Abstract
Paratuberculosis (Johne’s disease) is caused by Mycobacterium avium ssp. paratuberculosis (MAP), and affects both domestic and wild ruminants, including cattle, goats, sheep, and deer. In cattle, most infections occur during calfhood followed by a prolonged incubation period of 1–2 y or more before cows shed culturable numbers of MAP bacilli in their feces. As disease progresses, infected animals develop protein-losing enteropathy, intractable diarrhea, and weight loss. In a cohort of 32 clinically normal deer from a herd with a history of periodic clinical paratuberculosis, we found that subclinical infection was characterized by high rates of infection, common involvement of mesenteric lymph nodes, minimal lesion formation, few intralesional acid-fast bacilli, and low-level fecal shedding of MAP. The characteristics of subclinical paratuberculosis in white-tailed deer resemble those of cattle and red deer, although microscopic lesions were less common in subclinical deer than reported for subclinical cattle, and we did not see necrotizing granulomas as described in subclinical red deer and elk.
Introduction
Paratuberculosis (Johne’s disease) is caused by Mycobacterium avium ssp. paratuberculosis (MAP), a gram-positive, acid-fast bacillus and obligate intracellular pathogen. First described in Germany in 18955,21 and recognized in the United States in 1908, 5 paratuberculosis is a chronic enteritis of both domestic and wild ruminants, including cattle, goats, sheep, and deer. However, natural MAP infections have also been documented in many non-ruminant species,1,9,15 such as swine, 33 rabbits, 20 feral cats, 39 brushtail possums (Trichosurus vulpecula), 35 hedgehogs, hares, dogs, 32 birds, 9 and non-human primates. 30
The primary route of transmission is fecal–oral through ingestion of feces-contaminated milk, feed, or water. 21 In cattle, infection generally occurs at a young age, and although a small percentage of calves clear the infection, most calves will be infected for life. 24 Although most infections occur during calfhood, mature cows introduced to MAP-positive herds may also become infected. 14 Initial infection is followed by a prolonged incubation period of 1.5–2 y or more before cows shed culturable numbers of MAP bacilli in their feces; although in high-prevalence herds, 20% of cattle <2 y old may be shedding MAP in their feces. 56 Similarly, in some cases, infected young calves can be a source of infection for co-housed penmates. 8 As disease progresses, infected animals develop protein-losing enteropathy, intractable diarrhea, and weight loss, all hallmark clinical signs of bovine paratuberculosis.
MAP has been isolated from fallow deer (Dama dama),26,45,53 sika deer (Cervus nippon), 53 white-tailed deer (Odocoileus virginianus),22,25,49 key deer (Odocoileus virginianus clavium),34,44 axis deer (Axis axis), 45 red deer (Cervus elaphus),19,26,48,55 elk (Cervus elaphus canadensis),16,28 western roe deer (Capreolus capreolus),42,48 and tule elk (Cervus canadensis nannodes). 23 Paratuberculosis is well described in red deer in New Zealand where semi-intensive deer farming is well established and paratuberculosis is a serious problem.13,27,37 Elsewhere, both captive and free-ranging MAP-infected deer populations have been found.29,34,44,48,49 Both bovine and ovine strains of MAP exist, differing in host preference and necessary culture conditions for isolation, with the ovine strain being more difficult to isolate.7,46 Cattle are relatively resistant to infection with ovine strains; ovine strains readily infect sheep. Deer may be infected with either the bovine or ovine strain 10 ; however, deer are infected most commonly with the bovine type.11,12 Moreover, experimental infection studies demonstrate that red deer are more readily infected with the bovine strain than the ovine strain. 36
In general, clinical signs of paratuberculosis in deer are similar to those seen in cattle, specifically, chronic diarrhea, weight loss, and declining body condition. However, 2 clinical syndromes have been described in red deer: sporadic disease in mixed ages of deer characterized by low morbidity and high mortality, and severe outbreaks in young (8–15 mo old) deer, characterized by high morbidity and high mortality. 27 This latter syndrome is believed to be the result of high levels of pathogen exposure at a very young age. In white-tailed deer, there are various reports of experimentally induced paratuberculosis,38,58,59 and limited descriptions of natural infections.4,25,34,44,49 Accounts of natural infections are often case reports involving small numbers of deer. Even more limited is information on white-tailed deer herd-level infections. 60
The paucity of naturally occurring clinical cases in white-tailed deer makes conclusions regarding clinical syndromes, disease course, or fecal shedding patterns difficult. Although MAP has been isolated from subclinical white-tailed deer,4,44,60 observations are limited on MAP tissue colonization, lesion status, and fecal shedding of subclinical deer in a herd situation. 60 We examined a cohort of clinically normal white-tailed deer from a paratuberculosis endemic herd. Specifically, we examined the relationship between fecal shedding and the presence of lesions or culture-positive tissues in subclinical but MAP-infected white-tailed deer.
Materials and methods
Thirty-two clinically normal, female white-tailed deer were chosen randomly from a captive herd in which MAP infection was endemic. The presence of paratuberculosis in this herd had been established over a period of 3–4 y through autopsy, microscopic observation of granulomatous enteritis with acid-fast bacilli (AFB), and isolation of MAP from target organs of several deer with declining body condition and intractable chronic diarrhea.
Among the 32 selected deer, the mean age was 5 y (range: 3–12 y; Fig. 1). Fecal samples were collected 82 d before autopsy (fecal 1), 7 d before autopsy (fecal 2), and at the time of autopsy (fecal 3). For a period of 7 d prior to autopsy (between fecals 2 and 3), deer were housed indoors rather than on pasture, to prevent consumption of contaminated ingesta or water and decrease the likelihood of MAP “pass-through” with intestinal contents, in contrast to MAP fecal shedding associated with true infection. All procedures were conducted in accordance with recommendations in the Care and Use of Laboratory Animals of the National Institutes of Health and the Guide for the Care and Use of Agricultural Animals in Research and Teaching.18,50 Procedures were approved by the USDA National Animal Disease Center Animal Care and Use Committee.
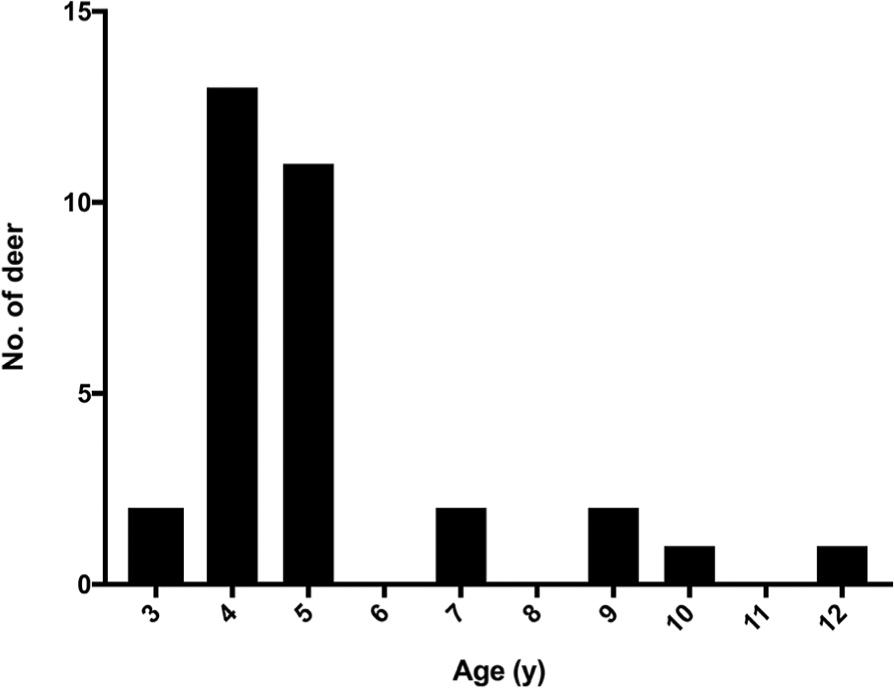
Age distribution of 32 clinically normal white-tailed deer from a herd with a history of clinical paratuberculosis.
Culture of feces and tissues for isolation of MAP
Tissues were frozen at −80°C until processed for isolation of MAP as described previously.47,54 For isolation of MAP from feces, 2.0 ± 0.2 g of feces were added to 35 mL of sterile water in a 50-mL conical tube. After vigorous shaking for at least 30 s to break up large clumps, the sample was allowed to sit at room temperature (RT) for at least 30 min to allow debris to settle to the bottom of the tube. Five milliliters of the supernatant were transferred to a new 50-mL conical tube containing 25 mL of 0.9% hexadecylpyridinium chloride (HPC) solution and mixed by shaking or vortexing. The suspension was incubated overnight at 37°C. Bacteria were collected by centrifugation at 900 × g for 30 min. The supernatant was discarded, and the pellet was resuspended in 1 mL of Johne’s antibiotic mix (100 µg/mL nalidixic acid, 100 µg/mL vancomycin, 50 µg/mL amphotericin B in sterile water) by vortexing for at least 15 s. The resulting sample was incubated overnight (12–24 h) at 37°C. The sample was then vortexed for a minimum of 15 s to resuspend the pellet and subsequently inoculated into broth (Trek para-JEM broth; Thermo Fisher Scientific, Waltham, MA) supplemented with 1 mL of Trek para-JEM GS, 0.5 mL of Trek para-JEM AS, 0.05 mL of Trek para-JEM Blue, and 2 mL of 100% egg yolk (prepared in-house). Samples were thoroughly mixed, and incubated for up to 60 d (Versa TREK system; Thermo Fisher). Positive cultures were confirmed by both Ziehl–Neelsen (ZN) acid-fast staining and IS900 PCR, as described below.
Culture confirmation using PCR
Proteinase K (100 µL, 10 mg/mL; MilliporeSigma, St. Louis, MO) was added to 500 mL of the liquid culture, and incubated at 50°C overnight. Bacteria were collected by centrifugation at 15,000 × g for 15 min. The lipid layer was removed using a sterile cotton swab and the remainder of the supernatant removed using a pipette. The pellet was resuspended in 175 µL of phosphate-buffered saline (PBS). The resuspended pellet was then transferred to an O-ring screw-cap tube containing 400 µL of TE (Tris–EDTA; MilliporeSigma), 400 µL of phenol–chloroform–isoamyl alcohol (MilliporeSigma), ~125 µL of 1.0-mm glass beads (Biospec Products, Bartlesville, OK), and ~1,258 µL of 0.1-mm glass beads (Biospec Products). Bacilli were then disrupted by shaking for 2 min in a bead beater (Biospec Products). The aqueous and organic phases were separated by centrifugation at 13,000 × g for 10 min. Up to 400 µL of the aqueous phase, containing the DNA, was transferred to a fresh tube containing 1.2 mL of DNA binding buffer (D6010-1-150; Zymo Research, Irvine, CA) and mixed. The DNA was captured on a spin column (Zymo-Spin IIC column C1011-50; Zymo Research) by centrifugation at 13,000 × g for 1 min. The column was washed (200 µL DNA pre-wash buffer, D3004-5-50; Zymo Research) by centrifugation at 13,000 × g for 1 min followed by another wash (500 µL fecal DNA wash buffer, D6010-2-100; Zymo Research). The filter was transferred to a new tube, the DNA eluted (100 µL elution buffer, D3004-4-10; Zymo Research), and centrifugation at 8,000 × g for 1 min. PCR detection of the IS900 element 31 was detected using the following primers 5’-AATCAACTCCAGCAGCGCGGCCTC and 5’-CCGCTAATTGAGAGATGCTGTAGG-3’ and probe 5’-/56-FAM/TCCACGCCC/ZEN/GCCCAGACAGG/31ABkFQ/-3’ in a quantitative PCR assay (PrimeTime Std; Integrated Technologies, Skokie, IL) using TaqMan universal PCR master mix (Thermo Fisher) according to the manufacturer’s instructions, with 5 µL of isolated DNA.
Direct fecal PCR testing
Fecal samples were frozen at −80°C until processed, when a 300-μg sample of feces was placed into 1 mL of PBS and mixed by vortexing for ~3 min. Solids were removed by centrifugation for 30 s at 100 × g. Avoiding solids, 175 mL of the supernatant was transferred to a tube containing 232 µL of MagMax lysis/binding solution (Thermo Fisher) prepared according to the manufacturer’s directions. The tubes also contained ~125 µL of 1.0-mm glass beads (Biospec Products), and ~1,258 µL of 0.1-mm glass beads (Biospec Products). Bacilli were then disrupted by shaking for 5 min in a bead beater (Biospec Products). The sample was clarified by centrifugation at 16,000 × g for 3 min. DNA was isolated from 20 µL of the clarified preparation (MagMAX total nucleic acid isolation kit; Thermo Fisher) as instructed by the manufacturer. Detection of the IS900 element by PCR was performed as described above.
Autopsy and tissue collection
Deer were euthanized by intravenous administration of sodium pentobarbital, and postmortem examinations conducted. Tissues were processed for microscopic analysis and isolation of MAP. Tissues collected included distal ileum, ileocecal valve, and lymph nodes associated with the ileocecal valve. Tissue samples (≤0.5 cm in width) were fixed by immersion in 10% neutral-buffered formalin for 24 h, and then transferred to 70% alcohol followed by standard paraffin-embedding techniques. Paraffin-embedded samples were cut to 4-μm thick sections, transferred to Superfrost Plus charged microscope slides (Thermo Fisher) and stained with hematoxylin and eosin. Adjacent sections were stained by the ZN technique for visualization of AFB. Near-adjacent unstained sections were used for in situ hybridization (ISH) analysis.
Messenger RNA chromogenic ISH
Messenger RNA ISH was performed (RNAscope ZZ probe technology; Advanced Cell Diagnostics, Newark, CA) on formalin-fixed, paraffin-embedded (FFPE) tissue sections (RNAscope 2.5 HD–RED kit; Advanced Cell Diagnostics) and a proprietary probe for MAP 16S mRNA (Cat 545801, Advanced Cell Diagnostics) as described previously.40,41
Results
Gross postmortem lesions were not present in the 32 deer examined. Microscopic granulomas were found in 1 of 32 deer; small granulomas composed of epithelioid macrophages, lymphocytes, and multinucleate giant cells were present in the ileum, ileocecal valve, and associated lymph nodes (Fig. 2). In this 1 animal, ZN staining demonstrated <3 AFB within macrophages of the lymph node microgranulomas (Fig. 3). Granulomas in all 3 tissues exhibited positive ISH labeling for MAP 16S mRNA.
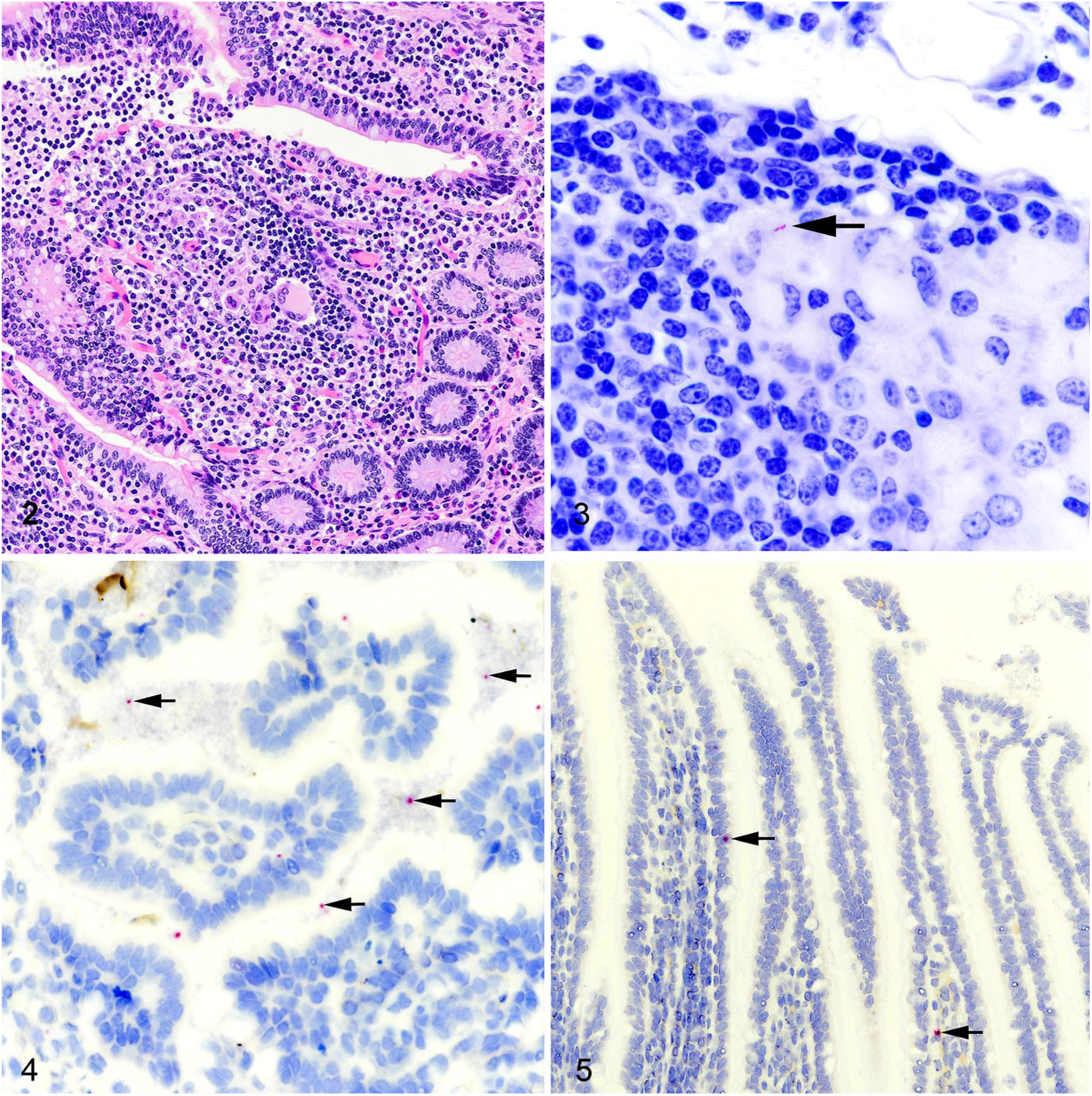
Granulomatous enteritis as a result of paratuberculosis in a white-tailed deer.
Although AFB were not seen in any of the tissues examined from the 31 remaining deer, positive ISH labeling was present in 19 of 31. Positive labeling for MAP RNA was generally limited to detached cells and cell debris within the intestinal lumens of the ileum and ileocecal valve (Fig. 4). Infrequently, positive labeling was displayed in cells within the epithelium or lamina propria (Fig. 5). In 18 of 19 deer with demonstrable labeling of MAP RNA, viable MAP was recovered from at least 1 tissue. In one deer, positive ISH labeling was present when no viable MAP were recovered from tissues or feces at any of the sampling times.
Isolation of MAP from tissues collected at autopsy was successful in 20 of 32 deer (Table 1). Lymph nodes associated with the ileocecal valve were most frequently affected (16 of 20), followed by ileum (8 of 20) and the ileocecal valve (7 of 20). In 4 of the 20 infected deer, all 3 tissues (ileum, ileocecal valve, and associated lymph nodes) contained viable MAP.
Results of isolation of Mycobacterium avium ssp. paratuberculosis (MAP) from feces and tissues, and direct PCR (Ct values) of MAP DNA from feces of 32 clinically normal deer from a herd with endemic MAP infection. Fecal samples were collected 3 times: 82 d (fecal 1) and 7 d (fecal 2) before autopsy and at the time of autopsy (fecal 3).

Ct = cycle threshold value; IC = ileocecal; LN = lymph node; − = no isolation of viable MAP; + = isolation of viable MAP; PCR = presence of detectable MAP DNA but negative for isolation of MAP.
Viable MAP were recovered from 1, 2, and 4 deer each at fecals 1, 2, and 3, respectively. Of the 5 total deer from which viable MAP were recovered from feces, 2 deer had MAP-containing samples at both fecals 2 and 3; the remaining 3 deer were positive at only 1 fecal collection time (Table 1).
Direct fecal PCR detected MAP DNA at various levels in 11, 2, and 2 deer each at fecals 1, 2, and 3, respectively (Table 1). None of the deer had culture-negative but PCR-positive feces at >1 sampling time. Three deer had culture-negative but PCR-positive feces and culture-positive feces at different sampling times (Table 1). Fifteen deer had no detectable MAP DNA in feces collected at any of the 3 fecal sampling points; however, 10 of these 15 deer had viable MAP recovered from at least 1 tissue at autopsy. The mean PCR cycle threshold (Ct) value for fecal samples that were both PCR-positive and culture-positive (data not shown) was 37.5 (95% confidence interval [CI]: 36.3, 38.6); the mean Ct among 15 fecal samples that were PCR-positive, but no viable MAP were recovered, was 38.0 (95% CI: 36.2, 38.8).
Discussion
The most common tissues from which we isolated MAP were the mesenteric lymph nodes associated with the ileocecal valve. In cattle, this is often a recommended tissue for sampling; although, in some cattle studies this lymph node has frequently been negative for the presence of MAP. 52 Mesenteric lymph nodes are also a common site for MAP colonization in farmed red deer, especially in subclinical or mild clinical cases.6,13,43,51 Similar to the findings of our study, of 619 farmed red deer from which MAP was isolated, 89.7% were identified from mesenteric lymph nodes, 583 (94.2%) of which were from subclinically affected animals. 13 In our study, the isolation of MAP from 80% of lymph nodes of infected deer is significant, given that infection of mesenteric lymph nodes is a definitive sign that MAP bacilli have crossed the intestinal epithelial barrier, traversed lymphatic vessels, and colonized the lymph node, and have not been passively carried through the intestine in MAP-contaminated ingesta or water.
Among deer in our study, MAP shedding was inconsistent. In cattle, both subclinical and clinical paratuberculosis may be characterized by low-level, intermittent fecal shedding. 57 Shedding of MAP is intermittent even in experimentally infected calves, and in calves infected through contact with experimentally infected calves. 8 Determination of the infectious nature of subclinical white-tailed deer through fecal shedding is unclear. In our study, high Ct values from direct fecal PCR and the intermittent presence of viable MAP or MAP DNA in fecal samples from the majority of the deer suggest that MAP shedding by these subclinical white-tailed deer was low. To further assess potential infectiousness, quantitative culture of feces would be required.
Cattle with paratuberculosis are described as passing through 4 stages. 57 Stage 1 represents silent infection of either young calves or adults. Stage 2 is subclinical disease of adults; stages 3 and 4 represent clinical and advanced clinical disease, respectively. 57 Stages 2–4 are associated with various degrees of fecal shedding of MAP, with advanced disease cases often shedding >108 CFU/g of feces. 5 The “iceberg” effect of Johne’s disease suggests that, for each advanced clinical case in a herd, there may be as many as 25 infected but unrecognized animals. 57 It is unclear if MAP-infected captive white-tailed deer herds demonstrate the “iceberg” effect or pass through the 4 stages of disease described in cattle. However, in our study there were deer representative of stage 1, silent infection with tissues colonized by MAP, but no fecal shedding, and stage 2 with low-level, intermittent shedding in the absence of clinical disease. In addition, historically, periodic cases consistent with advanced clinical disease (stage 4) had been recognized in this herd.
Microscopic lesions in subclinical cattle are similar to those seen in our study, composed of low-to-moderate numbers of epithelioid macrophages, lymphocytes, and Langhans-type multinucleate giant cells. 3 In our study, intralesional AFB were not present or present in very low numbers, as may be seen in subclinical cattle. 3 However, acid-fast staining of FFPE slides is known to be an insensitive means of demonstrating intralesional bacilli. 17 Detection of MAP nucleic acids through more sensitive methods such as ISH or PCR suggests the presence of more intralesional bacilli; however, it is important to note that nucleic acid detection methods reflect the presence of both viable and non-viable bacilli. 17
In our study, microscopic lesions were rarely seen (1 of 20 infected deer). In contrast, in a study of 15 subclinical, MAP-infected cattle, all had microscopic granulomas, including 7 without corresponding gross lesions. Similarly, compared to histopathology reports of American bison (Bison bison) with subclinical paratuberculosis, 2 deer in our study had far fewer granulomatous lesions; 1 of 20 subclinical deer compared to 12 of 23 subclinical bison. Moreover, 2 of 12 subclinical bison had multibacillary granulomas, although all lesions in our deer were paucibacillary.
A microscopic feature of paratuberculosis in naturally infected red deer and experimentally infected elk is the presence of necrotizing granulomas, with or without mineralization and variable numbers of intralesional AFB, causing the lesions to resemble those of tuberculosis caused by Mycobacterium bovis.10,13,59 Granulomas with necrosis have been reported as a feature of experimentally induced paratuberculosis in white-tailed deer38,58,59; however, it is not reported in naturally infected white-tailed deer, and was not seen in our study.
In this cohort of white-tailed deer from a herd with a known history of periodic clinical paratuberculosis, subclinical disease was characterized by high rates of infection, common involvement of lymph nodes associated with the ileocecal valve, minimal lesion formation, few intralesional AFB, and low-level fecal shedding of MAP. Limitations of our study include the evaluation of only ileum, ileocecal valve, and associated lymph nodes, thus missing infections of other portions of the small or large intestines or extraintestinal infection. Furthermore, rate of infection, tissue distribution, and lesion severity may all be influenced by exposure dose, repeated exposure, and age at exposure 52 ; as such, this cohort of subclinical white-tailed deer may not be representative of subclinical deer in situations of higher or lower exposure to MAP.
Footnotes
Acknowledgements
We thank animal caretakers, Lisa Ashburn, Hannah Schroeder, Robin Zeisneiss, and David Lubbers, for excellent animal care, and Shelly Zimmerman, Jayne Wiarda, Allison Lasley, and Hahley Fisher for excellent technical assistance.
Declaration of conflicting interests
Mention of tradenames or commercial products is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the U.S. Department of Agriculture.
Funding
Research funds were provided by the U.S. Department of Agriculture, Agricultural Research Service Project (CRIS 5030-32000-222).
