Abstract
West Nile virus infection poses a significant threat, especially during the warmer months when mosquitoes are abundant. Clinicians must remain vigilant for neuroinvasive illness in patients presenting with febrile symptoms and malaise following mosquito exposure. While magnetic resonance imaging and cerebrospinal fluid analysis aid in differential diagnosis, detecting West Nile immunoglobulin M in serum is crucial for definitive diagnosis. Treatment primarily involves supportive care due to the absence of established regimens, though promising outcomes have been reported with plasma exchange and intravenous immunoglobulin. We present the case of an 83-year-old resident of Alabama, an avid gardener living near a pond, who initially exhibited symptoms of productive cough, diarrhea, fever, and generalized malaise. However, within 48 h, he developed hypoxemia, functional quadriplegia, and bulbar palsy necessitating intubation. Diagnostic evaluations, including magnetic resonance imaging and positive West Nile virus immunoglobulin M in serum, confirmed West Nile virus-associated poliomyelitis viral syndrome, prompting intravenous immunoglobulin therapy. This case highlights the importance of promptly identifying and managing West Nile virus infection, especially in regions susceptible to mosquito-borne diseases, and being vigilant of the disease in non-endemic regions. The case also begs the question of the timing and efficacy of intravenous immunoglobulin and plasma exchange in West Nile virus infection and the fact that more data should be collected on these therapies.
Keywords
Introduction
West Nile virus (WNV) is a flavivirus transmitted by mosquitoes. WNV possesses zoonotic characteristics and belongs to the Flaviviridae family, encompassing roughly 75 distinct virus species. 1 WNV is part of the same serocomplex as the Japanese encephalitis virus and shares its classification with related viruses like St. Louis encephalitis virus (SLEV), Murray Valley encephalitis virus (MVEV), and Alfuy virus (ALFV).2,3 The first reported case dates back to 1937, isolated from a patient experiencing fever in the West Nile Province of Uganda.4–6 Over the past two decades, there has been a noticeable surge in both human and equine cases, underscoring the evolving threat posed by WNV. 6 In the United States, the data related to WNV surveillance are conveyed to the Centers for Disease Control and Prevention (CDC) using ArboNET, an internet-based arbovirus surveillance system jointly administered by the CDC and state health departments. According to their 2023 report, there have been a cumulative 2406 confirmed cases of human illness in 47 states for WNV disease, out of which 1599 cases have led to neuroinvasive human disease. WNV infections typically occur during the mosquito season, which begins in the summer and extends into the fall. 7 Unfortunately, there are currently no vaccines or medications available for the prevention or treatment of WNV in humans. However, the majority of individuals infected with WNV remain asymptomatic. 7 Approximately one out of every five individuals who contract the virus develops a fever along with other symptoms. Roughly 1 out of every 150 infected individuals face the potential of a severe and occasionally fatal illness. 7
Case presentation
The patient was an 83-year-old Caucasian male with a past medical history of hypertension, hyperlipidemia, and osteoarthritis who presented to our medical center with complaints of productive cough, diarrhea, fever, and generalized malaise for 4 days prior to the admission. He was a native of Alabama and lived in a house adjacent to a pond. He was frequently outside gardening and his vaccination status was unknown. On arrival, his initial neurologic exam demonstrated no significant findings; he had no focal deficits, he was alert and oriented and no cranial nerve deficits were noted either. The patient was found to be febrile on admission with a temperature of 102.5 F and had some rhonchi upon auscultation. He was initially treated with levofloxacin for suspected pneumonia. Within the next 48 h, the patient started to deteriorate; he rapidly became hypoxemic and functionally quadriplegic with bulbar palsy. This initially started with stroke-like symptoms, including facial droop and slurring of his speech which progressed to a minimally responsive state with an inability to protect his airway, disconjugate gaze, diplopia, and rigors. He was subsequently intubated and transferred to the intensive care unit. Initial imaging consisted of a CT head, CT angiogram, and CT perfusion which did not demonstrate any acute pathology. Given his initial presentation of flu-like symptoms with a sudden neurological decline, there were concerns for meningitis/encephalitis for which the patient underwent a lumbar puncture; this demonstrated a protein of 113, a glucose of 88, and 165 WBC. Cerebrospinal fluid (CSF) gram stain and culture were negative, as was the meningitis/encephalitis panel. A magnetic resonance imaging (MRI) of the lumbar spine was obtained (Figures 1–6) and demonstrated subtle cauda equina nerve root enhancement without mass or enlargement, which can be seen in meningitis and raised suspicion for West Nile encephalitis; MRI of the head was also obtained but was unremarkable. MRI of the cervical spine and brainstem were not obtained. Taking into consideration the patient’s history and initial presentation, rapid progression of symptoms, as well as the aforementioned MRI and CSF findings, WNV-associated poliomyelitis viral syndrome was suspected. WNV IgM was positive, IgG was not elevated, and Eastern Equine IgM was not positive. A 5-day course of intravenous immunoglobulin (IVIG) for poliomyelitis-like syndrome was started. Post-IVIG, the patient did not demonstrate any improvement in neurological status. An MRI of the lumbar spine (Figures 7–11) was repeated and demonstrated no significant interval change with stable, subtle enhancement of the nerve roots of the cauda equina. Ultimately, the patient required a tracheostomy and PEG tube (percutaneous endoscopic gastrostomy) for respiratory failure and profound encephalopathy. However, the patient continued to deteriorate clinically, and the family ultimately decided to withdraw care.
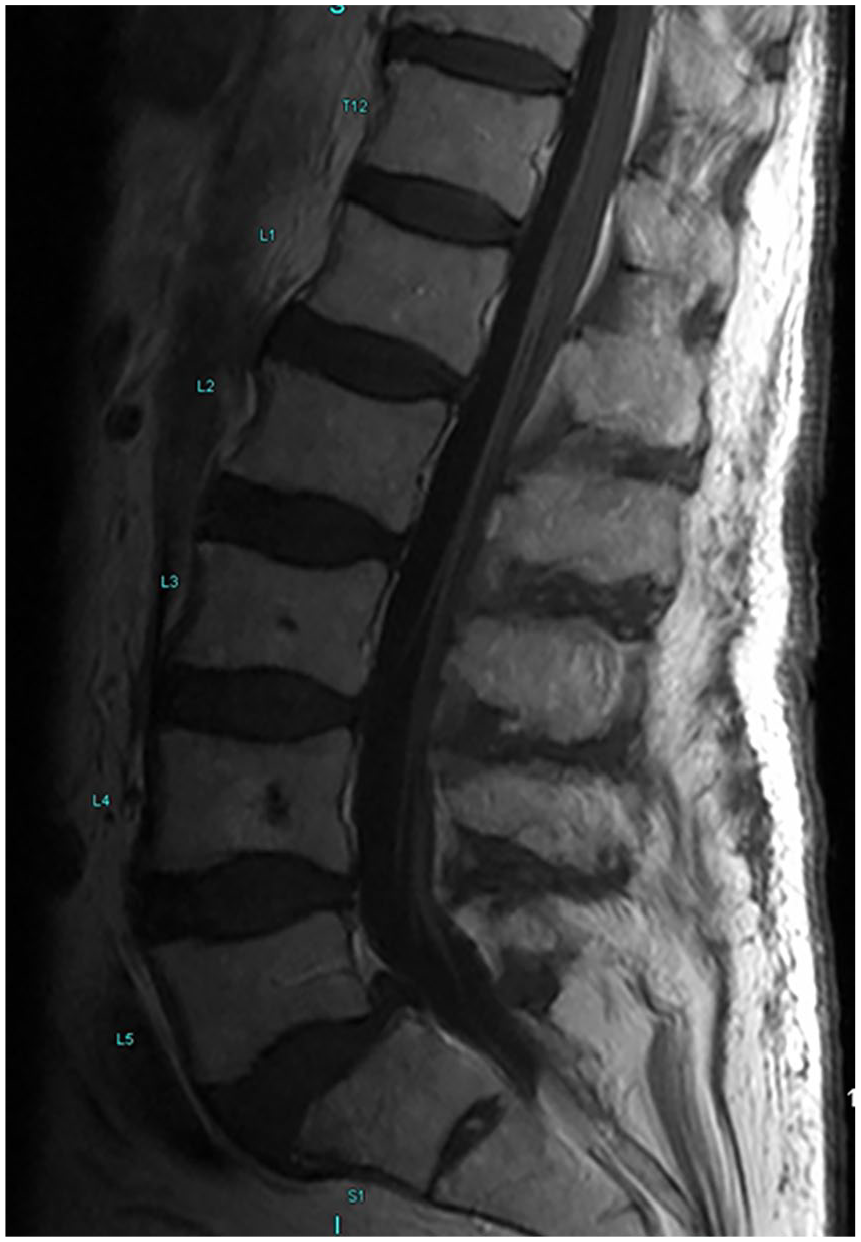
MRI lumbar spine with and without contrast prior to IVIG: lumbar spine sagittal view showing caudal nerve root enhancement.
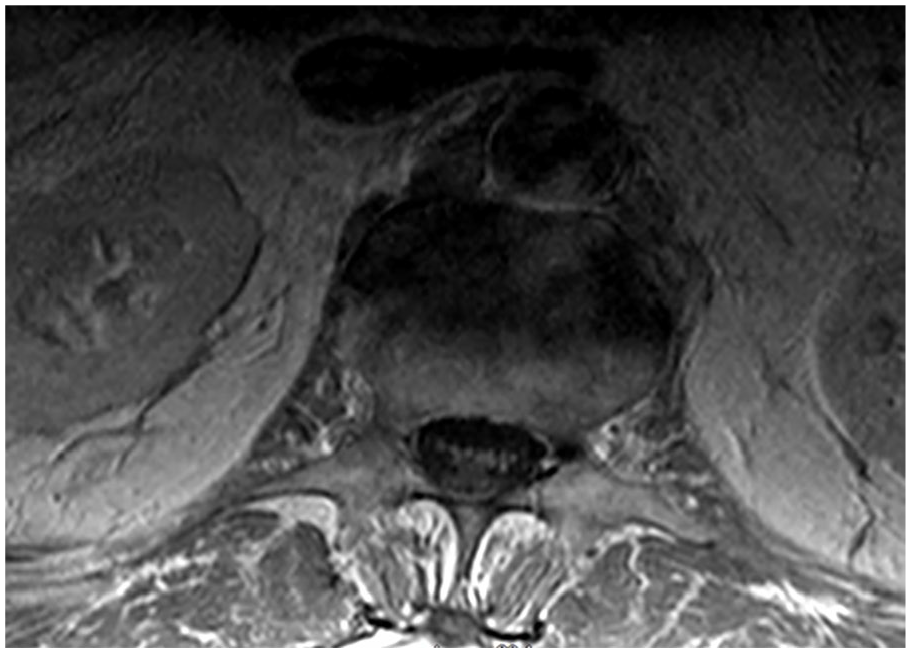
MRI lumbar spine with and without contrast prior to IVIG: T1 axial, L1-L2.

MRI lumbar spine with and without contrast prior to IVIG: T1 axial, L2-L3.

MRI lumbar spine with and without contrast prior to IVIG: T1 axial, L3-L4.

MRI lumbar spine with and without contrast prior to IVIG: T1 axial, L4-L5.
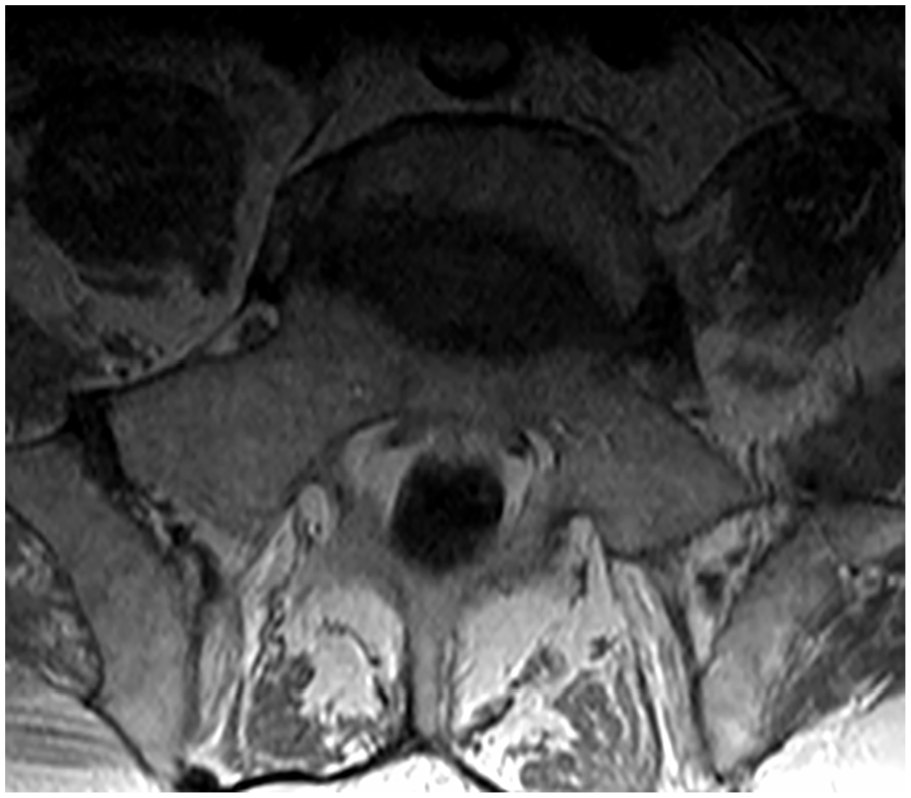
MRI lumbar spine with and without contrast prior to IVIG: T1 axial, L5-S1.
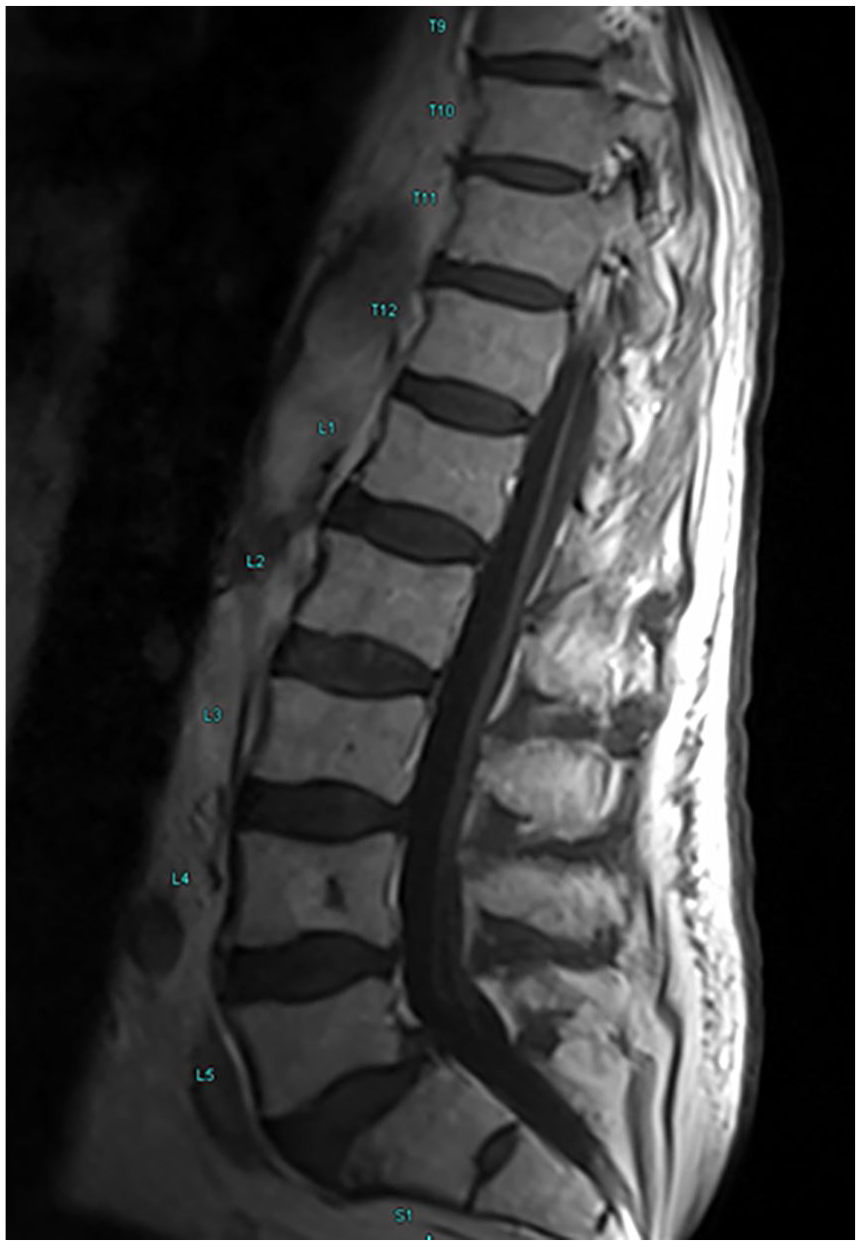
MRI lumbar spine with and without contrast post-IVIG: lumbar spine sagittal view with no significant interval change with stable subtle enhancement of the nerve roots of the cauda equina.

MRI lumbar spine with and without contrast post-IVIG: T1 axial, L1-L2.
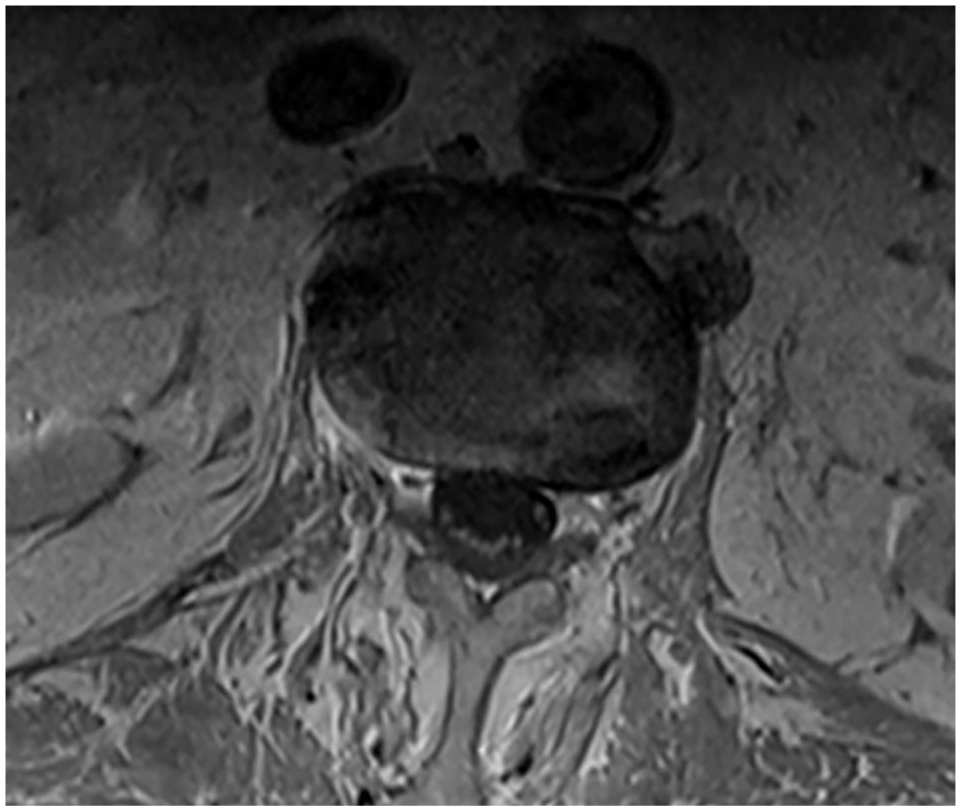
MRI lumbar spine with and without contrast post-IVIG: T1 axial, L2-L3.

MRI lumbar spine with and without contrast post-IVIG: T1 axial, L3-L4.
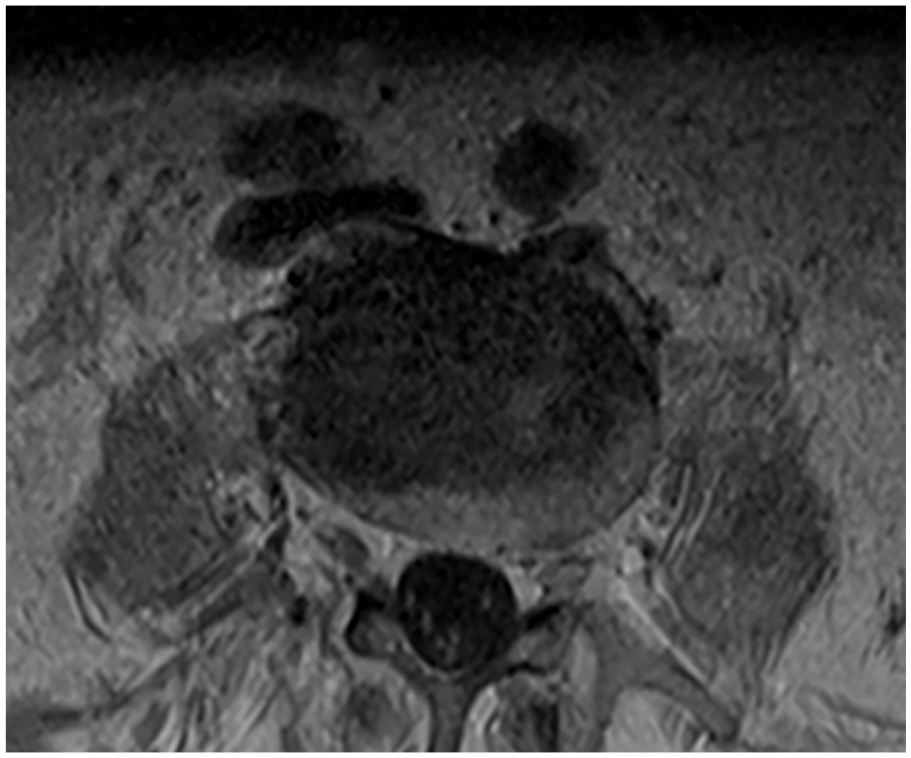
MRI lumbar spine with and without contrast post-IVIG: T1 axial, L4-L5.
Discussion
From the years 1999 to 2022, the CDC 7 has reported a total of 56,575 cases of WNV in the United States, with the most reported cases in 2002 with nearly 10,000 cases (Figure 12), with a larger proportion of cases being in males compared to females as well as the elderly (Figure 13). Approximately 25,777 cases required hospitalization, and 2776 resulted in death (Figure 14). Within the same timeframe, 28,684 cases resulted in neuroinvasive disease, of which 20,946 required hospitalization and 2641 resulted in death (Figure 15). Neuroinvasive disease tends to have a higher incidence per 100,00 in the Midwest United States (Figure 16). Petersen et al. describe the spread of WNV through birds and mosquitoes in the years 1999–2002 (Figure 17) with humans as incidental hosts with viral amplification occurring in early spring to late fall. 8

West Nile virus human disease cases by year of illness onset, 1999–2022.

West Nile virus human disease cases by age and sex, 1999–2022.

West Nile virus human disease cases reported by month of illness onset, 1999–2022, all disease cases.

West Nile virus human disease cases reported by month of illness onset, 1999–2022, neuroinvasive disease cases.

West Nile virus human neuroinvasive disease average annual incidence per 100,000 population by county of residence, 1999–2022.

West Nile Virus disease recognition by state.
According to Solomon et al., West Nile encephalitis can present with a variety of nonspecific symptoms including fevers, headache, malaise, fatigue, and gastrointestinal symptoms. 5 Mumoli et al. also describe multiple cases in which WNV neuroinvasive disease presented with nonspecific symptoms and eventually developed non-focal neurological symptoms; they emphasized the importance of having a high clinical suspicion in endemic areas, given that the disease can present like many other infections in the elderly. 9 The incubation of the virus is not well known but is thought to be 3–14 days, and only about 20% of patients develop symptomatic disease. 9 This could be a reason why we see an increased reported incidence of neuroinvasive cases compared to non-neuroinvasive cases. Severe neurological disease is uncommon, and advanced age increases the risk for neurological manifestations. 6 According to Leis and Stokic, a poliomyelitis syndrome with asymmetric paralysis is the most common neuromuscular manifestation of WNV. Fevers and meningoencephalitis are not always present. 10 Sejvar et al. identified patients with acute weakness and WNV infection; manifestations included a poliomyelitis-like syndrome, Guillain–Barré-like syndrome, and brachial plexus neuropathy. 11 Respiratory involvement leading to intubation and eventual tracheostomy placement was noted as well. 11 Another case of WNV manifesting as poliomyelitis is described by Glass et al. 12 ; they discuss how it can be differentiated from Guillain–Barré syndrome using electrodiagnostic studies demonstrating a pure motor illness.
Diagnosis of WNV includes MRI to examine for central nervous system (CNS) inflammation and IgM-capture enzyme-linked immunosorbent assay (ELISA). 13 Electromyograms and nerve conduction studies can be useful in those with neuromuscular involvement, but these studies were not performed on our patients. 13 CSF analysis may demonstrate an elevated protein level with lymphocytic pleocytosis and normal glucose; similar to what is exhibited in viral meningitis. 13 The ELISA for IgM has a sensitivity of 95% to 100% and this can be performed on the serum or CSF. 12 It should be noted that the IgM antibody cannot be detected until the end of the viremic period and that high antibodies in a patient with encephalitis or meningitis likely represent infection. To assess for CNS infection, intrathecal IgM antibodies can be used. 13
Although the mainstay of treatment for WNV is supportive care, 13 there have been some studies that demonstrate IVIG and plasma exchange as possible therapies.13,14 Malone et al. describe a case in which a patient with WNV-associated diaphragmatic paralysis and respiratory failure was successfully treated with IVIG and plasma exchange. 15 A similar case was described by Walid et. al in which a patient with a Guillain–Barré-like syndrome due to WNV was successfully treated with IVIG. 14
Due to the severity of its complications and death, prevention of WNV should be of the highest concern. Primary prevention can include mosquito repellents containing N, N-diethyl-meta-toluamide (DEET), avoiding areas where mosquitoes are active, protective clothing, and window screens. 12
Conclusion
WNV can cause a severe, life-threatening neuroinvasive illness in patients who develop a symptomatic infection. Clinicians should have high suspicion if a patient presents in the Summer and Fall months with a nonspecific febrile illness accompanied by generalized malaise after being in an environment with mosquitoes. Early recognition and identification are important, especially because encephalopathy in the elderly can be due to a multitude of reasons. MRI findings and CSF analysis are generally obtained to rule out other causes of meningitis or encephalitis, but a definitive diagnosis is made via the detection of West Nile IgM in the serum.4,6,13,16 For those who develop symptomatic infection with neurological complications, supportive care is the mainstay of treatment as there is currently no established treatment regimen; however, plasma exchange and IVIG have shown promise in some cases.6,13–15 This report highlights the importance of promptly identifying WNV infection so that effective treatment strategies can be sought out. This is especially important in regions susceptible to mosquito-borne diseases. Physicians should also be vigilant of the disease in non-endemic regions. The case also begs the question of the timing and efficacy of IVIG and plasma exchange in WNV infection. In our patient, these therapies did not help; could they have helped if administered sooner? Early recognition is also important if choosing to pursue the use of plasma exchange or IVIG, as many hospitals do not carry these therapies, and the patient may require prompt transfer. However, considering how fatal the infection can be, we emphasize the importance of primary prevention above all.
Footnotes
Author contributions
B.E. wrote the abstract and part of the case presentation for this paper. M.Q., along with S.J., were in charge of the discussion. S.M. wrote some parts of the case and the conclusion. S.L. was in charge of the figures and tables. T.D. managed the references. K.F. and F.A. were the attendings in charge of the project and provided final editing.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
Our institution does not require ethical approval for reporting individual cases or case series. We did not have to obtain approval from an ethics committee for this paper because the patient received the standard of care; no experiments were performed on the patient.
Informed consent
Written informed consent was obtained from a legally authorized representative(s) for anonymized patient information to be published in this article as the patient passed away from his illness.
