Abstract
This study evaluated the sensitivity of biotinyl–tyramide-based in situ hybridization (TISH) method by comparison with chromogenic in situ hybridization (CISH) and immunohistochemical staining (IHC) methods. This study also determined the effect of fixative and fixation time on the detection of Porcine reproductive and respiratory syndrome virus (PRRSV) in paraffin-embedded tissues. Lung samples were fixed in 4% paraformaldehyde (PFA) or 10% neutral buffered formalin (NBF) for various times before paraffin embedding. Of 30 paraffin-embedded lung samples, fixed for 1 day in 4% PFA or 10% NBF, 18 (60%) were positive for PRRSV by nested reverse transcription polymerase chain reaction (nRT-PCR). All 18 lung samples (100%) also were positive for PRRSV by TISH, but only 10 of these 18 specimens (56%) were positive for PRRSV by IHC and CISH. We demonstrated that TISH can detect PRRSV RNA in paraffin-embedded tissues after up to 90 days of fixation. PRRSV nucleic acids and antigens were better preserved in 4% PFA than in 10% NBF. Compared with CISH and IHC testing methods, TISH appeared to be more sensitive for the detection of PRRSV in paraffin-embedded tissues.
Introduction
Porcine reproductive and respiratory syndrome virus (PRRSV; order Nidovirales, family Arteriviridae, genus Arterivirus) emerged in North America in 1987 and in Europe in 1991.14,18,20 PRRSV is a small, enveloped, single-stranded RNA virus that contains 9 open reading frames (ORFs). 17 ORF5, which encodes the major envelope protein, is often used for phylogenetic analysis because this locus is the most variable region of the PRRSV genome.13,23 Based on genetic, antigenic, and pathogenic analyses, PRRSVs are divided into 2 distinct classes corresponding to the European genotype (EU-type) and the North American genotype (NA-type).2,10 The disease is known to result in reproductive failure in sows and respiratory problems in piglets, and is considered to be one of the most important viral pig diseases. An accurate diagnosis is essential for confirming the disease and epidemiology on a particular farm, thereby permitting the implementation of suitable PRRSV control strategies and the eventual elimination of this financially devastating disease.
Paraffin-embedded (PE) tissues are commonly used for laboratory diagnosis, and they can be stored for long periods, and are easy to transfer among laboratories without transmission of disease agents. Immunohistochemical staining (IHC), in situ hybridization (ISH), and nested reverse transcription polymerase chain reaction (nRT-PCR) can be used to detect PRRSV in PE tissues. To date, ISH is the most commonly used method for localization of specific viral nucleic acid sequences inside individual cells while preserving cell and tissue morphology, thus allowing simultaneous assessment of the morphological alterations associated with lesions. However, chromogenic ISH (CISH) with nonradioactive probes has limited sensitivity when applied to low-copy-number nucleic acid sequences. 16 In recent years, the biotinyl–tyramide-based ISH (TISH) method has become widely used; TISH permits the detection of low copy number nucleic acids even in PE tissues.6-8,21
Formalin is a commonly used fixative agent that inactivates most infectious agents and inhibits autolysis.9,19 Formalin fixation results in the cross-linking of tissue protein with DNA or RNA; overfixation can reduce the sensitivity of IHC and ISH. For routine IHC and ISH, solutions of 10% neutral buffered formalin (NBF) or 4% paraformaldehyde (PFA) are commonly used for fixation. However, to our knowledge, there are no published comparisons of the effects of these 2 fixatives on the detection of PRRSV by TISH, CISH, and IHC. Thus, the objective of this study was to evaluate the sensitivity of TISH in comparison with IHC and CISH for the detection of PRRSV. We additionally examined the effects of fixative and fixation time on the detection of PRRSV in PE samples from naturally infected pigs.
Materials and methods
Animals
Thirty pigs, 70–150 days of age, were collected from 2 farms in the Miyazaki and Kagoshima Prefectures in Japan. Both farms had a persistent problem with respiratory disease in weaning piglets. Clinical signs varied from sudden death to chronic pneumonia with weight loss and slow growth. The mortality rate of weaning piglets was 15–30%. The pigs used for the present study had been euthanized because of respiratory signs including coughing and dyspnea. Lung samples were fixed for 1 day in either 10% NBF (at room temperature) or 4% PFA (at 4°C). The 4% PFA was prepared 1 day before use. After fixation, lung sections were processed and embedded in paraffin by standard histologic procedures. PE lung tissues were examined for PRRSV by nRT-PCR, IHC, CISH, and TISH. To examine the effect of extended fixation on the sensitivity of TISH versus other direct detection assays, NBF- and PFA-fixed lung samples from a pig that tested positive for PRRSV by nRT-PCR, TISH, CISH, and IHC at 1-day fixation were maintained in the 2 fixatives for 7, 14, 21, 28, 60, 90, and 120 days before embedding.
Polymerase chain reaction from paraffin-embedded tissues
RNA was extracted from PE samples using a commercial kita in accordance with the manufacturer’s instructions. Two PCR primer pairs were used for nRT-PCR. The primers for nRT-PCR were designed as described previously.1,22
Immunohistochemical staining
Immunohistochemistry was performed as previously described, 5 using monoclonal anti-PRRSV antibody SR30b as the primary antibody and the EnVision systemc as the secondary antibody. Staining was visualized using the peroxidase stain diaminobenzidine (DAB) kit.d
In situ hybridization
Antisense complementary (c)RNA probes specific for PRRSV RNA were synthesized as previously described. 15 Digoxigenin-labeled cRNA probes were prepared using a commercial labeling kit.e ISH for PRRSV was performed as previously described 17 with slight modification. ISH can be divided into 5 phases: section preparation, tissue unmasking, hybridization, posthybridization washes, and detection. Briefly, deparaffinized sections were digested with 10 µg/mL of proteinase K at 37°C, postfixed in 4% PFA, treated with 0.1 N HCl, and acetylated with 0.25% acetic anhydride in 0.1 M triethanolamine (pH 8.0) for 10 min each. After treatment with 3% hydrogen peroxide for 1 hr, the sections were dehydrated and air-dried. Fifty microliters of a hybridization solutionf containing 50 ng cRNA probe for TISH and 200 ng cRNA probe for CISH was used and hybridized with the sections for 16–18 hr at 50°C. After hybridization, the sections were immersed in 50% formamide–2× saline–sodium citrate buffer (SSC) for 1 hr at 60°C followed by rinsing in 1× buffer containing 1 mmol/liter of Tris aminomethane (pH 7.5), 0.5 M NaCl, and 1 mmol/liter of ethylenediamine tetra-acetic acid for 10 min at 37°C. The sections were stringently washed sequentially in 2× SSC, 0.2× SSC, and 0.1× SSC for 40 min each at 60°C. CISH and TISH differed solely in the detection steps. For CISH, sections were incubated with 1.5% blocking reagentsg for 30 min, then reacted with a 1:400 diluted anti-digoxigenin antibody.h For TISH, sections were incubated with 0.5% casein at room temperature for 10 min. The biotinyl–tyramide signal was amplified by serial application of a 1:400 diluted anti-digoxigenin antibody,h 0.07 µM biotinylated tyramide solution, 7 and 1:500 diluted streptavidin antibodyi for 15 min each at room temperature. After each incubation period, the sections were washed 3 times with 0.01 M Tris–HCl (pH 7.5), 300 mM NaCl, and 0.5% Tween-20 for 5 min. Finally, color was developed using the DAB kitj and counterstained with hematoxylin. Known PRRSV-positive and PRRSV-negative pig lungs were used as controls for IHC and ISH by replacing the primary antibody or probe with phosphate buffered saline.
Results
At postmortem examination, multifocal areas of consolidation were disseminated throughout the lung lobes, and were accompanied by edema of the interlobular connective tissue in the cranial lobes with fibrinous adhesions to the thoracic wall. Bronchial lymph nodes were enlarged. Twenty-six of 30 pigs (89%) had interstitial pneumonia and/or fibrinosuppurative bronchopneumonia with streaming degenerate alveolar leukocytes (oat cells). Lung samples were collected for bacterial culture; Pasteurella multocida was detected in 15 pigs, Mycoplasma spp. in 6 pigs, and Streptococcus suis in 5 pigs. The pleura often had moderate numbers of neutrophils, macrophages, and abundant fibrin on the surface, with fibroplasia in chronic cases.
Among the 30 autopsied pigs, PRRSV was detected in PE lungs of 18 animals (60%) by nRT-PCR. All 18 of these lung samples (100%) also tested positive for PRRSV by TISH. PRRSV was detected in 10 of these 18 lung samples (56%) by both CISH and IHC. Consistent negative results were obtained from the remaining 12 PE tissues by TISH, CISH, and IHC. By all 3 techniques, positive cells exhibited a dark-brown reaction in the cytoplasm. Distinct positive labeling was scattered throughout the alveolar septa. The positive signal intensity varied among histological structures in one section and among pigs. The PRRSV nucleic acids were located within the cytoplasm of macrophages. Dark-brown cytoplasmic staining without background staining could be observed in infected macrophages. The intensity of PRRSV-positive cells in TISH was significantly higher than in CISH and IHC methods (Fig. 1). In 8 cases, IHC and CISH were not able to detect PRRSV, whereas TISH still showed clear positive signals. Positive signals were more often associated with acute lesions with necrotic macrophages versus subacute lymphohistiocytic interstitial pneumonia. Whatever the amplification procedure used, staining reactions were negative in the controls performed by omitting the specific viral probe.
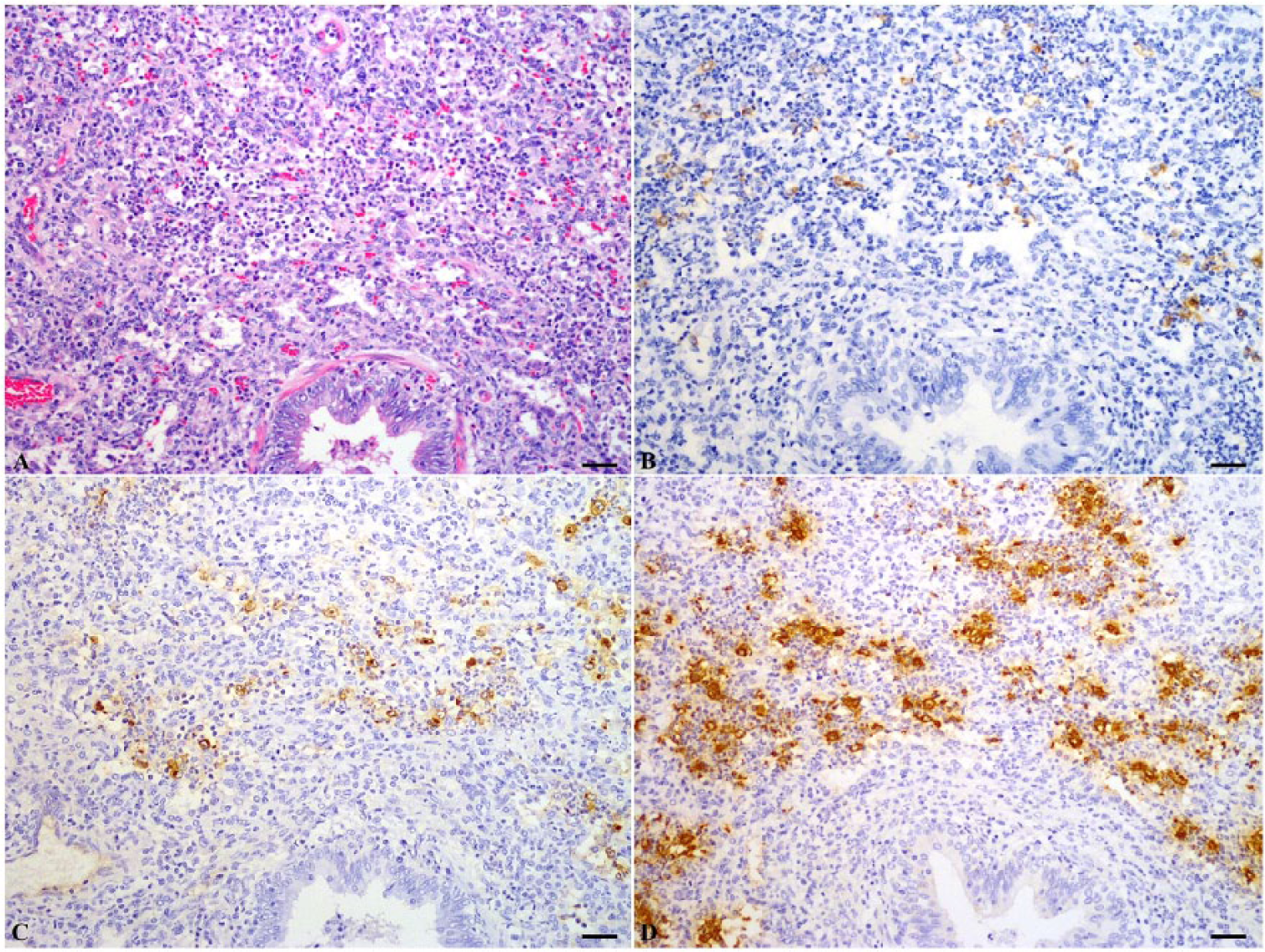
Serial sections of a lung tissue from a pig naturally infected with Porcine reproductive and respiratory syndrome virus. Lung sample was fixed in 4% paraformaldehyde for 1 day. Positive signal was visualized as dark-brown staining in the cytoplasm of macrophages.
In trials of prolonged fixation, RNA extracted from fixed tissue that had been stored in 4% PFA for up to 14 days or in 10% NBF for up to 1 day yielded positive results for both the first and second PCR (Table 1). PCR signals in PE lung tissues fixed in 4% PFA or 10% NBF for periods of 14–90 days yielded detectable positive staining only in the second PCR. On longer intervals of storage (90 days) in either of these fixatives, nominally PRRSV-containing tissues no longer yielded signal by the nRT-PCR assay. RNA accessibility was reduced with prolonged fixation, perhaps because of protein cross-linking. The accessible RNA obtained from fixed tissues was reduced with time, thus a reduction in TISH and CISH labeling was observed.
Effect of fixative and prolonged fixation on detection of Porcine reproductive and respiratory syndrome virus by nested reverse transcription polymerase chain reaction (nRT-PCR), biotinyl–tyramide-based in situ hybridization (TISH), chromogenic in situ hybridization (CISH), and immunohistochemical staining (IHC) in paraffin-embedded lung tissues.
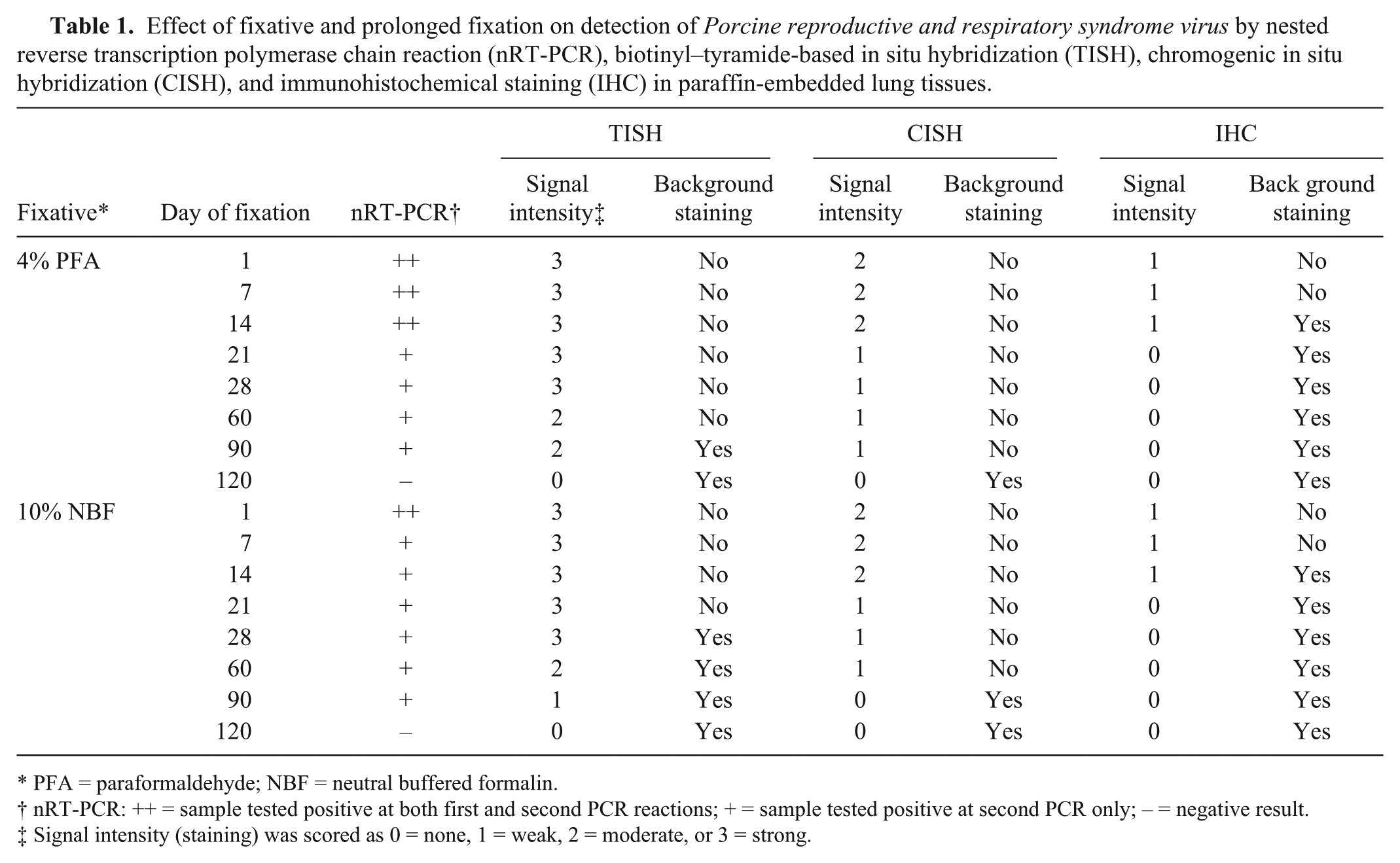
PFA = paraformaldehyde; NBF = neutral buffered formalin.
nRT-PCR: ++ = sample tested positive at both first and second PCR reactions; + = sample tested positive at second PCR only; – = negative result.
Signal intensity (staining) was scored as 0 = none, 1 = weak, 2 = moderate, or 3 = strong.
By TISH, positive signals were detected in tissues stored for up to 90 days in either type of fixative. Lung samples fixed in 4% PFA showed strong positive signals without background staining at day 1 and mild background staining at 28 days of fixation (Fig. 2A, 2B). Moderate positive signals and mild background staining was seen in tissues stored for up to 90 days in 4% PFA (Fig. 2C). Tissue fixed in 10% NBF produced strong positive signals with mild to moderate background stain at day 1and day 28 of fixation (Fig. 2D, 2E). Positive signals were strongly reduced in tissues stored for up to 90 days in 10% NBF (Fig. 2F). Fixation in 4% PFA increased positive signal intensity and reduced background staining compared to fixation in 10% NBF. IHC was strongly affected by prolonged fixation; weak positive signal was observed only up to 14 days fixation for tissues fixed in 4% PFA or in 10% NBF. After prolonged fixation for 120 days, PRRSV was not detected in any lung tissues by any of the attempted techniques. Moderate to strong background staining was seen in tissue samples subjected to more than 14 days prolonged fixation.
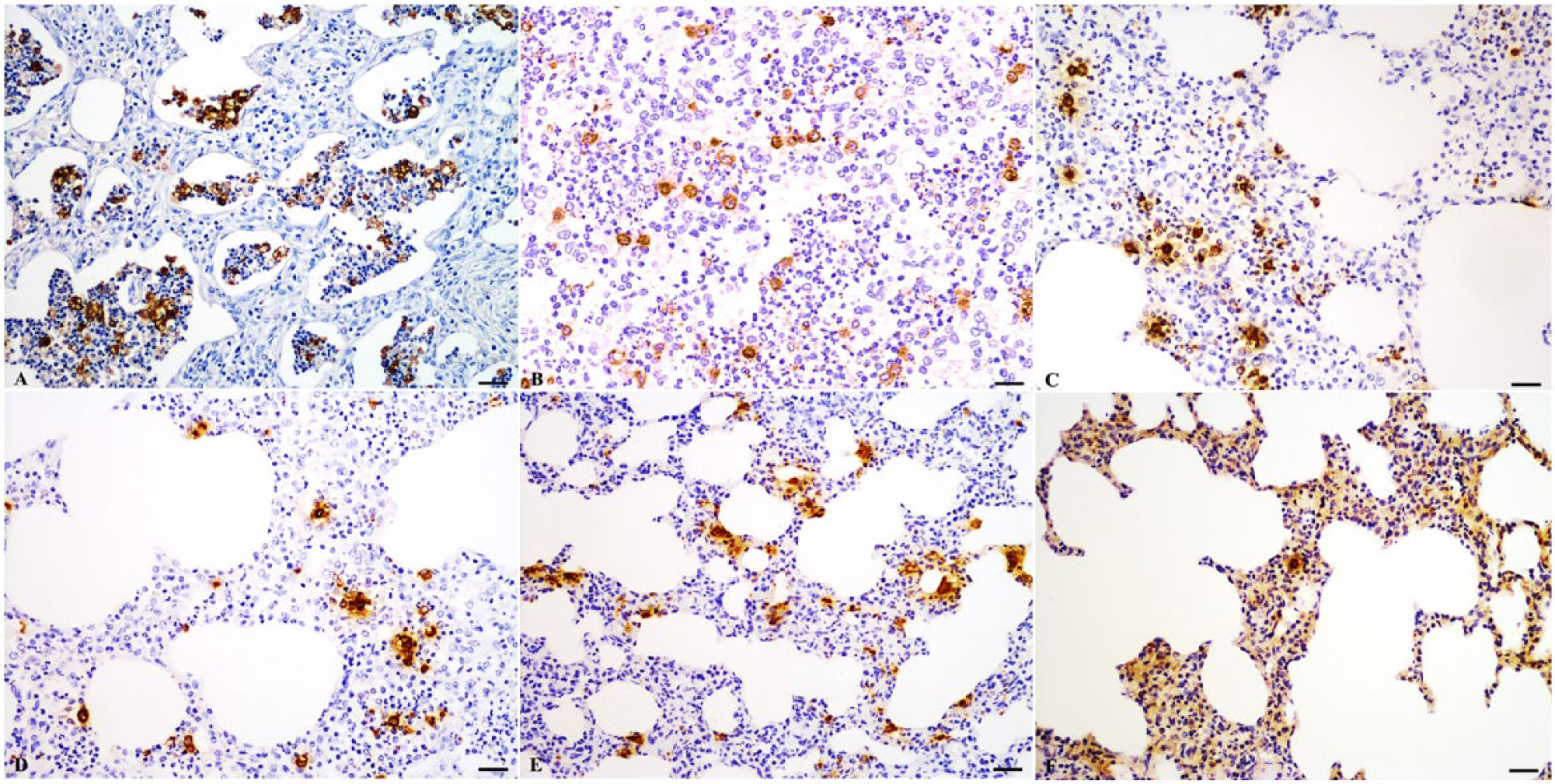
Lung; pig naturally infected with Porcine reproductive and respiratory syndrome virus (PRRSV). Effect of fixation time and fixative on the detection of PRRSV by biotinyl–tyramide-based in situ hybridization (TISH). Bar = 20 µm.
Discussion
Many experiments have been performed to test methods that detect low-copy-number nucleic acids even in PE tissues. The application of the RT-PCR method for recovery of RNA from PE tissues has revolutionized the opportunity for diagnostic analysis of fixed tissues.9,11 ISH has been widely used for research and routine examination to detect target nucleic acids. The development of TISH provides potential detection of low- and single-copy nucleic acid sequences.6 -8,16,21 In the present study, we compared the analytical sensitivity of TISH with CISH and IHC methods for the detection of PRRSV in infected lung tissue.
Previous studies3,4 comparing RT-PCR, IHC, and CISH demonstrated that RT-PCR was the most sensitive method for PRRSV detection. In the present study, the addition of the tyramide signal amplification step via TISH significantly increased the sensitivity relative to CISH in the detection of PRRSV. We did not observe a difference in the sensitivity of PRRSV detection by nRT-PCR or by TISH in PE tissues. However, RT-PCR alone cannot be used to evaluate the severity of a lesion, because RT-PCR does not provide spatial or structural information. In contrast, TISH provides cellular detail and histological architecture so that the number of PRRSV-infected cells and lesions may be observed simultaneously in the same section. TISH increases the detection sensitivity in comparison with CISH, while reducing background staining in the lung sections. The detection of false-positive signals is the most common problem encountered with ISH. However, the consistently negative results obtained in the present study with numerous negative controls indicate that false-positive reactions were not a problem with TISH in this study. Thus, this method promises to be extremely useful for detecting low-copy-number nucleic acids such as those of PRRSV in chronically infected pigs, as well as rare viral RNA in other diseases.
Previous work has demonstrated that the choice of fixative and duration of fixation significantly influence the efficiency of RT-PCR, IHC, and ISH.9,19 In the present study, PRRSV was not detected by IHC in tissue samples fixed for more than 14 days in either 4% PFA or 10% NBF. Prolonged formalin fixation has been shown to lead to masking of epitopes because of cross-linking, which reduces or even abolishes antigen detection. 19 Moreover, the results in this study showed that the selection of fixative (NBF or PFA) strongly influenced the sensitivity and background staining as detected by ISH. Thus, prolonged formalin fixation had a significant influence on the detectability of pathogenic agent in PE tissues, consistent with the results of previous studies.9,12,19
A previous study 9 that compared fixation methods for preservation of morphology and RNA in PE tissue derived from cancer cell implantation models demonstrated that among the formaldehyde fixatives, 4% PFA was most effective as preserving mRNA. In the present study, we found that PRRSV RNA and antigens were better preserved in 4% PFA than in 10% NBF. The results in this study indicate that fixation in 4% PFA for fewer than 14 days provides better preservation of RNA and morphology. In this study, 10% NBF was prepared from the stock commercial formaldehyde solution, whereas 4% PFA was prepared from paraformaldehyde powder the day before use. In conclusion, TISH is useful for the detection of low-copy-number PRRSV nucleic acids. Additionally, fixation in 4% PFA provides better preserved RNA and reduces background staining compared with traditional 10% NBF fixation.
Footnotes
RecoverAll total nucleic acid isolation kit, Life Technologies, Foster, CA. Rural Technologies Inc., Brookings, SD. REAL EnVision, Dako Denmark A/S, Glostrup, Denmark. Nacalai Tesque Inc., Kyoto, Japan. DIG RNA labeling kit, Roche Diagnostics GmbH, Mannheim, Germany. Maxim Biotech Inc., Rockville, MD. Blocking reagents, Roche Diagnostics GmbH, Mannheim, Germany. Anti-digoxigenin horseradish peroxidase, Fab fragments antibody; Roche Diagnostics GmbH, Mannheim, Germany. Peroxidase-conjugated streptavidin, Dako North America Inc., Carpinteria, CA. Liquid DAB+ substrate chromogen system, Dako North America Inc., Carpinteria, CA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a Japanese Society for the Promotion of Science (JSPS) Grant (no. 23580425).
