Abstract
The pathologic and peroxidase immunohistochemical features of West Nile flavivirus (WNV) infection were compared in four horses from the northeastern United States and six horses from central Italy. In all 10 animals, there were mild to severe polioencephalomyelitis with small T lymphocyte and lesser macrophage perivascular infiltrate, multifocal glial nodules, neutrophils, and occasional neuronophagia. Perivascular hemorrhages, also noted macroscopically in two animals, were observed in 50% of the horses. In the four American horses, lesions extended from the basal nuclei through the brain stem and to the sacral spinal cord and were more severe than the lesions observed in the six Italian horses, which had moderate to severe lesions mainly in the thoracolumbar spinal cord and mild rhombencephalic lesions. WNV antigen was scant and was identified within the cytoplasm of a few neurons, fibers, glial cells, and macrophages. WNV infection in horses is characterized by lesions with little associated antigen when compared with WNV infection in birds and some fatal human infections and with other important viral encephalitides of horses, such as alphavirus infections and rabies.
West Nile virus (WNV) is a member of the genus Flavivirus (family Flaviviridae) and was originally isolated from the blood of a woman with fever in the West Nile province of Uganda in 1937. 28 The virus is primarily transmitted and amplified between avian reservoir hosts by many species of mosquitoes in enzootic transmission cycles. During periods of favorable ecologic and climatic conditions, virus-carrying mosquitoes can transmit WNV to susceptible mammals and birds, causing epidemics and/or epizootics of encephalitis in humans and horses and mortality in certain domestic and wild birds. A recent review of the epidemiology of WNV has been published. 13 In Europe, Africa, and Eurasia, WNV has caused sporadic cases and outbreaks in humans and horses since the early 1960s, 10 but the virus was isolated for the first time in the United States in 1999. 3
West Nile virus was first recognized in Italy in a 1998 outbreak involving 14 horses from nine different farms in central Tuscany. 2 This epizootic occurred from August to October 1998, and 6 of the 14 affected horses presented with sudden tetraparesis or paraparesis, progressing to tetraplegia and recumbency; 8 animals recovered without significant consequences. Four of the six recumbent horses were euthanatized, and the other 2 died spontaneously. No cases of disease in human beings and other domestic or wild animals were observed during the equine epizootic in Tuscany. Fifty-seven cases of equine WNV encephalomyelitis were recognized from May to November 2000 in seven states in the northeastern United States. A total of 23 horses died or were euthanatized because of severe neurologic signs. 4 Preliminary reports suggested that a cluster of 25 cases of equine neurologic disease coinciding with the 1999 human cases of fatal viral encephalitis in New York City were most likely due to WNV infection. 34 Although it is still unclear how many horses developed WNV encephalomyelitis in 1999 in the counties nearby New York City, the virus spread rapidly in 1 year to the equine populations of the adjacent coastal states, and rapid additional virus diffusion is expected.
The clinical and neuropathologic features of spontaneous WNV infection in the horse have been recently reported in detail. 2 However, the lesions in horses from the northeastern United States have not been reported and the tissue distribution of WNV in horses has not previously been described. In this comparative study, which includes horses from Italy and the United States, we evaluated the lesions and the immunohistochemical distribution of WNV antigen in the nervous system and major organs of affected horses using serologic, virologic, and molecular methods. We also described the phenotypic characterization of the inflammatory cell infiltrates.
Materials and Methods
Complete postmortem examinations were carried out on six horses from Tuscany, central Italy. The horses were all females and ranged in age from 16 months to 12 years (horse Nos. 1–6; Table 1). These horses were examined during an epizootic of WNV infection in farms close to a wetland populated with numerous migratory birds and mosquitoes. 2 Postmortem examinations were also completed on 4 horses from the East Coast of United States (New Jersey, horse Nos. 7, 8, 10; New York state, horse No. 9; Table 1), that were submitted to the Department of Pathobiology, Section of Large Animal Pathology, New Bolton Center, University of Pennsylvania. The spinal cord of horse No. 8 was not examined because this animal was submitted with a presumptive diagnosis of viral encephalitis and had not been vaccinated for rabies.
Breed, age, gender, and origin of the 10 horses with WNV encephalomyelitis.

∗ F = female; M = male; MC = male castrated.
West Nile virus infection in horses from Italy was previously confirmed by virus isolation, reverse transcription polymerase chain reaction (RT-PCR) and serologic examination (horse Nos. 1–6).
2
Virus isolation on RK-13Ky cultured cells and identification with specific monoclonal antibodies recognizing a WNV structural glycoprotein (glycoprotein E) were conducted with a pool of fresh tissues (cerebral cortex, brain stem, cerebellum, spinal cord) from the four horses from the northeastern United States. Further virus identification for these tissues was obtained using a previously described RT-PCR technique
14
slightly modified to achieve results directly from pools of fresh tissues (E. Ostlund, personal communication). Representative tissue samples of major organs and the whole central nervous system (CNS) were fixed in 10% neutral buffered formalin solution and processed for histology. Tissue sections were stained with hematoxylin and eosin (HE), periodic acid–Schiff, Luxol fast blue, and crystal violet for Nissl substance and were processed with indirect immunoperoxidase staining using an automated slide processing unit (Autostainer Immunostaining System S3400, DAKO Corp., Carpinteria, CA). Tissue sections were mounted on Probe-On (Sigma Diagnostics, St. Louis, MO) glass slides coated with poly-
The leukocyte populations infiltrating the CNS were examined using antibodies recognizing CD3 of T cells (rabbit polyclonal antibody, dilution 1:100, DAKO), BLA.36 of B cells (murine monoclonal antibody, dilution 1:60, DAKO), and MAC 387 of macrophages (murine monoclonal antibody, dilution 1:1600, DAKO). Pretreatment included proteinase K (DAKO) for 5 minutes. The identity of cells labeled by each antibody was confirmed by the presence of morphologic features characteristic of each cell type. Positive controls were tissues from normal horses containing the appropriate cell types.
Results
Histologic lesions and peroxidase immunohistochemistry results are reported in Table 2. Results of clinical examination and neuropathology of the horses involved in the 1998 WNV epizootic in central Italy have been reported elsewhere. 2 Infection in all horses was characterized by mild to moderate, multifocal lymphocytic polioencephalomyelitis with constant involvement of the ventral and lateral horns of the thoracic and lumbar spinal cord and associated with moderate to severe hemorrhage in some horses. Slight to moderate lymphocytic and monocytic inflammation and scattered foci of microgliosis were observed in the medulla oblongata and pons and, to a lesser extent, in the basal nuclei, thalamus, and mesencephalon.
∗ Degree of inflammatory changes (lesions); - = none; +/- = occasional; + = mild; ++ = moderate; ++ = severe; NA = not available.
† Immunohistochemical results, amount of WNV antigen (virus): - = negative; + = single cell positivity; ++ = multiple cell positivity; NA = not available.
‡ Presence of perivascular hemorrhage.
§ Occasional presence of inflammatory lesions in some cortical areas associated with leptomeningitis.
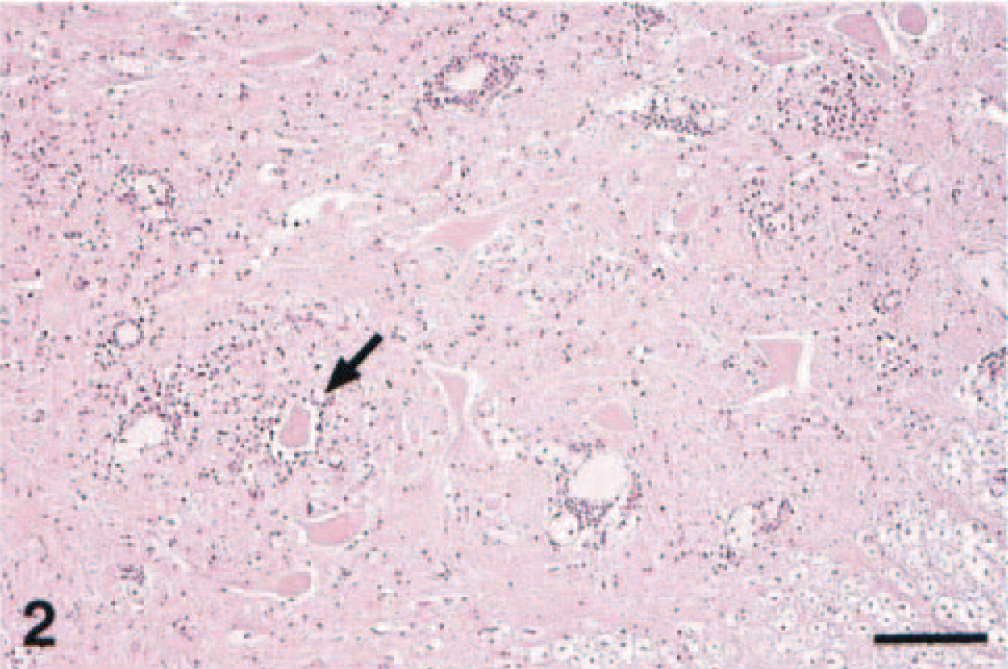
In one horse from the northeastern United States (horse No. 7), lesions were macroscopically evident, with petechiae sparsely distributed throughout the entire rhombencephalon and extending multifocally through the entire spinal cord (Fig. 1). These petechiae were especially prominent within the thalamus, the caudal brain stem, and the ventral horns of the spinal cord. All four American horses showed microscopic changes similar to those of the Italian horses. These changes were characterized by multifocal mild to severe perivascular cuffs comprised of lymphocytes and a few macrophages, with mild infiltration of lymphocytes and gliosis in the adjacent neuropil (Fig. 2). In the most severely affected areas, neuronal degeneration was prominent and characterized by central chromatolysis, cytoplasmic swelling, or cell shrinkage. Small, scattered microglial foci, occasional neuronophagia, and presence of small necrotic areas comprised of macrophages, neutrophils and cellular debris were also observed (Fig. 3). In all horses from the northeastern United States, these lesions involved the basal nuclei, gray matter of thalamus, midbrain, lower brain stem, and ventral and lateral horns of the spinal cord. Scattered and small microglial foci were sometimes observed in the white matter of the spinal cord. Swollen axons (spheroids) were also noted in the most severely affected spinal cord segments. Six of the 10 horses had a prominent diapedesis of leukocytes, with mild to severe perivascular hemorrhage, degenerative changes and occasional necrosis of the vessel wall with presence of a few neutrophils; within the CNS blood vessel lumina, there were marginating small lymphocytes and macrophages (Fig. 4).
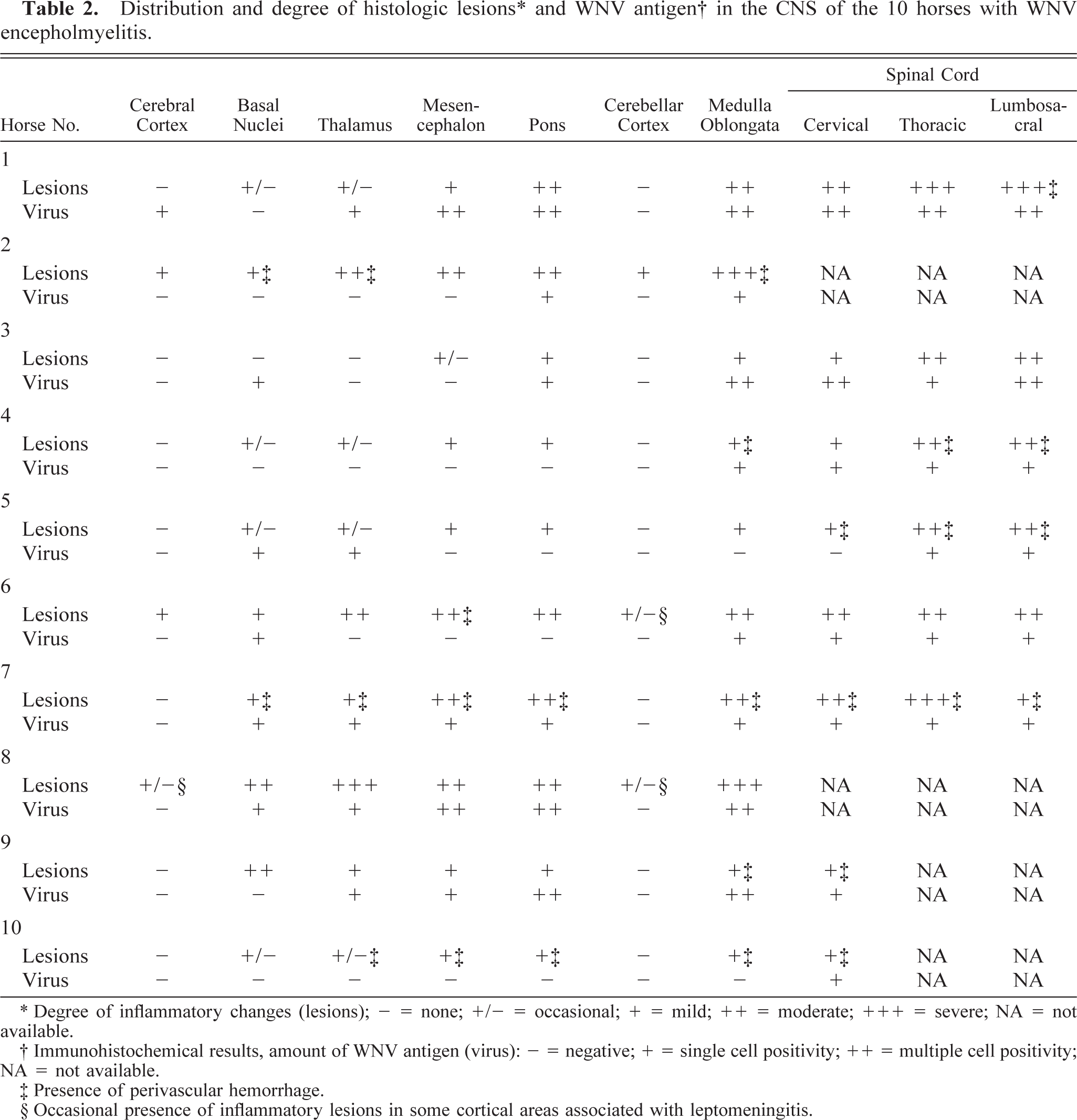
Left thalamus (left) and cervical spinal cord (right); horse No. 7. Severe, multifocal, acute hemorrhages mainly involve the gray matter. The ventral and medial edges of the thalamus have been trimmed, and the tissue is partially fixed in buffered formalin. Bar = 2 cm.
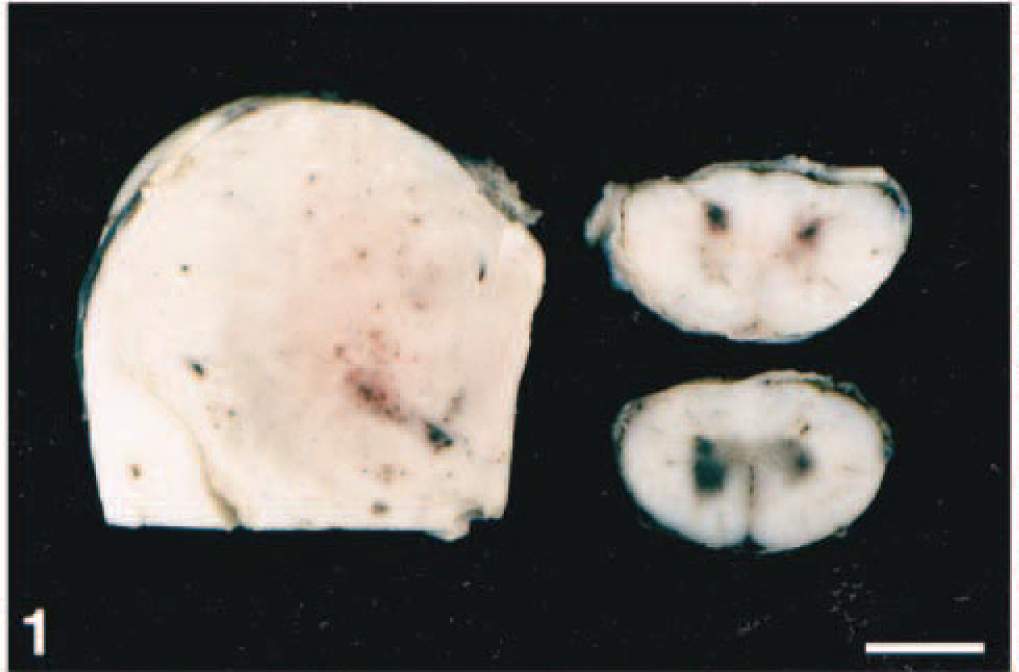
Thoracic spinal cord; horse No. 1. Lymphocytic cuffs and inflammation of the neuropil with neuronal degeneration and neuronophagia (arrow). HE. Bar = 200 µm.
Caudal brain stem; horse No. 8. Severe perivascular neutrophilic infiltrate with glial cells and a degenerate, shrunken, neuron surrounded by glial cells (bottom left). HE. Bar = 65 µm.
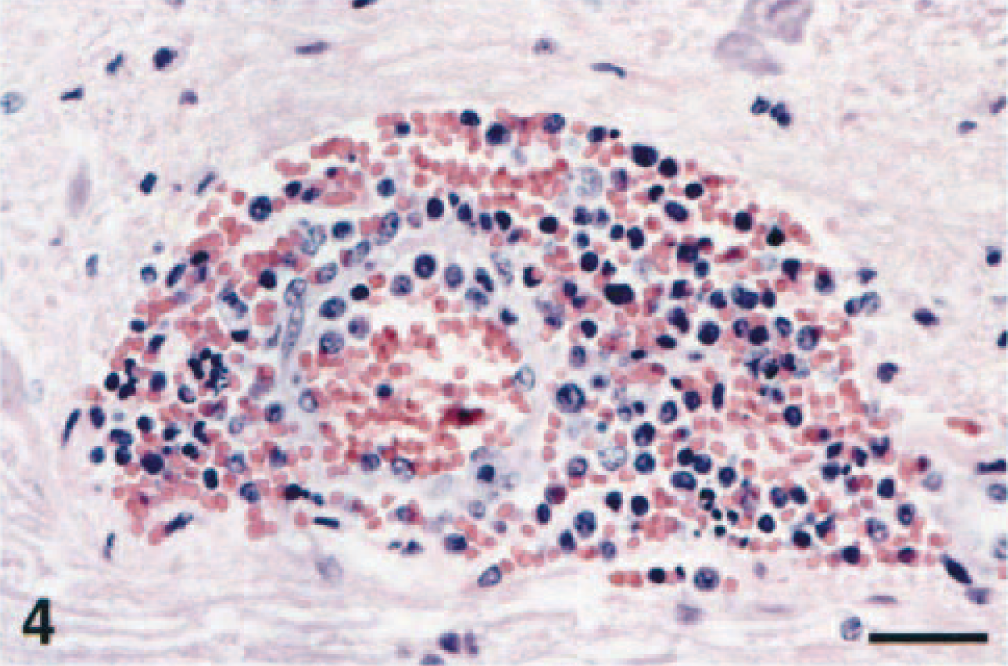
Caudal brain stem; horse No. 7. Margination of intravascular leukocytes, diapedesis, hyaline degeneration of the arteriolar wall, and prominent perivascular hemorrhage. HE. Bar = 65 µm.
Mild focal inflammation of cerebral and cerebellar cortical areas associated with mild leptomeningitis was occasionally observed. Choroid plexi, ependyma, and the pituitary and pineal glands were not affected. No lesions were detected in the remaining areas of the CNS, eyes, peripheral ganglia, or peripheral nerve samples. Age-related lesions in some older horses included neuronal lipofuscinosis, vascular siderocalcinosis, choroid plexus hyalinosis, and cholesterinosis. A small adenoma of the pars intermedia of the pituitary gland was observed in horse No. 9. Lesions in extraneural tissues of horses from the United States included mild scattered hemorrhages in the kidney medulla and atrophy of splenic lymphoid follicles (horse No. 7) and mild, focal and nonsuppurative myocarditis (horse No. 9).
In sites of inflammatory lesions, WNV antigen was mainly localized within the gray matter and had a finely granular appearance within the cytoplasm of a few morphologically normal and degenerate neurons. WNV antigen was also present in a large number of morphologically normal nerve fibers, axonal hillocks, glial cells, and spheroids of the medulla oblongata and spinal cord in all horses (Fig. 5). In the glial foci and areas of neutrophilic infiltration, small amounts of WNV antigen were contained within the cytoplasm of glial cells, macrophages, and occasional neutrophils. Occasionally, morphologically normal neurons diffusely harbored intracytoplasmic WNV antigen (Fig. 6) and viral antigen-positive glial cells also surrounded some neuronophagic neurons. No viral antigen was detected within the peripheral nervous system and extraneural tissues.
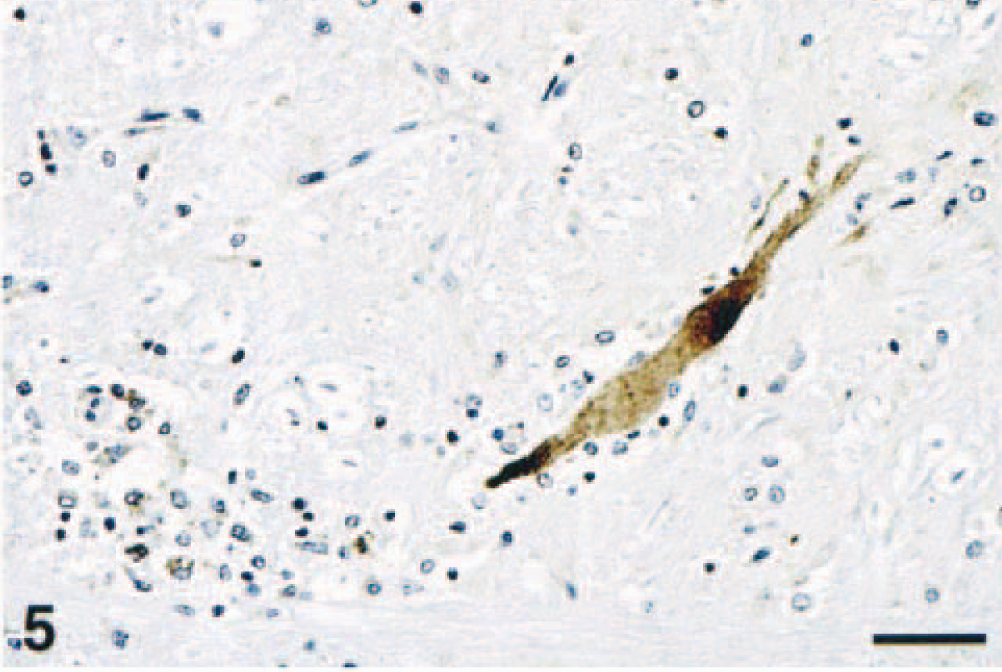
Medulla oblongata, reticular substance; horse No. 8. A prominent swollen axon and several macrophages (bottom left) contain WNV antigen. Peroxidase immunohistochemistry and hematoxylin. Bar = 65 µm.
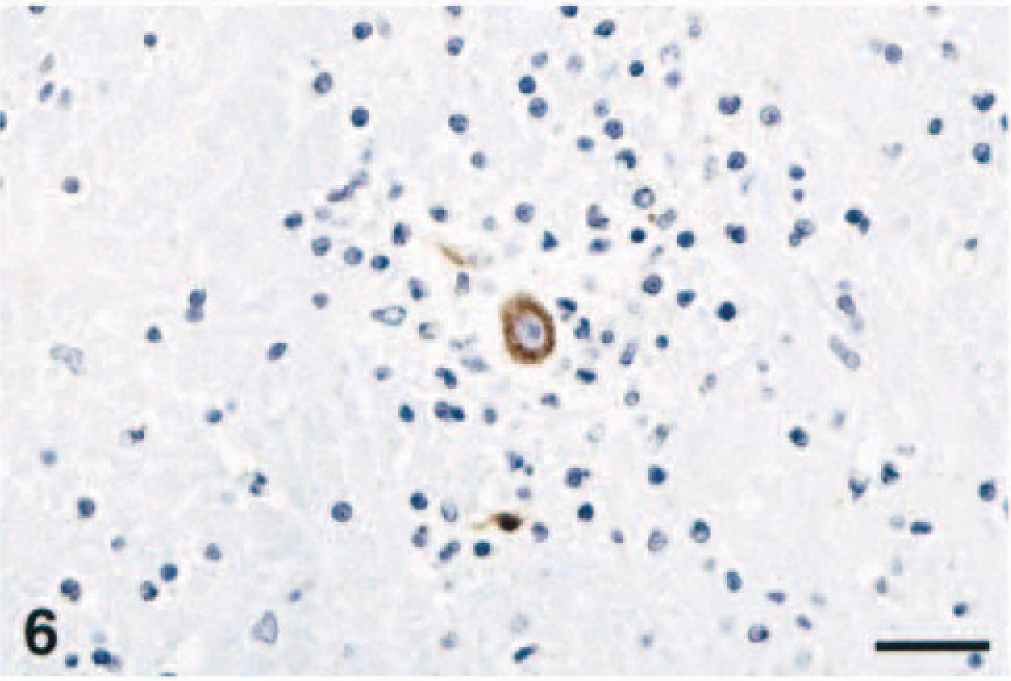
Basal nuclei; horse No. 1. Neuron infected with WNV surrounded by glial cells, lymphocytes, and a few macrophages. Neuronal cytoplasm, axon and a few glial cells contain WNV antigen. Peroxidase immunohistochemistry and hematoxylin. Bar = 65 µm.
Perivascular and leptomeningeal inflammatory infiltrate was composed almost exclusively of small T lymphocytes labeled for CD3 antigen. Occasionally, the perivascular infiltrates were composed purely of macrophages labeled for MAC 387, and rare macrophages contained intracytoplasmic WNV antigen. No BLA.36 positive leukocytes were identified.
Discussion
The histologic lesions and clinical signs observed in the affected horses were similar to those in the only previously described case of natural equine WNV infection where a detailed pathologic examination was carried out. 9 Lesion morphology and distribution were typical of polioencephalomyelitis, with prevalent involvement of lower brain stem and ventral horns of the thoracolumbar spinal cord, and were distinct from those of other equine encephalomyelitides. 2 The results obtained in the present study indicate that WNV exhibits a pronounced, if not exclusive, CNS tropism in the horse. In all cases, the quantity of WNV antigen was scant when compared with the extent of the inflammatory lesions. Perivascular infiltrate and hemorrhage were frequent findings, but no evidence of direct viral infection of the vessel wall was detected by immunohistochemistry.
The lack of apparent WNV antigen in extraneural tissues of the horses in this study is distinctly different from the widespread production of WNV antigen seen in infected birds 30 and indicates that WNV infection in horses is largely limited to CNS infection. Although some virus infection of other tissues may have occurred early in the course of disease and been cleared prior to death, the lack of antigen and virus-associated lesions in extraneural tissues makes this possibility unlikely. The Italian horses were examined 2 and 9 days following the onset of clinical signs, 2 and most American horses were euthanatized shortly after the onset of clinical disease; thus, limited numbers of virions may be sufficient to stimulate a significant CNS inflammatory response. The disassociation of virion density from lesion severity suggests an immunopathologic mechanism in the pathogenesis of WNV disease in horses.
Different genetic variants of WNV may exhibit different pathogenicity for mammals and avian species. The strain of WNV isolated in the United States and in Israel is highly pathogenic in many species of birds, 13, 30 whereas other WNV strains have not been associated with avian disease in nature. Strain variation also has been associated with differences in pathogenicity of WNV for cell cultures and mice, 18 and differences in virulence for human beings has been demonstrated for a number of viral strains. 21 Neurovirulence and neuroinvasiveness of encephalitogenic flaviviruses has been associated with changes of the amino acid sequence of viral glycoprotein E and with mutations in the nonstructural or untranslated regions of the flavivirus genome. 15 Our finding that horses from the United States exhibited more widespread lesions in the brain than did horses from Italy suggests that the US strain of WNV is highly pathogenic in horses as well as birds. The Italian isolate is considered closely related to the WNV isolate from a Moroccan equine epizootic in 1996. 14 No cases of avian or human disease have been associated with that particular WNV isolate. 32
West Nile virus experimental infection has been attempted in horses, mules, sheep, 30 donkeys, 24 dogs, 1 and pigs, 11 resulting in low-level and transient viremia that was no longer detectable in horses 4–8 days following inoculation. 19 Mild pyrexia was observed 3–6 days following WNV inoculation. A second mild pyretic phase was identified only in those animals that developed neurologic signs 5–22 days after inoculation. 19,20 Virus reisolation from different areas of the CNS, in relation to the route of inoculation and duration of the infection, suggested a descending neural kinetic of viral infection. 19 The onset, progression, and outcome of the disease depends on a number of factors, including species and route of inoculation, 33 infective virus burden, 12 age, 8, 23 and, as observed in humans, health status. 23 In the horses of this report, no influence of age was readily discernible, although other host factors may influence the course and outcome of flavivirus infection, including immune responses and genetic factors. 26 In the horses of this study, no other important diseases were detected, suggesting that a concurrent disease does not play a major role in the pathogenesis of WNV infection in horses.
The route of neuroinvasion by flaviviruses remains controversial. It has been suggested that low-level viremia in clinical hosts is inadequate to sustain endothelial cell viral replication, resulting in virus budding on the neuroparenchymal side. More likely, encephalitogenic arboviruses could invade the olfactory neuroepithelium during the viremic phase, subsequently infecting the olfactory neurons by retrograde axonal transport. 16 The negative finding of WNV antigen in the endothelial cells from all horses investigated here is consistent with this hypothesis. Once in the CNS, WNV may spread rapidly throughout the neuropil and may replicate in particularly sensitive areas, such as the hippocampus in mice, and the thalamus, substantia nigra, and cerebellum in monkeys and human beings. 17, 22
Contrary to findings in mammals, both spontaneous WNV infection of several bird species 30 and experimental infection in chickens 25 have been associated with lesions in multiple organs and, in crows and other wild birds, presence of large amount of viral antigen within either CNS and extraneural organs. Restricted localization of viral antigen within neurons, neuronal processes and necrotic areas, mainly of the brain stem and spinal cord, has been demonstrated by immunohistochemistry in an investigation of human WNV encephalitis. 27
Viral distribution varies considerably among different equine encephalomyelitides. In eastern equine encephalitis, alphavirus antigen is abundant and localized within the cytoplasm of neurons, glial cells, and extraneural cells, such as smooth muscle, myocardial fibers, dendritic cells. 7 In horses with rabies, rhabdovirus antigen tends to be abundant and widespread within the gray matter, including peripheral ganglia and retina, being located within the cytoplasm of neuronal cell bodies, nerve fibers, and some glial cells. 6 In rabies and alphavirus encephalitides, there is also prominent cerebral cortical involvement. 6, 7 EHV-1 induces ischemic myeloencephalopathy with perivascular lymphocytic cuffing secondary to endothelial necrosis and thrombosis associated with vasculitis, and small quantities of EHV-1 antigen are expressed within nucleus and cytoplasm of small muscular artery endothelium, myocytes, and pericytes, mainly in the spinal cord. 5
Considering the sporadic occurrence of WNV-associated disease in horses, moderate lesions, short viremia, and limited amount of viral antigen detected in tissues, horses may be incidental, dead-end hosts of this flavivirus. Consequently, horses most likely do not play a significant role in the epidemiology of WNV and may not pose a major risk to human health. However, special protection should be used during the dissection and examination of the CNS of WNV-infected horses. The limited tissue distribution of WNV antigen apparent in the horses of this study should be taken into account during postmortem examination. All six horses from Italy had localized lesions and viral antigen distribution almost exclusively limited to the lower brain stem and thoracolumbar spinal cord, with mild occasional involvement of the cerebrum. Although removal of the spinal cord from horses infected with neurotropic viruses can be potentially dangerous to the prosector and current regulations discourage the removal of the spinal cord from possible rabies cases, our findings demonstrate that failure to examine the spinal cord limits the ability to accurately identify WNV infection in horses.
In WNV-infected horses, low-level viremia, antibody production, and sub clinical illness seem to be the most common manifestations. Serologic investigations in a number of WNV equine disease outbreaks revealed that many horses had WNV antibodies, although only a few developed clinical signs. In clinically affected horses, a very serious and life-threatening disease develops, with a fatality rate of 43–45%. 13
WNV should be considered an important pathogen for horses, capable of inducing significant neurologic disease in temperate regions, where enzootic infection cycles may develop and be maintained. 10 The paucity of WNV antigen in equine nervous tissues indicates that multiple sections of brain and spinal cord labeled by immunohistochemistry should be examined, and negative results should be interpreted with significant caution. The use of histopathology, peroxidase immunohistochemistry, virus isolation, and RT-PCR techniques will increase the sensitivity for WNV identification, allowing further characterization of this increasingly important disease.
Footnotes
Acknowledgements
We are very grateful to the scientists who helped us. Dr. Vincent Deubel (Centre de Recherche Mérieux Pasteur, Lyon, France) provided one of the anti-WNV antibodies utilized in this study and identified the WNV of the Tuscany epizootic, Dr. Eileen N. Ostlund (National Veterinary Services Laboratories, Ames, IA) identified the American WNV isolates, Dr. D. E. Swayne (Southeast Poultry Research Laboratory, US Department of Agriculture, Athens, GA) provided the WNV avian positive controls, and Dr. Pamela Wilkins (Department of Clinical Studies, New Bolton Center) provided assistance in manuscript preparation. We thank Dr. Perry Habecker, chief of the pathology service at New Bolton Center, Dr. Daniela Ennulat (Schering-Plough), and Ralph Conti (New Bolton Center) for necropsy help. We are particularly grateful to Mrs. Jaqueline Ferracone (New Bolton Center) and Mrs. Lisa Baroncini (Dipartimento di Patologia Animale, University of Pisa) for superb assistance with immunohistochemistry and histology.