Abstract
Bacteria in the Mycobacterium tuberculosis complex and nontuberculous mycobacteria may affect a variety of animal species under human care and pose public health risks as zoonotic pathogens. A case of sudden onset of lethargy and increased respiratory effort in a 5-y-old, intact female reindeer (Rangifer tarandus) under managed care had progressed to severe dyspnea despite aggressive treatment. The animal was euthanized due to poor prognosis. Postmortem findings included: disseminated miliary nodules in the lungs, pleura, small intestine, liver, and spleen; enlarged mesenteric lymph nodes; dilated mesenteric and serosal lymphatic vessels; and renal infarcts. Histologically, granulomatous lymphadenitis and lymphangitis with intrahistiocytic acid-fast bacilli were observed. Mycobacterium sp. DNA was detected in lung via real-time PCR. Mycobacterial culture and sequencing identified Mycobacterium avium subsp. hominissuis (MAH) within pulmonary lesions. Infection with MAH has been reported in humans and many animal species; this nontuberculous mycobacterial infection may be an emerging concern in animals under managed care. To our knowledge, MAH infection has not been reported previously in reindeer.
Mycobacteria are non–spore-forming, non-motile, acid-fast bacteria of veterinary and public health concern, and include Mycobacterium tuberculosis, the predominant cause of tuberculosis (TB) in humans, and M. bovis, the principal cause of bovine TB. 17 Before the COVID-19 pandemic, TB was the leading cause of death from infectious disease in humans globally in the 21st century. 22 Organisms within the M. tuberculosis complex (MTBC) can cause zoonotic and reverse zoonotic disease in a wide range of animal hosts.15,23
Most Mycobacterium spp. are not included in the MTBC and are referred to as atypical mycobacteria or nontuberculous mycobacteria (NTM). NTM are environmental bacteria found in water, soil, and dust, and are most commonly responsible for opportunistic infections in immunocompromised individuals. 13 Clinically relevant NTM in humans include organisms in the Mycobacterium avium complex (MAC) that cause pneumonia in immunocompromised patients and disseminated disease in patients with acquired immunodeficiency syndrome (HIV/AIDS) worldwide.12,16 M. avium subsp. hominissuis (MAH) is one of the most significant MAC agents involved in human infection, and has been associated with chronic pulmonary disease and peripheral lymphadenomegaly in children. 16 MAH has also been reported in a variety of veterinary species; however, MAH infection has not been reported in a reindeer (Rangifer tarandus), to our knowledge.
A 5-y-old, intact female reindeer at a zoo was examined following acute onset of lethargy. On visual examination, the animal had a hunched posture and increased respiratory effort. The animal had been brought to the zoo as a juvenile with no significant medical history and was housed with 6 other female reindeer in an outdoor enclosure with dirt-mulch substrate and access to trees and brush. Given continued lethargy and onset of coughing despite initiation of minor supportive care (omeprazole), a physical examination was performed under anesthesia the following day.
CBC revealed an inflammatory leukogram with a left shift: WBC count of 3.47 × 109/L (RI: 1.8–3.1 × 109/L; 3,470 cells/µL [RI 1.8–3.1 cells/µL]), 2.71 × 109/L segmented neutrophils (RI: 0.91–2.08 × 109/L; 2.71 × 103/µL [RI: 0.91–2.08 × 103/µL]) with mild toxic change, and 0.03 × 109/L band neutrophils (0.03 × 103/µL). Serum biochemistry revealed elevated activities of aspartate aminotransferase (AST; 3.74 µkat/L [224 U/L]; RI: 0.72–2.12 µkat/L [43–127 U/L]), creatine kinase (14.6 µkat/L [872 U/L]; RI: 0.92–11.1 µkat/L [55–666 U/L]), and gamma-glutamyl transferase (GGT; 5.66 µkat/L [339 U/L]; RI: 0.1–0.72 µkat/L [6–43 U/L]). Aerobic bacterial culture from a swab taken from within the endotracheal tube following extubation was negative, and no severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) DNA was detected in a tracheal swab sample using a commercial PCR (University of Illinois–Veterinary Diagnostic Laboratory [UI-VDL], Urbana, IL, USA). Tracheal wash cytologic findings indicated acute inflammation, and bacterial infection was suspected. Antibodies to Aspergillus spp., Blastomyces spp., Coccidioides spp., Histoplasma spp., or Cryptococcus spp. were not detected in serum via agar gel immunodiffusion (Animal Health Diagnostic Center, College of Veterinary Medicine, Cornell University, Ithaca, NY, USA). Thoracic radiographs revealed a small, focal, alveolar pattern superimposed on the caudoventral cardiac silhouette. Laboratory testing and imaging supported a diagnosis of pneumonia, and differential diagnoses included primary infection (bacterial, viral, protozoal, or, less likely, fungal) or aspiration. A rigorous protocol of supportive care (Suppl. Table 1) was applied, during which the animal initially improved, based on visual assessment, before respiratory decline over the following days.
Six days following the initial workup, the patient was anesthetized again for additional testing including CBC (WBC count: 5.54 × 109/L [5,540 cells/µL], neutrophils: 4.49 × 109/L (4.49 × 103/µL), with reduced toxic change and resolved left shift) and serum biochemistry (mild changes consistent with cachexia: hypophosphatemia (0.9 mmol/L; RI: 1.74–2.78 mmol/L [2.8 mg/dL; RI: 5.4–8.6 mg/dL]), hypocalcemia (1.73 mmol/L; RI: 2.23–2.5 mmol/L [6.9 mg/dL; RI: 8.9–10 mg/dL]), hypoproteinemia (49 g/L; RI: 62–79 g/L [4.9 g/dL; RI: 6.2–7.9 g/dL]), increased AST (9.62 µkat/L; RI: 1.47–2.35 µkat/L [576 U/L; RI: 88–141 U/L]) and GGT (2.17 µkat/L; RI: 0.43–0.52 µkat/L [130 U/L; RI: 26–31 U/L]) activities, and hyperbilirubinemia (17.1 µmol/L; RI: 0–8.55 µmol/L [1.0 mg/dL; RI: 0–0.5 mg/dL]). A transtracheal wash was negative for bovine respiratory syncytial virus (BRSV), bovine alphaherpesvirus 1 (BoAHV1; infectious bovine rhinotracheitis), and bovine parainfluenza virus 3 via a commercial real-time PCR (rtPCR; UI-VDL). No serum antibodies were detected to BRSV or BoAHV1. Thoracic and abdominal computed tomography (CT) identified a severe, diffuse, unstructured, pulmonary interstitial pattern with linear soft-tissue striations (Suppl. Fig. 1A–C). Abdominal CT was unremarkable. Pulmonary changes were most severe around airways. Ventrally, a patchy alveolar pattern was also observed. These findings were consistent with severe generalized lower airway inflammation with interstitial pneumonia and ventrally distributed bronchopneumonia. Infectious pneumonia remained an important differential. Inflammatory (i.e., eosinophilic bronchopneumopathy) and toxic causes of interstitial pneumonia (i.e., atypical interstitial pneumonia due to ingestion of L-tryptophan, Perilla frutescens [perilla mint], or 4-ipomeanol from moldy sweet potatoes [Ipomoea batatas] infected with Fusarium solani) were considered less likely. 9
The animal was hospitalized and treated for 10 d (Suppl. Table 1). Minimal initial improvement was followed by progressive decline with severe dyspnea and lethargy on visual examination; therefore, euthanasia was elected.
Postmortem examination revealed gross lesions in various tissues of the thoracic and abdominal cavities highly suggestive of a disseminated infectious disease. The lungs failed to collapse upon entering the thorax. The pulmonary parenchyma was pliable and diffusely dark-red–purple with myriad, randomly distributed, pinpoint-to-2.5-cm, yellow-white, dull, granular, firm nodules that thickened and elevated the visceral pleura (Fig. 1A). The mediastinal, visceral, and costal parietal pleura had many similar, scattered, pinpoint-to-2-cm, often coalescing, nodules. Ziehl-Neelsen–stained impression smears of lung nodules contained foamy macrophages with rare intracytoplasmic, acid-fast bacilli. Mesenteric lymph nodes bordering a segment of proximal jejunum were markedly enlarged to 8 × 6 × 5 cm; >80% of the nodal parenchyma was discolored yellow-white, firm, and granular, or was replaced by small-to-moderate amounts of opaque yellow, mucoid material. The surrounding nodal parenchyma was dark-red. Lymphatic vessels extending from affected jejunal serosal surfaces to mesenteric lymph nodes were tortuous, markedly ectatic, and discolored yellow-tan (Fig. 1B). The segmentally affected jejunal serosa was up to 5-mm thick, with coalescing nodules similar to those noted in the lungs. Similar nodules were scattered randomly throughout the liver and spleen. Other gross findings included severe tracheal mural edema, submandibular edema, diffuse serous atrophy of fat, mild peritoneal effusion, and bilateral renal cortical infarcts.
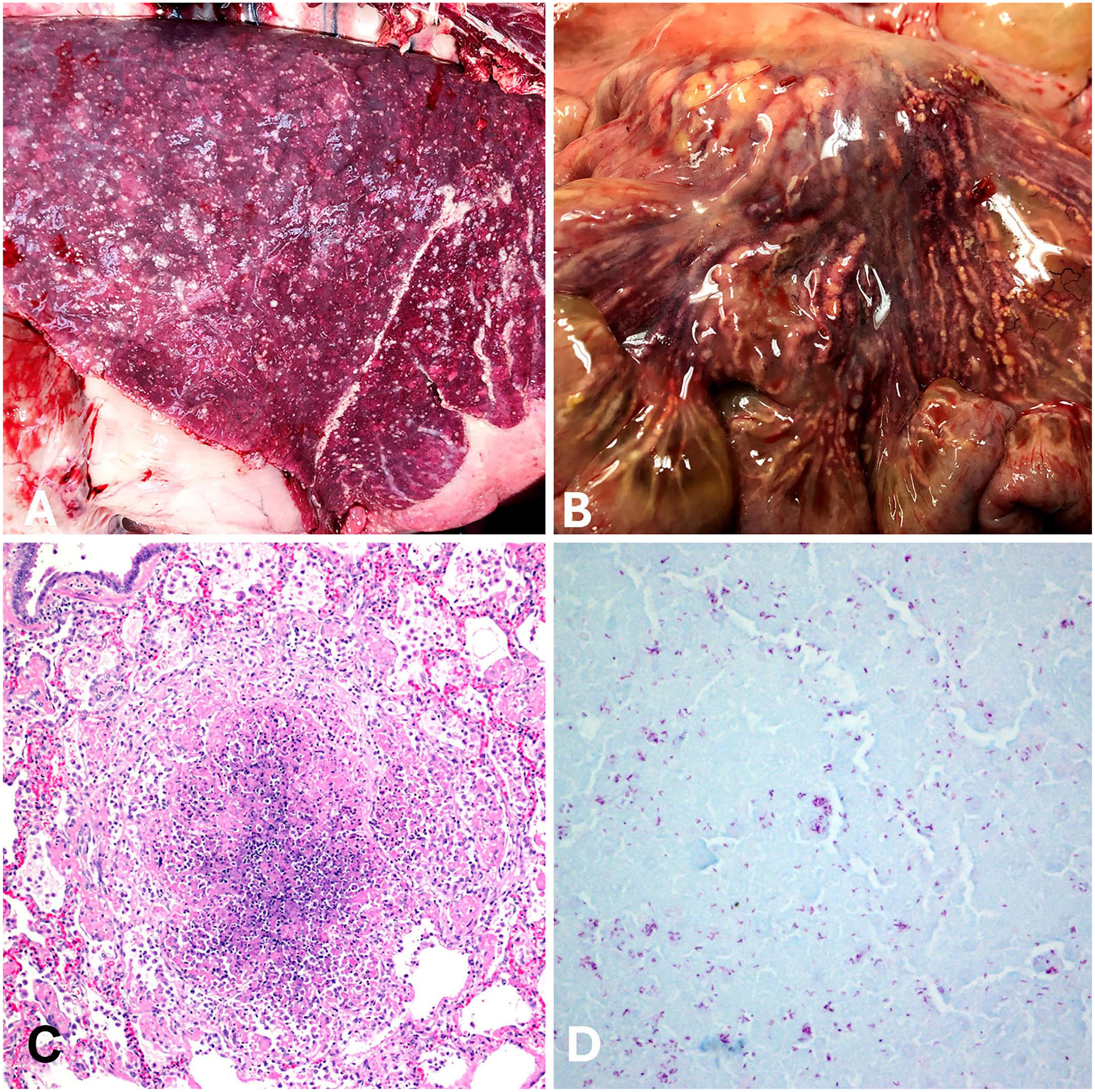
Disseminated nontuberculous mycobacteriosis due to Mycobacterium avium subsp. hominissuis disseminated infection in a reindeer (Rangifer tarandus).
Histologically, severe necrotizing, granulomatous pleuropneumonia was observed with many randomly distributed, discrete, variably sized nodules with abundant central necrotic debris bordered by moderate-to-high numbers of epithelioid macrophages and a few multinucleate giant cells that replaced the pulmonary parenchyma (Fig. 1C). Admixed with central necrotic debris and within the cytoplasm of macrophages and multinucleate giant cells were low-to-moderate numbers of acid-fast bacilli (Fig. 1D). Several affected areas included vascular profiles with transmural necrosis, absent endothelium, and thrombosis. Interlobular pulmonary septa were markedly expanded with edema, and lymphatic vessels were dilated and filled with proteinaceous fluid and/or inflammatory debris.
In histologic sections of mesenteric lymph nodes, >75% of the parenchyma was necrotic with severe lymphoid depletion. Necrotic tissue was bordered by fibrin, admixed karyorrhectic debris, and variable numbers of macrophages and multinucleate giant cells. The tracheobronchial lymph nodes had similar microscopic changes. Mesenteric lymphatics were ectatic and had inapparent endothelium; vessel walls were segmentally necrotic and/or expanded and obscured by mural fibrin, cellular debris, macrophages, and fewer neutrophils.
Segmentally, the proximal jejunum was expanded and distorted by severe transmural granulomatous inflammation. The mucosal surface was focally ulcerated; the submucosa and muscularis were markedly thickened with edema and granulomas; the corresponding serosa was markedly expanded by edema and wide tortuous tracts of necrosis and bordering granulomatous inflammation. Both the liver and spleen had many variably sized, randomly distributed foci of necrotizing and granulomatous inflammation similar to that noted in other tissues. Renal cortical infarcts observed grossly corresponded to locally extensive necrotizing and granulomatous vasculitis and nephritis.
Samples of fresh lung were positive for bacterial 16S rRNA via conventional PCR, and Mycobacterium sp. DNA was detected via a genus-specific commercial rtPCR assay (Zoological Pathology Program [ZPP], University of Illinois, Brookfield, IL, USA). Sequence analysis (University of Chicago DNA Sequencing Facility, Chicago, IL, USA) of Mycobacterium sp. DNA amplified from fresh lung tissue, utilizing primers targeting the heat-shock protein gene of the bacterium, was consistent with M. avium with >99% homology. The available amplified sequence at that time was insufficient to distinguish M. avium subspecies using publicly available libraries. Generic fungal, Nocardia sp., and Histoplasma capsulatum rtPCR assays were all negative (ZPP).
Based on preliminary results (i.e., positive rtPCR for a Mycobacterium sp. and acid-fast bacilli observed on impression smears of the lung), regional authorities were contacted for guidance. Mesenteric lymph node and lung tissue samples were sent to the National Veterinary Services Laboratories (NVSL; Ames, IA, USA) for mycobacterial culture and MTBC direct PCR. Direct PCR was negative, and the cultured isolate was identified via Sanger-sequencing the regions of the 16S and RNA polymerase subunit beta (rpoB) genes. An 818-bp 16S gene sequence and a 689-bp rpoB gene sequence were both homologous to M. avium spp., with the more discriminatory rpoB gene sequence grouping among M. avium subsp. hominissuis sequences. Sequence identification of MAH was confirmed with maximum-likelihood analysis.
Given our initial suspicion of infectious disease and possible zoonosis in this case, proper biosecurity and safety measures were employed, including use of appropriate personal protective equipment and decontamination protocols. Exposed personnel underwent intradermal TB skin testing within 2 wk of exposure to the sick animal as a precaution, and no reactors were detected. Pending final diagnosis, access to the reindeer habitat was restricted, and the herd was screened for TB (Dual-path platform Mycobacterium tuberculosis assay; NVSL) under the direction of state veterinary authorities. All herdmates tested negative.
In addition to humans, MAH has been detected in other mammals including cattle, 19 swine, 5 deer (Cervus spp.), 7 mule deer (Odocoileus hemionus), 6 sitatunga (Tragelaphus spekii), 18 dogs, 8 domestic rabbits, 10 horses, 11 lowland tapirs (Tapirus terrestris), 13 an African elephant (Loxodonta africana), 1 and less commonly in birds: blue-fronted Amazon parrot (Amazona aestiva) 20 and cattle egret (Bubulcus ibis). 4 Animals infected with MAH can have variable clinical signs and gross and microscopic lesions affecting various organs and tissues. Red deer (Cervus elaphus) infected with MAH and M. avium subsp. paratuberculosis (MAP) had mixed lesion distribution, with some individuals having abnormalities restricted to the intestinal tract (purulent, caseous, or granulomatous lesions of different sizes); others also had pulmonary, hepatic, and mediastinal lymph node involvement. 7 M. kansasii in a reindeer and in a bontebok (Damaliscus pygargus dorcas) led to systemic inflammation, and the organism was cultured from the lungs, kidneys, liver, and lymph nodes.14,21 A tundra reindeer (Rangifer tarandus tarandus), in which MAP was detected by PCR in intestinal and lung tissue, had associated lesions of granulomatous ileitis, granulomatous mesenteric lymphadenitis, and focal granulomatous pneumonia with caseous necrosis. 2 This, with our case, may be evidence of MAP leading to disseminated lesions in reindeer.
Our case is an example of an atypical mycobacterial infection with a spectrum of lesions including features suggestive of both atypical NTM (e.g., M. avium) and MTBC infections. Lesions observed in the lymphatic vessels of the mesentery and intestinal serosa, mesenteric lymph nodes, and small intestinal wall were reminiscent of paratuberculosis due to MAP in domestic and wild ruminants. However, disseminated infection with pneumonia, as seen in our case, is not typical for MAP and is more consistent with MTBC infection. Caseous lymphadenitis caused by Corynebacterium pseudotuberculosis is another possible differential for similar pulmonary and lymph node lesions that have been seen in roe deer (Capreolus capreolus) and red deer. 3 Ancillary tests were necessary to confirm infection with the NTM organism MAH. Pathologic features may have been reflective of a unique or enhanced disease susceptibility in this individual reindeer and/or elevated susceptibility of the entire taxa as suggested by disseminated disease reported in other cervids.
Our searches of PubMed and Google Scholar using the search terms “reindeer/Rangifer tarandus disease/health/Mycobacterium”, “Mycobacterium avium subsp. hominissuis”, and “wildlife Mycobacterium avium subsp. hominissuis” returned no cases of this non-tuberculous mycobacteria affecting reindeer, which suggests that MAH infection has not been reported previously in a reindeer. The broad taxa range and expanding list of species that have been affected by MAH suggests that this organism is an emerging pathogen of importance in animals under managed care.
Supplemental Material
sj-pdf-1-vdi-10.1177_10406387241307644 – Supplemental material for Disseminated nontuberculous mycobacteriosis due to Mycobacterium avium subsp. hominissuis infection in a reindeer from a zoo
Supplemental material, sj-pdf-1-vdi-10.1177_10406387241307644 for Disseminated nontuberculous mycobacteriosis due to Mycobacterium avium subsp. hominissuis infection in a reindeer from a zoo by John M. Winter, Jennifer N. Langan, Jennifer A. Landolfi, Tyler Thacker and Katie W. Delk in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We thank the animal care and hospital staff for their efforts with this case. The University of Illinois–Veterinary Diagnostic Laboratory histology laboratory provided all slide-preparation services. Dr. Megan Colburn contributed to pathology interpretations.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
