Abstract
Using eye samples of nine 9-week-old experimentally West Nile virus (WNV)–infected red-legged partridges (
Flaviviruses are small single-stranded RNA viruses with a virtually worldwide distribution. West Nile virus (WNV), a pathogen of importance for birds, has a wide tissue distribution. The eye is one of the target organs in infected avian hosts. Visual impairment and ocular lesions have been described mainly in hawks,
4
–6
but little is known about the mechanisms by which this virus reaches the eye and produces ocular lesions.
4
Another flavivirus, Bagaza virus (BAGV), recently caused an outbreak among game birds in southwestern Spain, producing neurologic signs in common pheasants (
For the present study, we collected right eye samples of nine 9-week-old red-legged partridges (Nos. 1–9) subcutaneously inoculated with 107 pfu of a cell culture–passaged New York/1999 (NY99) WNV strain. Experimental infections were done with approval of IACUC of the Instituto Nacional de Investigación Agraria y Alimentaria INIA (INIA permit number 2011-022) These birds were euthanized on days 3 (No. 2), 7 (No. 4), 10 (Nos. 6 and 7), and 14 (Nos. 8 and 9) postinoculation (dpi) or found dead during the experiment presumably due to WNV infection (2 dpi [No. 1], 7 dpi [No. 3], and 8 dpi [No. 5]). Eye samples were examined to follow the appearance of lesions and antigen during the time course of the infection. Moreover, we took right eye samples of 6 sexually mature red-legged partridges (Nos. 10–15) and 3 common pheasants (Nos. 16–18, corresponding to references 1, 2, and 3 from Gamino et al 3 ) that died or were euthanized in a moribund state during the recent BAGV outbreak in Spain. 3 In these samples, the objective was to compare microscopic lesions and BAGV antigen distribution between the 2 species. The aim was to expand our recent description of species-specific tissue tropism of BAGV and to investigate the lesions causing blindness observed in the affected partridges.
Hematoxylin and eosin stain and viral antigen detection by immunohistochemistry (IHC) were performed on eye sections. For the IHC, a rabbit polyclonal antibody against WNV that has been shown to cross-react with other flaviviruses (product 81–015; BioReliance, Rockville, Maryland) was used by applying the protocol described previously 3 but using 3-amino-9-ethylcarbazole (AEC) as a visualizing method.
These sections were examined under a light microscope by 2 different investigators (V.G. and U.H.). For comparative purposes between species (BAGV) or among different dpi (WNV), the distribution and abundance of the viral antigen were subjectively scored as absent, mild (focal or multifocal, <10% cells stained), moderate (multifocal, 10%–50% cells stained), or marked (diffuse, >50% cells stained).
WNV and BAGV infection was confirmed in several organs (brain, heart, spleen, and kidney) of all experimentally and naturally infected birds by real-time reverse transcription polymerase chain reaction (RT-PCR) and sequencing as previously described. 2,3
Unlike BAGV-infected partridges, none of the experimentally WNV-infected birds showed visual impairment.
Experimentally WNV-infected partridges that were euthanized had an increased number of lymphoplasmacytic, histiocytic, and, less frequently, heterophilic inflammatory cells in the vascular space of the choroid, pecten, and ciliary body on various dpi (Table 1). Mild inflammation in the connective tissue of the sclera (7 dpi, No. 4) and ciliary body (7 and 14 dpi, Nos. 4 and 9, respectively) and endothelial cell swelling in the choroid (10 dpi, No. 7), periocular muscle, and pecten (14 dpi, Nos. 8 and 9) were also detected (Table 1 and Fig. 1). Compared with euthanized birds, fatally WNV-infected partridges showed mononuclear inflammatory cells and muscular degeneration in the iris on days 2 and 7 pi (Nos. 1 and 3, respectively) and mild optic neuritis on 7 dpi (No. 3) (Table 1). In both euthanized and fatally infected birds, WNV antigen was only detected in photoreceptor (rods and cones) processes in the retina. The immunostaining was mild and appeared earlier in euthanized than in fatally infected partridges (Table 1 and Fig. 2).
Lesion and Antigen Distribution in the Eyes of Experimentally WNV-Infected Red-Legged Partridges, Collected Between Days 2 and 14 Postinoculation (dpi).

HE, hematoxylin and eosin; IHC, immunohistochemistry; NA, no tissue analyzed; WNV, West Nile virus. Lesion evaluation in HE-stained sections: −, lesion absence; +, lesion presence: (a) infiltrating inflammatory cells, (b) myofibers degeneration, (c) increased number of inflammatory cells in the vascular space, and (d) endothelial cell swelling. Immunostaining scores: −, absent; +, mild; ++, moderate; +++, marked.
*Partridges found dead.
†Euthanized partridges.
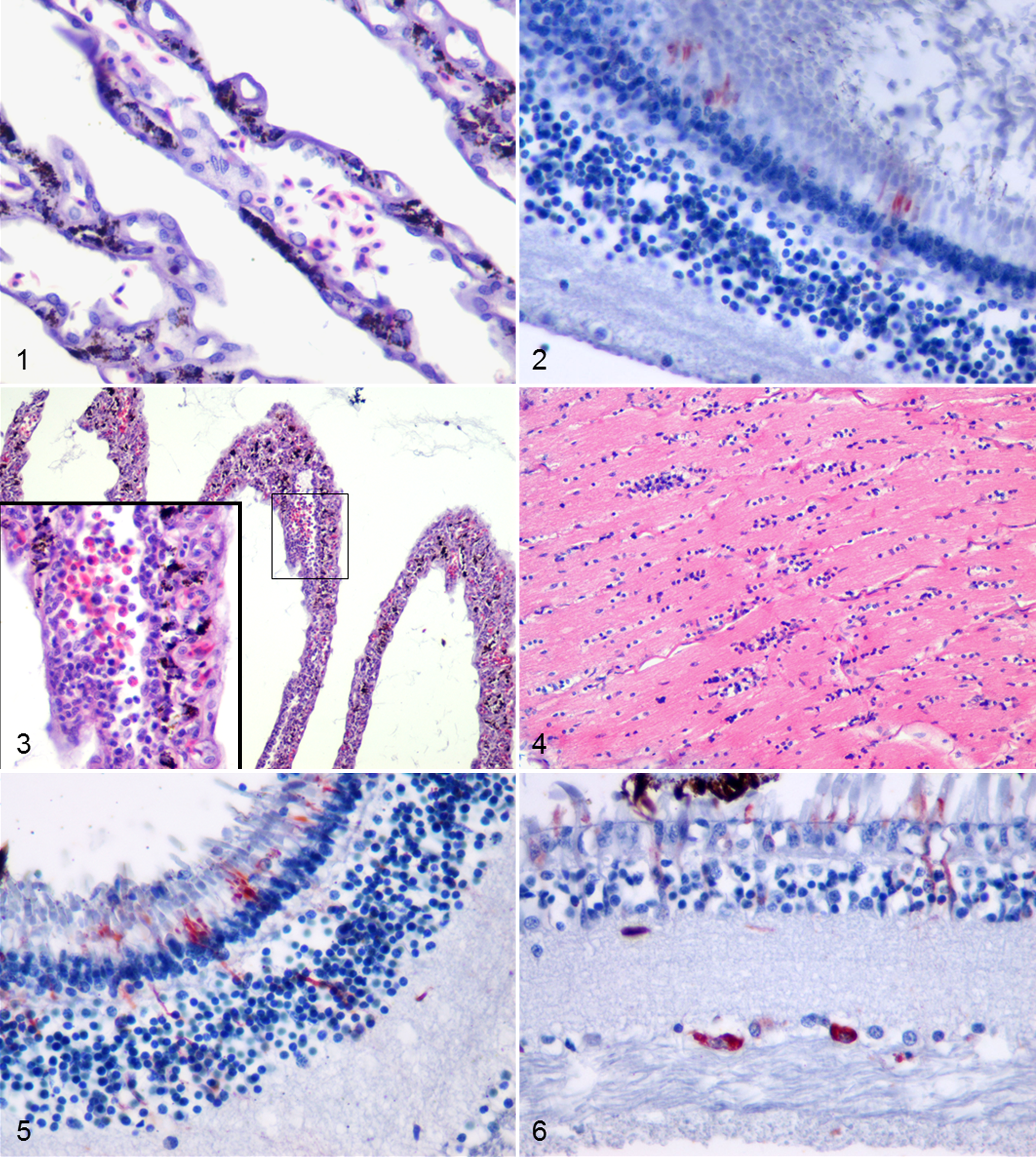
In BAGV-infected partridges, we observed an increased number of mononuclear or mixed inflammatory cells in the vascular space. These cells also markedly infiltrated the periocular muscle, iris, ciliary body, choroid, pecten, and optic nerve. In addition, myofibril degeneration (Nos. 11–14) and endothelial cell swelling (Nos. 13 and 14) were detected but only in the periocular muscle. In pheasants, inflammation was less intense, with the exception of the pecten and the optic nerve (Figs. 3, 4), and absent in the periocular muscle. Endothelial cell swelling was observed in the choroid and pecten of case No. 16. In both species, BAGV antigen was detected only in the retina. Positive staining was observed in photoreceptor processes in the rods and cones layer, in the cells of the outer nuclear layer, in cells and vertical cell processes in the inner nuclear layer, and in ganglion cells (Figs. 5, 6). As can be observed in Fig. 7, the concentration of BAGV antigen detected in the retina of partridges was higher than in pheasants.
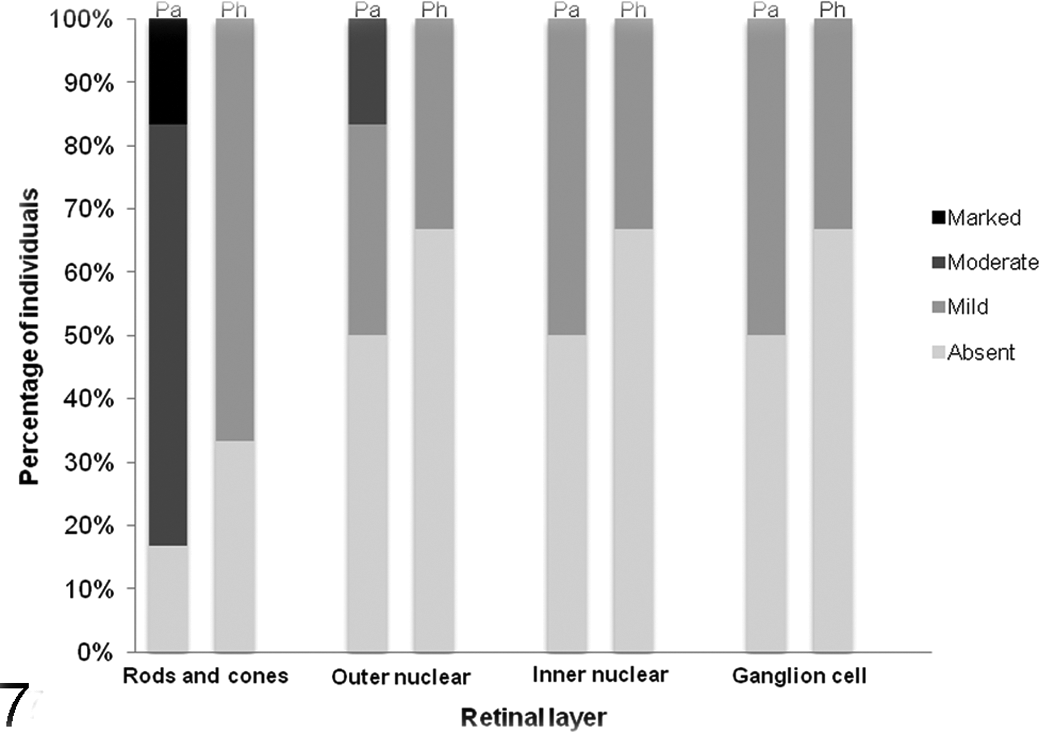
Immunostaining scores of Bagaza virus antigen in different layers of the retina of red-legged partridges (Pa) and common pheasants (Ph) naturally infected with the virus.
In the present study, we observed inflammation in ocular structures and WNV antigen in the retina of experimentally WNV infected partridges. These findings are similar to those described in the eyes of naturally infected hawks. 4 –6 Ocular infection of flaviviruses is thought to occur either hematogenously during viremia or by extension from the central nervous system (CNS) to the retina via the optic nerves. 4 Studying eye samples of the experimentally WNV-infected partridges, microscopic lesions appeared early in the course of the infection and, in euthanized animals, WNV antigen was detected as early as 3 dpi. Considering that WNV RNA, viral antigen, and microscopic lesions were detected later in the CNS of the same birds (7–10 dpi, data not shown), we assumed that WNV distribution into the eye occurred hematogenously.
In this study, we also found differences in the inflammatory response and antigen abundance in the eyes of BAGV-infected partridges and pheasants. The mild inflammatory reaction and the scarce BAGV antigen presence in the retina of pheasants could be related to the apparently lower virulence of this virus in this species. 1,3 On the contrary, the higher abundance and wider distribution of BAGV antigen in the retina of infected partridges accompanied by a more severe inflammatory reaction may be the cause of the more prominent effect of BAGV on visual capacity in this species. This matches clinical observations of apparent blindness in BAGV-infected partridges absent in the experimental WNV infection. Nevertheless, pathological differences could be related to the experimental conditions in the WNV infection, the age of the infected birds, or the virus.
In conclusion, virus replication and development of lesions in ocular structures of flavivirus-infected gallinaceous birds vary with the virus and host species involved. The degree of retinal involvement apparently determines the development of impaired vision, as evidenced here in the case of BAGV-infected red-legged partridges. Under field conditions, visual impairment could have an important impact on the survival of individuals that adds to primary mortality from the disease.
Footnotes
Acknowledgments
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants AG2008-02504GAN and SAF-2008-04232 funded by the Spanish Ministry for Science and Innovation, grants FAU2008-00006 and RTA2011-00036 from the Instituto Nacional de Investigación y Tecnología Agraria y Alimentaria (INIA), and The Network of Animal Disease Infectiology and Research Facilities, NADIR-EU-228394, funded by the EU.
