Abstract
A retrospective study on pig lung tissues from 60 cases of proliferative and necrotizing pneumonia (PNP) was performed to determine the presence of porcine reproductive and respiratory syndrome virus (PRRSV), swine influenza virus (SIV), and porcine circovirus type 2 (PCV2) in these lesions. Cases selected included 30 cases diagnosed between 1988 and 1992 and 30 cases diagnosed between 1997 and 2001. In each group of 30 cases, 10 were from suckling piglets, whereas the other 20 were from postweaned animals representing either nursery or grower-finisher pigs. Immunohistochemistry using a monoclonal antibody to influenza virus type A was used to determine the presence of SIV, and in situ hybridization was used for the detection of PRRSV and PCV2 nucleic acids. PRRSV was detected in 55 of the 60 cases examined (92%), PCV2 in 25 cases (42%), and SIV in only 1 case (2%). In 30 cases (50%), PRRSV was the only virus detected, whereas in 25 other cases (42%), a combination of PRRSV and PCV2 could be detected in the lungs with PNP lesions. PCV2 could not be detected in the lungs of suckling pigs with PNP. All PCV2-positive cases were found in postweaned pigs and were always in combination with PRRSV. In this latter age group, PCV2 was detected in 63% of the cases (25/40). Data from our study indicate that SIV is rarely identified in PNP and that PCV2 infection is not essential for the development of PNP lesions. The results of the present study demonstrate that PRRSV is consistently and predominantly associated with PNP and should be considered the key etiologic agent for the condition.
Keywords
In the late 1980s, a new type of porcine pneumonia of unknown etiology called proliferative and necrotizing pneumonia (PNP) was described in Quebec, Canada.
20
Noncollapsed, plum-red, grayish, tan, or mottled lungs with a rubbery texture characterize gross lesions (Fig. 1). Histologically, there is a lymphohistiocytic interstitial pneumonia with a marked proliferation of type 2 pneumocytes and many coagulates of necrotic cells in the alveoli. Hyaline membranes, intra-alveolar lymphoplasmacytic infiltrates, and bronchiolar necrosis are also observed in some cases. The recognition of PNP occurred simultaneously with the emergence of a new disease in the swine population, called at the time “mystery swine disease” and now known as porcine reproductive and respiratory syndrome (PRRS). The latter syndrome is caused by the PRRS virus (PRRSV), an RNA virus originally isolated in the Netherlands
24
and classified as a member of the
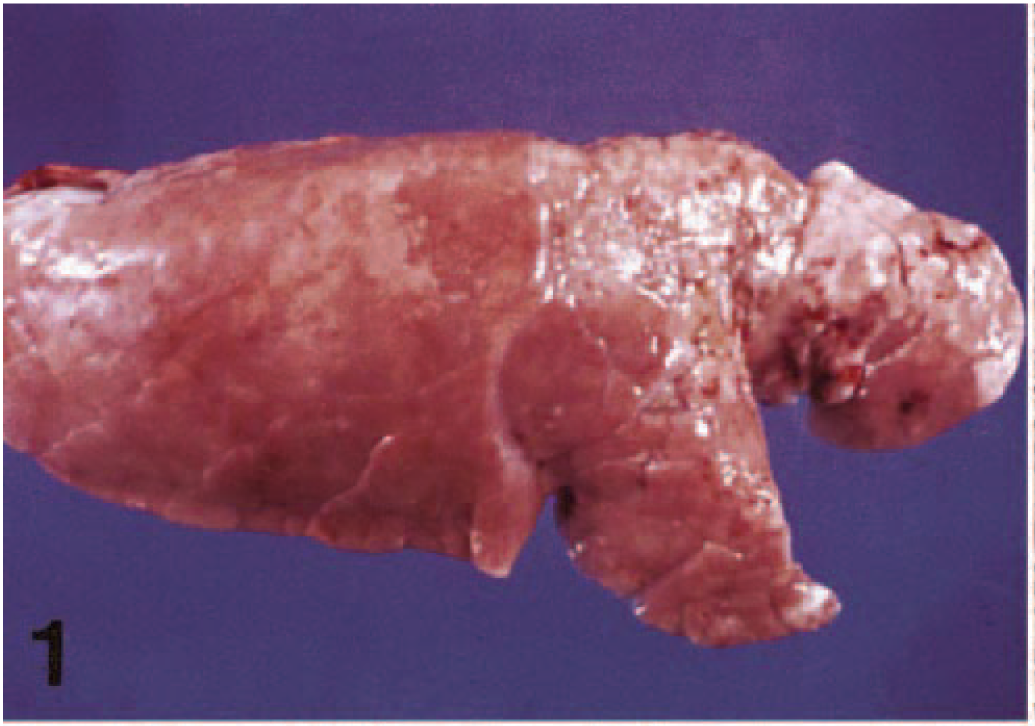
Lung; pig. Gross lesions of PNP characterized by a tan-colored, noncollapsed lung with a rubbery texture.
PNP lesions were initially thought to be caused by new strains of swine influenza virus (SIV) type A (H3N2 and H1N1). 3 , 7 Subsequently PRRSV was repeatedly isolated from lungs with PNP lesions in the absence of SIV. 17–19 Experimental transmissions with several of these PRRSV isolates caused lung lesions similar to those observed in field cases of PNP, but milder. 18 , 19 The frequent association of PRRSV with PNP lesions and the rare involvement of SIV were also confirmed by immunohistochemical studies on PNP field cases collected between 1988 and 1993. 16
There is a wide spectrum of pulmonary lesions that may be observed in field cases of PRRSV infection. These lesions may range from a mild and rather nonspecific multifocal interstitial and proliferative pneumonia 23 to the severe and histologically distinctive PNP. Differences in pathogenicity among PRRSV isolates have been reported and could explain the variation in the severity of the disease observed in the field. 10 Recently it has been suggested that PNP might be the result of coinfection with PRRSV and porcine circovirus type 2 (PCV2). 4 , 6 , 21 PCV2 is the postulated primary etiologic agent of postweaning multisystemic wasting syndrome (PMWS). 1 , 5 However, in a recent field study, the presence of PRRSV nucleic acid was demonstrated by polymerase chain reaction (PCR) in all cases of PNP examined, whereas PCV nucleic acid was detected in only one-third of these cases. 15
The purpose of this work was to determine, from archival material, the presence of PRRSV, PCV2, and SIV in the lungs of pigs with PNP.
Materials and Methods
Selection of cases
A total of 60 cases of porcine PNP was examined in the study. Cases were selected based on specific lung lesions, age of the animals, year of submission, and availability of lung tissue blocks for further testing until the predefined number of animals in each category was reached. Cases were stratified as follows: 30 cases diagnosed between 1988 and 1992 and 30 cases diagnosed between 1997 and 2001. In each group of 30 cases, 10 were from suckling piglets, whereas the other 20 were from postweaned animals representing either nursery or grower-finisher pigs.
For each formalin-fixed, paraffin-embedded lung tissue block, at least four serial 5-μm-thick sections were prepared. One section was processed for histopathology, one for PRRSV in situ hybridization (ISH), one for PCV2 ISH, and finally, one for SIV immunohistochemistry.
Histopathology
All selected cases were reviewed histologically to verify the presence or absence of bronchial/bronchiolar necrosis.
In situ hybridization
ISH for the detection of PRRSV nucleic acid was performed using a 254-bp PCR product obtained from the PRRSV LHVA-93-3 isolate. 13 For the detection of PCV2 nucleic acid, a 263-bp DNA fragment was generated by PCR from the open reading frame (ORF) 2 of the PCV2 isolate LHVA-V53. 12 For use as probes, PCR products were purified and labeled by random priming with digoxigenin-dUTP using a commercial kit (Boehringer Mannheim Canada, Laval, Quebec, Canada). ISH was performed essentially as previously described. 13 In brief, tissue sections were heated at 65 C for 20 minutes, deparaffinized, rehydrated, and immersed in 0.2 M HCl for 20 minutes. After rinsing, sections were treated with proteinase K (25 μg/ml) for 15 minutes at 37 C. After washing, sections were prehybridized for 60 minutes at 42 C with a hybridization solution (50% formamide, 2× standard sodium citrate, 1× Denhardt's solution, 1 mM ethylenediamine-tetraacetic acid, and 100 μg/ml denaturated herring sperm DNA). Hybridization was carried out in the same solution incorporating 10% dextran sulfate, 250 μg/ml denaturated herring sperm DNA, and the denaturated labeled probe. The PCV2-labeled probe was used at a 1:30 dilution. Hybridization was carried out overnight at 42 C in a humid chamber. After stringent washes, the detection of the PCV2 probe was performed using an anti-digoxigenin conjugated with alkaline phosphatase (Boehringer Mannheim) diluted 1:400 for 2 hours at 37 C. Sections were finally incubated with nitroblue tetrazolium salt and 5-bromo-4-chloro-3-indolylphosphate and counterstained with hematoxylin. The detection of the PRRSV digoxigenin-labeled probe and the signal amplification were performed using a commercial kit (TSA Plus DNP System, NEN Life Science Products, Boston, MA). This kit was used to amplify the signal, allowing the use of the probe at a 1:100 dilution. Known tissue blocks of previous experimental studies, obtained from noninfected pigs and pigs infected with PRRSV or PCV2, were used as negative and positive controls.
Immunohistochemistry
An immunoperoxidase assay was performed for the detection of SIV using a monoclonal antibody (Chemicon International, Inc., Temecula, CA) directed to the nucleoprotein of influenza A virus and the avidin-biotin complex method. In brief, lung sections were deparaffinized, rehydrated, and digested with 0.05% protease (Protease XIV, Sigma Chemical Co., St. Louis, MO) in phosphate-buffered saline (PBS) for 15 minutes at 37 C followed by endogenous peroxidase blocking in 3% peroxide for 5 minutes. After washing, blocking was done with a 5% solution of normal horse serum for 30 minutes. Primary mouse anti–influenza A virus monoclonal antibody, diluted 1:1,000 in PBS, was added to the lung sections and incubated for 16 hours at 4 C in a humidified chamber. After two washes, the slides were flooded with biotinylated horse anti-mouse linking antibody 1:200 (Vector Laboratories, Burlingame, CA) for 30 minutes. The sections were washed twice in PBS, treated with peroxidase-conjugated avidin (Vector Laboratories) for 30 minutes, washed twice in PBS, and then incubated with 3-amino-9-ethylcarbazole (Vector Laboratories) for 25 minutes. Sections were stained with Mayer's hematoxylin counterstain. Controls included the testing of lung sections from a known SIV-negative pig and from two known SIV-positive pigs.
Results
The age of the animals studied ranged between 3 days and 3 weeks for suckling piglets and between 6 and 14 weeks for postweaned pigs. Histologically, the two hallmarks of severe PNP were 1) the presence of large numbers of coagulates of necrotic cells with acidophilic and granular debris within alveolar spaces and alveolar canals and 2) marked hypertrophy and proliferation of type 2 pneumocytes with varying degrees of lymphohistiocytic interstitial inflammation (Fig. 2). In the present cases studied, significant damages to the bronchial and bronchiolar epithelium were noted in 6 of the 60 cases (10%), all from postweaned animals.
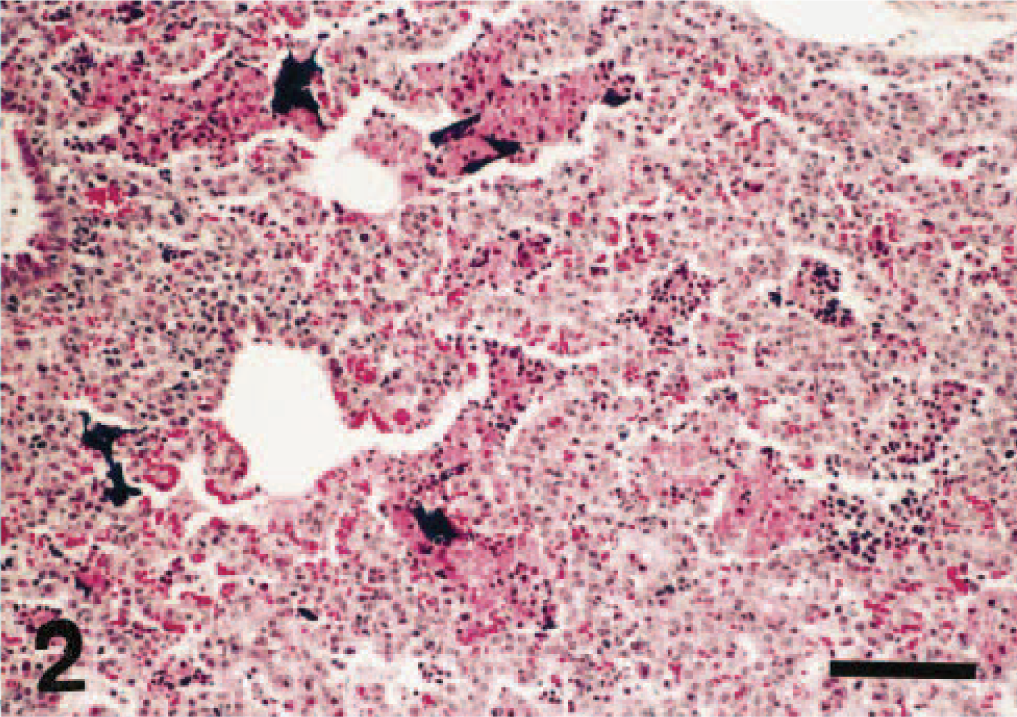
Lung; pig. Histologic section from a lung with PNP. Note the proliferation of pneumocytes and the presence of many coagulates of necrotic cells within the alveoli. The dark-blue blots (DNA material) are the result of an extensive karyolysis. Hematoxylin–phloxine–saffron stain. Bar = 90 μm.
Globally, the PRRSV was detected in 55 of the 60 cases examined (92%), PCV2 in 25 cases (42%), and SIV in only one case (2%). In 30 cases (50%), PRRSV was the only virus detected, whereas in 25 other cases (42%), a combination of PRRSV and PCV2 could be detected in the lungs with PNP lesions (Table 1). The detection rates of PRRSV and PCV2 in the lungs of pigs with PNP according to the study period and the age group are shown in Table 2. These data show that PCV2 could not be detected in the lungs of suckling pigs with PNP. All PCV2-positive cases were found in postweaned pigs and were always in combination with PRRSV. In this latter age group, PCV2 was detected in 63% of the cases (25/40). The viral detection rates were similar for the two selected study periods.
Viral agents detected in 60 lungs of pigs with lesions of PNP.
Detection rates of PRRSV and PCV2 in 60 lungs of pigs with lesions of PNP according to the study period and the age group.
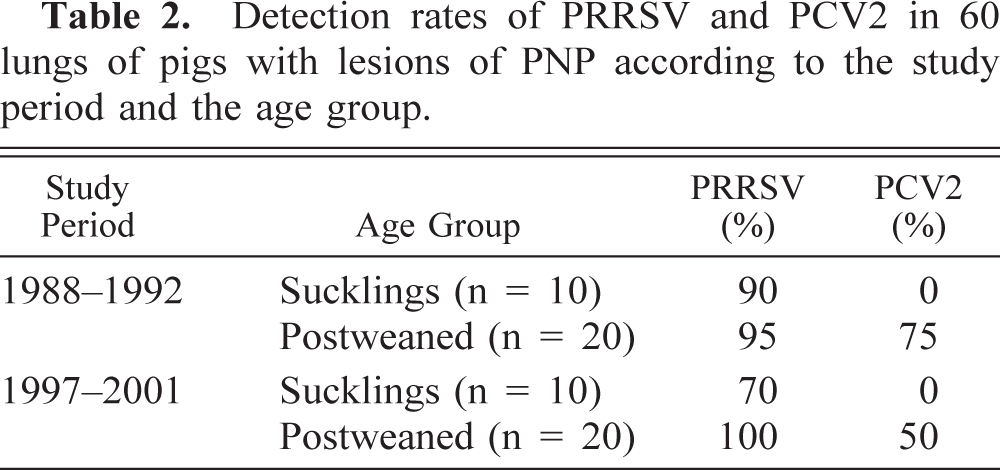
In the lung tissues where PRRSV was demonstrated by ISH, the labeling was most often observed in the cytoplasm of macrophages within the alveolar spaces and septa. Labeling was also occasionally found within intra-alveolar debris. The distribution of PRRSV-positive cells in the lung tissue was generally focal or multifocal. The number of labeled cells varied from case to case and from an area of the lung to another within the same case, but generally it ranged from a few to a moderate number of cells (Fig. 3).
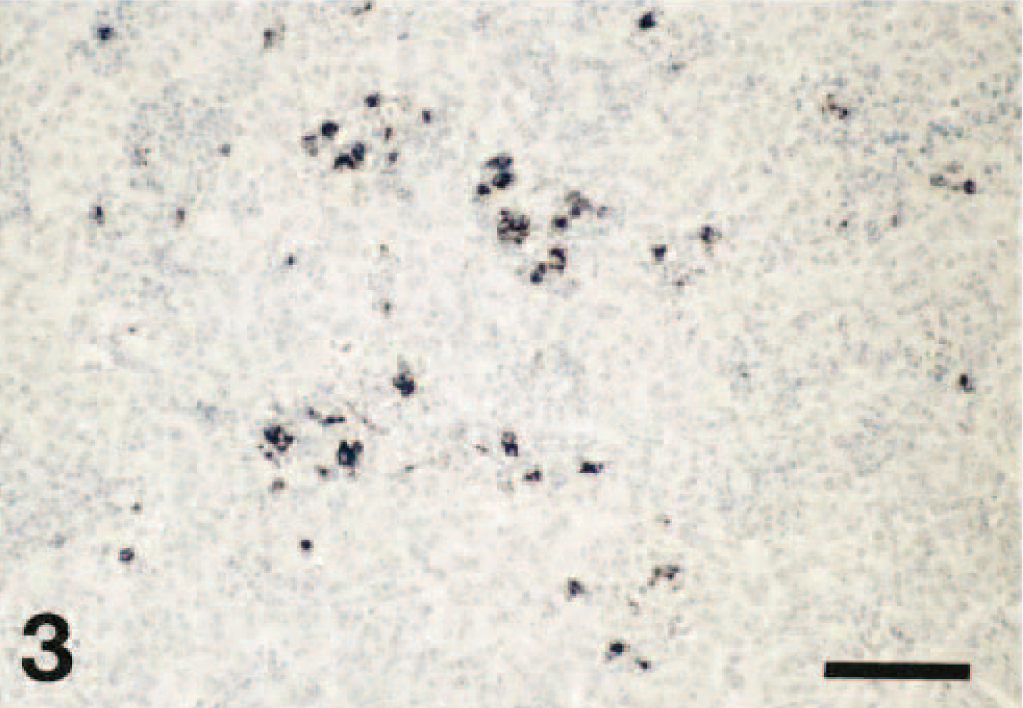
Lung; pig. In situ hybridization demonstrating PRRSV nucleic acid in a moderate number of macrophages in the alveolar spaces and septa. Hematoxylin counterstain. Bar = 90 μm.
In lung tissues in which PCV2 was demonstrated by ISH, the labeling varied from slight to very marked. PCV2 nucleic acid was most consistently found in macrophages (mainly intracytoplasmic) and in debris within the alveolar spaces as well as in mononuclear cells present within the alveolar septa and the bronchial/bronchiolar walls (Fig. 4). Positive staining was also occasionally noted in blood monocytes, in fusiform cells around airways, and in the nuclei of bronchial/bronchiolar epithelial cells, namely in five of the six cases in which bronchiolar necrosis was observed histologically (the other case was positive for SIV). PCV2-positive cells were generally more widely distributed throughout the lung tissue than were PRRSV-positive cells. Furthermore, in lungs infected by both viruses, the distribution of PRRSV-positive and PCV2-positive cells was different.
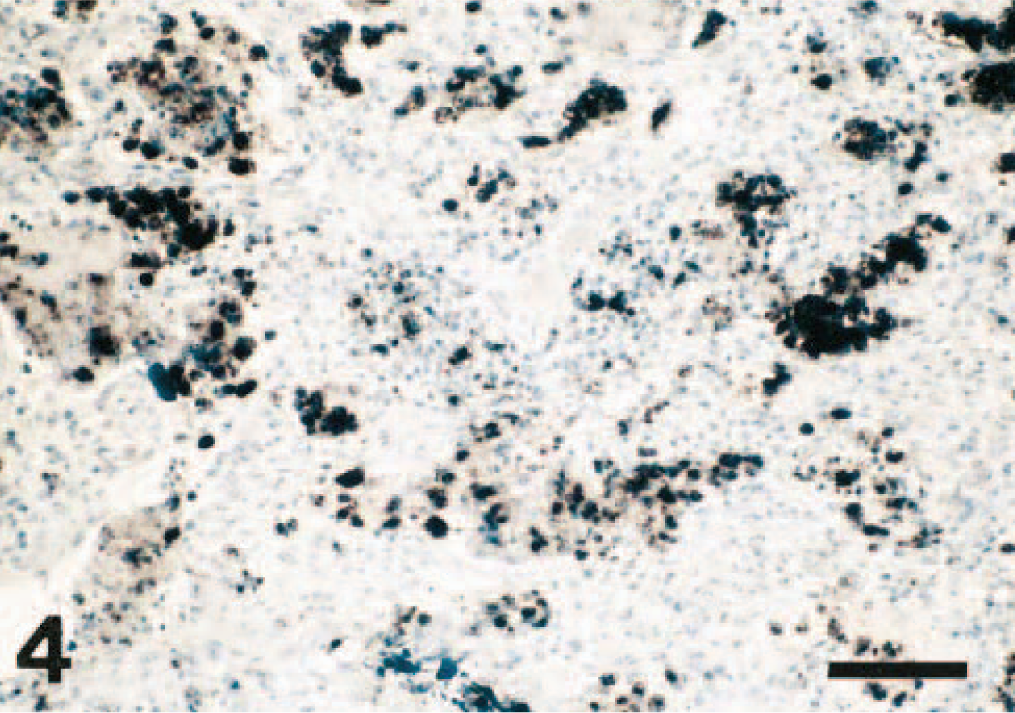
Lung; pig. In situ hybridization demonstrating PCV2 nucleic acid in large numbers of intra-alveolar macrophages and in intra-alveolar debris. Hematoxylin counterstain. Bar = 90 μm.
Discussion
The etiology of PNP of swine or the factors responsible for the development of this severe form of pneumonia have not yet been totally elucidated since the original description of the disease several years ago. 20 In the present study, PRRSV, as detected by ISH, was the virus most consistently present in the lungs of pigs with PNP, with a rate of detection of 92%. Other studies using different viral detection techniques such as immunohistochemistry, PCR, or virus isolation have also shown a close association between PRRSV and this pneumonia. 15–19 On the other hand, SIV was detected in only one of the 60 field cases examined, and this rare involvement of SIV in PNP is also in agreement with a previous study on PNP. 16
In this study, PCV2 was detected by ISH in 42% of the cases examined. This detection rate for PCV2 is similar to those recently reported in cases of PNP 15 and in the lungs of 632 pigs with respiratory disease complex, regardless of the type of pneumonia observed. 21 In our study, PCV2 was only detected in the lungs of postweaned pigs with PNP. The absence of PCV2 in the lungs of suckling pigs examined in this study is consistent with the postnatal natural infection by this virus, which generally occurs several weeks after birth. 14 , 22 PCV2 was always found in combination with PRRSV in the lungs of postweaned animals with PNP, with a detection rate of 62.5% for this age group. German workers have recently reported a detection rate of 85.4% for the combined PRRSV and PCV2 infection in the lungs of pigs with PNP. 21 The age of the animals selected in the latter study was however not mentioned. The high detection rates of PCV2 in postweaned PNP cases might simply be a reflection of the very high occurrence of PCV2 infection in grower pigs independent of disease. 14
The amount of PCV2 nucleic acid was so abundant in some of the lungs examined in our study that it would be difficult to believe that this virus has no deleterious effect on the lung tissue or in the development of these pulmonary lesions. Bronchial and bronchiolar damages found in some of the cases studied appeared to be associated with the presence of PCV2 in nuclei of bronchial/bronchiolar epithelial cells. Recently it has been shown experimentally that concurrent infection with PRRSV and PCV2 produces a more severe pneumonia than that caused by either one of these viruses alone. 11 Data from our study indicate that although PCV2 infection might potentially contribute to the severity of this type of pneumonia, it is not essential to its development because all neonatal piglets and approximately a third of the postweaned animals with PNP were PCV2 negative.
Results of the present study in addition to several other laboratory and epidemiologic data support the role of PRRSV as the principal etiologic agent of PNP. All factors, however, involved in triggering this severe pulmonary reaction have still to be fully understood. Experimentally, significant differences in the pathogenicity of PRRSV isolates resulting sometimes in severe extensive pneumonic lesions involving over 80% of the lung have been reported. 9 , 10 The infectious dose, the susceptibility of the host, and the environment of the animals are no doubt additional factors that may contribute to the severity of the disease. In the present study, the degree of pneumonia observed histologically often far exceeded the amount of PRRSV nucleic acid detected in the lungs. Similarly, after experimental PRRSV infection, it was noted that antigen distribution was very patchy in the lung and that often it was not detected in extensive areas of interstitial pneumonia typical of PRRS. 8 These findings suggest that the pulmonary lesions are not all due to the direct cytopathic effect of the PRRSV and that inflammatory responses consecutive to the release of enzymes and cytokines from PRRSV-damaged or -infected macrophages are also possibly involved significantly. These latter mechanisms need however to be more fully investigated. The present work demonstrates that coinfection with PCV2 is not a determining factor in PNP and that PRRSV is consistently and predominantly associated with PNP and should be considered the key etiologic agent of the condition.
Footnotes
Acknowledgements
We are grateful to J. Cardin and J. Deslandes for their excellent technical assistance.
