Abstract
Mixed germ cell sex cord-stromal tumors (MGSCTs) of the testis are rare in dogs. We describe the histopathology and immunohistochemical characteristics of an MGSCT associated with a Leydig cell tumor in a cryptorchid testis. Histologically, MGSCT consisted of two nodules of seminiferous tubules lined by germ cells and Sertoli cells in variable proportions. Germ cells had variable size and nuclear features, with frequent giant cells. Germ cells were evenly mixed with Sertoli cells or located in the center of tubules. Markers that labeled mainly germ cells and few or no Sertoli or Leydig cells were calretinin, KIT, and PGP 9.5. E-cadherin, GATA-4, inhibin-α (INH-α), and neuron-specific enolase (NSE) were predominantly detected in Sertoli cells, whereas melan A was particularly expressed in Leydig cells and vimentin in all three cell types. OCT3/4 was not detected in any cell type. Although more cases of canine MGSCT need to be examined, our results suggest that an immunohistochemical panel of E-cadherin, GATA-4, INH-α, KIT, NSE, PGP 9.5, and melan A will help distinguish the three main cell types in canine testicular germ cell and sex cord-stromal tumors.
Testicular tumors are common in dogs; however, mixed germ cell sex cord-stromal tumors (MGSCTs) are rarely reported. 21, 27, 35 These neoplasms consist of intimately associated germ cells and Sertoli cells. In dogs, they are usually unilateral, with only one report of bilateral involvement. 27 In a survey of 262 testicular neoplasms in dogs, 18 MGSCTs were found, and only 1 also contained a Leydig cell tumor in the same testis. 21 The current report describes the histopathologic and immunohistochemical features of a canine testis with MGSCT and concurrent interstitial (Leydig) cell tumor.
A 9-year-old male Shetland Sheepdog had a cryptorchid testis. The cryptorchid testis was removed, fixed in 10% neutral buffered formalin, and submitted to the Purdue University Animal Disease Diagnostic Laboratory for histopathologic review. The other testis was not submitted.
On gross examination, the testis had multiple nodules (Fig. 1A). Microscopically, two 1.5 × 1.9 cm and 0.8 × 0.5 cm, respectively, unencapsulated nodules were composed of seminiferous tubules distended by two cell populations. (Fig. 1B). One population of cells (Sertoli cells) formed multiple layers within tubules, with the outer cells oriented perpendicularly to the basement membrane (Fig. 1C). In some regions, the cells were streaming and lacked polarity. Sertoli cells were columnar, with abundant granular to indistinctly vacuolated, pale eosinophilic cytoplasm and distinct cell margins. Neoplastic Sertoli cells had a small to medium-sized oval nucleus, with finely stippled chromatin and one to three small nucleoli. There was moderate anisocytosis and anisokaryosis. The mitotic index was 1 per ten 400× fields. Neoplastic Sertoli cells often lined tubules with a second population of polygonal to round cells interpreted as germ cells. Germ cells were at the center of many seminiferous tubules, but in some tubules they were mixed with neoplastic Sertoli cells. Germ cells had a scant to moderate amount of amphophilic to basophilic, sometimes vacuolated, cytoplasm. Few PAS-positive granules were observed in rare germ cells. Nuclei were round to oval with smooth nuclear membrane, although in some karyomegalic cells the nuclear membrane was irregular. Germ cells had single or multiple nucleoli. Three germ cell sizes were observed. The most common cell type was 12 to 16 μm in diameter; the other two cell types were much less common and consisted of 1) lymphocyte-like cells 5 to 8 μm in diameter with condensed nuclei or 2) bizarre giant cells 50 μm in diameter or larger (Fig. 1D). Mitoses were common in the germ cell population, with up to 6 per 400× field. The percentage of germ cells and Sertoli cells within a tubule varied, with some containing mostly Sertoli cells and others with a preponderance of germ cells. Tubules with only one cell population were also observed. Most tubules lined mainly by Sertoli cells were surrounded by more abundant fibrous stroma than were those with mainly germ cells. The stroma contained scattered lymphocytes, plasma cells, and foamy and pigment-laden macrophages. Some seminiferous tubules in the periphery of this mass were lined by a single layer of nonneoplastic Sertoli cells with no spermatogenesis. Histologic findings were consistent with MGSCT. 16 The testis had another, smaller (0.6 × 0.5 cm), circumscribed but unencapsulated mass composed of solid cords and packets of variably sized polygonal cells with eosinophilic cytoplasm containing single or multiple clear, distinct vacuoles that rarely contained yellow-brown pigment granules (Figs. 1E, 1F). These cells had a round to oval nucleus, finely stippled chromatin, and usually one prominent central nucleolus. There were rare binucleate cells. Mitotic figures were not apparent. Sparse fibrovascular stroma contained distended blood vessels. Morphologic features were typical of interstitial (Leydig) cell tumor. 16 The final diagnosis in this testis was MGSCT with an interstitial (Leydig) cell tumor.
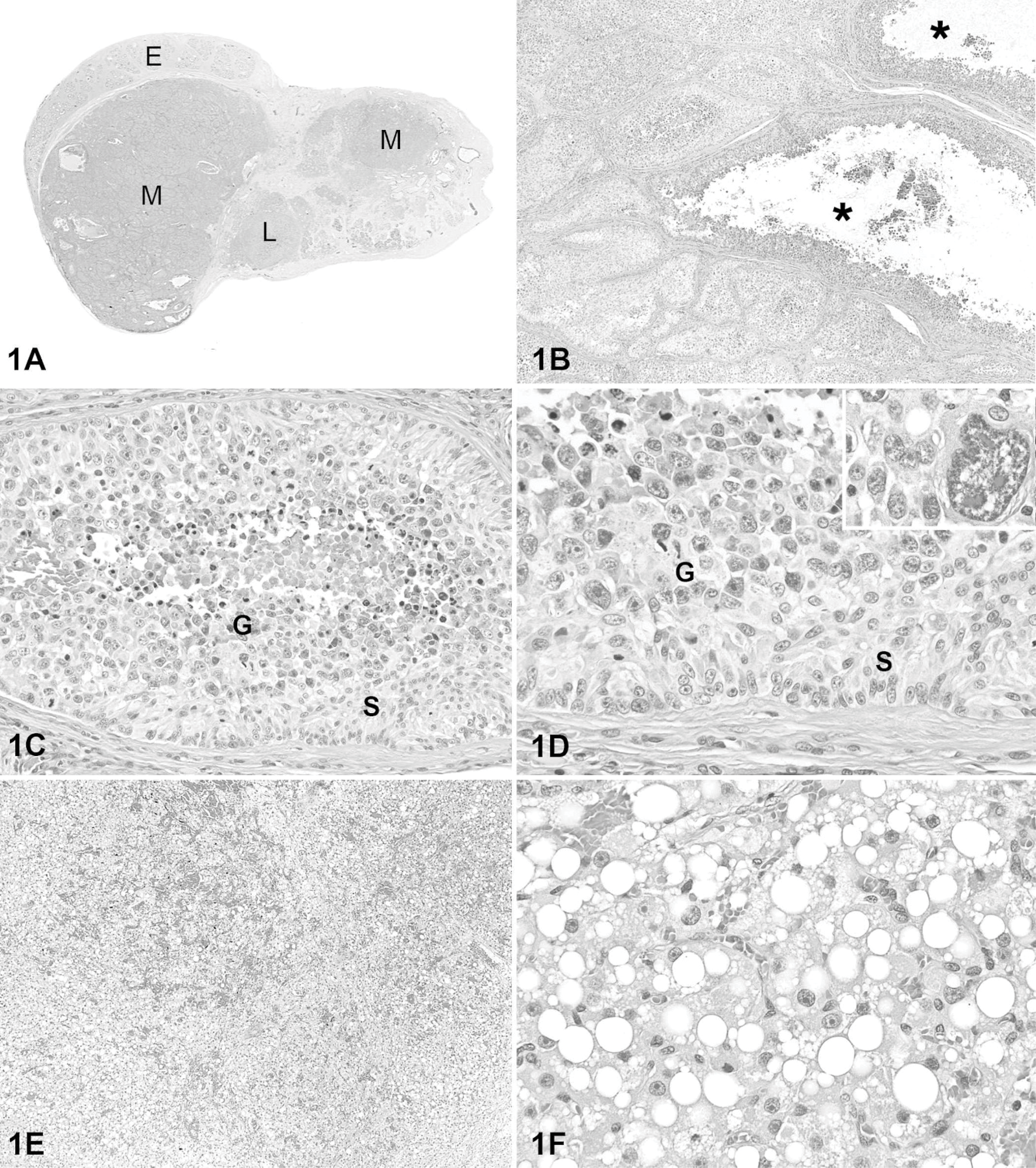
Mixed germ cell sex cord-stromal and Leydig cell testicular tumors; dog.
To further characterize the tumor cells, formalin-fixed, paraffin-embedded tissue sections were used for immunohistochemistry (Table 1). Heat-induced epitope retrieval (HIER) in a decloaker chamber was used for all antibodies except cytokeratins (proteinase K), PGP 9.5 (no epitope retrieval), and vimentin (proteinase K). Staining protocols have been published previously. 23– 25 Staining was graded as: 0 (no staining), 1+ (<10% positive cells), 2+ (10%–50% positive cells); and 3+ (>50% positive cells). The location of the staining (membrane, cytoplasmic, nuclear) was also recorded. Positive tissue controls from dog were used for each antibody. The negative reagent control was rabbit (for polyclonal antibodies) or mouse immunoglobulins (for monoclonal antibodies). Peroxidase-DAB detection systems were used to demonstrate the immunologic reaction. Sections of two formalin-fixed, paraffin-embedded normal testes also were immunostained with the same antibodies.
Antibody reagents used in immunohistochemistry.
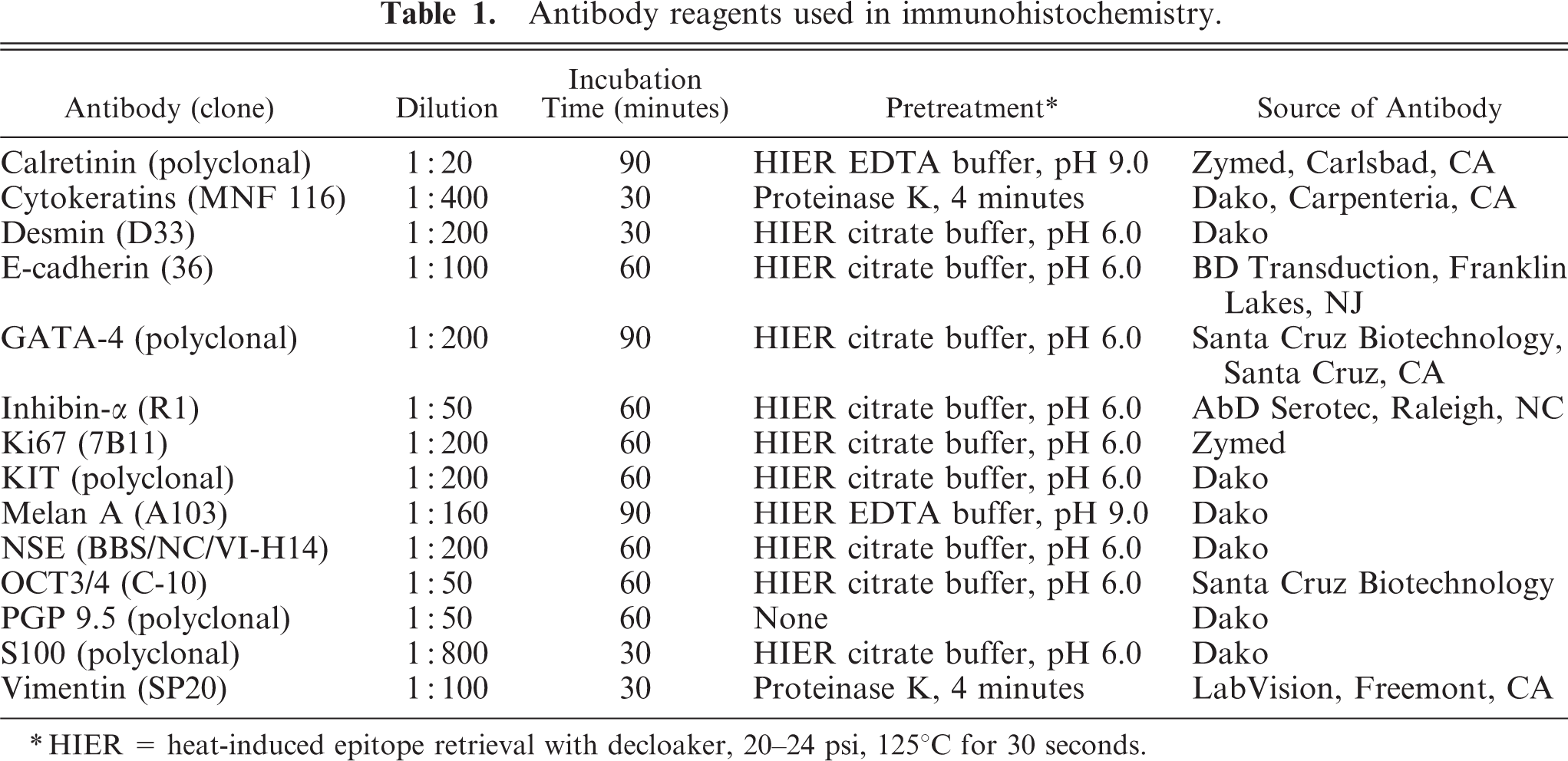
∗HIER = heat-induced epitope retrieval with decloaker, 20–24 psi, 125°C for 30 seconds.
Immunohistochemical staining results of mixed germ cell sex cord-stromal and Leydig cell tumors and normal testis.∗
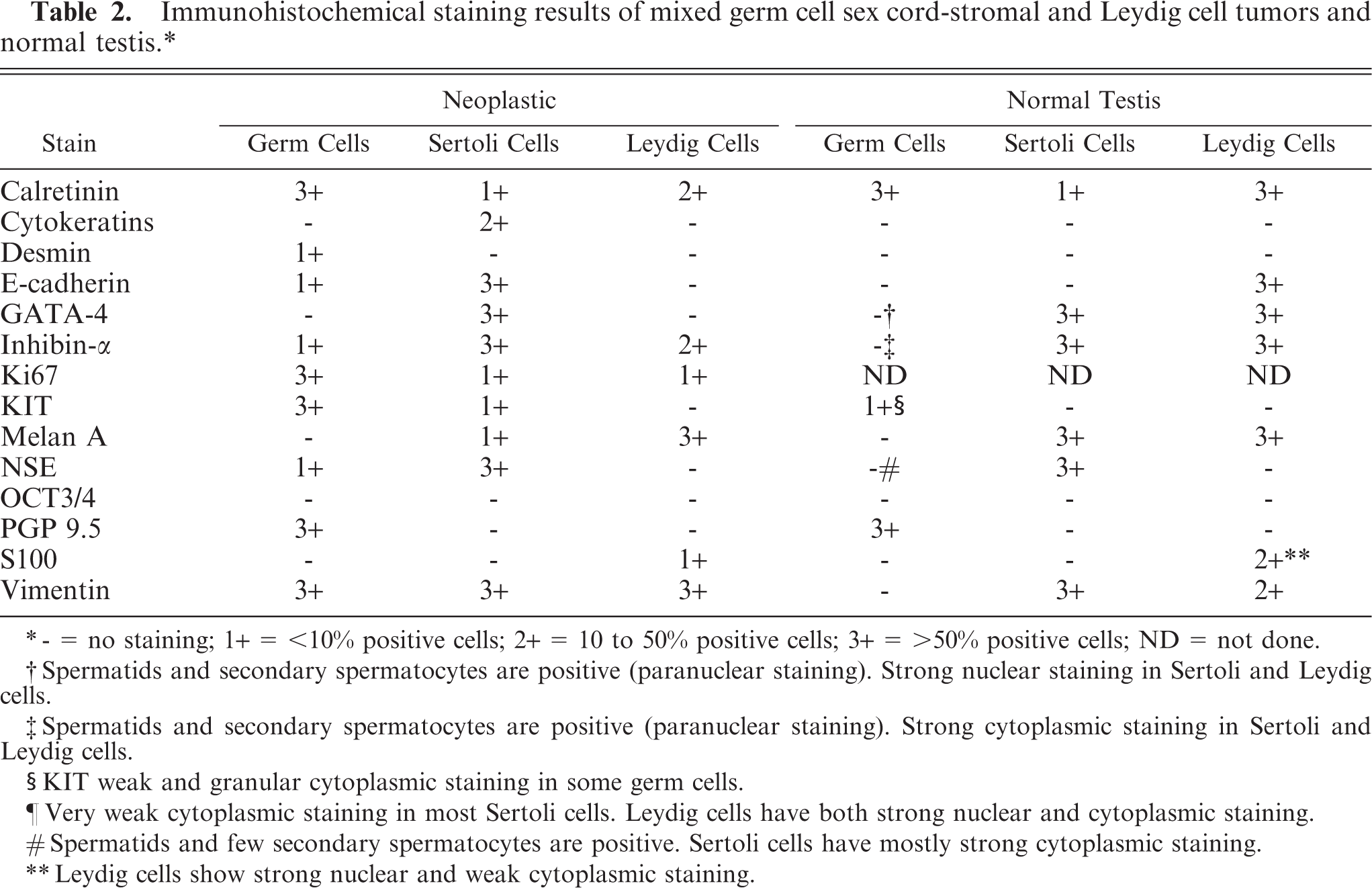
∗- = no staining; 1+ = <10% positive cells; 2+ = 10 to 50% positive cells; 3+ = >50% positive cells; ND = not done.
†Spermatids and secondary spermatocytes are positive (paranuclear staining). Strong nuclear staining in Sertoli and Leydig cells.
‡Spermatids and secondary spermatocytes are positive (paranuclear staining). Strong cytoplasmic staining in Sertoli and Leydig cells.
§KIT weak and granular cytoplasmic staining in some germ cells.
¶Very weak cytoplasmic staining in most Sertoli cells. Leydig cells have both strong nuclear and cytoplasmic staining.
#Spermatids and few secondary spermatocytes are positive. Sertoli cells have mostly strong cytoplasmic staining.
∗∗Leydig cells show strong nuclear and weak cytoplasmic staining.
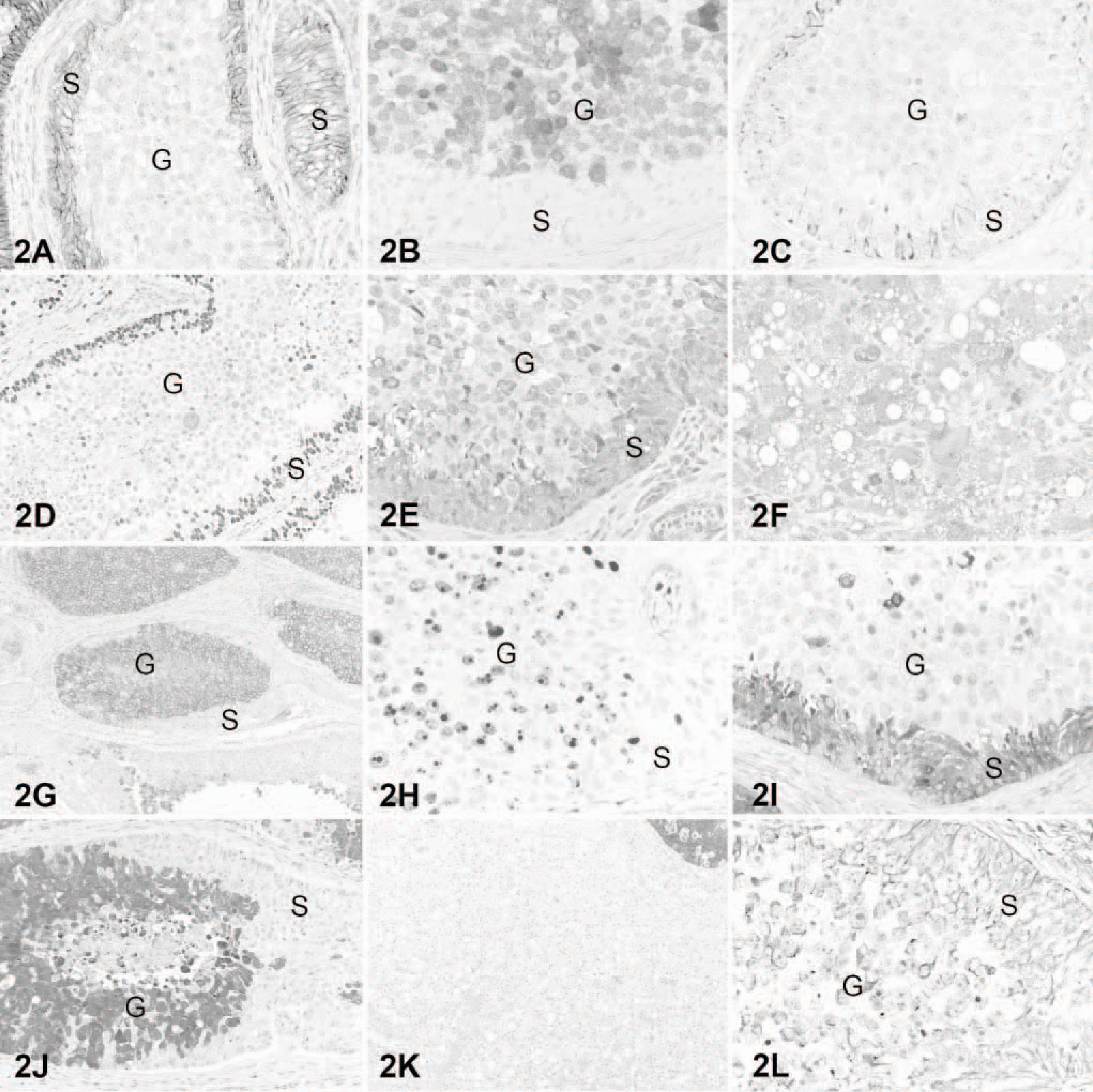
Mixed germ cell sex cord-stromal and Leydig cell testicular tumors; dog. Comparison of immunohistochemical staining of the mixed germ cell sex cord-stromal (
We have described the histologic and immunohistochemical characteristics of a canine MGSCT associated with a Leydig cell tumor. Canine MGSCTs are rare, with only a few reports. 21, 27, 35 To our knowledge, concurrent MGSCT and Leydig tumor in the same testis has been reported in only one dog. 21 We have added to the descriptions of canine MGSCTs with detailed histopathologic and extensive immunohistochemical characterization. In our dog, neoplastic germ cells had microscopic features compatible with human spermatocytic seminoma (variable cell size, giant cells, high mitotic rate, lack of cytoplasmic glycogen) rather than seminoma. 29 The germ cell compartment of at least one of the reported canine MGCSTs, diagnosed as gonadoblastoma, had microscopic features of seminoma. 27 The affected testis was descended and had microscopic features of gonadal dysgenesis. 27 In our case, the affected testis was cryptorchid with no evidence of dysgenesis.
The immunohistochemical profile of the current canine MGSCT has some similarities with other canine MGSCTs. NSE was expressed strongly in the Sertoli cell compartment and rarely in germ cells. A previous study of multiple MGSCTs only found consistent NSE staining in Sertoli cells. 21 Those authors did not find reactivity in the germ cell or Sertoli tumor compartments for cytokeratins and desmin, but strong staining for vimentin. 21 In our case, there was patchy staining of Sertoli cells for cytokeratins and strong staining of a few germ cells for desmin. These differences could reflect the use of different antibodies, detection systems, or antigen retrieval methods. Strong staining of germ cells for KIT in our MGSCT was also present in the germ cell compartment of a canine gonadoblastoma. 27 GATA-4 is a marker of Sertoli cell differentiation in mice and pigs. 19, 37 To our knowledge, there are no reports of immunohistochemical characterization of canine testicular tumors with antibodies to GATA-4. In a series of testicular tumors, we found that this marker is consistently expressed in Sertoli cell tumors and not in germ cell tumors (J. Ramos-Vara personal observation). The cause for the variation in immunostaining between MGSCT and normal testicular cells (Table 2) is unknown. However, upregulation or downregulation of specific genes might result in de novo expression or loss of expression of specific proteins.
Based on our and others' results, KIT and PGP 9.5 are excellent markers for canine testicular germ cells; GATA-4, NSE, INH-α, and E-cadherin are good markers to distinguish the stromal component of MGSCTs; melan A and INH-α are excellent but not completely specific markers for interstitial (Leydig) cell tumors. In summary, for the characterization of canine MGSCTs we recommend an antibody panel including E-cadherin, GATA-4, INH-α, KIT, NSE, and PGP 9.5. Although immunohistochemistry did not assist in the diagnosis of the MGSCT in the case examined, we conclude that the use of immunohistochemistry in testicular tumors may help in distinguishing difficult cases, such as malignant Sertoli cell tumor, which may be interpreted as seminoma. 6 However, to validate the use of these markers in canine testicular tumors, large series of germ and sex cord-stromal tumors need to be examined.
Footnotes
Acknowledgement
We appreciate the technical expertise of Dee DuSold on immunohistochemistry.
