Abstract
Myeloid sarcoma (MS) is a solid tumor of granulocytic origin with extramedullary localization. This tumor is rare in humans and animals. The diagnostic approach is heterogeneous, and the definitive diagnosis may be difficult to achieve. Primary MS has never been described as a spontaneous neoplasm in companion dogs. Two purebred and 1 mixed-breed dogs, 6- to 11-year-old, developed round cell tumors in the mediastinum, lymph nodes (LNs) and tonsils, and LNs, respectively. Granulocytic origin and exclusion of lymphoid lineage were confirmed by flow cytometry, supported by immunohistochemistry or immunocytochemistry. Pivotal to the diagnosis were positive labeling for myeloid (CD11b, CD14) and hematopoietic precursors (CD34) markers, along with negative labeling for lymphoid markers. Blood and bone marrow infiltration were not detected at initial diagnosis, excluding acute myeloid leukemia. The behavior of these tumors was aggressive, resulting in poor clinical outcomes, even when chemotherapy was attempted.
Keywords
Myeloid sarcoma (MS) is a rare tumor of granulocytic origin developing outside of the bone marrow (BM) and peripheral blood (PB). It can precede clinical or hematological signs of acute myeloid leukemia (AML), or represent a separate event 4 or, more commonly, be diagnosed concomitantly with AML.2,7,9 A diagnosis of MS may be suspected based on patient’s clinical history and imaging results,10,13 but the definitive diagnosis is confirmed by PB and BM assessment and demonstration of granulocytic origin by immunohistochemistry on tissue specimens. Also, cytology supported by immunocytochemistry, flow cytometry (FC), and/or molecular analysis has been performed to obtain a definitive diagnosis. 15 Herein, we report for the first time 3 cases of MS in dogs.
Case 1 was a 7-year-old, spayed-female Doberman presented for a cough of 2 weeks duration. The laboratory diagnostic workup, including complete blood count (CBC) and biochemistry, was unremarkable. Thoracic radiographs showed pleural effusion and a mediastinal mass. Echocardiography was unremarkable. Cytologic evaluation of the effusion was performed by a European College of Veterinary Clinical Pathologists diplomate in a private laboratory. The effusion was characterized by high cellularity (14.5 × 103 WBC/µl) with pleomorphic round cells, admixed with small mature lymphocytes, macrophages, and reactive mesothelial cells. Reactive lymphoid effusion was the main differential, but analysis identified 42% CD45+CD11b+ cells consistent with a myeloid origin; 43% of them coexpressed CD14 (monocytes) and 16% coexpressed CD34 (hematopoietic precursors). Less than 15% of the cells expressed lymphoid markers, consistent with reactive B-cells and T-cells (Table 1 and Supplemental Materials “Flow cytometry and immunohistochemistry methods”). As almost no segmented neutrophils or monocytes were observed in the cytological specimens, FC findings were considered consistent with a myeloid neoplasm, and BM evaluation was recommended. One week later, the dog developed a hematologic profile compatible with acute leukemia (leukocytosis, anemia, thrombocytopenia, and 50% atypical unclassified cells) as reported by a private laboratory. The dog died 20 days later. Necropsy led to a diagnosis of round cell tumor with multiple organ involvement (Supplemental Materials “Case 1”). Relevant gross findings included enlargement of peripheral and internal lymph nodes (LNs) and tonsils, splenomegaly, serohemorrhagic thoracic effusion, and a white-tan, multilobulated neoplastic mass adherent to the lung lobes (Fig. 1a, b). Microscopically, the LNs, spleen, and BM were effaced by dense sheets of 10 to 16 µm diameter (occasionally up to 30 µm), undifferentiated neoplastic round cells (Fig. 1c, d) with high nuclear to cytoplasmic ratios, an incomplete rim of lightly eosinophilic cytoplasm, round nuclei with granular chromatin, and 1 to 3 round basophilic nucleoli. In the liver, neoplastic cells were in hepatic sinusoids, surrounded central hepatic veins, and infiltrated and bridged portal spaces. In the kidney, neoplastic cells were in the periglomerular and cortical interstitium. Neoplastic cells also filled renal, meningeal, and cerebral vascular lumens (Supplemental Figures S1a–d). Owing to the widespread distribution of the disease at necropsy, it was impossible to determine the primary origin of the tumor. However, the presence of the mediastinal mass preceding the development of peripheral leukemia and the FC characteristics consistent with a granulocytic neoplasm, led to the diagnosis of MS.
Flow cytometric phenotype of neoplastic cells in 3 cases of canine myeloid sarcoma.
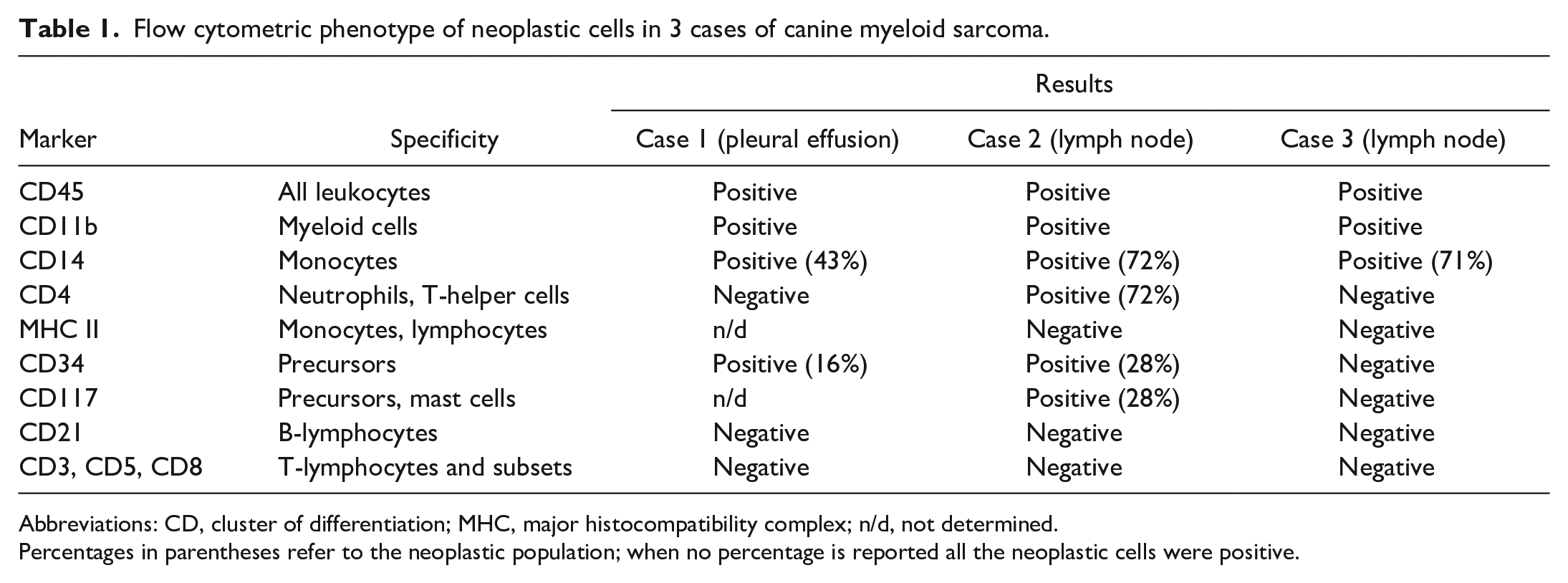
Abbreviations: CD, cluster of differentiation; MHC, major histocompatibility complex; n/d, not determined.
Percentages in parentheses refer to the neoplastic population; when no percentage is reported all the neoplastic cells were positive.
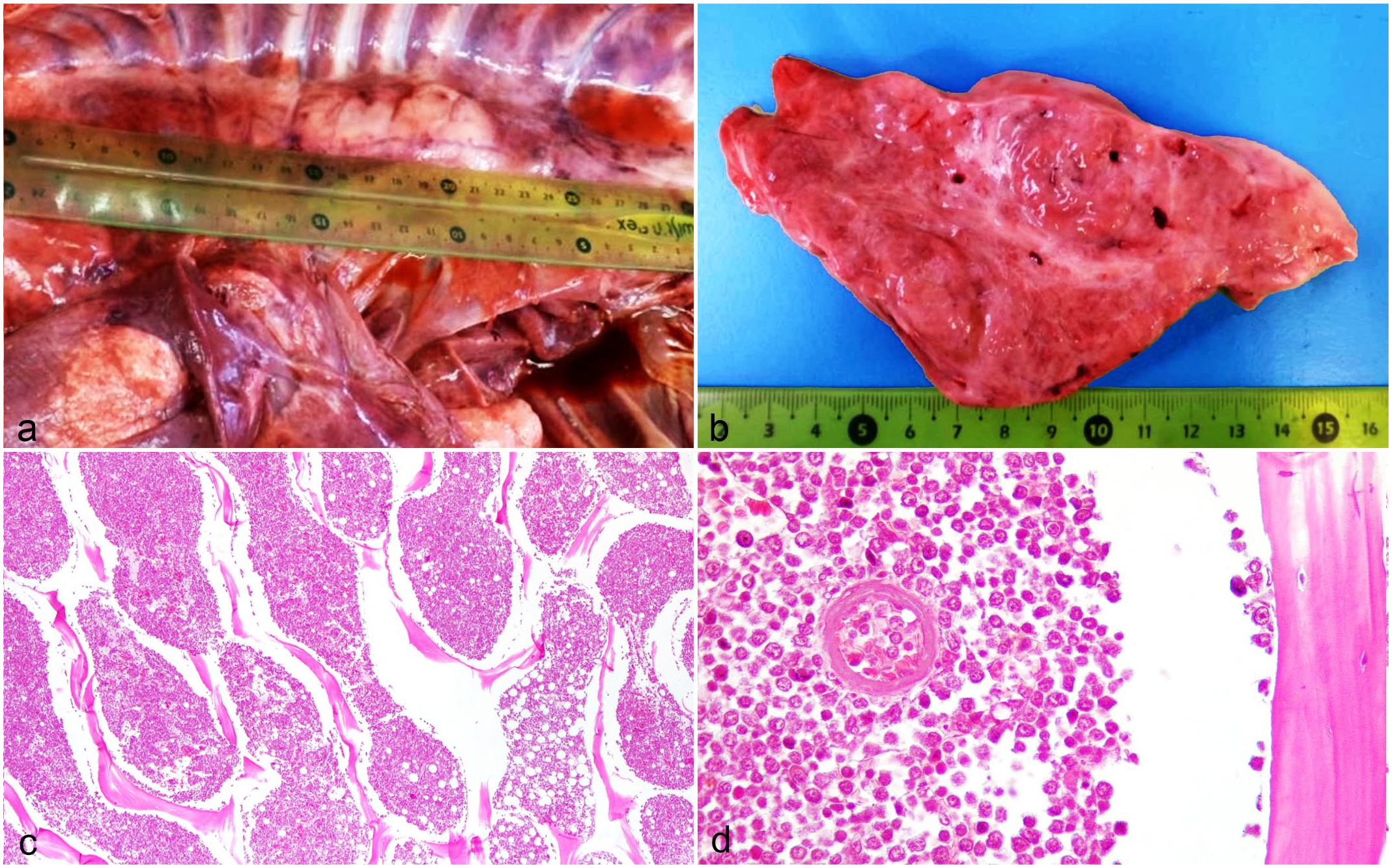
Macroscopic and histopathologic features of myeloid sarcoma in 1 dog. Case 1. (a) Thorax. A multilobulated white-tan mass measuring approximately 14 cm × 7 cm involving the cranial mediastinum and the thoracic inlet. The lung lobes adjacent to the mass are atelectatic, and there is mild serosanguineous thoracic effusion. (b) Cut section of the mass. The tissue appears lardaceous and dissected by fibrous stalks. (c) Histology of bone marrow sampled at necropsy. Highly cellular bone marrow with diffuse infiltration by neoplastic round cells (Fig. 1d) substituting the normal cellular components and fat. Demineralized sample; hematoxylin and eosin (HE). (d) Bone marrow at higher magnification, the neoplastic cells have high nuclear to cytoplasmic ratio, with an incomplete rim of variably eosinophilic cytoplasm, round nuclei with granular chromatin, and 1 to 3 round basophilic nucleoli. One vessel lumen contains neoplastic cells. Demineralized sample; HE.
Case 2 was a 6-year-old, intact female golden retriever that presented with generalized lymphadenopathy and bilateral tonsillar enlargement. The CBC was unremarkable. Thoracic radiographs and abdominal ultrasound revealed enlargement of iliac LNs. Lymphoma was suspected based on clinical examination. Cytology of prescapular and popliteal LNs revealed 37% large blast cells with small amounts of basophilic cytoplasm, single round or indented nuclei, fine granular chromatin, and prominent nucleoli, admixed with numerous myeloid cells at different stages of maturation showing dysplastic/toxic changes; occasional small undifferentiated cells with red-purple granules were present (Fig. 2a). The differential diagnosis included extramedullary myelopoiesis and myeloid leukemia. FC from a nodal aspirate revealed 2 prevalent populations: 1 CD11b+CD14+CD4+CD34−CD117− (38%) and 1 CD11b+CD14−CD4−CD34+CD117+ (15%). Both populations lacked major histocompatibility complex (MHC) II and lymphoid markers (Table 1, Supplemental Table S1, and Supplemental Materials “Flow cytometry and immunohistochemistry methods”). Thus, the immunophenotype was consistent with a predominance of granulocytic cells. Based on blood smears and FC, the PB was devoid of immature myeloid cells. A CBC was repeated 10 days later (Supplemental Table S2, day 13) and showed no abnormalities except for onset of mild anemia. Simultaneously, a BM aspirate from the iliac crest was examined. A slight left shift of the granulocytic lineage with blast cells accounting for 5% was the main alteration on cytology; CD34+ cells accounted for 1.9% in the FC. The finding of myeloid cells in the nodes, the absence of PB cytopenias or dysplastic features, together with the low number of blast cells in the BM, led to the exclusion of myelodysplastic syndrome and AML, and prompted suspicion of MS. Twenty days later (day 35), the patient developed grade I neutropenia, 6 and 46% dysplastic myeloid cells and rare unclassifiable cells (<1%) were observed on a PB smear (Supplemental Table S2). The right popliteal LN was surgically removed for diagnostic purposes, and histopathology and immunohistochemistry were performed. Nodal architecture was partially effaced, and the cortex and paracortex were expanded by dense sheets of medium to large, round neoplastic cells with a high mitotic count and several atypical mitoses. Neoplastic cells did not stain with toluidine blue (mast cells) and were negative for CD20 (a B lymphocyte marker) and CD3 (a T lymphocyte marker) by immunohistochemistry (Supplemental Material “Flow cytometry and immunohistochemistry methods”). On day 49, CBC, cytology, and FC of LNs, PB, and BM were repeated. Nodal FC results overlapped with those obtained previously. In the PB, 55% dysplastic myeloid cells and 6% undifferentiated cells were found on cytology (Fig. 2b, c), and 3% CD34+ cells were found by FC. In the BM, 44% blasts and myeloid dysplastic cells were found on cytology (Fig. 2d), and 6% CD34+ cells were found by FC. An LN cell suspension was used for immunocytochemistry 11 and polymerase chain reaction for antigen receptor rearrangement (PARR).5,14 Immunocytochemistry confirmed the myeloid origin of neoplastic cells according to the positive labeling for CD11a and CD11b. However, PARR detected a T-cell receptor-y (TCRy) oligoclonal rearrangement indicative of lymphoid lineage.
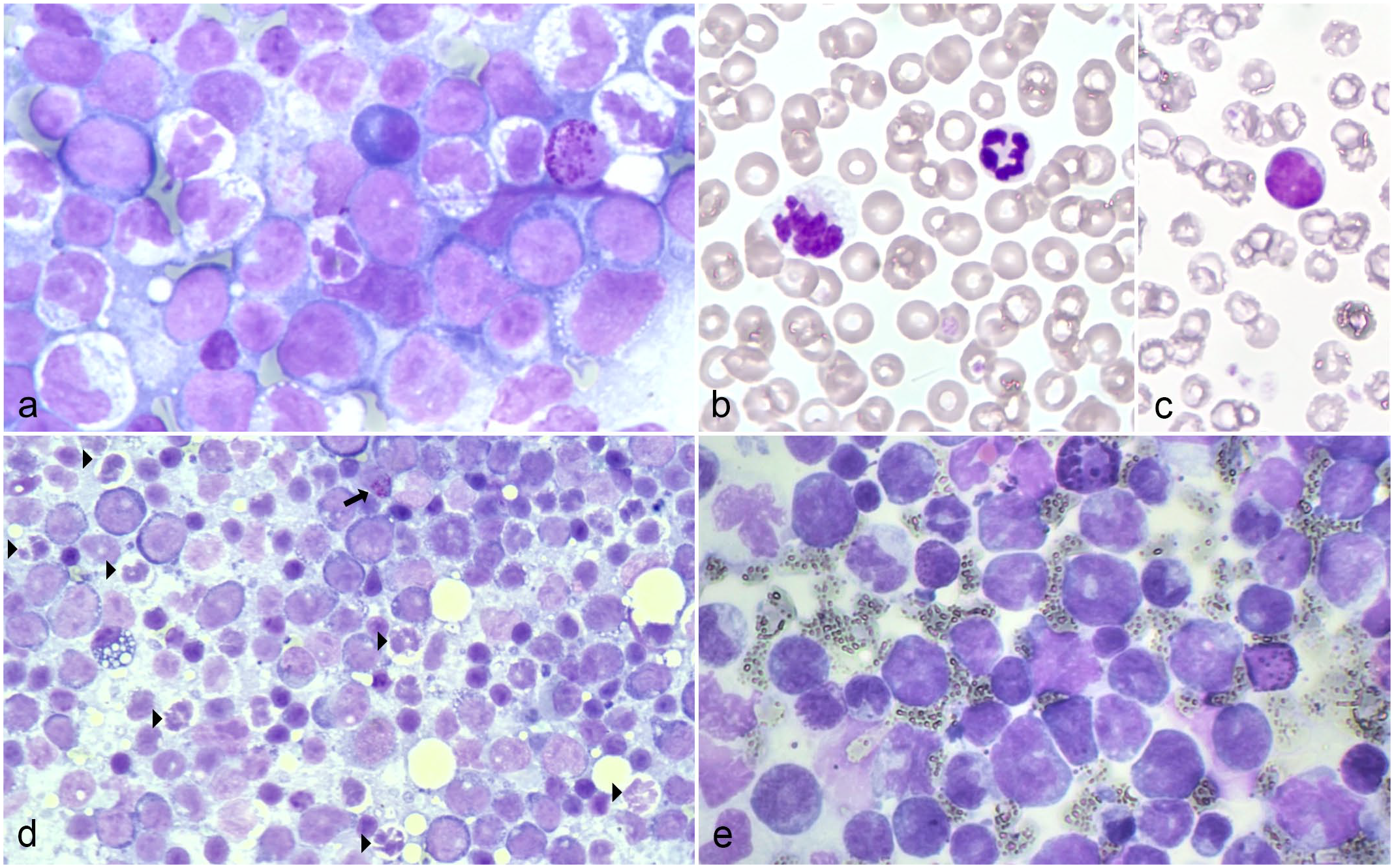
Cytological features of myeloid sarcoma in 2 dogs. May-Grunwald Giemsa (MGG). (a) Right popliteal lymph node, case 2. Mixed population of large blast cells and myeloid cells at different maturation stages showing toxic/dysplastic changes; 1 undifferentiated cell with red-purple granules is also visible on the right. (b, c) Peripheral blood, day 49, case 2. (b) One normal neutrophil and a dysplastic cell and (c) 1 undifferentiated cell. (d) Bone marrow, day 49, case 2. High number of blast cells and dysplastic neutrophils (arrowheads); 1 undifferentiated cell with red-purple granules is also present (arrow). (e) Lymph node, case 3. A prevalent population of large blast cells, often with irregular and indented nuclei, is present; 2 undifferentiated cells with red-purple granules are also visible.
Serial CBCs demonstrated progressive worsening of the pancytopenia and increased undifferentiated cells in the following 2 months of monitoring (Supplemental Table S2). The granulocytic/monocytic origin of the circulating undifferentiated cells was confirmed via FC, leading to a diagnosis of AML. Despite the poor prognosis, the owner opted for chemotherapy (cytosine arabinoside, 200 mg/m2 daily for 3 days, repeated weekly), but the dog died after 2 weeks of treatment, 3 months after the initial presentation. A necropsy was not performed.
The presence of solid lesions (enlarged LNs) composed of immature myeloid cells and the initial lack of PB and BM involvement supported a tentative diagnosis of MS. AML developed a few weeks later.
Case 3 was an 11-year-old, neutered male, mixed-breed dog referred for generalized lymphadenopathy. The dog was being treated with corticosteroids (Prednicortone, 1 mg/kg daily, given orally) for 1 month due to a cytological diagnosis of lymphoma. The CBC and thoracic radiographs were unremarkable. Abdominal ultrasound revealed intra-abdominal lymphadenomegaly and splenomegaly. Nodal cytology and FC were performed. On cytology, most cells were large, with a scant to moderate amount of basophilic cytoplasm; round, indented, or irregular nuclei; fine granular chromatin; and rare inconspicuous nucleoli. Occasional large cells with morphological features of granulocytic or monocytic differentiation and small undifferentiated cells with red-purple granules were also present (Fig. 2e).
On FC, 70% of nodal cells expressed CD11b and IgG, and most of them (71%) also expressed CD14, whereas other markers, including lymphoid, monocytic, megakaryocytic, and hematopoietic precursor markers, were not expressed (Table 1, Supplemental Table S1, and Supplemental Materials “Flow cytometry and immunohistochemistry methods”). Thus, FC results were consistent with a granulocytic/monocytic lineage with aberrant IgG expression. PB and BM were also investigated via FC, and all parameters were within normal limits with no evidence of neoplasia. Based on clinical, cytological, and FC results, MS was diagnosed. The dog continued corticosteroid therapy and remained clinically stable for 10 days but spontaneously died 1 month after diagnosis. The owners declined necropsy.
To the authors’ knowledge, these are the first cases of putative MS reported in client-owned dogs. To date, MS has not been clearly documented in veterinary medicine, neither has it been unequivocally categorized within myeloid neoplasms. 8 These cases have many similarities with the human counterpart.
Initially, lymphoma was the main clinical and cytological differential diagnosis in cases 1 and 3, but FC excluded a lymphoid origin and provided indications for a tumor of granulocytic lineage. The granulocytic origin of the cells was more easily identified by morphological assessment in case 2.
Based on clinical presentation, cytology, and FC, a myeloid neoplasia involving immature cells was diagnosed in all 3 cases. AML and MS represented the 2 main differential diagnoses. In all 3 cases, no alterations were found in the CBC at the time of diagnosis, and FC allowed definitive exclusion of PB involvement in cases 2 and 3. Furthermore, in case 2, serial FC examination of PB allowed for the detection of AML initiation and clinical development, which occurred subsequent to the MS but concurrent with the development of cytopenias, a common hematological pattern in acute leukemias. 8 In case 1, FC was not performed on PB and BM samples; thus, minimal levels of infiltration might have remained undetected. Because BM was not evaluated at the time of initial diagnosis, aleukemic leukemia could not be ruled out, but it was considered unlikely due to the presence of a single mass and the lack of peripheral cytopenias.
Case 1 presented with pleural effusion and a mediastinal mass. Mediastinal masses have been previously reported in lymphoid neoplasms and AML in dogs, without differences in the radiographic appearances. 3 Owing to the intrinsic difficulties in sampling thoracic lesions for histopathology, only pleural effusion was examined via cytology and FC. A similar diagnostic workup has been reported in humans. 1
Interestingly, PARR testing resulted in an oligoclonal TCRy rearrangement in case 2, giving a false positive result pointing toward lymphoma. Similar results have been previously reported in myeloid neoplasms. 12 Thus, PARR should be used to confirm or exclude the presence of neoplasia, not to assess the lineage of neoplastic cells.
In all 3 cases, the dogs were asymptomatic or had minimal clinical signs at diagnosis, whereas their clinical conditions deteriorated as soon as AML occurred. Thus, close monitoring of PB and BM infiltration may be useful to predict survival in dogs with MS. Neither chemotherapy nor palliative corticosteroids provided meaningful clinical responses. The longer survival of case 2 might be due to an earlier diagnosis or to a less aggressive neoplasm, rather than to the treatment itself.
In conclusion, MS should be considered as a rare and possibly underestimated differential diagnosis in the presence of a solid mass composed of immature round cells in the absence of PB abnormalities. In these cases, lymphoma is often the default interpretation, and histology alone may be not sufficient to reach a final diagnosis. The diagnostic workup can benefit from cytologic evaluation when granulocytic differentiation (possibly with dysplasia) is recognizable. However, the lineage of the neoplastic cells must be confirmed by immunophenotyping including granulocytic and monocytic markers in addition to lymphoid markers. Positive labeling for CD11b and negative labeling for lymphoid markers confirmed via FC or immunohistochemistry, together with clinical presentation and morphological features, should be considered the best tools for MS diagnosis.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858241257897 – Supplemental material for Granulocytic neoplasm suggestive of primary myeloid sarcoma in 3 dogs
Supplemental material, sj-pdf-1-vet-10.1177_03009858241257897 for Granulocytic neoplasm suggestive of primary myeloid sarcoma in 3 dogs by Alessandra Ubiali, Valeria Martini, Stefano Comazzi, Selina Iussich, Barbara Miniscalco, Alessia Poggi, Emanuela Morello, Paola Roccabianca, Barbara Rütgen, Clarissa Zamboni and Fulvio Riondato in Veterinary Pathology
Footnotes
Acknowledgements
We wish to thank the private veterinarians who sent their samples to our FC facilities or referred their patients to our oncology service.
Authors’ Note
This manuscript has been prepared in the Uniform Requirements format.
Author Contributions
VM, FR, AP, and SC performed flow cytometric analysis. PR and SI performed histologic and immunohistochemical evaluations. CZ performed immunocytochemistry. BR performed PARR test. BM and FR performed cytologic and hematologic evaluations. EM and AU oversaw clinical and therapeutic evaluations. The manuscript was written by AU, VM, and FR with contribution from the other authors.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
