Abstract
Simultaneous detection of porcine reproductive and respiratory syndrome virus (PRRSV) and porcine circovirus 2 (PCV2) was achieved by a double-labeling technique using a combination of immunohistochemistry and in situ hybridization in five pigs with naturally occurring porcine dermatitis and nephropathy syndrome (PDNS). Both PRRSV and PCV2 were isolated from a homogenate of pooled skin and kidney from three pigs. PRRSV RNA was demonstrated by reverse transcription polymerase chain reaction (PCR) in skin, kidney, lymph node, and tonsil homogenates from all pigs. PCV2 DNA was demonstrated by PCR in kidney, lymph node, tonsil, liver, and lung homogenates from all pigs. For double-labeling studies, the tissue samples were processed sequentially, first by immunohistochemistry and then by in situ hybridization. The most consistent and intense staining for PRRSV and PCV2 was in the kidney, lymph node, and tonsil. PRRSV antigen and PCV2 DNA were also detected in the skin. This morphologic study is the first to confirm the presence of both PRRSV and PCV2 in the same tissues in pigs with naturally occurring PDNS.
Keywords
Porcine dermatitis and nephropathy syndrome (PDNS) is a relatively new and often fatal disease of unknown etiology that primarily affects recently weaned and feeder pigs from 1.5 to 4 months of age. 21,23 The syndrome was first recognized in the United Kingdom in 1993. 21 However, it has since been reported in several countries, including the United States. 8,18,19,23 Significant gross and microscopic lesions are present consistently in the skin and kidneys of swine with PDNS, although other organs may also be affected. 8,23 In fatal cases, cutaneous lesions consist of severe necrotizing vasculitis affecting the dermis and subcutis, characterized by leukocytoclastic inflammation involving capillaries, small and medium-size venules, and arterioles, accompanied by epidermal necrosis and ulceration and dermal hemorrhage. 8,21,23 The kidneys are often enlarged and pale, with surface petechiation. 21 Microscopic renal lesions consist of exudative glomerulonephritis, interstitial nephritis, and necrotizing arteritis. Renal and inguinal lymph nodes are usually enlarged and red. 18
The vasculitis associated with PDNS is thought to involve an immune-mediated mechanism. 23 However, the etiology and pathogenesis remain undetermined. 11,18,21 Recent studies have shown that porcine reproductive and respiratory syndrome virus (PRRSV) and porcine circovirus (PCV) are associated with some cases of PDNS. 19,23 In cases of acute and chronic PDNS, PRRSV antigen has been detected utilizing monoclonal antibody–based immunohistochemical procedures in perivascular macrophages of the skin and renal tissues. 23 In other cases of PDNS, PCV DNA has been detected in monocyte/macrophage lineage cells of lymphoid tissues by in situ hybridization. 19 The close association of PCV DNA or PRRSV antigen within lesions from a wide range of tissues in diseased pigs has led to speculation that coinfection of PCV and PRRSV may play a important role in the pathogenesis in PDNS.
Double-labeling techniques are used for the simultaneous demonstration of either two different cellular components or two different cell types or tissue structures. 20,24 The combination of immunohistochemistry and in situ hybridization allows the detection of nucleic acid and the simultaneous localization of antigen. The objective of the present study was to develop a double-labeling technique, combining immunohistochemistry and in situ hybridization techniques, to simultaneously demonstrate the cellular distribution of PRRSV antigen and PCV DNA in formalin-fixed, paraffin-embedded tissues from pigs with PDNS.
Five pigs from different herds were selected for this study based on the presence of gross and microscopic lesions characteristic of PDNS, as previously described. 8,23 Three pigs (Nos. 1, 3, 4) were euthanatized, and two pigs (Nos. 2, 5) were found dead. At necropsy, samples of skin, superficial lymph nodes, brain, left and right myocardium, anterior and posterior lung lobes, thymus, tonsil, liver, kidneys, spleen, pancreas, adrenal glands, stomach, small intestine, and colon were fixed in 10% neutral buffered formalin, embedded in paraffin, cut at 5 µm, and stained with hematoxylin and eosin for routine microscopic examination. Skin, kidney, lymph node, spleen, tonsil, liver, and lung were stored frozen at −70 C for subsequent virus isolation and polymerase chain reaction (PCR) testing.
Attempted isolation of PRRSV and PCV2 from homogenates of skin and kidney from three pigs (Nos. 1, 3, 4) was performed by inoculation of MARC-145 cells and PCV-free PK-15 cells, respectively, as previously described. 2,16,23 Bacterial culture was performed on lung, liver, kidney, and small and large intestine from all five pigs.
PRRSV nucleic acid in tissue samples was detected by reverse transcription PCR (RT-PCR) as previously described. 6 PCR for PCV2 nucleic acid in tissue samples was performed as follows. DNA was isolated from a 25-mg tissue sample using a commercial kit (Qiagen, Santa Clarita, CA) according to the manufacturer's instructions. The forward and reverse primers were 5′-CGGATATTGTAGTCCTGGTCG-3′ and 5′-ACTGTCAAGGCTACCACAGTCA-3′, respectively. These primers can detect PCV2 only. 9 PCR was performed for 30 cycles with the following parameters: denaturation at 94 C for 20 seconds, annealing at 61 C for 20 seconds, and elongation at 72 C for 45 seconds. 9
Three serial sections were prepared from each of the formalin-fixed tissues. One was processed for PRRSV immunohistochemistry, one for PCV2 in situ hybridization, and one using the double-labeling technique. Immunohistochemistry for PRRSV and in situ hybridization for PCV2 were carried out as previously described. 4,5 For the double-labeling technique, immunohistochemistry was sequentially followed by in situ hybridization. To ensure adequate suppression of residual alkaline phosphatase in the tissue sections used for immunohistochemistry, the slides were washed in Tris-ethylenediaminetetraacetic acid buffer (10 mM Tris-HCl, 1 mM ethylenediaminetetraacetic acid [EDTA], pH 8.0), for 10 minutes at room temperature. After incubation with 50 mM EDTA for 10 minutes at 85 C, sections were thoroughly washed in maleic acid buffer (100 mM maleic acid, 150 mM NaCl, pH 7.5) for 5 minutes to remove the EDTA. Tissue sections were then subjected to the in situ hybridation procedure. Positive control tissues were collected from 76-day-old pigs experimentally infected with PRRSV and from a 6-week-old pig naturally infected with PCV. 4,5,7 Negative tissue controls were collected from two 1-day-old colostrum-deprived pigs not exposed to PRRSV and PCV.
Gross skin lesions consisted of several round to irregularly shaped red to purple macules and papules that coalesced over the perineum and distal limbs to form large irregular patches. The kidneys were enlarged and had pale cortices with multiple red circular hemorrhagic cortical foci that were 2–4 mm in diameter. Superficial and internal lymph nodes were enlarged and often hemorrhagic. Gastric ulceration was observed in two pigs (Nos. 1, 3). Microscopically, the most significant lesion was the presence of severe, fibrinoid, necrotizing vasculitis affecting the dermis, subcutis, and renal pelvis and medulla. Necrotizing vasculitis was also found in lymph nodes, stomach, spleen, and liver. In lymph nodes, there was lymphoid depletion and occasional necrosis of lymphocytes in both the cortex and paracortex. Numerous multinucleated giant cells were scattered in the cortex and paracortex. Four pigs (Nos. 1, 3–5) had interstitial pneumonia of variable severity characterized by alveolar septal thickening and hypercellularity attributed to infiltration by macrophages and type 2 pneumocyte hyperplasia.
Both PRRSV and PCV2 were isolated from the skin and kidney in three pigs (Nos. 1, 3, 4). PRRSV RNA was detected consistently by RT-PCR in skin, kidney, and lymph node from all five pigs (Table 1). The tissue homogenates tested by PCR were positive for PCV2 DNA for all five pigs (Table 2). Culture of lung, liver, small and large intestine, and kidney in all five pigs failed to detect pathogenic bacteria.
Reverse transcription polymerase chain reaction (RT-PCR) and immunohistochemistry results for porcine reproductive and respiratory syndrome virus (PRRSV) in five pigs with naturally occurring porcine dermatitis and nephropathy syndrome.
∗2 - = no signal; + = positive signal.
†+ = virus isolated; ND = not done.
Polymerase chain reaction (PCR) and in situ hybridization results for porcine circovirus 2 (PCV2) in five pigs with naturally occurring porcine dermatitis and nephropathy syndrome.
∗- = no signal; + = positive signal.
†+ = virus isolated; ND = not done.
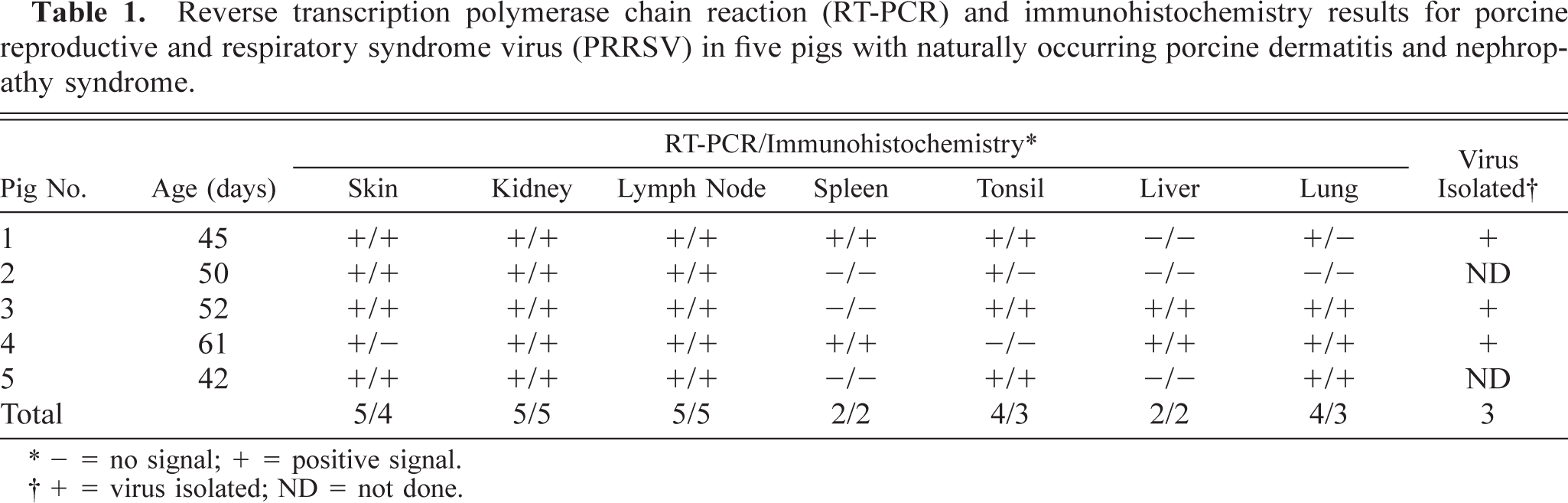
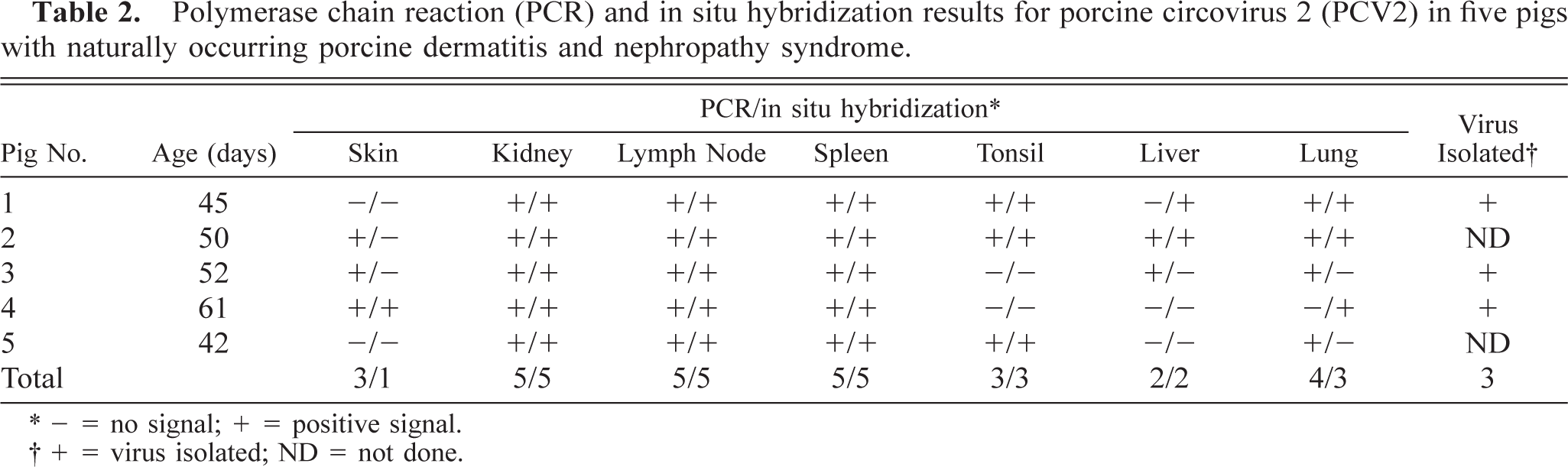
Immunohistochemistry and in situ hybridization results are summarized in Tables 1 and 2. Positive cells contained a red (immunohistochemical) or dark brown (in situ hybridization) reaction product for PRRSV and PCV2, respectively. PRRSV antigen was located almost exclusively within the cell cytoplasm, whereas PCV2 DNA was observed primarily in the cytoplasm but occasionally in the nucleus. PRRSV antigen and PCV2 DNA were detected in the skin, kidney, lymph node, spleen, tonsil, liver, and lung (Tables 1, 2). PRRSV antigen was detected in all PDNS cases. PRRSV antigen was located almost exclusively in the cytoplasm of mononuclear cells and was often located around blood and lymphatic vessels in the deep dermis and subcutis (Fig. 1). PCV2 DNA was detected by in situ hybridization in the skin of one PDNS pig (No. 4). PRRSV antigen and PCV2 nucleic acid were detected in kidney in all five PDNS pigs. PRRSV antigen was most often detected in the glomeruli (Fig. 2). Occasional PRRSV-positive mononuclear cells were found in the renal medulla and pelvic interstitium. Positive hybridization signals for PCV2 were detected in renal tubular epithelial cells (Fig. 3), fusiform interstitial cells, and macrophage-like cells, which were usually localized around vessels of the renal pelvis and among infiltrating nonpositive mononuclear cells in the interstitium of the renal cortex and medulla.
Skin; pig No. 1. PRRSV antigen (red reaction product) is in scattered mononuclear cells within the dermis adjacent to a lymphoid aggregate. Immunohistochemistry. Vector red, hematoxylin counterstain. Bar = 55 µm.
Kidney; pig No. 2. PRRSV antigen (red reaction product) is in multiple cells in the glomerulus. Proteinaceous fluid/fibrin expands Bowman's space. Double labeling for immunohistochemistry and in situ hybridization. Vector red, nitroblue tetrazolium/5-bromocresyl-3-indolylphosphate, methyl green counterstain. Bar = 55 µm.
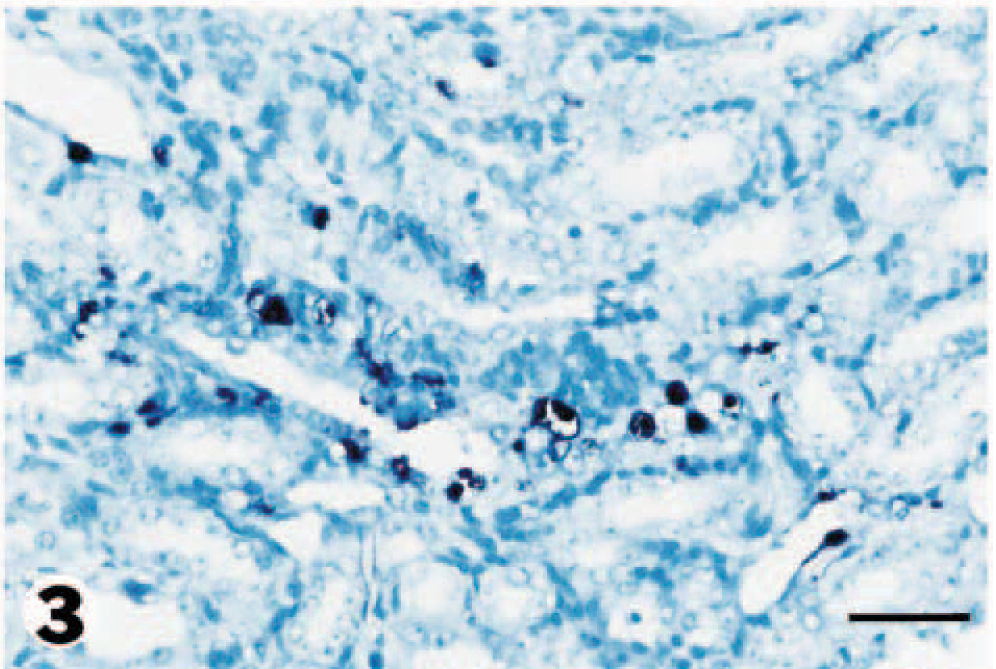
Kidney; pig No. 2. PCV2 DNA (dark brown reaction product) is detected in renal tubular epithelial cells. In situ hybridization. Nitroblue tetrazolium/5-bromocresyl-3-indolylphosphate, methyl green counterstain. Bar = 55 µm.
The inguinal and perirenal lymph nodes in all five PDNS pigs contained both PRRSV antigen and PCV2 nucleic acid. The inguinal lymph node was most consistently positive. Intense specific staining for both viruses was most often seen in cells within necrotic germinal centers. Cells staining positive for both viruses included macrophage-like cells with one or two large round nuclei and abundant cytoplasm (Fig. 4). PRRSV antigen was much less frequently detected in perifollicular regions and trabeculae. PRRSV antigen and PCV2 nucleic acid were detected in the tonsil of three PDNS pigs. PRRSV antigen was very patchy in distribution and was most readily detected within and adjacent to the crypt epithelium, whereas PCV2 nucleic acid was multifocal in distribution and was most commonly detected within tonsillar follicles (Fig. 5). In tonsillar crypt epithelium, PRRSV antigen was present in the cytoplasm of large round or irregularly shaped cells that resembled macrophages or dendritic cells. Large macrophages within lymphoid follicles contained PCV2 nucleic acid. Kupffer cells and hepatocytes were the predominant PCV2-infected cells in the liver. PCV2-positive cells were concentrated around periarteriolar lymphoid sheaths in the spleen. Positive cells generally had large oval nuclei and a moderate amount of cytoplasm, compatible with the macrophage lineage. The distribution of positive cells in the spleen was very patchy. Positive signal for PRRSV and PCV2 was occasionally detected in pulmonary interstitial macrophages. Sections from the two negative control pigs lacked positive staining for PRRSV and PCV2 by immunohistochemistry and in situ hybridization techniques, respectively.
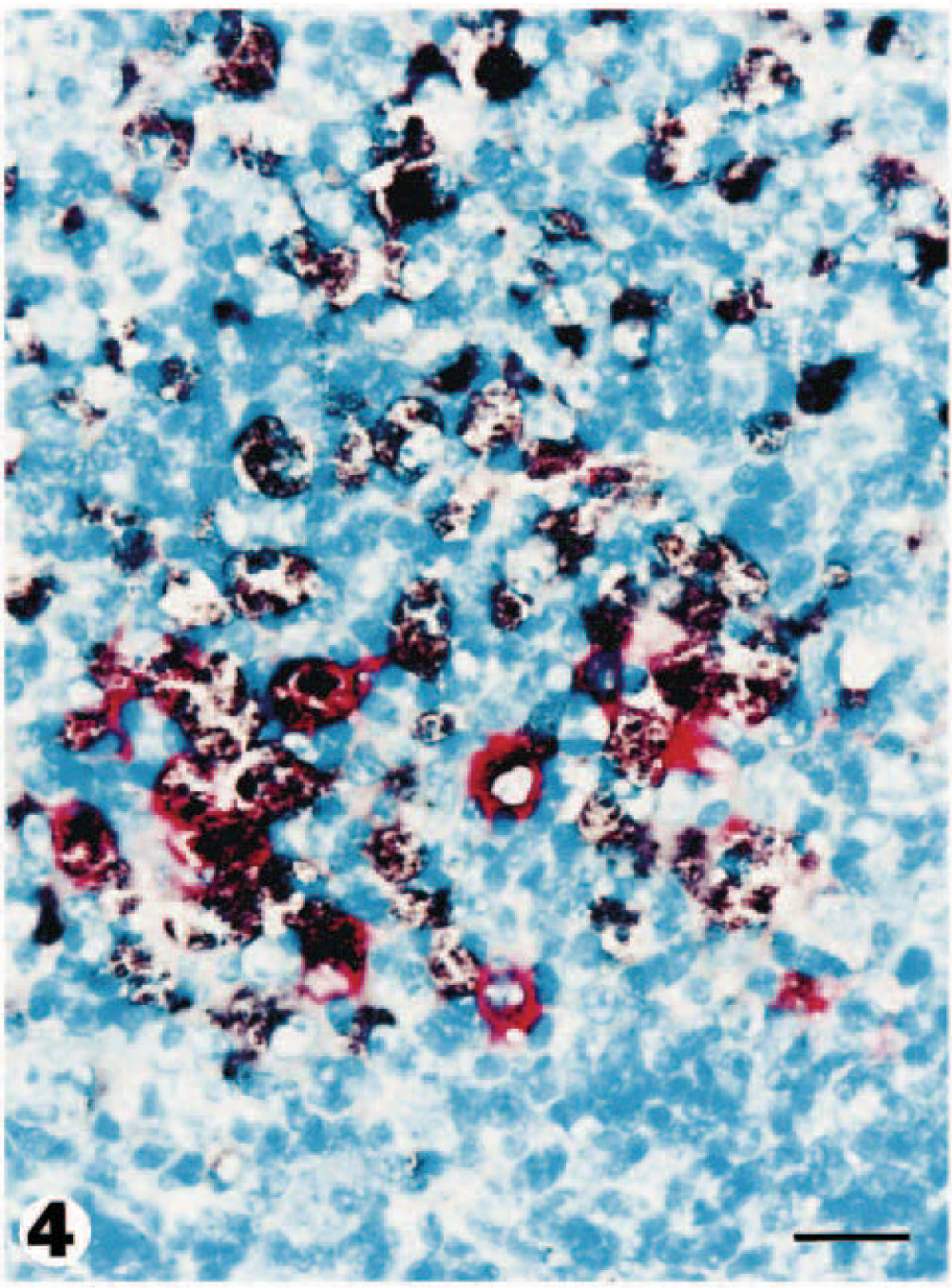
Lymph node; pig No. 3. PRRSV antigen (red reaction product) and PCV2 DNA (dark brown reaction product) is detected simultaneously in macrophages laden with necrotic debris. Double labeling for immunohistochemistry and in situ hybridization. Vector red, nitroblue tetrazolium/5-bromocresyl-3-indolylphosphate, methyl green counterstain. Bar = 30 µm.
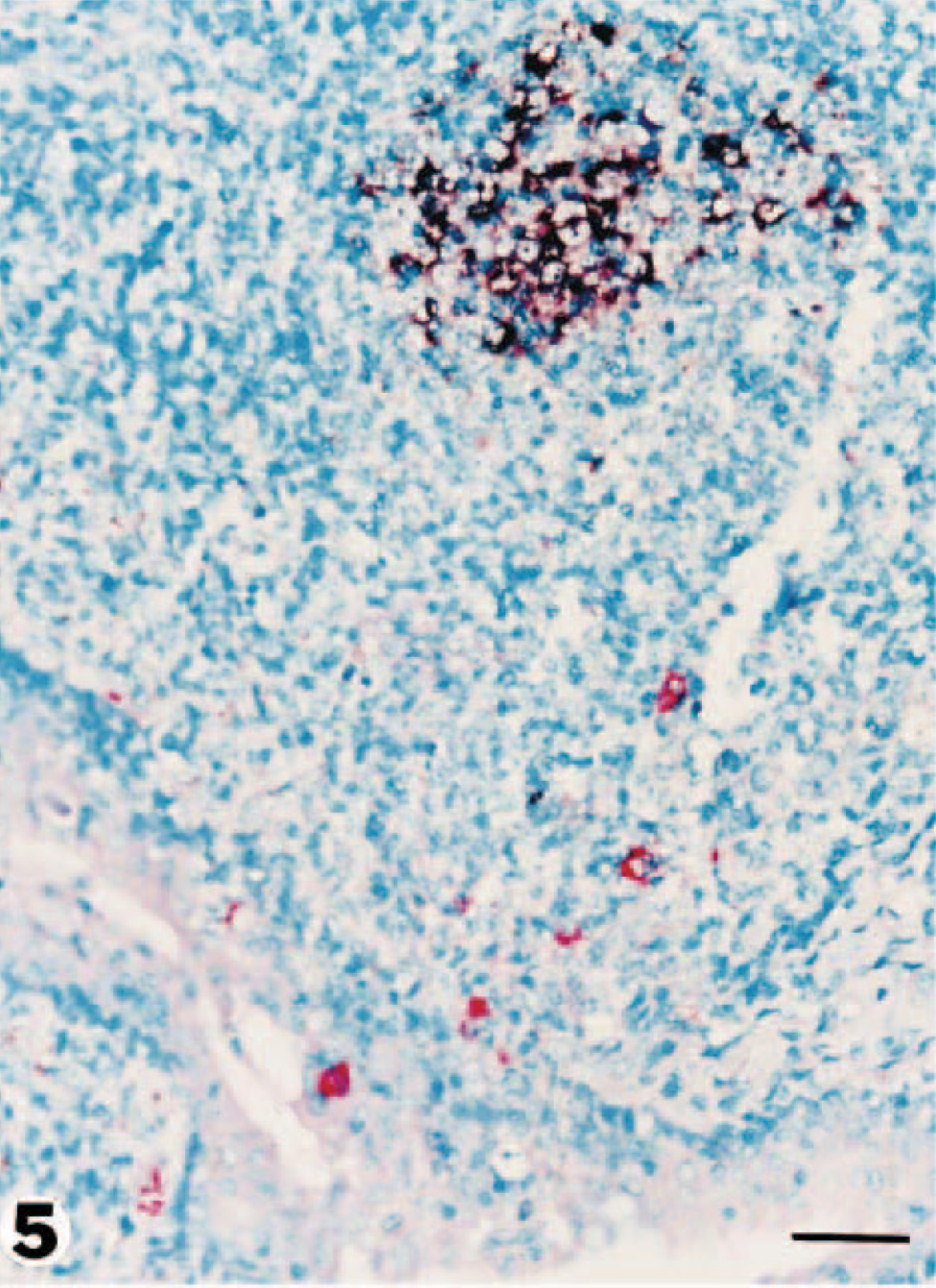
Tonsil; pig No. 1. PRRSV antigen (red reaction product) is present both in crypt epithelium and in scattered mononuclear cells. PCV2 DNA (dark brown reaction product) appears confined to a germinal center. Double labeling for immunohistochemistry and in situ hybridization. Vector red, nitroblue tetrazolium/5-bromocresyl-3-indolylphosphate, methyl green counterstain. Bar = 55 µm.
In this study, coinfection of PRRSV and PCV2 in the same tissue section was confirmed by the simultaneous detection and differentiation of PRRSV and PCV2 by immunohistochemistry and in situ hybridization, respectively. The order of application of the techniques used in this study is important. The initial application of immunohistochemistry avoids the possibility that the viral antigen could be denatured during the various incubation steps of the in situ hybridization procedure. 20,24 However, when peroxidase immunohistochemistry with diaminobenzidine (DAB) is performed, the DAB polymer may interfere with in situ hybridization. 3 Several investigators have demonstrated the superiority of digoxigenin-labeled probes over other nonradioactive detection systems for in situ hybridization. 10,12,17 Thus, we chose to combine immunohistochemistry using alkaline phosphatase with nonradioactive in situ hybridization using a digoxigenin-labeled probe.
Positive staining using both immunohistochemistry and in situ hybridization techniques provides strong evidence that cells infected with either PRRSV or PCV2 were present in most tissues. When interpreting positive immunohistochemistry and/or in situ hybridization signal in cells of the macrophage lineage, it is important to determine whether the cell is truly permissive for viral infection or whether the positive signal merely reflects phagocytized viral antigen/DNA from neighboring infected permissive cells. Previous reports have alluded to the possible involvement of PRRSV and PCV2 in the pathogenesis of PDNS, based on serologic, histopathologic, and immunohistochemical observations. 19,23 This study provides strong supportive evidence for PRRSV and PCV2 coinfection, including detection of viral nucleic acid sequence by RT-PCR or PCR in several tissues and isolation of both PRRSV and PCV2 in cell cultures. The skin from two pigs in this study was positive for PCV2 by PCR but negative by in situ hybridization. We have no explanation for this discrepancy. PCR may be more sensitive than in situ hybridization for the detection of viral nucleic acid; however, a comparative study of the sensitivity of these methods is needed.
The putative mechanisms of viral infection–related vascular diseases are many and varied, and more than one mechanism may be operational in a vasculite syndrome. The two major mechanisms for viral infection–associated vasculitis are direct viral invasion and damage, and humoral-mediated injury and/or cellular-mediated injury to the vessel wall. 15 One possible mechanism for the development of leukocytoclastic vasculitis in hepatitis C virus–infected humans is vascular deposition of high-molecular-weight immune complexes, which initiate activation of endothelial cells and inflammatory cell infiltration, leading to altered vascular permeability and vessel wall damage. 1 Because PRRSV antigen and PCV2 DNA were not detected in endothelial cells in the present and previous studies, 19,23 a direct effect of these two viruses on endothelial cells subsequent to endothelial cell infection is an unlikely mechanism of vasculitis in PDNS. These observations suggest that PRRSV and/or PCV2 likely induce vascular lesions indirectly, possibly via formation of immune complexes.
The clinical relevance of coinfection with PRRSV and PCV2 and the roles of these viruses in the pathogenesis of vasculitis in PDNS remain unclear. Human immunodeficiency virus–infected patients frequently have concurrent cytomegalovirus or hepatitis C virus infection, each of which is itself a potential causes of vasculitis; hence, the cause of vasculitis may be multifactorial in any given case. 14 One patient with cutaneous vasculitis was coinfected with both hepatitis C and G viruses. 22 In our study, PCV2 DNA was most prevalent in macrophages in germinal centers of lymph nodes. Coinfection with PCV2 may have resulted in enhanced PRRSV replication in macrophages, or vice versa. PRRSV and PCV2 may cause PDNS directly or they may act in a synergistic fashion to trigger another as yet unidentified mechanism for the induction of PDNS. Because many pigs coinfected with PRRSV and PCV do not develop PDNS, 13 further studies are needed to define the etiopathogenic role of these two agents in PDNS.
Footnotes
Acknowledgements
The research reported here was supported by contract research funds of the Research Institute for Veterinary Science from the College of Veterinary Medicine of Seoul National University and The Brain Korea 21 Project, Republic of Korea.
