Abstract
CD1 molecules are a family of cell surface-associated glycoproteins now recognized as having a role in antigen presentation. These glycoproteins are distinct from yet have some similarities to classical major histocompatibility complex class I and class II molecules. The role of these molecules has been studied in detail over recent years, with an explosion of interest following the demonstration that they can present nonprotein antigens to certain subpopulations of T cells. The purpose of this paper is to provide an overview of current knowledge of the function of the CD1 family with specific emphasis on the potential role in the pathogenesis of certain diseases. Although much of the current research in this field has inevitably concentrated on mice and humans, this work also has potential significance for veterinary species.
The CD1 family comprises five distinct genes, A–E, which are variably conserved across the species. The family can be broadly divided into two groups—the CD1A-C group (group 1) and the CD1D group (group 2). Sequencing of CD1E (only identified in humans and guinea pigs to date) indicates that this is an intermediate group with similarities to both group 1 and group 2. 13,16 Mice and rats only possess members of the CD1D group, and humans, sheep, and rabbits possess members of both groups. Recent molecular studies in the pig have identified a gene (pCD1.1) that is relatively dissimilar to other CD1 isotypes and moreover contains a major histocompatibility complex (MHC) class I-like cytoplasmic tail. 15 A summary of current knowledge of CD1 genes and proteins across species is presented in Table 1. With the apparent exception of the guinea pig, 16 there is evolutionary conservation of CD1d antigens, consistent with a key role for these molecules in the immune response. 11 More recent studies have begun to extend the initial immunologic characterization of these molecules and to address the significance of this axis of the immune system in the pathogenesis of a range of diseases.
Interspecies summary of the documented presence (X) of members of the CD1 family.

∗ Data obtained on the basis of mAb cross-reactivity (specific isotype identity not proven).
† The porcine gene, pCD1.1, is most similar to human CD1A but is nevertheless relatively dissimilar to other CD1 molecules. 15
The details of the immunology of these molecules have been extensively reviewed recently. 9,36,37 Here, I provide only a brief overview as a prerequisite to the understanding of the pathologic significance of CD1. I then review the role of CD1 in disease pathogenesis with more detailed discussion of the implications and potential significance for the future in terms of veterinary pathology.
CD1 Structure and Function
A highly relevant feature of these molecules in their remarkable lack of polymorphism, totally unlike classical MHC class I and class II molecules, which are inherently polymorphic. In the early days of CD1 research, this lack of variability in MHC-related molecules was perceived to reflect possible functional redundancy of the population 27 ; however, it is now clear that this feature provides vital clues to the role of these molecules. CD1 molecules are structurally similar to MHC molecules, in particular to MHC class I molecules, with which they share a closely similar exon arrangement and corresponding morphology (Fig. 1). The recent resolution of the crystal structure of mouse CD1 demonstrated that the structure is more closely related to that of MHC class I than to that of MHC class II. The binding groove, although significantly narrower, is substantially larger because of increased depth and it has only two major pockets that are almost completely hydrophobic. 56 This structure provides a classical antigen-presenting architecture allowing fragments of antigenic material to be presented on the cell surface, where appropriate T cells can be triggered on recognition of the antigen.
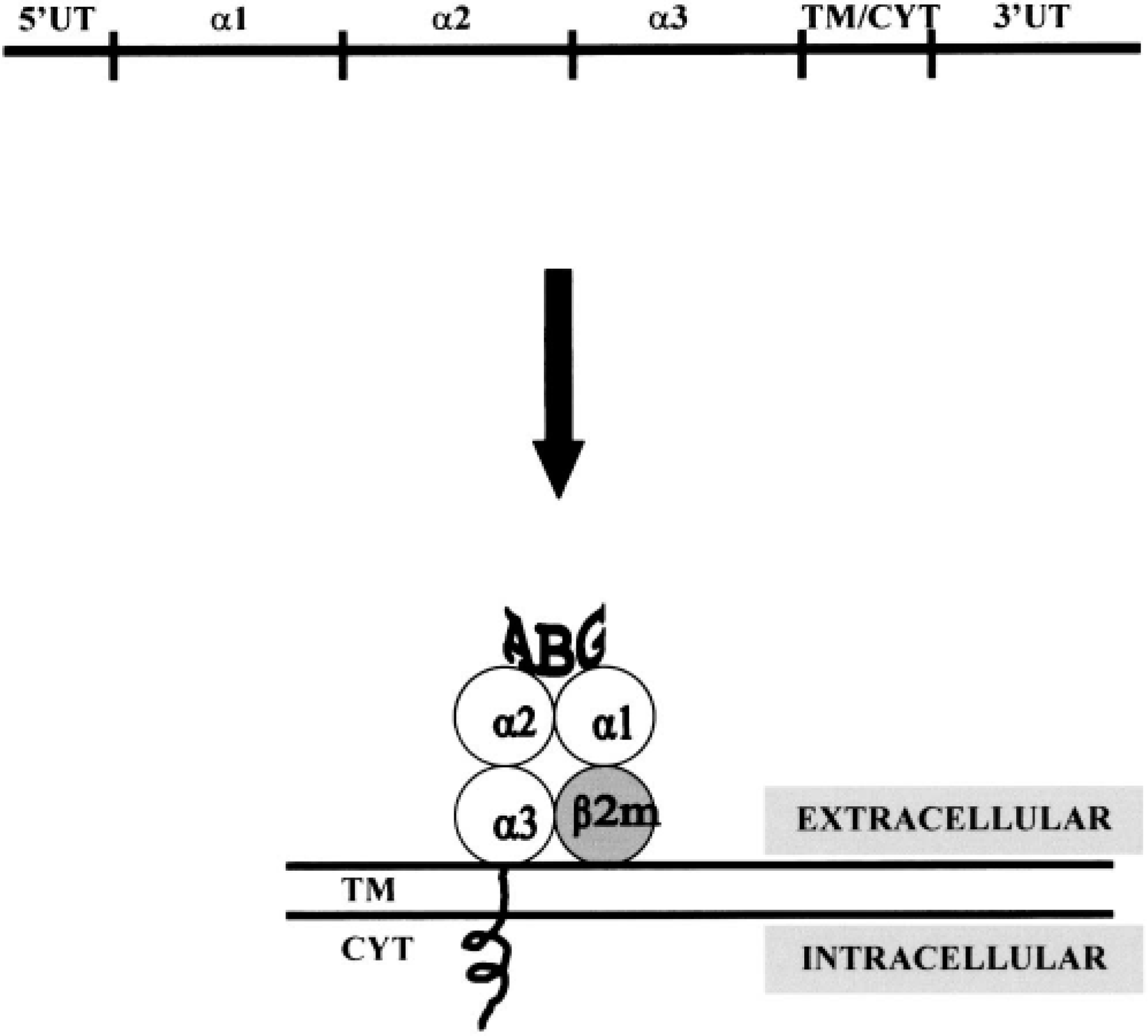
Exon arrangement of CD1 and translation to protein structure showing antigen-binding groove (ABG) created by the alpha 1 and alpha 2 domains. Note also the noncovalent association with β2-microglobulin. TM = transmembrane; Cyt = cytoplasmic; UT = untranslated.
T-cell Recognition of CD1
There has been much work carried out on the nature of the T cells capable of recognizing CD1, a detailed discussion of which is outside the scope of this review; however, the gamma delta (γδ), double negative (DN), and natural killer (NK) T cells have all been linked with recognition of CD1 molecules. The key feature common to many of these cells is expression of a relatively invariant T-cell receptor (consistent with recognition of conserved antigen and an invariant presentation molecule). The DN subset is characterized by lack of expression of both CD4 and CD8 molecules. These cells can express either αβ or γδ receptors and many are relatively invariant.
The γδ T-cell population has attracted much interest with regard to potential recognition of CD1 molecules. These cells have marked interspecies differences in numbers and location and are present in notably large numbers in young ruminants. 21 In humans, a major subset of γδ T cells recognize CD1c molecules, 48 although significant functional studies on the relationship between CD1 molecules and γδ Τ cells have yet to be carried out in animals. NK T cells are a specialized population of T cells that coexpress receptors of the NK lineage. These cells are distinct from natural killer cells, with which they share some common receptors. 8 The NK T cells have a remarkable capacity to rapidly secrete large amounts of cytokines (notably Interleukin 4 [IL-4] and Interferon γ [IFNγ]) and can thus rapidly regulate Th1/Th2 differentiation. 7 Many of these cells are reactive to CD1 molecules. 6 A further intriguing feature of these cells is autoreactivity; exogenous antigen is not always required to elicit a CD1-restricted response.
The functional relevance of this CD1 recognition by specific T-cell subsets provides the immune system with a mechanism for rapid response to nonpolymorphic molecules, consistent with a key role in innate immunity prior to the onset of the definitive acquired immune response.
CD1 Expression
In broad terms, CD1 expression can be divided into two categories on the basis of anatomic localization: strong expression on cortical thymocyte populations and expression on a variety of antigen-presenting cells (APCs), notably dendritic cells, B cells, and monocytes. There is interisotype and interspecies variation in CD1 expression, and monoclonal antibodies (mAbs) can be used to readily demonstrate this feature across many species. With the exception of the mouse (which for reasons of limited expression is not a good model for the system), the animal species with one of the best characterized CD1 families is the sheep, in which studies of expression at the cellular and molecular level have been carried out. 18,39,40 CD1 molecules have been identified using mAbs in other species, including the cat, 53 dog, 34,35 pig, 17,42 and horse. 45 Most CD1 molecules are expressed at high levels in the thymus. Using the mAb IAH-CC20 (which recognizes CD1b 23 ), strong expression of CD1 is evident in cortical thymocytes of ruminants, dogs, and horses (Fig. 2).
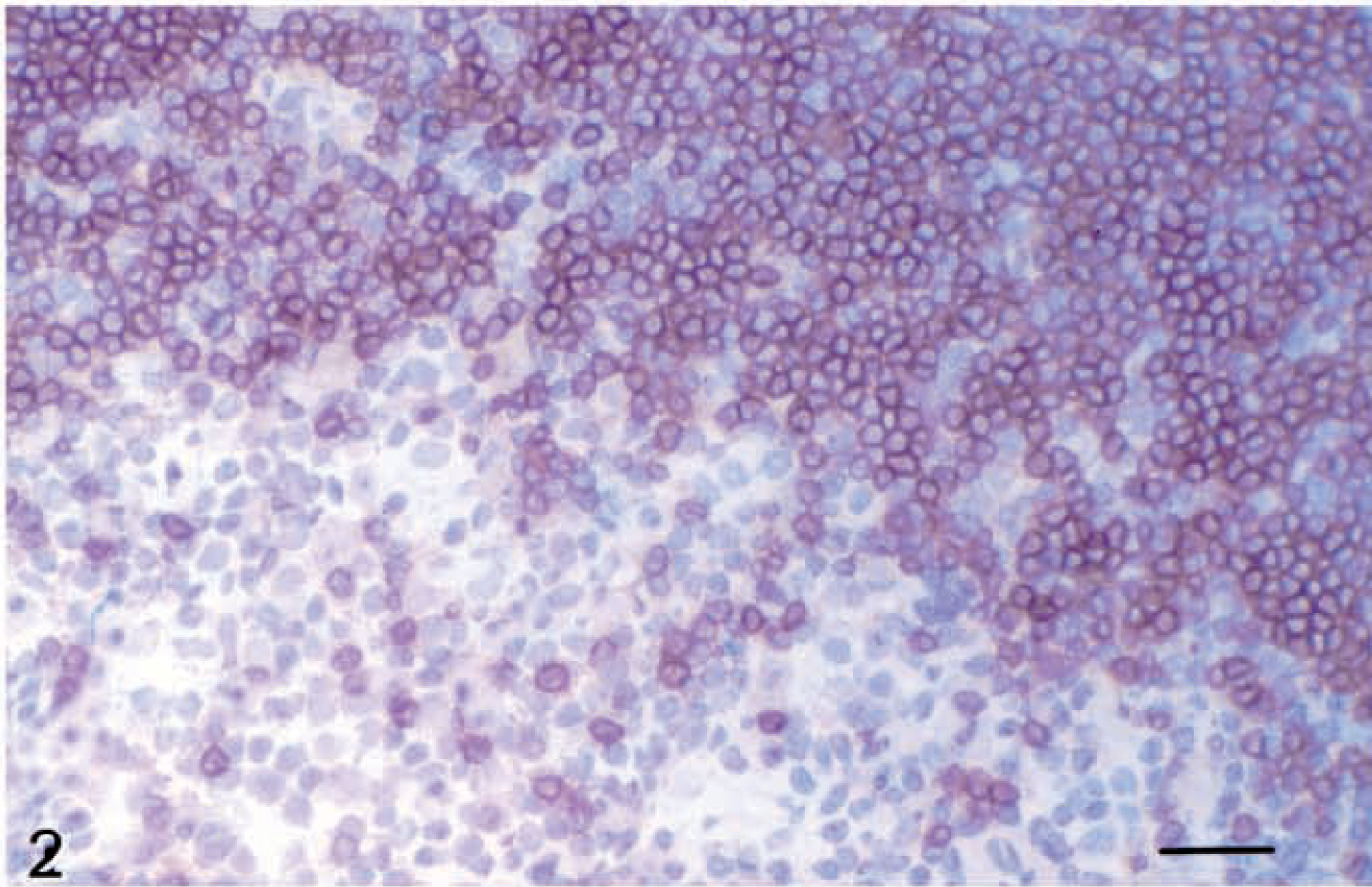
Thymus; horse. Strong staining of cortical thymocytes for group 1 CD1 (mAb CC20). Avidin–biotin–peroxidase complex method, Mayer's hematoxylin counterstain. Bar = 50 µm.
Figure 3 demonstrates CD1 staining on canine dendritic cells using the mAb IAH-CC20. In addition to the use of mAbs to study surface expression of these molecules, our work in sheep has utilized in situ hybridization to identify expression in a wide range of cells including B cells (e.g., in bronchus/bronchiole-associated lymphoid tissue, Fig. 4) and in central nervous system (CNS) microglial cells and neuronal cell bodies. 41
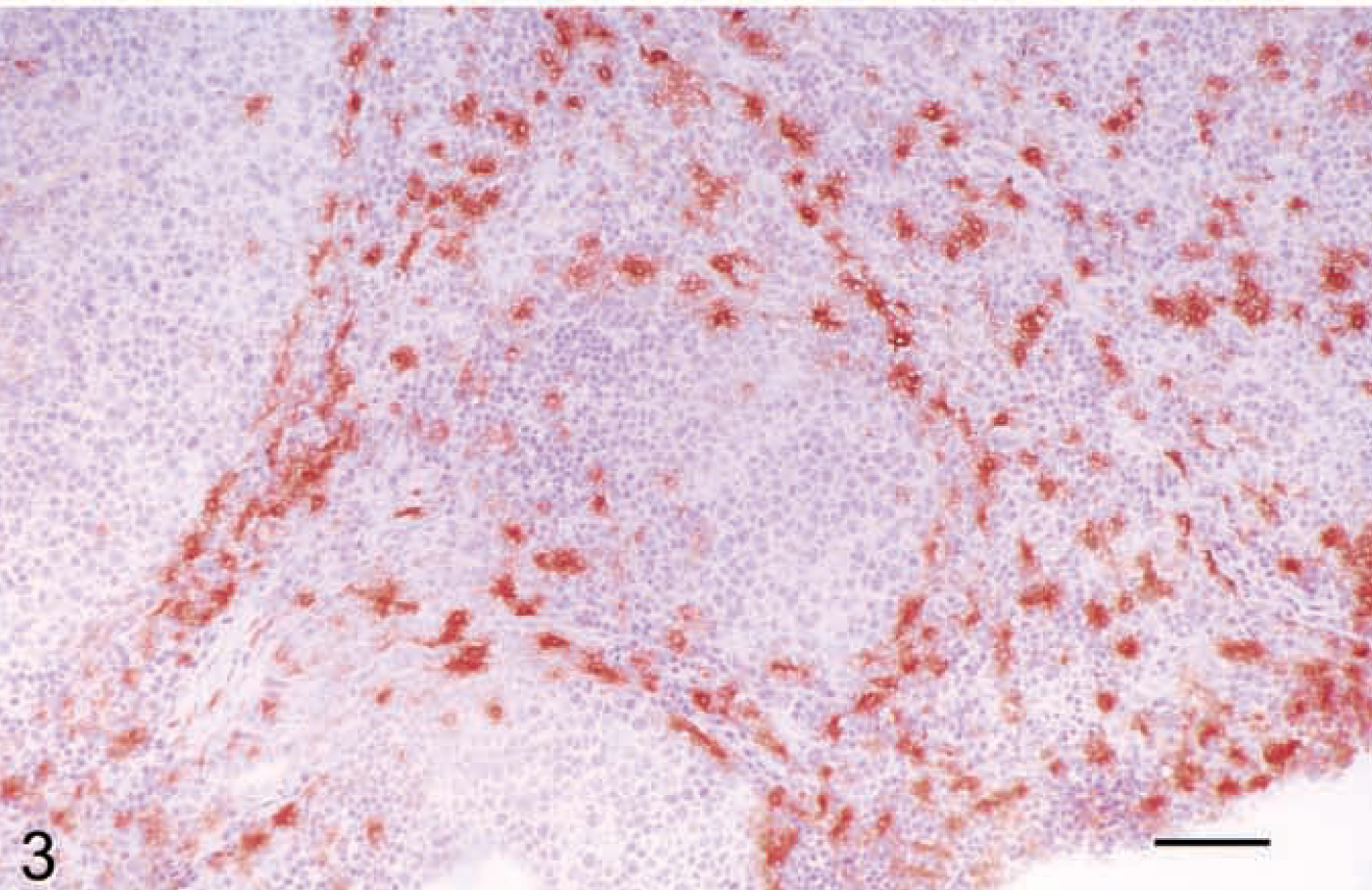
Lymph node; dog. Staining of paracortical dendritic cells for group 1 CD1 (mAb CC20). Avidin–biotin–peroxidase complex method, Mayer's hematoxylin counterstain. Bar = 100 µm.
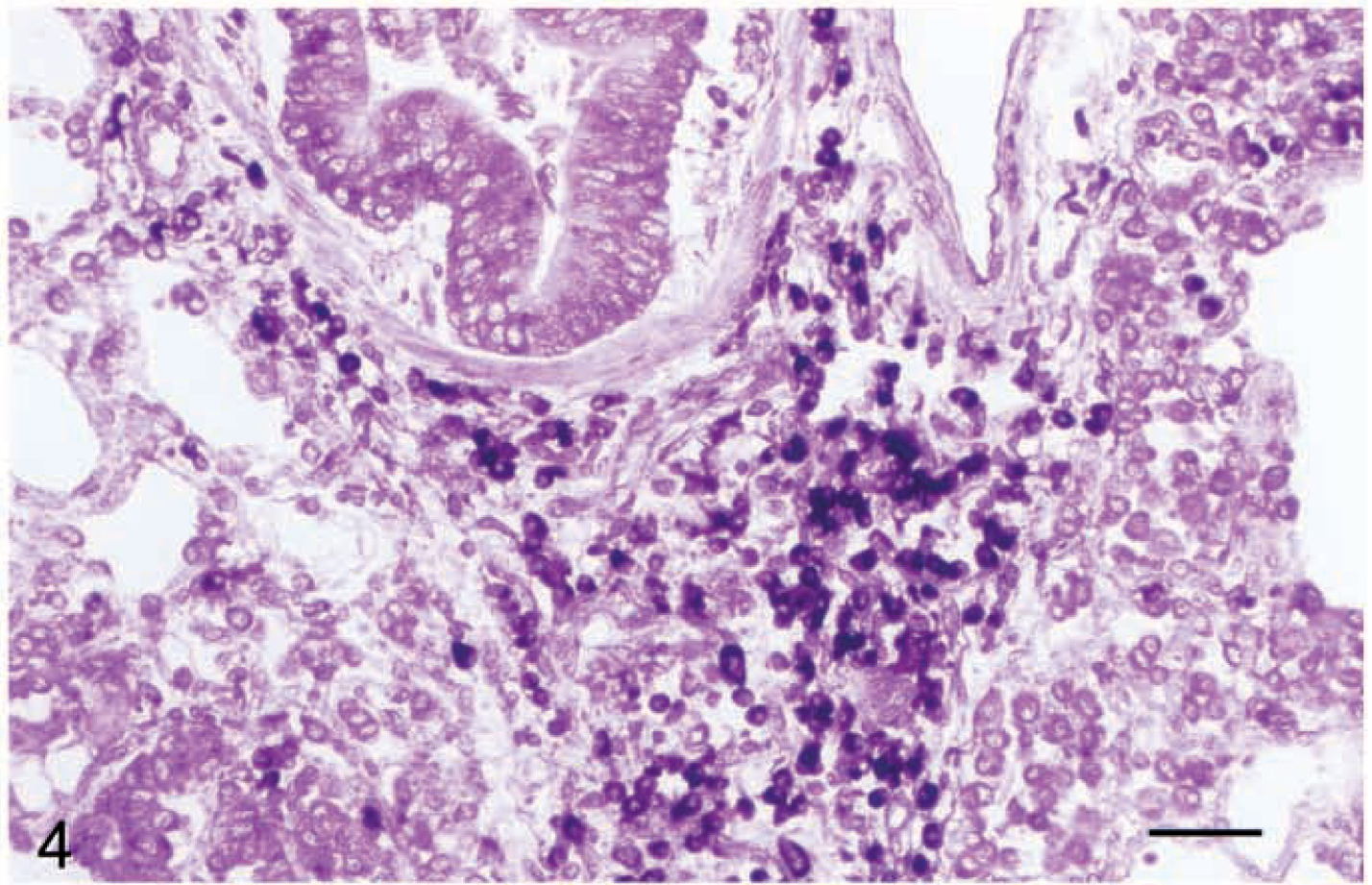
Lung; sheep. In situ hybridization using conserved (alpha 3 domain) antisense digoxigenin-labeled riboprobe showing positive bronchiole–associated lymphoid tissue lymphocytes. Developed using antidigoxigenin alkaline phosphatase; nitroblue tetrazolium/bromochloroindelyl phosphate method. Bar = 50 µm.
CD1 and Antigen Presentation
Although the precise mechanism for antigen presentation has not currently been elucidated, it is now generally accepted that CD1 molecules have evolved to provide an extra arm to the immune response by virtue of their ability to present lipid/glycolipid-derived and hydrophobic antigens. The recent resolution of the crystal structure of mouse CD1d demonstrated a deep antigen-binding groove, implying that presentation occurs in a fashion broadly similar to that of classical MHC molecules. 56 In simplistic terms, the presentation of lipid by CD1 molecules can be broadly divided into two categories: that derived from infectious agents (principally bacterial and parasitic) and self lipid. Research to date suggests that bacteria-derived antigens are presented by group 1 CD1 molecules rather than group 2 molecules. 43 Figure 5 illustrates examples of proposed mechanisms and downstream consequences of this presentation, whereby presentation of bacterial antigens elicits IFNγ production and a Th1-type response, with ultimate killing of infected macrophages 14,29,50 (Fig. 5A). In contrast, presentation of self lipid may be more likely to elicit a Th2-type response, with resultant rapid production of cytokines by responding populations of NK T cells (Fig. 5B).
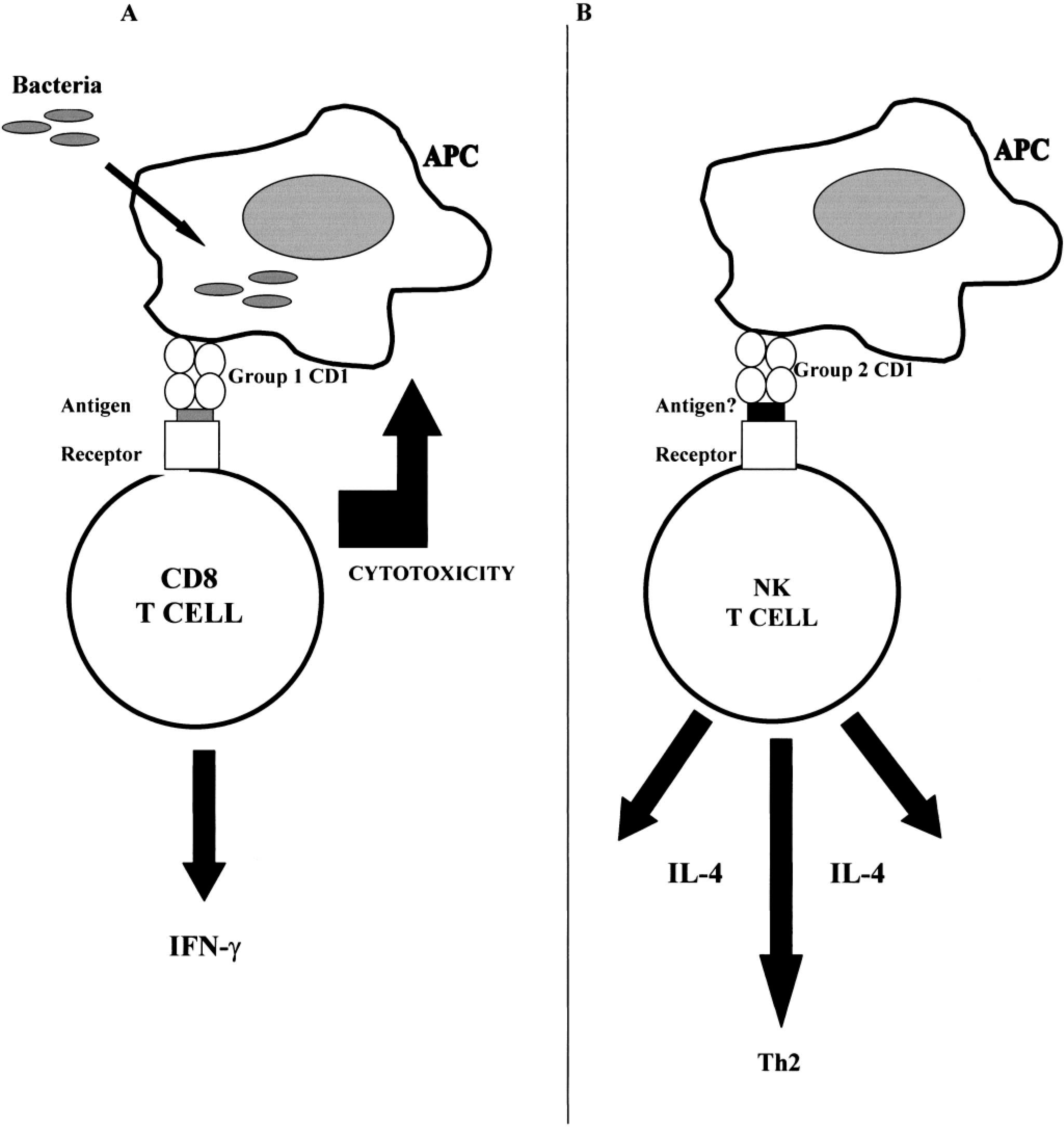
Schematic of different potential roles of group 1 versus group 2 CD1 molecules. Current evidence suggests that group 1 CD1 recognition is directly involved in removal of infected APCs (
Presentation of foreign lipid
Mycobacteria
In terms of specific infectious agents, most research has focused on mycobacterial disease, with detailed biochemical analysis of the microbial lipids involved. 12,20,32,33 The identification of the capacity for CD1 molecules to bind hydrophobic ligands, notably bacteria-derived lipid/glycolipid, has been extended to show definitively that CD1 molecules effectively sample mycobacterial glycolipids from intracellular sites within infected cells. 43 Infection of APCs with Mycobacterium tuberculosis downregulates the expression of human CD1b, suggesting the presence of a novel evasion mechanism of M. tuberculosis that could contribute to persistence of intracellular infection by avoiding immune recognition. 51
Research has only relatively recently turned from this basic immunologic characterization toward assessment of the role and significance of this CD1-mediated presentation in vivo in naturally occurring disease situations. In humans, study of the role of CD1 in the pathogenesis of leprosy has revealed that expression of CD1 is correlated with effective immunity to Mycobacterium leprae. 46,47 Investigation of CD1 expression in sheep with ovine paratuberculosis has shown a similar upregulation of CD1 expression in association with the tuberculoid form of the disease. 38 In addition, a recent study investigated both the expression of CD1 molecules (group 1) and γδ T cells in a model of ovine paratuberculosis. 4 Levels of γδ T cells increased to a significant level, and a decrease was observed in the number of CD1 cells in the jejunal Peyer's patch of the infected group. Although this decrease was not significant, it may represent a mild in vivo illustration of a downregulation mechanism employed by M. tuberculosis.
Overall, these findings are consistent with a role for CD1 in ensuring the generation of an effective cell-mediated immune response against mycobacteria. In addition to these studies of mycobacterial disease of humans and ruminants, mice treated with anti-CD1 mAbs show exacerbated tuberculosis lesions compared with controls. 52
Other pathogens
Although the majority of research has inevitably focused on the potential for CD1 molecules to present mycobacterial antigens, it seems unlikely that this family of molecules has been retained throughout evolution solely to provide a protective mechanism against this one class of bacteria. Obvious other candidate organisms would include organisms with similar structural elements, such as Rhodococcus and Nocardia. The palmitate hypothesis has been proposed 20 whereby palmitate anchors covalently attach capsular polysaccharides from a variety of organisms into the groove of CD1. In support of this hypothesis, preliminary studies have identified CD1-restricted T-cell populations that proliferate in response to Haemophilus influenzae type b antigen. 19 Other recent studies have shown that CD1d deficiency impairs host resistance to the spirochaete Borrelia burgdorferi (an organism containing proinflammatory lipid antigen). 28 Glycosylphosphatidylinositol (GPI) has been eluted from murine CD1d, 25 and this compound is present in several protozoan parasites, including Leishmania. CD1–NK T-cell interactions are involved in regulating immunogobulin G responses to the GPI-anchored surface antigens of Plasmodium and Trypanosoma. 44 These studies suggest that the specific pathogens identified thus far as having an association with CD1-presenting molecules are likely to represent the tip of the iceberg.
Presentation of self-derived lipid
In addition to the role in certain infectious diseases, CD1 molecules have also been implicated in other diseases by virtue of their capacity to present nonmicrobial lipid. The best example of this presentation is the CD1 expression associated with human atherosclerotic plaques. 30 CD1 expression in these areas may indicate a role in the genesis of the lesions and may provide a mechanism for sustained T-cell activation.
A further area in which CD1 presentation of lipid has been investigated is in tumors associated with glycolipid antigens. More specifically, the relationship between the number of infiltrating CD1a-positive cells and prognosis has been investigated in tumors, including breast carcinomas, with some descriptions of an association of these cells with an improved prognosis. In particular, there is evidence that CD1a-positive cells may have a role in antigen capture and presentation within these tumors. 5,22
Considering this association with lipid and lipid-related antigens, it is perhaps not surprising that studies of CD1 expression in certain CNS diseases have also identified upregulation of CD1 in this lipid-rich environment. Specifically, studies on patients with acute inflammatory demyelinating polyradiculoneuropathy showed upregulation of CD1a and CD1b on endoneurial macrophages and on myelinated nerve fibers. 26 Chronic-active multiple sclerosis lesions also show prominent CD1b immunoreactivity on perivascular inflammatory cells and on hypertrophic astrocytes. 3
Role of CD1 in Immune-mediated Disease
In addition to the role in presentation of lipid-derived antigens (either lipid from pathogens or internally derived lipid), a role for CD1 molecules in immune-mediated disease has also been shown. In particular the two broad areas of allergic/atopic disease and autoimmune disease have been investigated.
NK T cells reactive with group 2 CD1 molecules can produce large amounts of IL-4, 6,54 thus supporting the hypothesis that these molecules are involved in allergic diseases by virtue of skewing the immune response towards the Th2 pathway (Fig. 5B). Also in support of this hypothesis, studies on alveolar macrophages isolated from asthmatic humans have shown overexpression of CD1, 2 and it has been hypothesized that both γδ T cells and CD1 expression contribute to atopic status in some individuals. 49
Studies on contact hypersensitivity in sheep have shown CD1 expression both on and in close association with vascular endothelium, consistent with a role in antigen presentation during the dermatitis reaction. 24 In humans, studies on lesions of psoriasis have identified overexpression of CD1d in chronic-active plaques. 10 In contrast to the restricted and low level of expression in normal skin, CD1d expression was induced in these lesions and also in response to physical trauma and contact sensitizing agents.
With regard to autoimmune disease, most data are from studies on genetically modified mice, 31,55 e.g., genetically engineered mice predisposed to development of lupus exhibit CD1-dependant production of autoantibody. 55 Nevertheless, there is sufficient evidence to suggest that there may be a role for the CD1 axis in genuine naturally occurring autoimmune and allergic disease.
CD1 Expression in Reactive and Neoplastic Populations
In addition to the role of CD1 as an antigen-presenting molecule and resultant involvement in infectious and immune-mediated disease, expression of CD1 has also been used to confirm the histogenesis of certain neoplastic populations. CD1 expression by neoplastic cells has been demonstrated in canine cutaneous histiocytoma, establishing the origin of this tumor as a Langerhans cell histiocytosis. 35 Although this example illustrates the use of CD1 mAbs for simplistic cell phenotyping, more recent studies have defined canine cutaneous and systemic histiocytosis as two clinical manifestations of a reactive population of CD1-positive dermal dendritic cells, and immune dysregulatory mechanisms are thought to be involved in the genesis of the lesion. 1 It is tempting to speculate that CD1 as a recognized antigen-presenting molecule expressed on potent APCs (dendritic cells) may be involved in lesion development.
Conclusion
CD1 molecules are an intriguing family of antigen-presenting molecules whose importance in the pathogenesis of certain diseases is only beginning to be elucidated. Although there is much focus currently on their role in presentation of mycobacteria-derived lipids, it is unlikely that the evolutionary conservation that is a feature of this family relates to a role solely as presenters of molecules from such a specific group of pathogens. It is more likely that the diseases in which CD1 molecules have been shown to be important represent the tip of the iceberg. Further research is needed into the relevance of this arm of the immune response to immune-mediated (in particular autoimmune) disease and how these molecules might guide the immune response along advantageous or detrimental pathways. This research would be aided by investigation of CD1 expression patterns in various diseases in domestic rather than laboratory species, in particular in view of the recognized dissimilarities between the rodent CD1 system and those of other veterinary species. Many questions pertaining to the CD1 system remain to be answered. In particular it would be desirable to establish the CD1 status of domestic animals with the aim of facilitating subsequent investigations into the role of these molecules in infectious and immune-mediated disease.
Footnotes
Acknowledgements
I am grateful to J. Hopkins for critical review of the manuscript.
