite, Vector Laboratories, Burlingame, CA) diluted with 0.05 M Tris HCl containing 10% Block Ace for 1 hour at 37 C. The sections were then washed with 0.05 M Tris HCl three times for 5 minutes each and treated with 2% avidin–biotin complex for 30 minutes at 37 C. Sections were then reacted with 0.5% 3,3′-diaminobenzidine tetrachloride (Sigma Chemical Co., St. Louis, MO) and 0.01% H2O2 to visualize the bound antibody. Control sections were exposed to normal rabbit serum (diluted 1:4,000) instead of inhibin antibody at 37 C overnight.
Radioimmunoassay for plasma inhibin
Concentrations of ir-inhibin in canine plasma were measured using a rabbit antiserum against purified bovine inhibin and 125I-labeled 32-kd bovine inhibin, as described previously. 8 Plasma samples were corrected from normal dogs (2/3) and prior to surgery from dogs with Leydig cell tumors (5/5), Sertoli cell tumors (3/5), and seminomas (3/5) (Table 1). In one dog with a Leydig cell tumor, a plasma sample was also collected after surgical excision of the tumor. The results were expressed in terms of 32-kd bovine inhibin. The sensitivity of the assay was 7.8 pg/tube (78 pg/ml). The intraassay coefficients of variation were 8.0%. All data were presented as means ± SEM. Student's t-test was used to compare the evaluations of concentration of plasma inhibin in four groups composed of dogs with Leydig cell tumors, Sertoli cell tumors, seminomas, and normal testes. All difference with values of P < 0.05 were considered significant.
Results
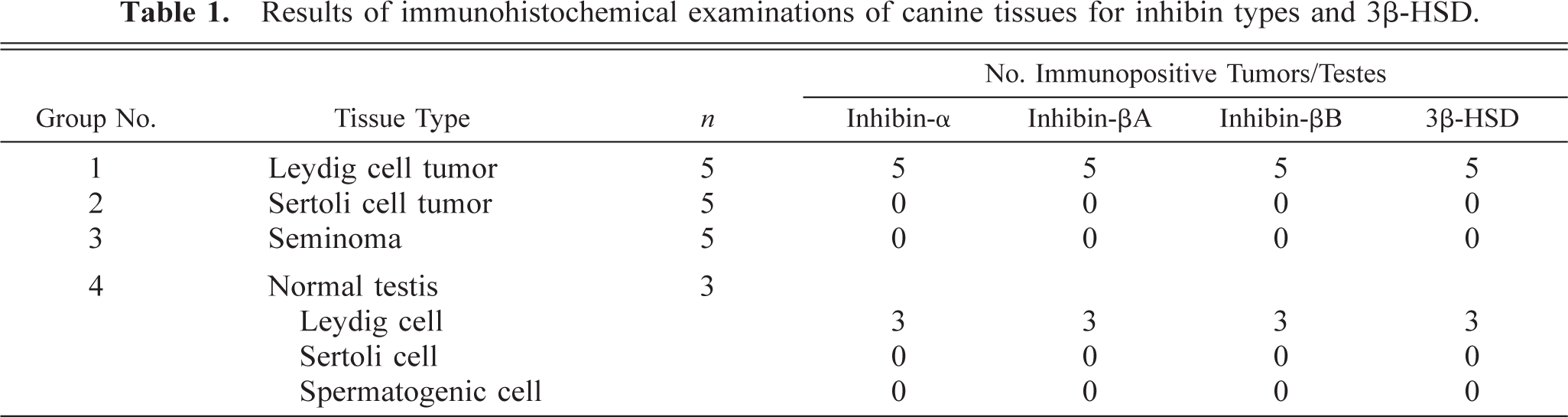
Testicular tumors were classified according to the World Health Organization recommendation. 9 Leydig cell tumors were classified into three patterns: solid diffuse type, pseudoadenomatous type (Fig. 1), and cystic-vascular type. Sertoli cell tumors were classified into tubular and diffuse type. Cytoplasm of tumor cells in seminoma was scant and slightly acidophilic. The nuclei were variable in size, round, and hyperchromatic. Mitotic figures were very common. In all normal testes, spermatocytogenesis was seen in the seminiferous tubules. The interstitial cells of Leydig formed well-vascularized groups of polyhedral cells, which occupied the spaces between the seminiferous tubules.
Leydig cell tumor; dog. Pseudoadenomatous pattern consists of the neoplastic cells, which are columnar in shape and have abundant cytoplasm, showing an acidophilic and coarsely vacuolated appearance. HE. Bar = 50 µm.
Immunohistochemistry
The results of immunohistochemical examination of inhibin-α, -βA, and -βB chains and 3β-HSD in testicular tumors and normal testes are summarized in Table 1.
Leydig cell tumors
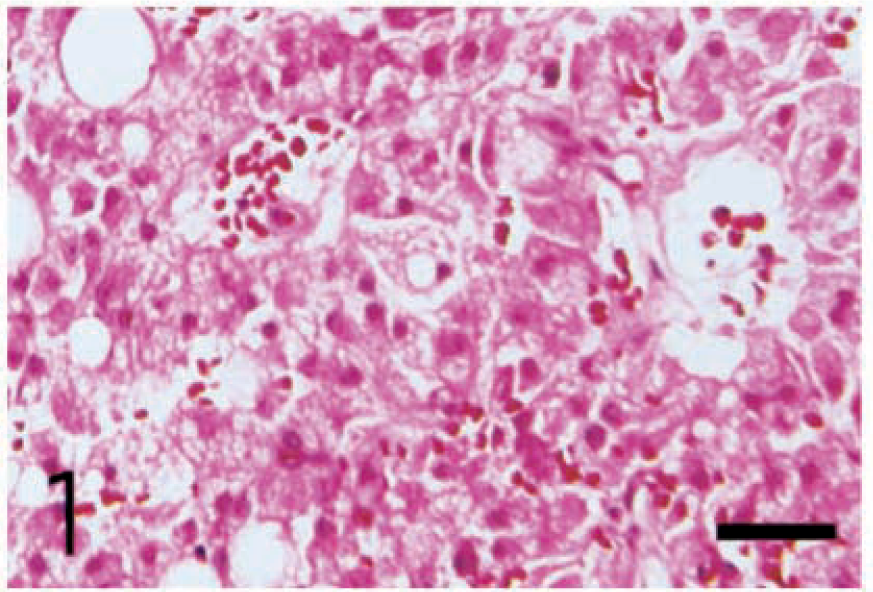
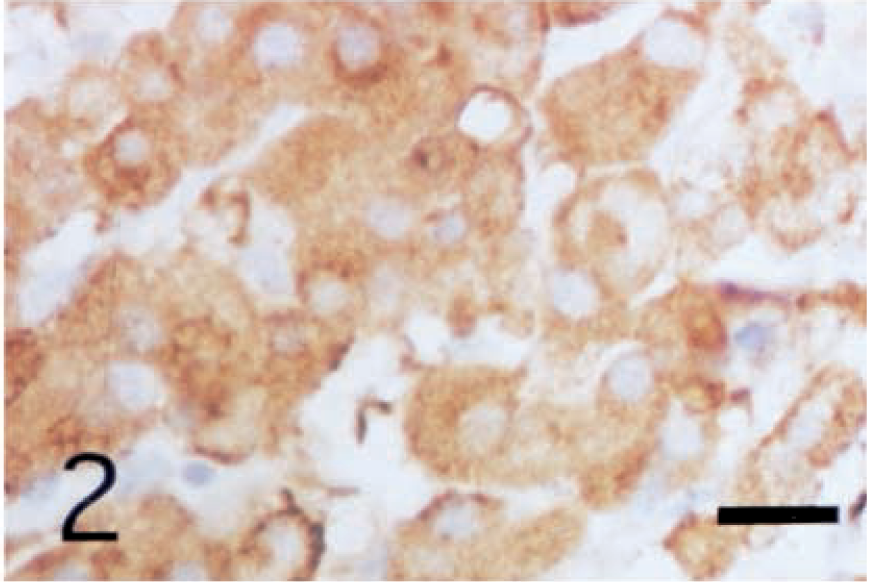
The cytoplasm of all neoplastic cells, regardless of their histologic tumor patterns, showed immunohistochemical positive reactivity against anti-inhibin-α (Fig. 2), anti-inhibin-βA (Fig. 3), and anti-inhibin-βB (Fig. 4) chains and anti-3β-HSD antibodies. The reactivity varied in intensity within individual cells, without regard to their morphologic characteristics.
Leydig cell tumor; dog. Cytoplasm of the neoplastic cells in pseudoadenomatous tumor shows positive reactivity with anti-porcine inhibin-α chain antibody. ABC complex method, Mayer's hematoxylin counterstain. Bar = 25 µm.
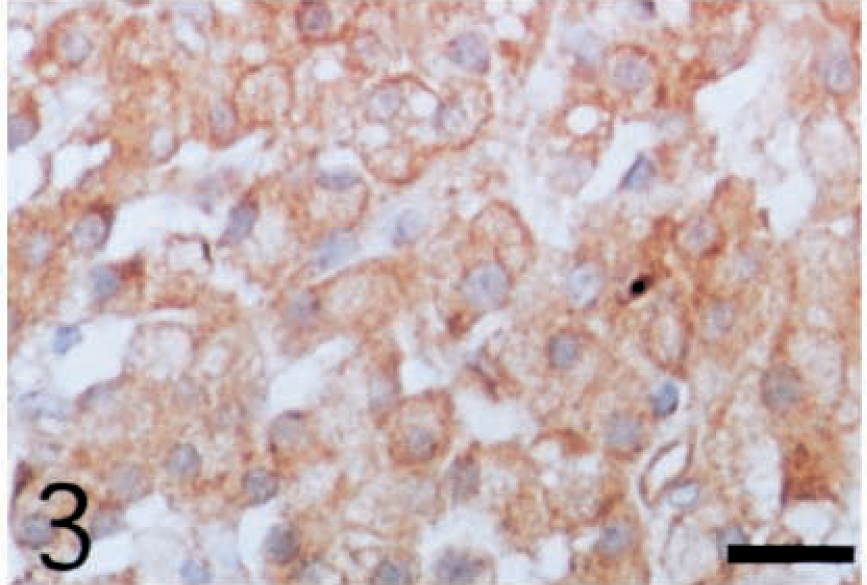
Leydig cell tumor; dog. Cytoplasm of the neoplastic cells shows positive reactivity with antihuman inhibin-βB chain antibody. ABC complex method, Mayer's hematoxylin counterstain. Bar = 25 µm.
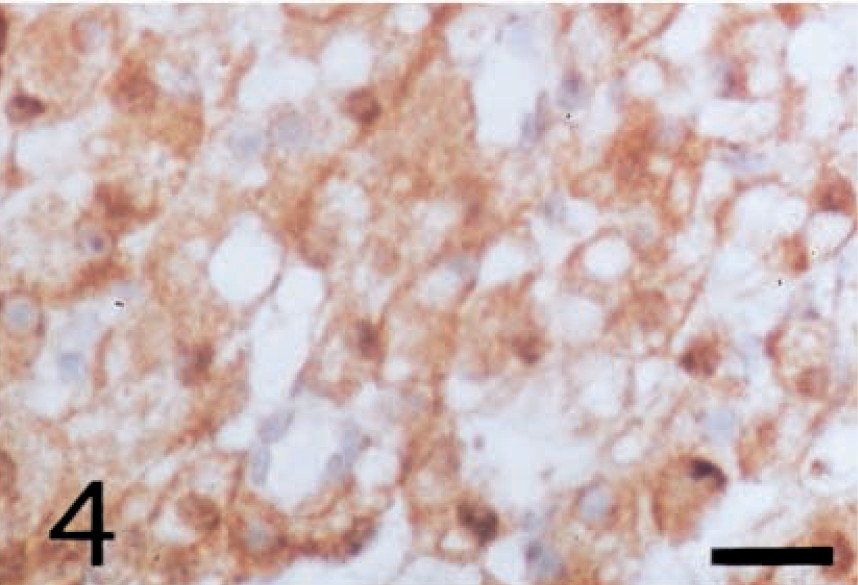
Leydig cell tumor; dog. Cytoplasm of the neoplastic cells shows positive reactivity with anti-porcine inhibin-βA chain antibody. ABC complex method, Mayer's hematoxylin counterstain. Bar = 25 µm.
Sertoli cell tumors
Neoplastic Sertoli cells did not react to anti-inhibin-α, -βA, and -βB chains or 3β-HSD antibodies.
Seminoma
Neoplastic cells in all seminomas showed no immunohistochemical reactivity for any antibodies.
Control testis
The interstitial cells of Leydig were positively stained with anti-inhibin-α (Fig. 5), -βA (Fig. 6), and -βB (Fig. 7) chains and with 3β-HSD (Fig. 8) antibodies. In seminiferous tubules, Sertoli cells and spermatogenic cells were not stained with any antibodies.
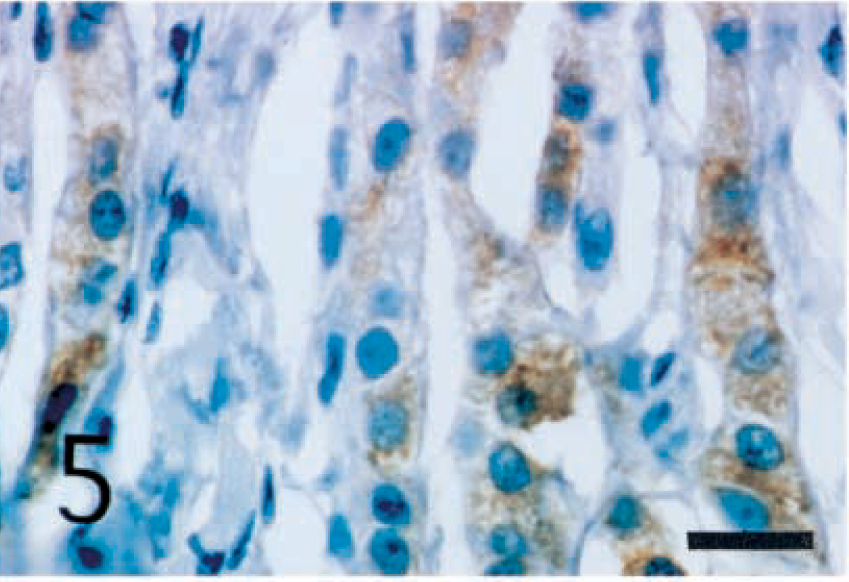
Normal testis; dog. Interstitial cells of Leydig show strong reactivity with anti-porcine inhibin-α chain antibody. ABC complex method, Mayer's hematoxylin counterstain. Bar = 25 µm.
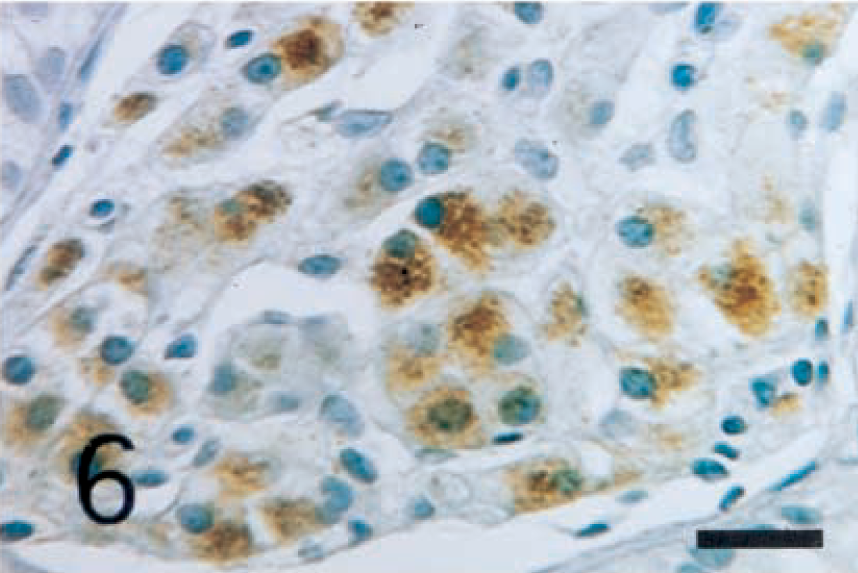
Normal testis; dog. Interstitial cells of Leydig show strong reactivity with anti-human inhibin-βB chain antibody. ABC complex method, Mayer's hematoxylin counterstain. Bar = 25 µm.
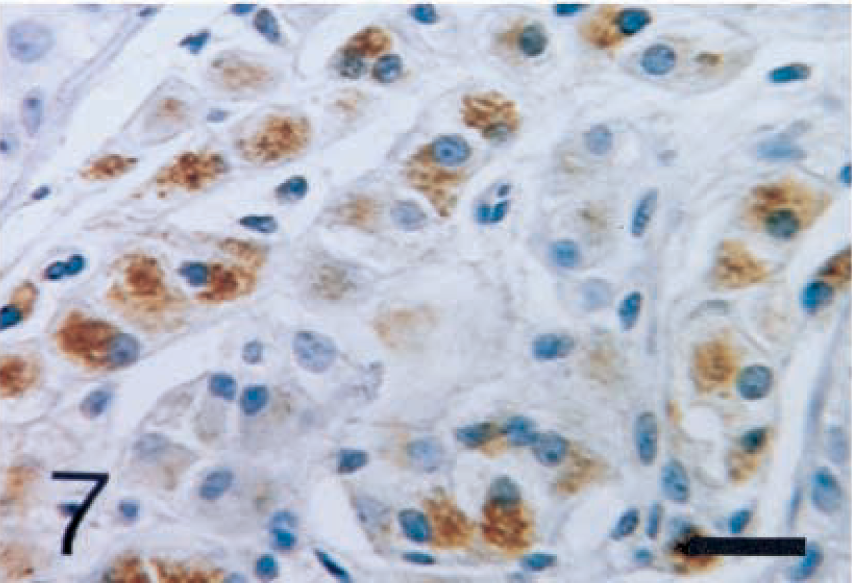
Normal testis; dog. Interstitial cells of Leydig show strong reactivity with anti-porcine inhibin-βA chain antibody. ABC complex method, Mayer's hematoxylin counterstain. Bar = 25 µm.
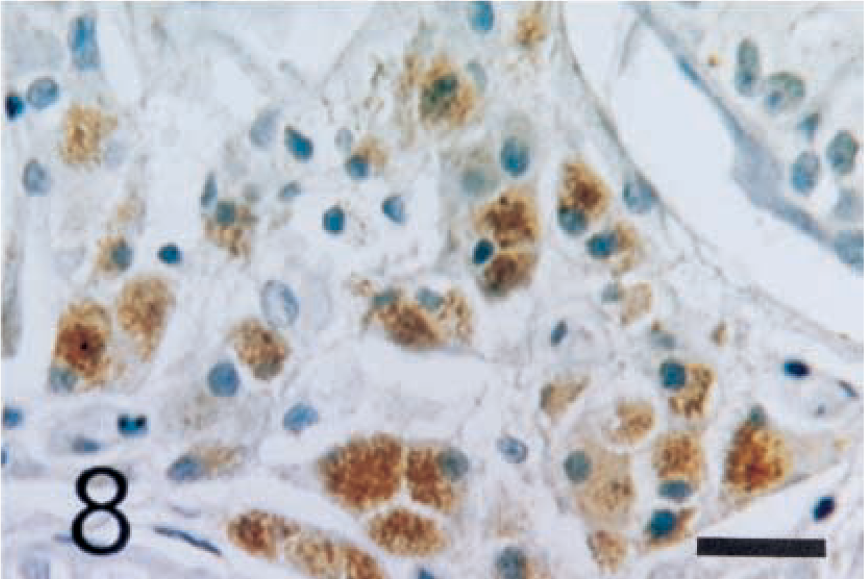
Normal testis; dog. Interstitial cells of Leydig show strong reactivity with anti-human 3β-HSD antibody. ABC complex method, Mayer's hematoxylin counterstain. Bar = 25 µm.
Concentrations of plasma inhibin
The results of radioimmunoassay for plasma inhibin are summarized in Fig. 9. Leydig cell tumors had a higher concentration of ir-inhibin than did Sertoli cell tumors and seminomas and normal testes. Differences in plasma concentration of ir-inhibin were significant (P < 0.05) between the Leydig cell tumor group and the other three groups (Fig. 10). In one dog, the concentration of ir-inhibin in the plasma was markedly decreased by surgical excision of the Leydig cell tumor (Fig. 9).
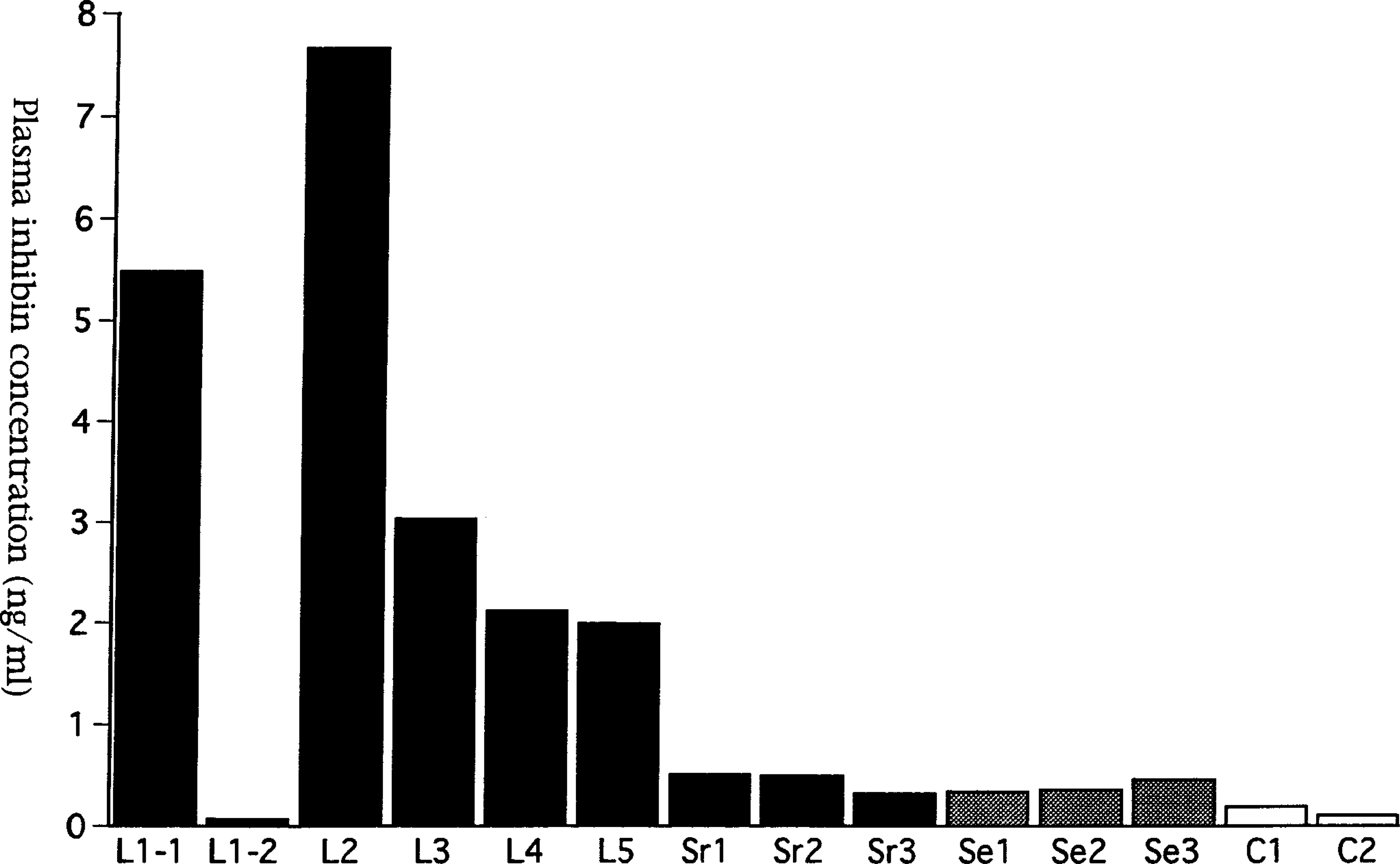
Results of radioimmunoassay for plasma inhibin of dogs. Concentrations of inhibin in Leydig cell tumor (columns L1–L5) are higher than those in Sertoli cell tumor (columns Sr1–Sr3) and seminoma (columns Se1–Se3) and control dogs (columns C1 and C2). The concentration of inhibin in the plasma is markedly decreased after surgical removal of the Leydig cell tumor in one dog (columns L1-1 and L1-2).
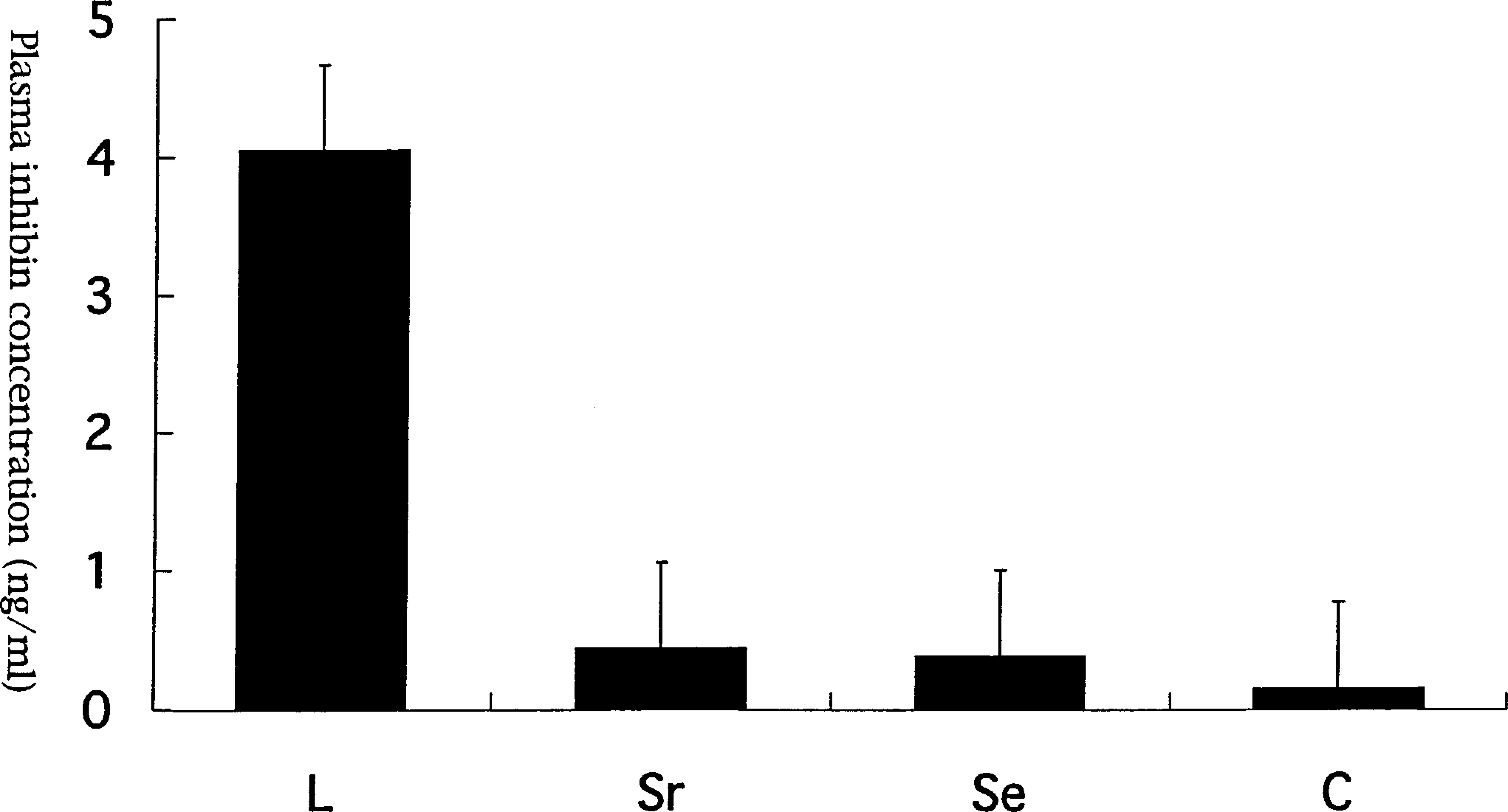
There are significant (P < 0.05) changes in plasma concentration of inhibin between Leydig cell tumor (column L) and Sertoli cell tumor (column Sr), seminoma (column Se), and control dogs (column C).
Discussion
In the human fetal testis, both inhibin-α and -βB chains were immunodetectable in Sertoli and Leydig cells at early gestation; however, the immunostaining was stronger in Sertoli cells than in Leydig cells at midgestation. 12,16,19 Inhibin in the human adult testis is mainly synthesized by Sertoli cells, with small amounts produced also in the Leydig cells. 12,19 In the rat testis, the majority of inhibin-α and -βB subunits are immunolocalized to the fetal-type Leydig cells during fetal and neonatal life, but following birth, immunoexpression in the Sertoli cells of both subunits increases markedly. 17 One research group reported an absence of immunostaining for the βA-subunit in human or rat testes. 5 Immunoreactive inhibin-α, -βA, and -βB subunits have been reported present in multiple cells in the testes of fetal, neonatal, and adult rats, and amounts of immunostainable subunits in the seminiferous epithelium are differentially regulated. 18 In the stallion, immunostaining against inhibin-α and -βB subunits revealed strong reactivity in the cytoplasm of Leydig cells and a weak reaction in Sertoli cells in the seminiferous tubules. 13,14 Immunoreactivity against the βA-subunit was also detectable in both types of cells. 13,14 3β-HSD is localized only in the Leydig cells and not in the Sertoli cells and germ cells. 13 However, localization of inhibin in canine testes has not been reliably detected. In one study, inhibin was localized in the adult testis by a immunoperoxidase technique using specific antiserum to human testicular inhibin. 5 Expression of inhibin was localized in the Sertoli cells, spermatogenia, and primary spermatocytes of dog testis. However, weak positive immunoreactivity indicating the presence of inhibin was also found in Leydig cells. 6 In the present study, inhibin was localized in normal canine testes to the interstitial Leydig cells, as in horses, 13,14 a different location than that observed in adult humans and rats. 12 The coexpression of inhibin-α, -βA, and -βB subunits in the canine interstitial cells of Leydig is consistent with the secretion of both inhibin A and inhibin B, as is the case in horses. 13 Furthermore, 3β-HSD was localized only in the Leydig cells. These findings suggest that inhibin is mainly synthesized in Leydig cells in adult canine testes.
Although little is known about the cells secreting inhibin in primary testicular tumors of humans and the older animals, 3,6,10 inhibin immunoreactivity has been biochemically estimated in human and canine gonadal tumors. Sertoli cell tumors in dogs and Leydig cell tumors in humans contained high concentrations of inhibin-like material. 3 Inhibin-α was usually demonstrated in the human Leydig cell tumors and occasionally in the Sertoli cell tumors by immunohistochemical techniques. 10 Peripheral levels of immunoassayable inhibin in dogs with Sertoli cell tumors were higher than those in normal dogs. 6 Furthermore, such tumors contained increased levels of inhibin and mRNA for the inhibin-α and -β subunits as compared with normal testes. The mRNA for the inhibin-βA subunit was not detected in tumors and testes of normal dogs. Thus, secretory products from Sertoli cell tumors were thought to be inhibin B. 6 In rat primary testicular tumors, Leydig cell tumors secreted immunoreactive and bioactive inhibin, which could be immunoneutralized by an antibody against inhibin subunits. 4 However, these reports did not include histologic and immunohistochemical studies. Therefore, the cells secreting inhibin subunits in human, canine, and rat primary testicular tumors remain unclear.
In the present study, immunohistochemical expressions of inhibin-α, -βA, and -βB subunits were detected in Leydig cell tumors but not in any Sertoli cell tumors or seminomas. 3β-HSD was localized only in the Leydig cell tumors. The immunohistochemical findings differ from the results of previous studies 3,5,6 because of the different methods of analysis used. Results of radioimmunoassay for plasma inhibin concentration suggest that inhibin is secreted from the neoplastic interstitial cells of Leydig because plasma concentrations of inhibin in dogs with Leydig cell tumors were significantly higher than those in dogs with Sertoli cell tumors or seminomas, and in one dog, the level of inhibin was markedly decreased by the removal of the Leydig cell tumor.
Immunohistochemical expression of inhibin-α, -βA, -βB subunits and 3β-HSD was localized in the interstitial cells of Leydig of normal testes and of Leydig cell tumors. This finding is supported by the result of radioimmunoassay for plasma inhibin. Inhibin in the adult canine testis is mainly synthesized by Leydig cells and may be inhibin A and inhibin B. 3β-HSD should be a useful marker to distinguish Leydig cell tumors from other tumors in canine testis.
Footnotes
Acknowledgements
We thank Dr. N. Ling for donating the inhibin-α chain, Dr. W. Vale for donating the anti-inhibin-βA chain and anti-inhibin-βB chain, and Dr. J. I. Mason for donating anti-3β-HSD antibodies. This study was supported in part by a Grant-in-Aid for Scientific Research (No. 10839014) from of the Ministry of Education, Science and Culture, Japan.
