Abstract
Senescent cells accumulate with age but tissue-based studies of senescent cells are limited to selected organs from humans, mice, and primates. Cell culture and xenograft studies have indicated that senescent cells in the microenvironment may play a role in tumor proliferation via paracrine activities. Dogs develop age-related conditions, including in the testis, but cellular senescence has not been confirmed. We hypothesized that senescent cells accumulate with age in canine testes and in the microenvironment of testicular tumors. We tested the expression of the established senescence markers γH2AX and p21 on normal formalin-fixed, paraffin-embedded testes from 15 young dogs (<18 months of age) and 15 old dogs (7–15 years of age) and correlated the findings with age-dependent morphological changes. A statistically significant age-dependent increase in the percentage of p21-expressing cells was observed for testicular fibroblasts (4-fold) and Leydig cells (8-fold). However, p21-expressing cells were still a rare event. In contrast, the percentage of γH2AX-positive cells did not increase with age. P21- and γH2AX-expressing cells were rare in the microenvironments of tumors. Age-dependent morphological changes included an increased mean number of Leydig cells per intertubular triangle (2.95-fold) and a decreased spermatogenesis score. To our surprise, no age-related changes were recorded for interstitial collagen content, mean tubular diameter, and epithelial area. Opposed to our expectations based on previous in vitro data, we did not identify evidence of a correlation between age-associated accumulation of senescent cells and testicular tumor development. Understanding the role of the microenvironment in senescence obviously remains a challenging task.
Cellular senescence is defined as irreversible growth arrest and develops in response to diverse stressors. 14 In young individuals, cellular senescence is an important mechanism to prevent uncontrolled proliferation of cells after DNA-damaging insults. However, at later stages in life, senescent cells are also thought to drive age-related pathologies, including cancer. 3 This seeming paradox has been explained by the theory of antagonistic pleiotropy, also known as the pay later theory, which states that some genes offer a benefit in young (re)productive years while having negative effects that manifest at an age beyond the reproductive phase. 57
Current knowledge on senescence has primarily been gained from cell culture studies.
14
In 1965, Hayflick and Moorhead
16
introduced the concept of replicative senescence, showing that human embryonic fibroblasts had a limited life span, after which they permanently arrested their cell cycle, termed the
Previous studies, mostly in mice and men, have shown that senescent cells accumulate with age, with few studies in other species (eg, baboons). 11,17,34,42,55 The most commonly investigated tissue is the skin, followed by a few studies on brain, liver, pancreas, and hematopoietic stem cells. 4,11,21,34,39,42,55 Senescent cells all exhibit the same characteristics: an altered, enlarged morphology; irreversible cell cycle arrest with resistance to apoptosis; and the expression of a senescence-associated secretory phenotype. 3 This phenotype includes numerous paracrine activities such as the secretion of proinflammatory cytokines, as well as chemokines, growth factors, and proteases that have both deleterious (ie, tumor-promoting) and beneficial (ie, tissue repair) effects strongly dependent on the physiological context. 3,8,50 Studies with mice have shown that the senescence-associated secretory phenotype has the ability to promote optimal repair of damaged skin and liver with restriction of fibrosis. 1,27,29,45 Other studies, however, have suggested that senescent cells drive age-related degenerative and hyperplastic pathologies, 3 such as prematurely aged skin, neurodegeneration, joint diseases, benign prostate hyperplasia, and atherosclerotic lesions. 2,7,43,44,46,52,53
The theory that accumulated senescent cells create a microenvironment that is favorable for tumor development and growth was supported by co-culture as well as xenograft studies. Senescent fibroblasts stimulate the growth of preneoplastic as well as neoplastic but not normal epithelial cells. 31 Also, injection of senescent fibroblasts significantly stimulates the proliferation of mouse and human epithelial tumor cells in immunocompromised mice. 31 To date, there is a substantial lack of in vivo data that would support this theory.
The identification of biomarkers for senescent cells is rather challenging as no known single marker is considered specific. 10 Senescence-associated growth arrest starts with the DNA damage response, characterized by activation of ATM/ATR, leading to phosphorylation of Ser-139 of histone H2AX molecules (γH2AX). γH2AX then facilitates the activation of the p53/p21 or p16INK4a/pRB (retinoblastoma protein) tumor suppressor pathways. 5 In the present study, 1 upstream marker was selected, γH2AX, which is known to be essential in any DNA damage response and thought to be one of the primary and most important initiators of senescence. For downstream marker selection, we focused on the classical replicative senescence pathway associated with telomere-initiated senescence. 18,36 Telomere shortening triggers senescence through the p21-pathway but not p16. Furthermore, it has been shown that p16-mediated senescence is not associated with a senescence-associated secretory phenotype. The senescence-associated secretory phenotype is assumed to be responsible for tumor development (eg, via metalloproteinases [MMP]-3, vascular endothelial growth factor [VEGF], interleukin [IL]–6, IL-8, amphiregulin, growth related oncogenes [GROs]). 9 The phosphorylated histone protein H2AX and p21 have often been used as senescence markers in murine and human tissues. 14,17,32,48,55 So far, the presence of cellular senescence has not been confirmed in the dog, although aging dogs develop several age-related diseases, in particular in the testis, that appear at least in some aspects similar to those in humans.
Consequentially, we hypothesized that senescent cells accumulate with age in canine testes and in the microenvironment of testicular tumors. Hereby, senescent cells could play a role in tumor proliferation via paracrine activities. We further expected morphological changes that reflect the aging of canine testes (ie, decreased mean tubular diameter, increased relative collagen content, or increased Leydig cell number per area). Thus, in the present study, we tested the 2 most established markers of senescence on canine tissues and correlated their expression with age-dependent morphological changes. Furthermore, we asked whether the proportion of senescent marker-positive cells changes in the microenvironment of testicular tumors. The same was investigated for cryptorchid testes without tumors, as they are predisposed for tumor development, possibly due to premature aging.
Materials and Methods
Case Selection
Healthy normal testes from 15 young dogs (range, 9–18 months of age; 9 purebred and 6 mixed breed) and 15 testes from old dogs (range, 7–15 years; 12 purebred and 3 mixed breed) were collected (Suppl. Table S1). Dogs were considered old in accordance with the Goldston method, which correlates different weight classes with the age at which they are considered geriatric as smaller dog breeds live longer than larger breeds (Suppl. Table S2). 15 All young dogs were castrated to prevent uncontrolled reproduction or testes were collected from routine diagnostic necropsy of animals with no testicular pathology. Old dogs had been castrated for reasons such as benign prostatic hyperplasia, perineal herniation, testicular tumor, or epididymitis of the contralateral testis. Only healthy testes were included in this study, with exclusion of possibly diseased contralateral testes.
For evaluation of the tumor microenvironment, routine diagnostic tissue samples from 48 canine testicular tumors were included with 15 Leydig cell tumors (range, 8–13 years; mean, 10.5 years; 10 purebred, 5 mixed breed), 15 seminomas (range, 7–15 years; mean, 10.1 years; 13 purebred, 2 mixed breed), and 18 Sertoli cell tumors (range, 6–16 years; mean, 10.2 years; 12 purebred, 3 mixed breed), of which 3 (range, 8–11 years; 1 purebred, 2 mixed breed) had developed in cryptorchid testes. Supplemental Tables S1 and S3 contain case history and clinical information.
Also, cryptorchid testes from 3 dogs (≤2 years of age; 2 purebred and 1 mixed breed, Suppl. Table S1) were collected. Two of the dogs were castrated due to cryptorchidism and 1 collected from a dog during routine diagnostic necropsy.
Tissue samples were formalin-fixed, longitudinally cut, and paraffin-embedded, and 4-μm-thick sections were stained with hematoxylin and eosin (HE) or using the periodic–acid Schiff (PAS) reaction for independent blinded evaluation (R.K., A.B., and S.E.M.).
Histopathology and Quantification
Interstitial collagen content per area, tubular diameter, epithelial area, and number of Leydig cells per intertubular triangle (ITT) were recorded in all healthy testes of young and old dogs as well as in 3 cryptorchid testes. In addition, the spermatogenetic activity of 10 young and 10 old canine testes as well as the 3 young cryptorchid testes was evaluated using the modified Johnsen score. 24,41 These parameters had been reported to reflect species- and study-dependent age-associated changes. 33,41
To measure the relative interstitial collagen content, slides were stained with picrosirius red, scanned (200× magnification with ScanScope CS2; Leica Biosystems Imaging, DB Maarn, the Netherlands), and analyzed using image analysis software (eSlide Manager/Genie; Leica Biosystems Imaging). The percentage of collagen tissue (stained red) was measured in relation to the total area, including the tubular lumina, as well as in relation to the tissue area excluding the tubular lumina. All artifacts were excluded.
For the evaluation of tubular diameters as well as epithelial areas, 100 round tubular cross sections of each testis were examined at 100× total magnification with an Olympus BX41 microscope equipped with a Color View II Camera (Olympus Soft Imaging Solutions, Münster, Germany). Total areas of entire tubuli were put in percentual relation to areas of tubular lumina (epithelial area/tubular area) and tubular diameters were determined using the CellSens software (Olympus Soft Imaging Solutions). Ten round tubuli were examined in 10 randomly chosen representative fields.
Leydig cells were counted in each testis in 10 random ITTs on PAS-stained sections. An ITT is the area within a triangle formed by 3 touching tubuli (Suppl. Fig. S1).
Spermatogenetic activity was determined by the modified Johnsen score. This procedure (1970) 24 assigns to each of 100 tubular sections a score from 1 to 10 depending on the presence of the main cell types arranged in the order of maturity (spermatogonia, spermatocytes, spermatids, spermatozoa, and Sertoli cells) and was modified by Peters et al 41 for application to canine testes (Suppl. Fig. S2). HE-stained sections of 10 young and 10 old canine testes as well as 3 young cryptorchid testes were evaluated. For each testis, 100 round tubular cross sections were analyzed per ten 400× fields total magnification.
Immunohistochemistry
Paraffin-embedded tissues were cut at a 4-μm thickness, mounted on adhesive glass slides, and dewaxed in xylene, followed by rehydration in descending graded alcohols. Endogenous peroxidase was quenched with 3% H2O2 in phosphate-buffered saline (PBS) for 30 minutes at room temperature. Antigen heat retrieval was performed in citrate buffer (pH 6) for 12 minutes (600 Watt) in a microwave oven followed by a cooling period of 15 minutes. Nonspecific antibody binding was blocked by addition of 8% Roti-Immunoblock (Roth, Karlsruhe, Germany) and 20% goat serum for 30 minutes at room temperature. Monoclonal mouse anti-human γH2AX (phospho S139) antibody (dilution 1:1000; Abcam, Cambridge, England) and monoclonal mouse anti-human p21 (WAF1/Cip1; dilution 1:50; DAKO, Glostrup, Denmark) were incubated for 1 hour at room temperature. For slides stained for p21, cultured primary canine fibroblasts from passage 11 were used as positive controls. Slides were incubated with a secondary biotinylated goat anti-mouse antibody (dilution 1:200; Vector Laboratories, Burlingame, CA) for 30 minutes at room temperature. Color was developed by incubating the slides with freshly prepared avidin-biotin-peroxidase complex (ABC) solution (Vectastain Elite ABC Kit; Vector Laboratories), followed by repeated washes and exposure to diaminobenzidine tetrachloride (Merck, Darmstadt, Germany). The sections were counterstained with hematoxylin, dehydrated in ascending graded ethanols, cleared in xylene, and coverslipped. All 3 antibodies showed a nuclear staining. Negative controls were included in each run with commercial mouse immunoglobulins (Bio-Genex, Fremont, CA) instead of primary antibodies. Germ cells during chromatin remodeling, associated with double-strand breaks, served as internal positive control for γH2AX. 12,23
Fibroblasts and Leydig cells positive for γH2AX or p21 were counted in 200 cells of that particular cell type per random 400× fields total magnification. To evaluate the microenvironment of tumors, areas of abundant intratumoral stroma were used for the microenvironment count. In tumors with minimal amounts of stroma, fibroblasts directly surrounding the tumor at a maximum distance of 200 μm were examined, based on studies from cell culture, which had demonstrated a maximum range of approximately 400 μm for effective cell-cell interactions. 20
Statistical Analyses
Statistical significance analyses and graphical illustrations were performed using GraphPad Prism version 7.00 for Windows (GraphPad Software, La Jolla, CA). Data were tested for Gaussian distribution using the Shapiro-Wilk test. Gaussian distributed data (morphological parameters except Johnsen score) were tested with an unpaired
The minimum number of cells to be evaluated for γH2AX or p21 positivity as well as the number of intertubular triangles and tubuli were calculated based on preliminary evaluations of selected samples.
Results
Morphologic Parameters of Young, Old, and Young Cryptorchid Canine Testis
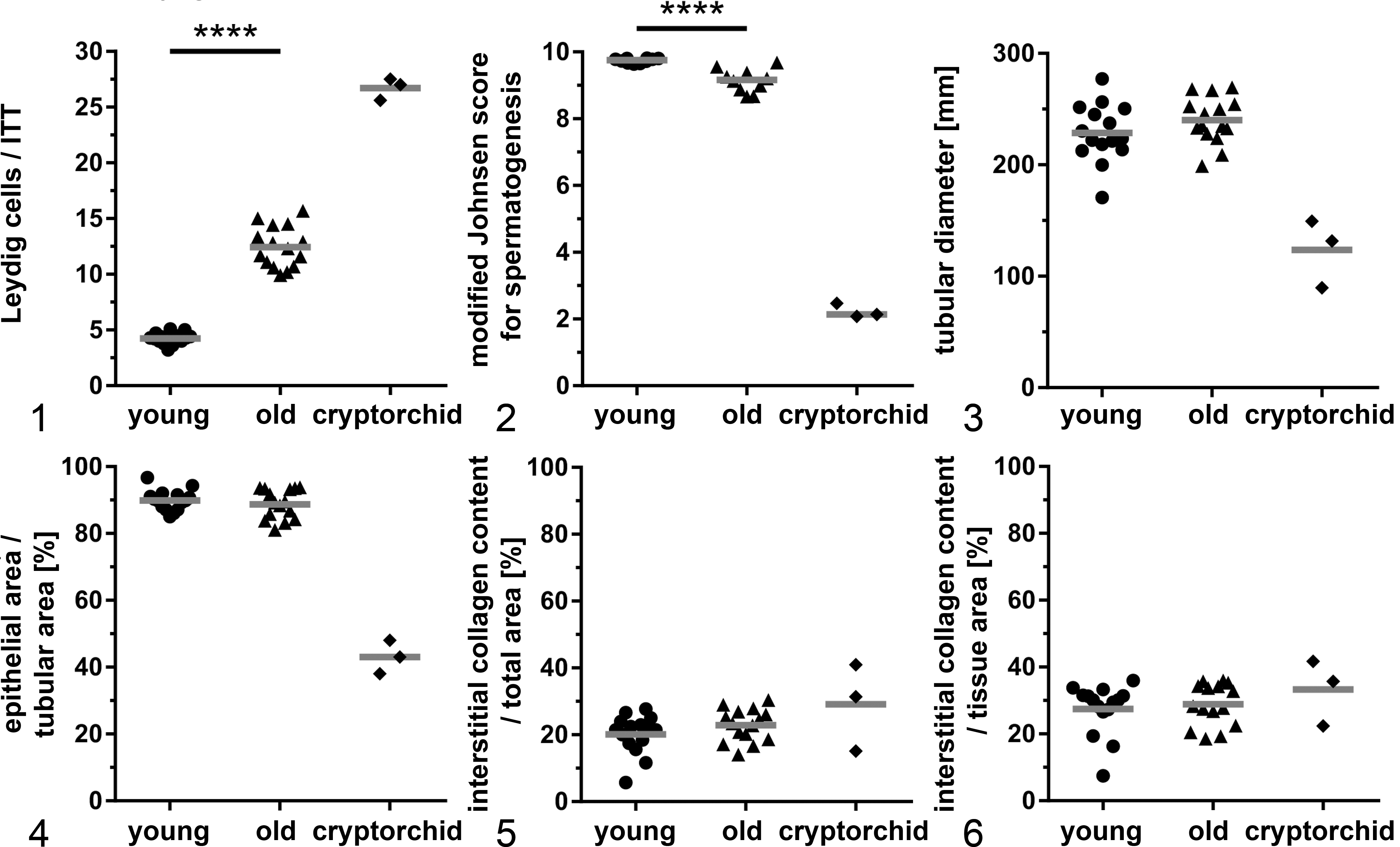
The mean number of Leydig cells/ITT was significantly increased from 4.2 ± 0.5 cells in young to 12.4 ± 1.8 cells (
Results of Morphological Changes for Young, Old, and Cryptorchid Testes.
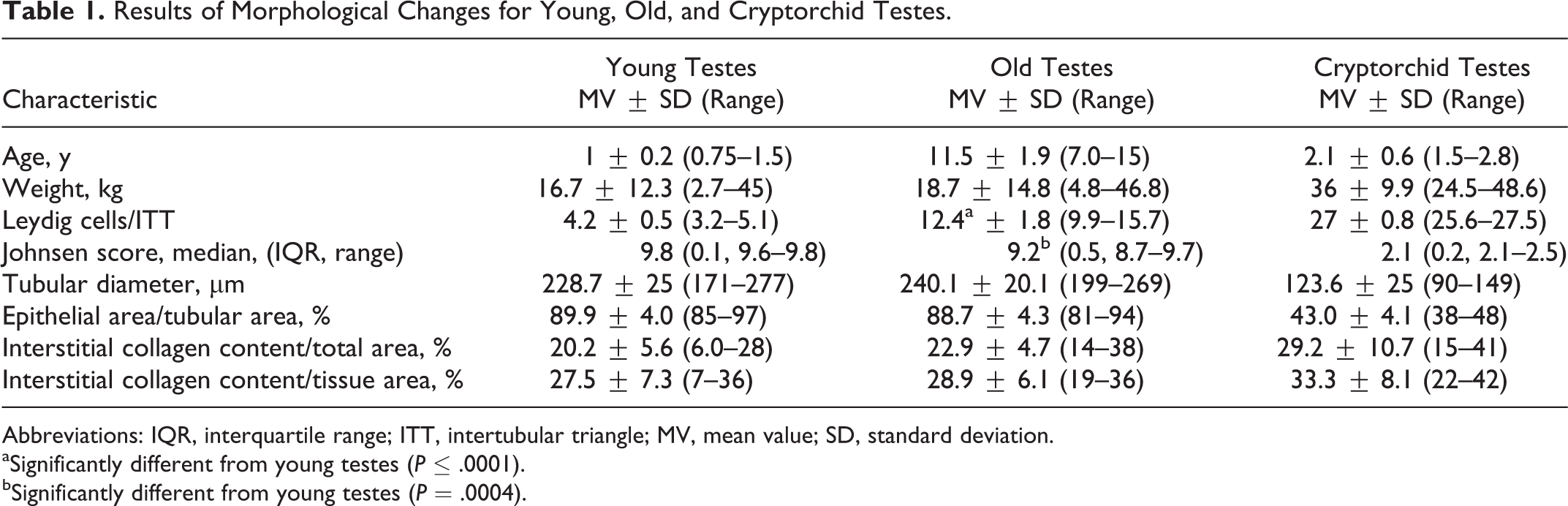
Abbreviations: IQR, interquartile range; ITT, intertubular triangle; MV, mean value; SD, standard deviation.
aSignificantly different from young testes (
bSignificantly different from young testes (
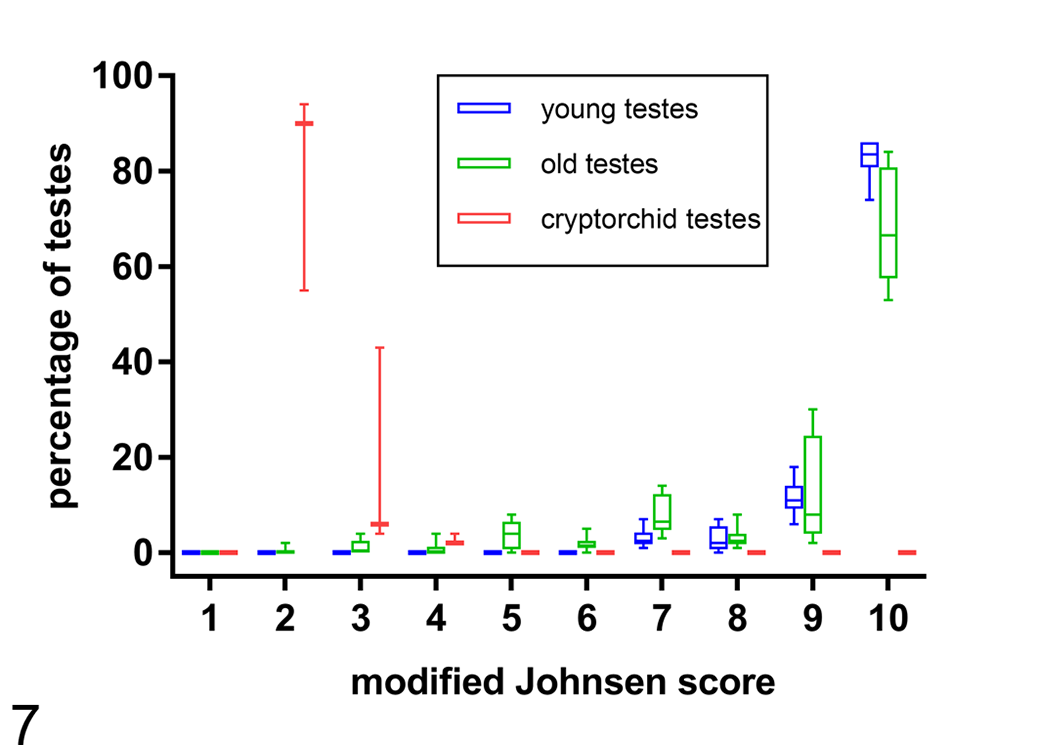
Modified Johnsen scores for young, old, and cryptorchid canine testes. Old testes were similar to young testes with a few higher scores. Cryptorchid testes had only low scores in comparison to young and old testes. Box plot (box: 25th/75th percentile; whiskers: minimum and maximum; horizontal line: median).
When compared to young descended testes, young cryptorchid testes had an increase in the mean number of Leydig cells/ITT from 4.2 ± 0.5 cells in young to 26.7 ± 0.8 cells in cryptorchid testes (Fig. 1). Young cryptorchid testes also had increased interstitial collagen content per total area (young noncryptorchid, 20.1% ± 5.6%; young cryptorchid, 29.1% ± 10.7%), increased interstitial collagen content in relation to epithelial tissue (young, 28.9% ± 6.1%; cryptorchid, 33.3% ± 8.1%) (Figs. 5, 6), a decrease in mean tubular diameter (young, 228.7 ± 25.0 μm; cryptorchid, 123.6 ± 25.0 μm; Fig. 3), a decrease in mean epithelial area/tubular area (young, 89.9% ± 4.0%; cryptorchid, 43.0% ± 4.1%; Fig. 4), and a low median modified Johnsen score (young, 9.76; cryptorchid, 2; Figs. 2, 7). No spermatozoa were detected in the epididymis of any of the 3 cryptorchid testes.
Senescence Marker Expression in Testicular Cells
P21 and γH2AX immunolabeling were exclusively localized to the nucleus. A significant relative increase was noted for p21-positive cells from young to old canine testes in fibroblasts from a median of 0.5% (IQR, 0.8) to 2.0% (IQR, 1.5) (
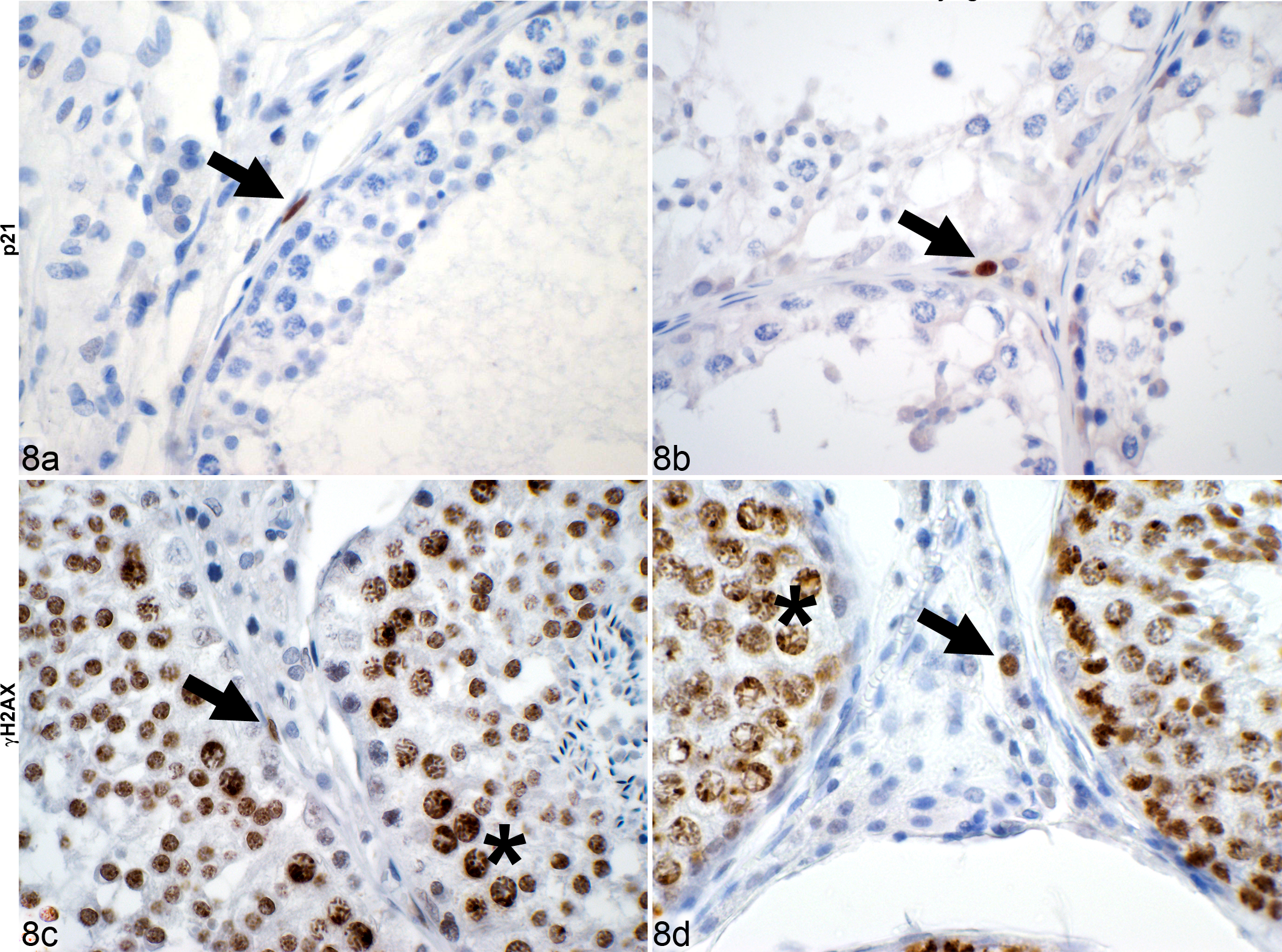
Testis, dog. Immunohistochemical detection (arrows) of positive fibroblasts (a, c) and Leydig cells (b, d) in canine in testicular interstitium. Germ cells (asterisk) served as an internal control for γH2AX due to chromatin remodeling-associated double-strand breaks. Immunohistochemistry for p21 (a, b) and γH2AX (c, d).
For γH2AX, less than 1% of fibroblasts and Leydig cells were positive, which was not significantly different when comparing young and old canine testes (Figs. 8c,d, 11–12). Cryptorchid testes had no immunolabeling for p21 or γH2AX in fibroblasts or Leydig cells (Figs. 9–12).
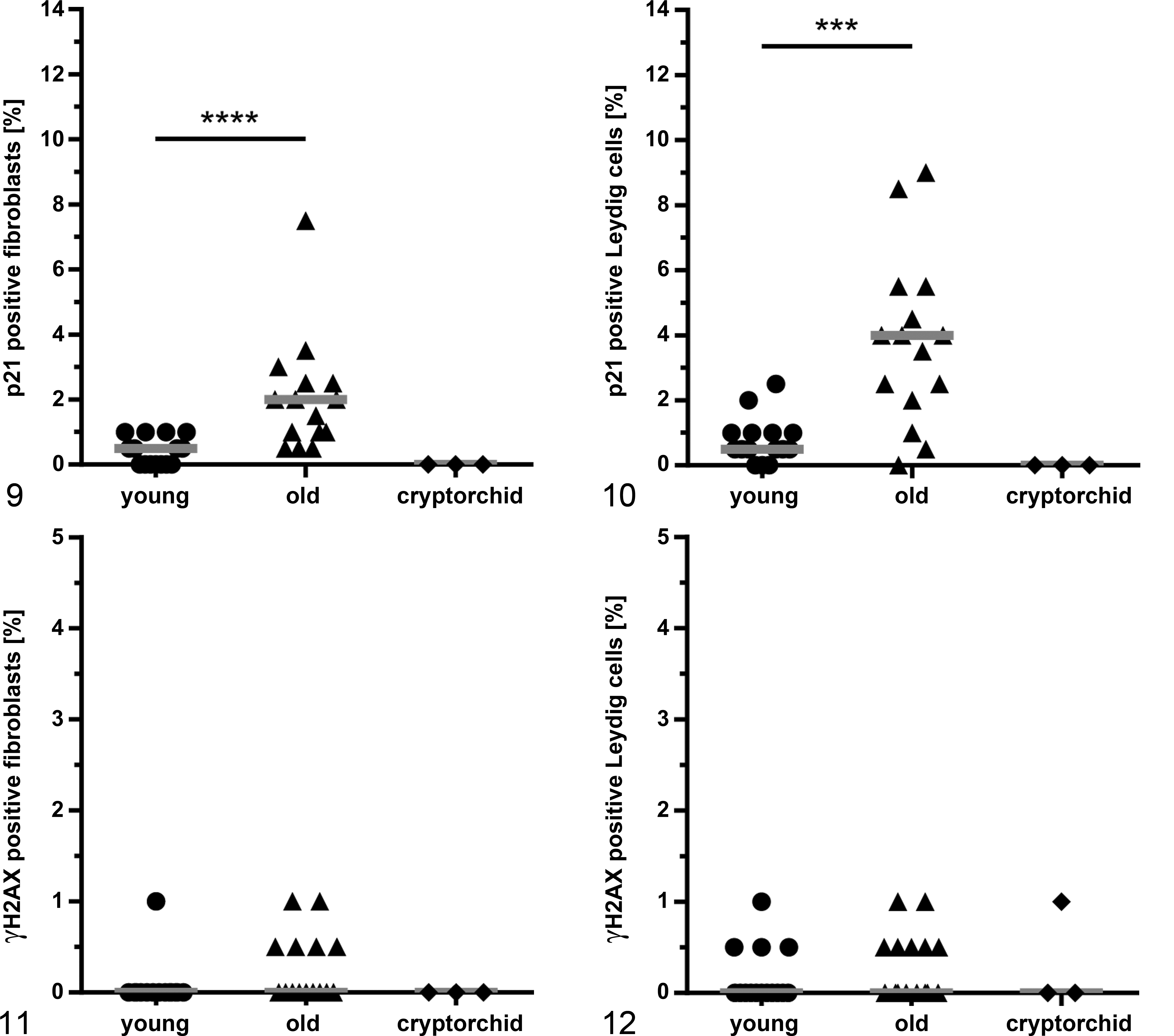
Senescence Marker Expression in the Microenvironment of Testicular Tumors
The number of γH2AX- and p21-positive fibroblasts was low (≤1% positive cells) in the microenvironments of all 3 tumors. We compared the microenvironments of all 3 tumors with fibroblasts in young and old individuals without tumors (Figs. 13, 14). For p21, in comparison to fibroblasts from old dogs with a median of 2.0% (IQR, 1.5), there were even significantly fewer positive cells in the microenvironment of Leydig cells tumors (
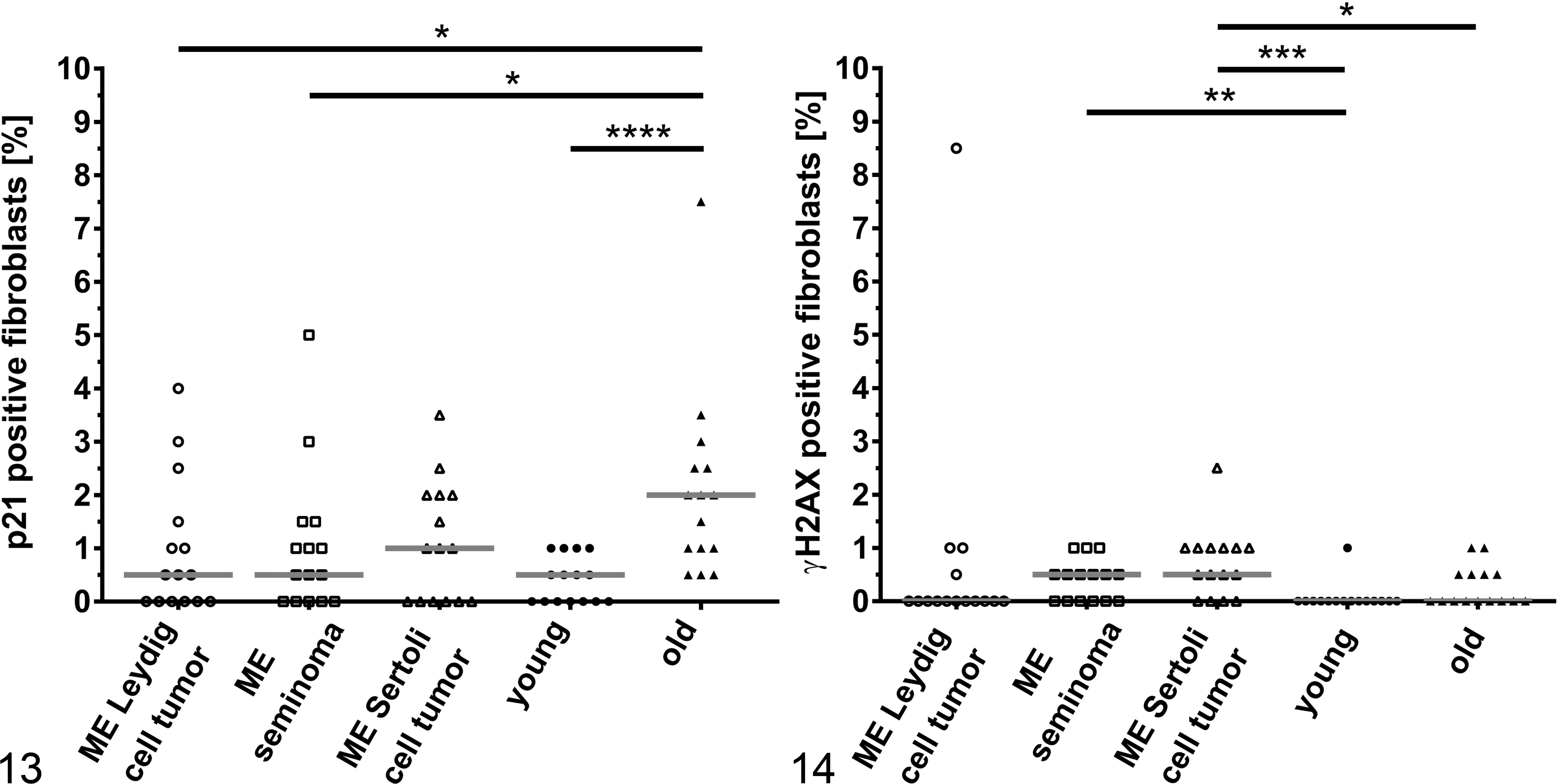
Fibroblasts from the microenvironments of seminomas (median, 0.5%; IQR, 0.5;
The microenvironment of cryptorchid Sertoli cell tumors had similarly few positive cells in comparison with the noncryptorchid Sertoli cell tumors with a median of 0.5% (IQR, 0.5) p21-positive cells and 1.0% (IQR, 0.8) γH2AX-positive cells.
Discussion
Based on previous in vitro and ex vivo data, we hypothesized that senescent cells accumulate with age in canine testes and in the microenvironment of testicular tumors. Thus, senescent cells could play a role in tumor proliferation via paracrine activities. Reflecting the aging process, we expected morphological changes in canine testes, such as decreased mean tubular diameter, increased relative collagen content, or increased mean Leydig cell number per area. Therefore, we tested whether the established markers of senescence, γH2AX and p21, could reveal an age-dependent increase of senescent cells in canine testes that might correlate with established age-related morphological changes.
This is the first report on age-dependent expressional changes of established senescence markers in canine tissue. Our results provide comparative value to published data on senescence in human, murine, and primate tissue (ie, skin, brain, liver, pancreas, and hematopoietic stem cells).
In accordance with published data on age-related accumulation of senescent cells, 11,17,34,42,55 an age-dependent, statistically significant increased percentage was noted for p21-expressing testicular fibroblasts and Leydig cells. However, although statistically significant, senescent cells were still rare and of yet unknown biological significance. When considering the reference data, it becomes clear that increased numbers of allegedly senescent cells seem to be a rare event. Supporting our data, Wang et al 55 detected a significant yet small increase in γH2AX-positive cells in the dermis of mice from 1.1% ± 0.2% in young animals to 4.8% ± 2.5% in old animals. Similarly, for human skin, Ressler et al 42 found an increase in p16INK4a-positive cells in the dermis from 0.6 cells per millimeter basement membrane in young donors, 2.9 cells per millimeter for middle-aged donors, and 6.3 cells per millimeter for old donors.
In addition, photographs of immunohistochemically labeled tissues in previous reports identified senescent cells in aged individuals often as single cells, which supports the assumption of only a slight increase in detectable senescent cells with age. 11,40,42,54 Whether these cells possess the secretory phenotype with all due consequences as observed in replicative senescent cells in vitro as well as the amount of senescent cells necessary to achieve an effect remains speculative. 19 To date, it is unclear how long they are present before they are disposed of, thus limiting the possibilities to seize them. 31
We did not detect an increase in γH2AX-positive fibroblasts or Leydig cells in the aged canine testis. There could be several reasons for this finding. First, whereas p21 is a component of numerous senescence pathways, many of these are independent of double-strand breaks as detected by γH2AX. Other triggers such as oncogenic signaling, reactive oxygen species (ROS), or cell fusion may initiate the senescence pathway. 36 Second, it has been shown that aging in various tissues progresses at different paces, indicating a certain tissue- and cell-type specificity for cellular senescence. Wang et al 55 found a significant increase in γH2AX-positive cells in murine lung, spleen, small intestine, liver, and dermis but not in testis and other select tissues. Similarly, findings in primates indicate an increase in senescent cells in the dermis but not in the skeletal muscle. 22 And third, when following the senescent cascade, p21 is very close to the senescent state, whereas double-strand breaks are early events and may also lead to apoptosis or even be repaired. 5,48 It remains unknown how many of these alleged senescent cells will not only transiently express the markers but actually enter the senescent state with essentially irreversible cell cycle arrest.
To our surprise, only few age-related morphological changes were found in old canine testes in our study, and senescence marker expression was meager to nonexistent. This raises the question of whether the canine testis may not age or does not show signs of aging within the age range included here. There was no significant decrease in epithelial area or tubular diameter and no significant increase in interstitial collagen content, all of which have been controversially discussed for the canine testis. However, a limiting factor in our retrospective study and also in previous studies of canine testes is that a more complex, stereological evaluation was impossible for several reasons. These include limited availability of tissues in cross-sectional paraffin blocks, lack of random embedding procedures, and lack of information on, for example, testicular weights and breed in several cases. 41,49 Although some studies came to the same conclusion as we do, 41,49 veterinary pathology reference texts still state the contrary, 13 presumably based on the observation that in humans, epithelial area and tubular diameter are known to decrease with age. 25,26,28,30,37,38 Significant changes in the aging canine testes were only seen in the mean number of Leydig cells/ITT, which increased significantly (2.95-fold) in aged testes. Leydig cell hyperplasia is considered common in old dogs. 33 In contrast, in humans and rats, Leydig cells decrease with age, suggesting complex species-specific differences. 6,38,56 In addition, we noted a slight albeit significant decrease in spermatogenesis in old canine testes and a minor increase in tubular irregularities (scores ≤8). Although this contradicts findings by Peters et al 41 that spermatogenesis does not decrease significantly with age in dogs, old canine testis still had a mean score above 9, and abundant morphologically normal spermatozoa were present wherever an epididymis was available. However, no conclusions can be drawn on the functional quality such as vitality or motility of the spermatozoa produced. In humans, it has been shown that increased paternal age negatively affects sperm quality as well as fertility and reproductive outcomes. 47 Furthermore, many studies have shown that the canine aging testis is prone to age-associated neoplasia, with a prevalence of up to 65% depending on the study quoted. 35 In our study, the mean age was 10.2 years for dogs with testicular tumors, 10.5 years for Leydig cell tumors, 10.1 years for seminomas, and 10.2 years for Sertoli cell tumors. Although it is rather unlikely that the canine testis does not age in general, the mechanism might differ from other organs or tissues, or aging may occur at ages beyond those studied here. Thus, the detection of significantly increased albeit still few p21-positive cells and no relevant γH2AX labeling is not completely surprising.
Although the relative number of p21-expressing cells increases with age, no significant increase was noted for p21 in the microenvironments of any of the testicular tumors tested in comparison to fibroblasts from young dogs. When comparing them to old dogs, there were a significantly lower number in the microenvironment of Leydig cell tumors and seminomas. This could possibly be explained by non-tumor-associated variances in p21-expressing cells among individuals of different ages. This raises the possibility that p21-mediated senescence may not be relevant for tumor development in aged canine testes.
We detected a significantly higher number of γH2AX-positive cells in the microenvironment of seminomas and Sertoli cell tumors in relation to fibroblasts from young dogs. For the microenvironment of Sertoli cell tumors, it was even significantly higher in comparison to fibroblasts from old dogs (
Both markers are expressed in only very few cells with ≤1% positive fibroblasts in the microenvironment of all 3 canine testicular tumors, and an increase in positive cells with age is either small (p21) or not measurable (γH2AX). We thus speculate that p21- or γH2AX-positive senescent fibroblasts may have no impact on tumor development in the canine testes. However, we focused on the classical age-related, replicative senescence pathway associated with telomere-initiated senescence via p21-mediated cell cycle arrest. Other pathways, including those triggered by ROS, oncogenic signaling, or loss of tumor suppressors, are likely associated with additional senescence marker expression (eg, p16). 36 Thus, senescence might have been underestimated in our study when considering such alternative pathways.
As with all retrospective studies, certain methodological limitations could not be excluded here. For example, fixation, processing, dehydration, and embedding of the tissues may have varied slightly between the samples compared here. However, due to sufficient quality of our samples as assessed, for example, by preserved morphology, lack of epithelial detachment, and sufficient immunoreactivity, we presume that their influence on the results of our study was negligible.
Conclusions
Our data did not support several previous hypotheses and expectations on structural changes as well as the role of senescence in aged canine dogs. Species-specific differences may be involved: for example, in contrast to humans, aged dogs presented a significantly increased mean number of Leydig cells per intertubular triangle (2.95-fold) and no decrease in the mean tubular epithelial area or tubular diameter compared to young dogs. Only a slight but significant decrease was found in the spermatogenesis score. Different to what was expected, no significant increase in interstitial collagen content was identified in testes of aged dogs. Although both p21 and γH2AX are considered senescence markers, only the relative number of p21-positive cells increased with age in healthy canine testes. Again, opposed to our expectations based on previous in vitro studies, we did not identify any evidence of a correlation between the age-associated accumulation of senescent cells and testicular tumor development.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985819843683 - Aging and Senescence in Canine Testes
Supplemental Material, DS1_VET_10.1177_0300985819843683 for Aging and Senescence in Canine Testes by Sophie E. Merz, Robert Klopfleisch, Angele Breithaupt and Achim D. Gruber in Veterinary Pathology
Footnotes
Author Notes
This article is part of the doctoral thesis of S. E. Merz.
Acknowledgments
We thank Bernhard Sörensen with team, especially Thomas Lottermoser; Thomas Göbel with team; and Georg König with team for their cooperation. We thank Charlene Lamprecht for her technical support and Roswitha Merle for her statistical support. We also thank Hannah Pischon for her continuous support and thoroughness.
Author Contribution
A. Breithaupt and A. D. Gruber contributed equally to this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
