Abstract
Granulosa cell tumors (GCTs) are common ovarian neoplasms in the mare and bitch that can be challenging to diagnose on histopathology. Inhibin has long been the standard immunohistochemical (IHC) marker for GCTs; however, anti-Müllerian hormone (AMH) has not been evaluated widely as an IHC marker in the bitch and mare. We compared the efficacy of AMH and inhibin as IHC markers in canine and equine GCTs. We selected retrospectively 18 equine and 15 canine cases. All equine tumors were dominated by a cystic pattern; canine tumors often had solid patterns. Both inhibin and AMH had similar punctate cytoplasmic patterns of immunolabeling, although labeling intensity was variable; distribution and intensity of labeling were unrelated to the histomorphologic pattern. Labeling for AMH occurred in 12 of 15 canine and 18 of 18 equine cases. Labeling for inhibin occurred in 15 of 15 canine and 18 of 18 equine cases. AMH in equine GCTs often had stronger immunolabeling than inhibin, and granulosa cells were labeled more extensively. Inhibin and AMH performed comparably in bitches, but AMH had more diffuse immunolabeling than inhibin in mares.
Granulosa cell tumor (GCT) is a commonly diagnosed ovarian neoplasm in the mare and bitch, belonging to a broad class of sex-cord stromal tumors. Although in mares, the classic honeycomb pattern imparted by the cystic gross and histomorphology of GCTs often allows for a straightforward diagnosis, canine GCTs exist in a broader variety of histomorphologic patterns.5,7,12 Inhibin, a dimeric glycoprotein of the tumor growth factor–beta superfamily expressed by granulosa cells, has long been the standard immunohistochemical (IHC) marker for GCTs. 3 Anti-Müllerian hormone (AMH), a homodimeric protein that belongs to the same superfamily and is expressed by the granulosa cells of preantral and small antral follicles in the postnatal ovary of females of many mammal species, has not been evaluated as an IHC marker in the dog and horse, to our knowledge. 2
In mares, AMH is a reliable serum marker to monitor GCTs.1,11 In 2 case studies, expression of AMH by GCTs was correlated with the serum concentrations and demonstrated consistent labeling in the GCTs.1,2 Expression of AMH and inhibin has additionally been evaluated in bovine GCTs, revealing positive immunolabeling for AMH in all GCT cases assessed. 6 Based on these studies, we investigated the potential use of AMH as an IHC marker in the bitch and mare, compared with inhibin immunohistochemistry.
A retrospective search of equine and canine GCTs was performed in the biopsy database of the New York State Animal Health Diagnostic Center (College of Veterinary Medicine, Cornell University, Ithaca, NY, USA) between 2004 and 2019. We initially selected 20 equine and 19 canine cases, refined to 18 equine and 15 canine cases based on histomorphologic criteria assessed on H&E-stained sections and availability of tissue blocks. No clinical information was available for 5 of 18 horses and 5 of 15 dogs. For those that did have clinical histories, behavioral issues were most reported in mares (8 of 18), including stallion-like behavior and conspecific aggressiveness. The most frequent concern in the evaluated dogs was abnormal vaginal discharge (4 of 15) and abnormal cycling (3 of 18). Blood serum results were available in 6 of 18 horses, including: elevated AMH and testosterone (1 of 18), elevated inhibin (1 of 18), elevated AMH (1 of 18), and elevated AMH, inhibin, and testosterone (1 of 18). Testosterone and inhibin levels were each within the RI in 2 of 18 mares. AMH was reportedly within the RI in 2 of 18 mares. A positive ELISA result for AMH was reported in a single dog (1 of 15).
Formalin-fixed, paraffin-embedded tissues were processed for immunohistochemistry; glass-mounted paraffin scrolls were dewaxed (Bond dewax solution; Leica), followed by heat epitope retrieval for 30 min, and application of peroxide block (Leica) for 5 min. The slides were then immunolabeled for AMH (anti-Müllerian inhibiting substance [MIS] antibody, sc-166752; Santa Cruz Biotechnology), at a dilution of 1:200 for 90 min, and inhibin-α (NCL-L-inhibinA; Leica), at a dilution of 1:100 for 15 min. Hematoxylin was then applied for 5 min as a counterstain. Labeling was performed with an automated immunohistochemistry system (Bond Max; Leica). Positive control tissue consisted of normal canine and equine ovary as well as internal positive control. Labeling for both antibodies was cytoplasmic and granular.
In canine GCTs, the most common histologic pattern was solid (11 of 15), followed by mixed solid and cystic (3 of 15), and cystic (1 of 15). In equine GCTs, the most common pattern was cystic (10 of 18), followed by mixed solid and cystic (8 of 18). Call–Exner bodies occurred in 5 of 15 canine GCTs and 6 of 18 equine GCTs. Neoplastic granulosa cells in neoplasms in a solid pattern were polygonal to vaguely tapered, with variable amounts of clear-to-light eosinophilic, bland to faintly granular cytoplasm, organized in variably well-defined nests supported by a delicate fibrovascular stroma. In cystic patterns, neoplastic granulosa cells lining the cysts had similar cytoplasmic features, were polygonal-to-cuboidal, and organized in a piled-up, disorganized fashion. In all patterns, the degree of atypia, including anisokaryosis and anisocytosis, was mild-to-moderate, and the mitotic count varied widely.
Labeling for AMH occurred in 12 of 15 canine GCTs; 4 cases had immunolabeling of >50% of the neoplastic population, and 8 samples had immunolabeling in <50% of the neoplastic population (Figs. 1, 3, 5; Table 1). Labeling for AMH occurred in 18 of 18 equine GCTs; 17 samples had immunolabeling in >50% of the population, and 1 sample had immunolabeling in <50% of the neoplastic population (Figs. 7,9, 11; Table 2).
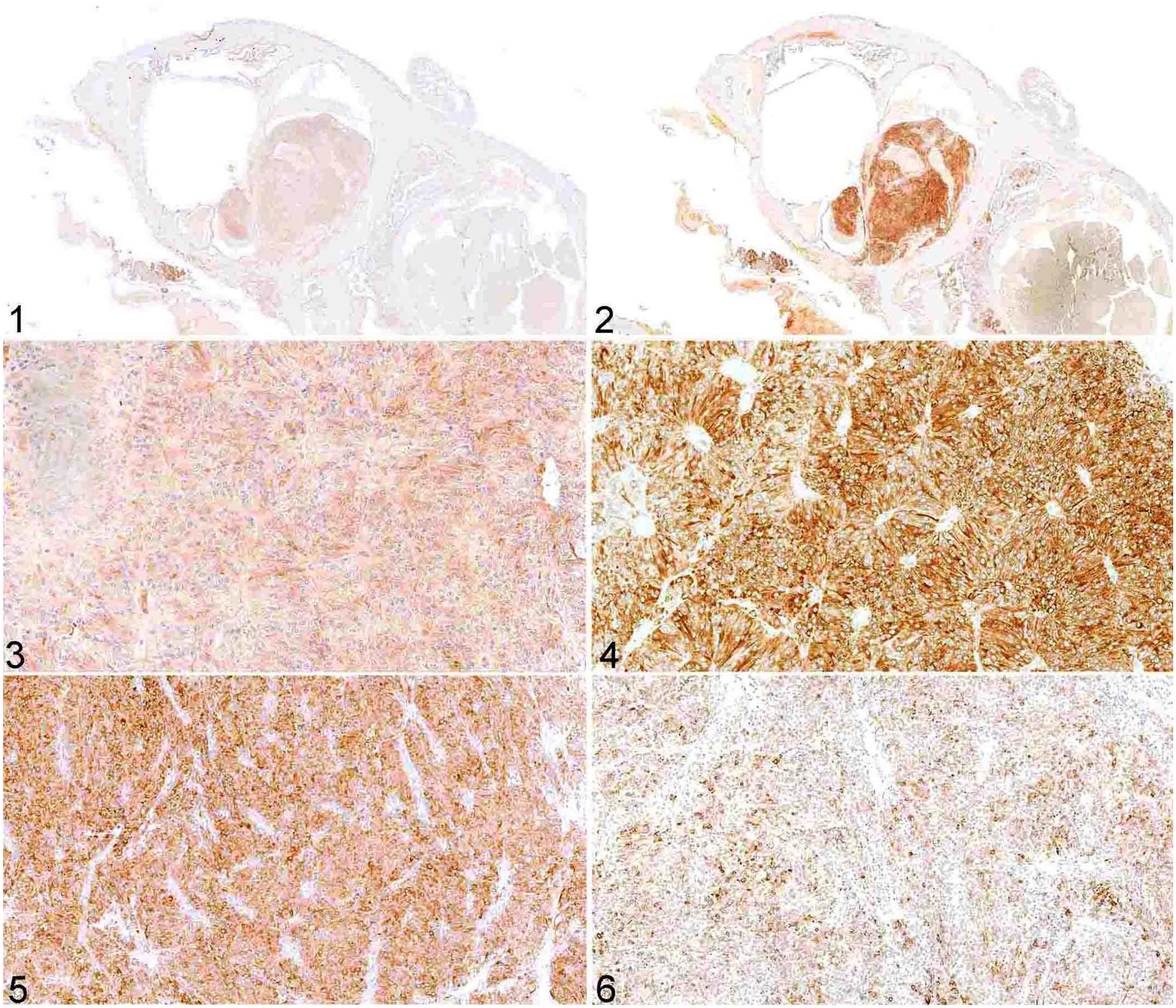
Canine granulosa cell tumor; immunohistochemical staining in serial sections.
Inhibin and anti-Müllerian hormone (AMH) immunohistochemistry in canine granulosa cell tumors.
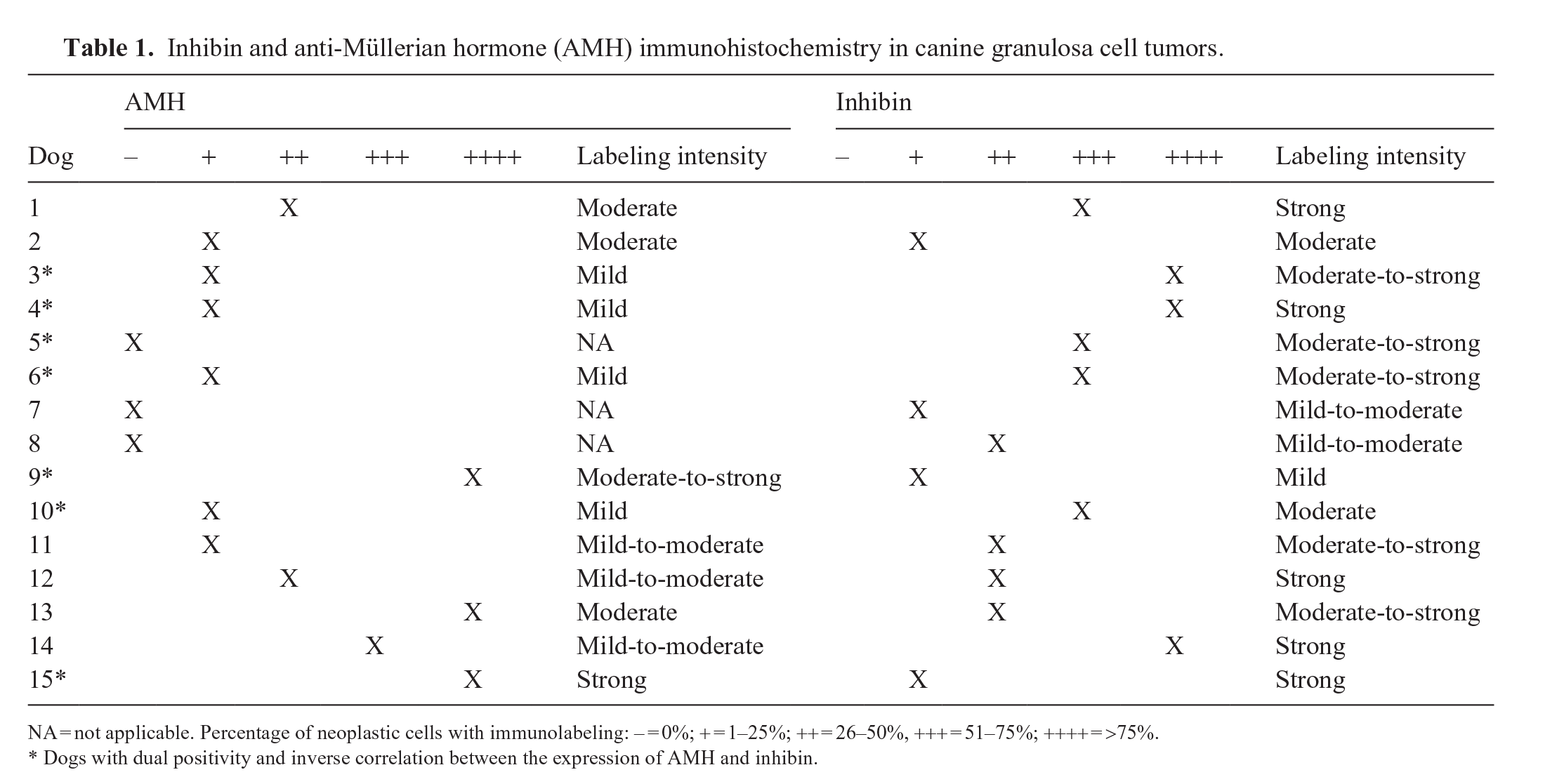
NA = not applicable. Percentage of neoplastic cells with immunolabeling: – = 0%; + = 1–25%; ++ = 26–50%, +++ = 51–75%; ++++ = >75%.
Dogs with dual positivity and inverse correlation between the expression of AMH and inhibin.
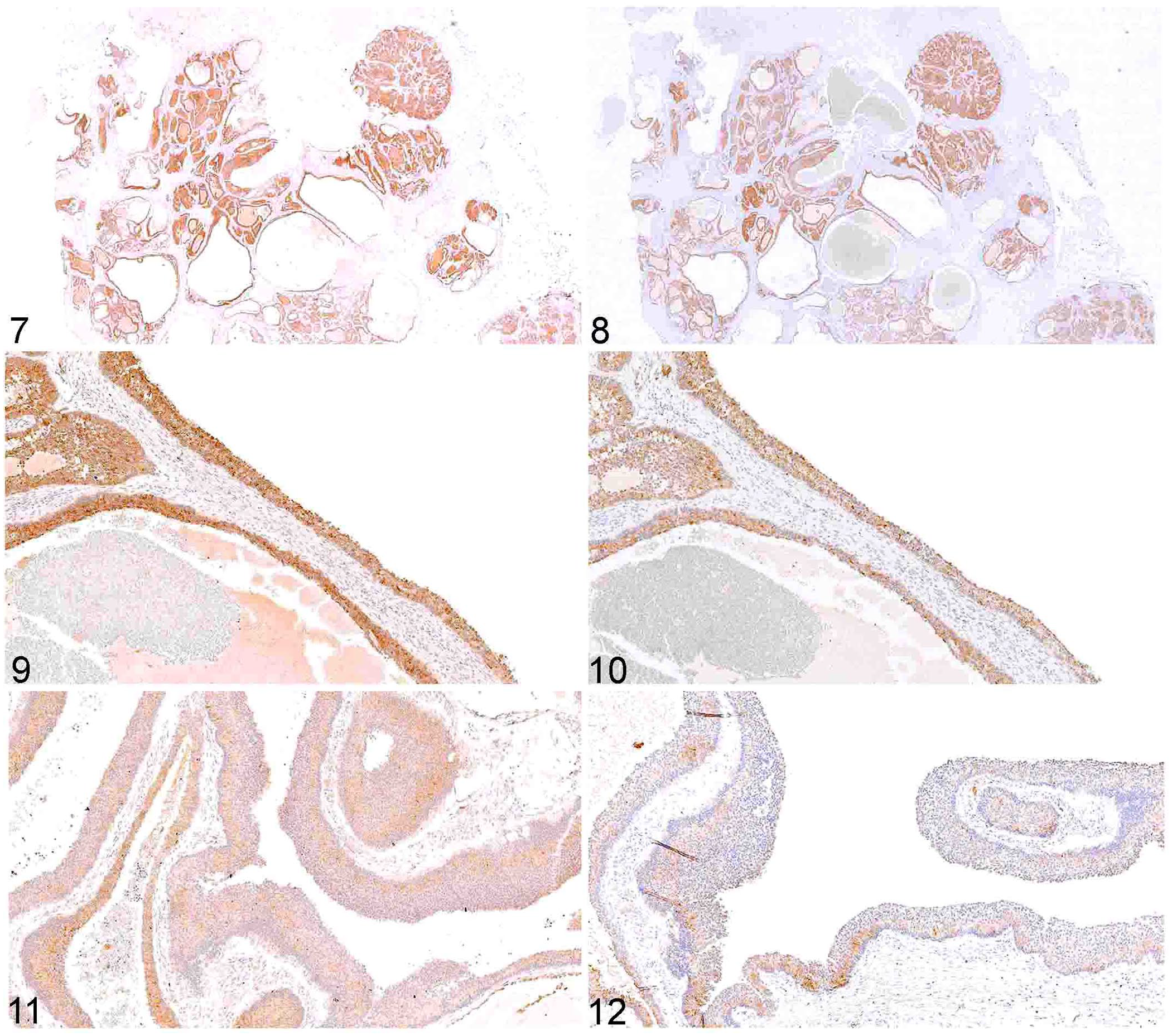
Equine granulosa cell tumor; immunohistochemical staining in serial sections.
Inhibin and anti-Müllerian hormone (AMH) immunohistochemistry in equine granulosa cell tumors.
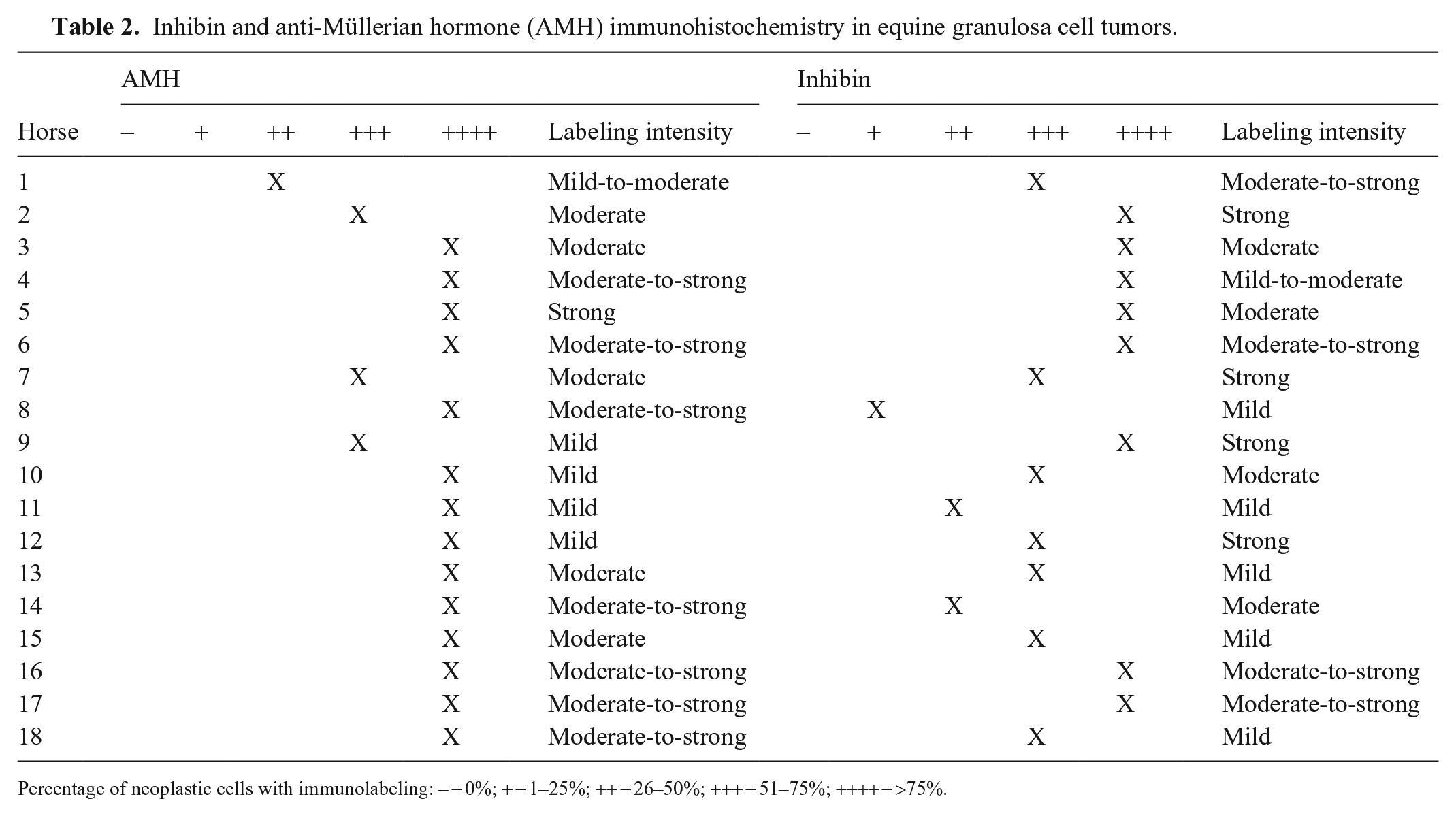
Percentage of neoplastic cells with immunolabeling: – = 0%; + = 1–25%; ++ = 26–50%; +++ = 51–75%; ++++ = >75%.
Labeling for inhibin occurred in 15 of 15 canine GCTs; 7 samples had immunolabeling of >50% of the neoplastic population, and 8 samples had immunolabeling in <50% of the neoplastic population (Figs. 2, 4, 6; Table 1). Labeling for inhibin occurred in 18 of 18 equine GCTs; 15 samples had immunolabeling of >50% of the neoplastic population, and 3 samples had immunolabeling in <50% of the neoplastic population (Figs. 8, 10, 12; Table 2).
In equine GCTs, the intensity of the labeling was stronger for AMH than for inhibin. In comparison, the intensity of the labeling was variable in canine GCTs for both AMH and inhibin. In 7 canine GCTs with labeling for both proteins, an inverse correlation between the expression of AMH and inhibin was observed; in these tumors, neoplasms with a solid pattern had stronger labeling for inhibin than for AMH, and cystic and mixed tumors had stronger labeling for AMH than for inhibin (Table 1). Whether this difference in expression is related to the degree of differentiation, density of neoplastic cells, or even represents a prognostic factor, is unclear.
Although immunohistochemistry is not always necessary in the diagnosis of well-differentiated GCTs, increasing the available panel of IHC markers for these tumors is critical not only to accurately diagnose poorly differentiated GCTs, but also in differentiating such neoplasms from malignant ovarian epithelial tumors.5,6,8,12 The use of additional markers in poorly differentiated ovarian tumors, such as CK7, has been reported as an accurate means to differentiate ovarian epithelial tumors (positive) from GCTs (negative), especially given that Call–Exner bodies, although fairly specific for GCTs, are not a consistent feature.9,12
The results that we obtained for AMH correlate with studies conducted in equine and bovine GCTs and suggest that AMH may be used in a broader range of veterinary species for immunohistochemistry studies.1,2,10 Furthermore, a study conducted in nonhuman primates reported occasional negative results for inhibin in simian GCTs (8 of 11), similarly to our canine GCT results (12 of 15). 4 Given the consistent results obtained in both species and the positive correlation with inhibin staining, AMH can be considered a valid IHC tool to be used in the diagnosis of both canine and equine GCTs.
Footnotes
Declaration of conflicting interests
The authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors declared that they received no financial support for their research and/or authorship of this article.
