Abstract
After the introduction of West Nile virus (WNV) into North America, bird mortalities associated with West Nile disease have dramatically increased in this continent and, to a lesser extent, in Europe. The different West Nile disease incidence in birds in these 2 continents demands an explanation, and experimental studies can provide important information. The authors inoculated thirteen 9-week-old red-legged partridges (
Keywords
West Nile virus (WNV) is a globally distributed mosquito-borne flavivirus whose main reservoir is birds. 14 Before the 1990s, this arbovirus was considered a pathogen of limited importance for birds. 35 While West Nile disease has a high impact with significant mortality in bird species in the North American continent, 15 outbreaks in Europe are sporadic and have no impact on the population of the affected species. Several hypotheses have been proposed to explain the contrasting ecoepidemiology, such as different virulence of circulating strains or varied susceptibility of Palearctic and Nearctic bird species to WNV infection. 1
Experimental infections of wild birds can greatly enhance the understanding of the ecoepidemiology and virulence of different WNV strains in its natural host.
24
In fact, susceptibility to both New and Old World WNV strains has been evaluated in various bird species.
24
However, this information is unbalanced, as there are few experimental studies done in birds native to Europe while American species are fairly well represented.
8,10,17,28
The study carried out in the red-legged partridge (
Material and Methods
Virus
This study used a cell culture–passaged WNV New York/1999 strain (GenBank accession No. KC407666.1) isolated from a flamingo. 7,19
Experimental Infection
As described in Escribano-Romero et al, 10 recently hatched red-legged partridge chicks were raised in the experimental farm of the Instituto de Investigación en Recursos Cinegéticos until they were 8 weeks of age. At that time, they were transported to the biosafety level 3 facilities of the Centro de Investigación en Sanidad Animal of the Instituto Nacional de Investigación y Tecnología Agraria y Alimentaria. There, the birds were given 5 days for acclimatization. Prior to the experiment, oropharyngeal/cloacal swabs and blood of all birds tested negative for WNV-specific RNA by quantitative reverse transcription polymerase chain reaction RT-qPCR and negative for WNV antibodies by commercial ELISA (ID Screen, West Nile Competition, IDVet, Montpellier, France), respectively. For the present study, thirteen 9-week-old partridges were subcutaneously inoculated in the cervical region with 107 plaque-forming units (PFU) / individual of the WNV New York/1999 strain diluted in up to 0.1 ml of Dulbecco’s Minimum Essential Medium (supplemented with 2mM L-glutamine, 100 U/ml of penicillin, and 100 μg/ml of streptomycin). Animals were observed daily for clinical signs or death. For the specific purpose of our study, we prescheduled euthanasia at 3, 7, and 10 days postinoculation (dpi). Three birds were found dead during the experiment, and the 6 remaining ones were euthanized at 14 dpi. Euthanasia was performed by inhalation anesthesia with isoflurane (Isoba Vet; Intervet/Schering-Plough, Madrid, Spain) and intravenous injection of embutramide (T61; Intervet/Schering-Plough). All animals were handled in strict accordance with the guidelines of the European Community 86/609/CEE, and the protocols were approved by the Committee on Ethics of Animal Experimentation of the Instituto Nacional de Investigación y Tecnología Agraria y Alimentaria (permit No. 2010-005). Food and water were provided ad libitum throughout the experiment.
Sample Collection
Necropsies were performed on all dead and euthanized individuals. Samples of brain, heart, liver, spleen, and kidney were collected into sterile polypropylene tubes and stored at –70°C until analysis by RT-qPCR. Additional samples were fixed in 10% neutral buffered formalin—namely, brain, oral mucosa, thymus, trachea, lung, heart, liver, spleen, kidney, adrenal gland, testicle, ovary, small and large intestine, pancreas, cecal tonsils, bursa of Fabricius, spinal cord, pectoral muscle, and skin with feather follicles. For this study, samples from the partridges found dead (
Virus RNA Detection
WNV RNA was extracted with a commercial Kit (Speedtools RNA Virus Extraction Kit; Biotools B&M Labs SA, Madrid, Spain) and quantified by RT-qPCR. 16 For RNA quantification, a standard curve was generated with previously titrated WNV (106–10-1 PFU), and samples were considered negative when cycle threshold ≥37, equivalent to 9 PFU/sample (103 PFU/ml). 10
Histopathology
Formalin-fixed tissue samples were trimmed, embedded in paraffin, and processed to obtain 4-µm sections that were stained with hematoxylin and eosin. These were independently examined by 2 investigators (U.H. and V.G.) to determine the presence of WNV infection–associated lesions. When lesions were present, these were graded according to their distribution (focal, multifocal, or diffuse) and severity (mild, moderate, or marked).
Immunohistochemistry
Tissue sections were also mounted on pretreated slides (VECTABOND Reagent; Vector Laboratories, Inc, Burlingame, CA, USA) for immunohistochemistry (IHC). Details of antibodies and reagents used for WNV antigen detection and inflammatory cell characterization in the brain are given in Supplemental Table 2. Endogenous peroxidase activity was inhibited with a peroxidase blocking reagent (Dako EnVision+ System-HRP [AEC]; DakoCytomation, Carpinteria, CA, USA) (CD3, GFAP), or with 3% H202 diluted in methanol (WNV antigen, RCA-1, CD79αcy); slides were rinsed with 0.1% Tris-buffered saline (TBS) / Tween20 (TBS 0.05 M, pH 7.5); nonspecific primary antibody labeling was blocked with 2% albumin from bovine serum albumin (BSA; Sigma-Aldrich Chemie, Steinheim, Germany) diluted in 0.1% TBS/Tween20; and sections were counterstained with Mayer hematoxylin. WNV antigen detection was carried out as described recently. 11
Tissue sections of red-legged partridges positive for WNV by RT-qPCR served as positive controls. Negative controls included substitution of the primary antibody by 2% BSA-0.1% TBS/Tween20 and a negative rabbit antibody (product 81-015; BioReliance, Rockville, MD, USA), as well as tissue sections of a noninfected (WNV RT-qPCR negative) red-legged partridge. Sections of spleen and bursa of Fabricius of the same species served as IHC positive controls for T and B cells. In this case, negative controls included substitution of the primary antibody by 2% BSA-0.1% TBS/Tween20 and a brain section of a WNV RT-qPCR–negative partridge. A brain section of a noninfected partridge of the same age served as reference for RCA-1 and GFAP. Virus antigen staining was graded under the light microscope according to its distribution and abundance. Moreover, we studied the distribution of inflammatory cells within the brain parenchyma at a magnification of 200×. We counted the number of CD3+ T cells and RCA-1+ microglial cells/macrophages in 30 randomly selected fields (in each brain region: cerebrum, cerebellum, optic lobe, and pons and medulla oblongata) at different dpi at a magnification of 400×. Changes in the morphology, distribution, and relative abundance of GFAP+ astrocytes during the course of the infection were also evaluated.
Results
Morbidity and Mortality
The 3 partridges that died at 2 (No. 1), 7 (No. 3), and 8 dpi (No. 5) showed depression, ruffled feathers, and recumbency starting 24 hours before death.
Macroscopic Lesions
Macroscopic lesions were detected in every necropsied partridge. Differences among birds that died or were euthanized were minimal, with the exception of cachexia observed only in fatally infected birds and characterized by the absence of fat deposits and atrophy of the pectoral muscles. Lesions were more widespread between 7 and 10 dpi, and the most affected organs were heart, spleen, liver, and kidney. The most commonly observed lesions were diffuse pallor of the myocardium (Nos. 3, 6, 7) and of the hepatic (Nos. 4, 8, 9), splenic (Nos. 2, 5, 9) and renal parenchyma (Nos. 1, 5, 6), as well as splenomegaly (Nos. 2, 4). In the liver, the partridge found dead at 8 dpi (No. 5) had multifocal irregular reddish areas, and 1 of the birds euthanized at 10 dpi (No. 6) had multifocal pinpoint yellowish foci.
Other macroscopic lesions were observed, such as dilation of intestinal loops (7 of 9) and congestion of serosal vessels (4 of 9; mainly in duodenum and cecum), as well as distension of crop with a whitish pseudomembrane covering the mucosa (2 of 9).
Virus RNA Detection
The heart was the first tissue in which viral RNA was detected by RT-qPCR (3 dpi, No. 2; Table 1). The kidney and spleen were positive from 7 and 10 dpi (Nos. 3, 6, 7), respectively, but we did not detect viral RNA in the brain until 10 dpi (Nos. 6, 7). At 14 dpi, every organ was negative (Table 1).
Detection of WNV Genome in Tissues of Experimentally WNV-Infected Red-legged Partridges According to dpi.a

Abbreviations: dpi, days postinoculation; n/d, tissue sample not analyzed; PFU, plaque-forming units; WNV, West Nile virus.
aPositive quantitative real-time reverse transcription polymerase chain reaction results are expressed in PFU/ml. Tissue sample with cycle threshold ≥37 equivalent to ≤9 PFU/sample or 103 PFU/ml.
bPartridges found dead.
cEuthanized partridges.
Histopathology
Microscopic lesions were observed early in the infection, but their severity increased after 7 dpi (see Suppl. Table 1). The most severely affected tissues were the brain, spinal cord, and heart. In fatally infected animals, spleen, liver, kidney, large intestine, pancreas, and bursa of Fabricius also showed moderate lesions. There were no microscopic findings in the trachea, proventriculus, cecal tonsils, testicles, ovaries, adrenal gland, and skin and feather follicles.
Main microscopic findings were the presence of inflammatory infiltrates, cellular degeneration, and necrosis. While fatally infected animals showed moderate to marked lymphoid cell depletion and necrosis in lymphoid organs (characterized by karyorrhexis of lymphoid cells, accumulation of necrotic debris, and deposition of fibrin; Nos. 3, 5), this was rarely observed in the euthanized ones. In the heart, there was multifocal shrinkage and hypereosinophilia of cardiomyocytes, with occasional loss of cross striation, and pyknosis or karyorrhexis of the nucleus (Fig. 1). Mild edema and multifocal inflammatory infiltrates composed of lymphocytes, plasma cells, and macrophages were also observed (Fig. 1). The lesions were more severe in the animals that died between 7 and 10 dpi, particularly in fatally infected animals (Nos. 3, 5). Massive hepatic necrosis characterized by individualized, shrunken, rounded, hypereosinophilic hepatocytes with pyknotic or karyorrhectic nuclei was observed in the partridge found dead at 7 dpi (Fig. 2). The partridge euthanized at 7 dpi (No. 4) had multifocal random areas of hepatocyte necrosis and deposition of fibrin, as well as multifocal infiltration of small numbers of lymphocytes, plasma cells, macrophages, and heterophils, mainly in the sinusoids. Scattered Kupffer cells with a brownish pigment in the cytoplasm (likely hemosiderin) were also detected. Mild hemosiderosis was a common feature, also in the spleen. As early as 3 dpi (partridge No. 2), tubular epithelial cells in the renal cortex were hypereosinophilic, shrunken, detached from the basement membrane, and showing pyknosis and/or karyorrhexis of the nucleus. These changes were more severe in the partridge found dead at 7 dpi (No. 3), in which the tubular lumen often contained eosinophilic granular to fibrillary material (protein casts). Multifocal lymphoplasmacytic and histiocytic interstitial nephritis was also observed in this bird and in 1 of the partridges euthanized at 10 dpi (No. 6). Multifocal infiltration of small numbers of lymphocytes in the septum of the air capillaries of the lung was observed in partridge Nos. 4 and 7 (euthanized at 7 and 10 dpi, respectively). Multifocal infiltration of lymphocytes, plasma cells, and macrophages was observed in the muscularis externa of the gizzard of the birds that died at 8 dpi (No. 5) and were euthanized at 10 dpi (Nos. 6, 7) and in the lamina propria of partridge No. 7. Similar inflammatory cells were found in the muscularis externa of the duodenum and large intestine. Large numbers of heterophils were also present in the lamina propria of the duodenum of the partridges euthanized at 14 dpi (Nos. 8, 9) and the large intestine of 2 partridges euthanized at 10 and 14 dpi (Nos. 6, 8, respectively). The 2 fatally infected partridges (Nos. 3, 5) had necrosis of acinar pancreatic cells, characterized by individualized cells displaying shrinkage, hypereosinophilia, occasional loss of zymogen granules, and vacuolation of the cytoplasm, as well as pyknosis and/or karyorrhexis of the nucleus and loss of polarity. In the CNS, a nonpurulent encephalitis characterized by small to moderate numbers of lymphocytes, plasma cells, and macrophages, mainly expanding the Virchow-Robin space (perivascular cuffing), was observed from 7 dpi on. Increased numbers of glial and microglial cells (gliosis) that often formed aggregates (glial nodules) were also detected (Fig. 3; Suppl. Table 1). In the cerebrum, optic lobe, and spinal cord of fatally infected partridges (Nos. 3, 5) and 1 of the partridges euthanized at 10 dpi (No. 7), there were scattered neurons with hypereosinophilic contracted cytoplasm and pyknotic, occasionally karyorrhectic nuclei (degeneration and necrosis). Mild to moderate Purkinje cell necrosis was also observed in these animals (Fig. 4). Additionally, multifocal neurons and Purkinje cells were surrounded by low numbers of glial cells (satellitosis), mainly between 8 and 10 dpi. Another common finding that appeared as early as 3 dpi was endothelial cell swelling.
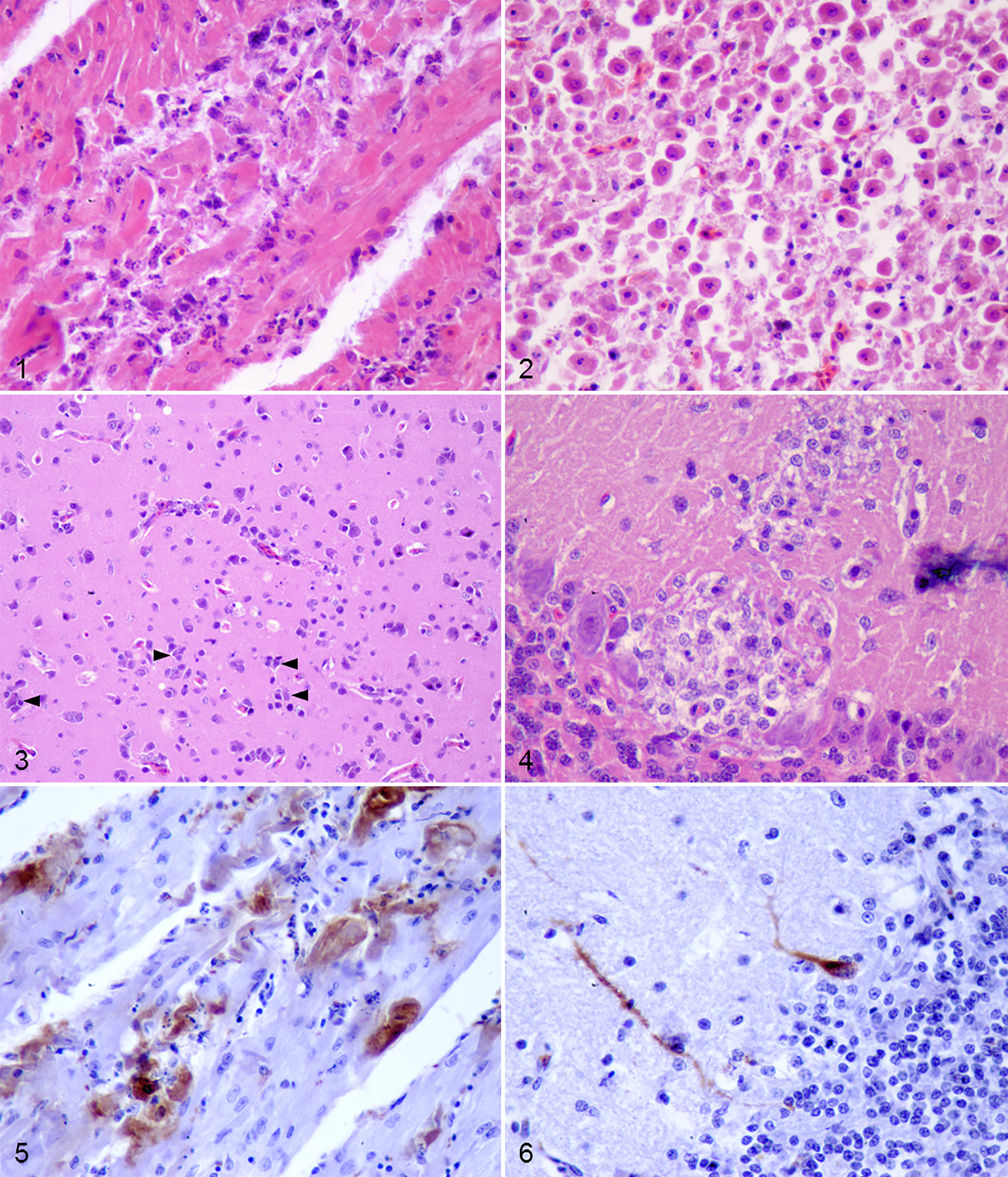
Experimental West Nile virus (WNV) infection, red-legged partridges.
Incidental findings included intestinal coccidiosis in 2 fatally WNV-infected birds (Nos. 3, 5) and crop mycosis in 1 fatally infected (No. 3) and 5 euthanized (Nos. 2, 6–9) partridges.
Immunohistochemistry
Virus Antigen Detection
Although WNV antigen detected by IHC was relatively scarce, it was widespread, especially between 7 and 10 dpi (Table 2). Positive immunostaining was observed in the cytoplasm of cardiac myofibers (Fig. 5), glomerular mesangial and tubular epithelial cells of the kidney, and acinar cells of the pancreas as early as 3 dpi (Table 2). In the spleen, WNV antigen was detected in macrophages (3 and 8 dpi) and in few smooth muscle cells of vascular walls (10 dpi). Only the 2 birds found dead at 7 and 8 dpi showed positive staining in hepatocytes and Kupffer cells and in proventricular gland epithelial cells. In 1 of the partridges euthanized at 10 dpi (No. 6), WNV antigen was detected in inflammatory cells in the lamina muscularis of the gizzard and in muscular fibers of the lamina muscularis of the large intestine. In the CNS, the 2 birds euthanized at 10 dpi showed positive staining within the cytoplasm of 1 Purkinje cell and very few dendrites, as well as in scarce glial cells in the cerebellum (Fig. 6). At 14 dpi, viral antigen was detected only in case No. 8 in 1 cardiac myofiber (Table 2).
Antigen Distribution in Tissues of Experimentally WNV-Infected Red-legged Partridges According to dpi.a
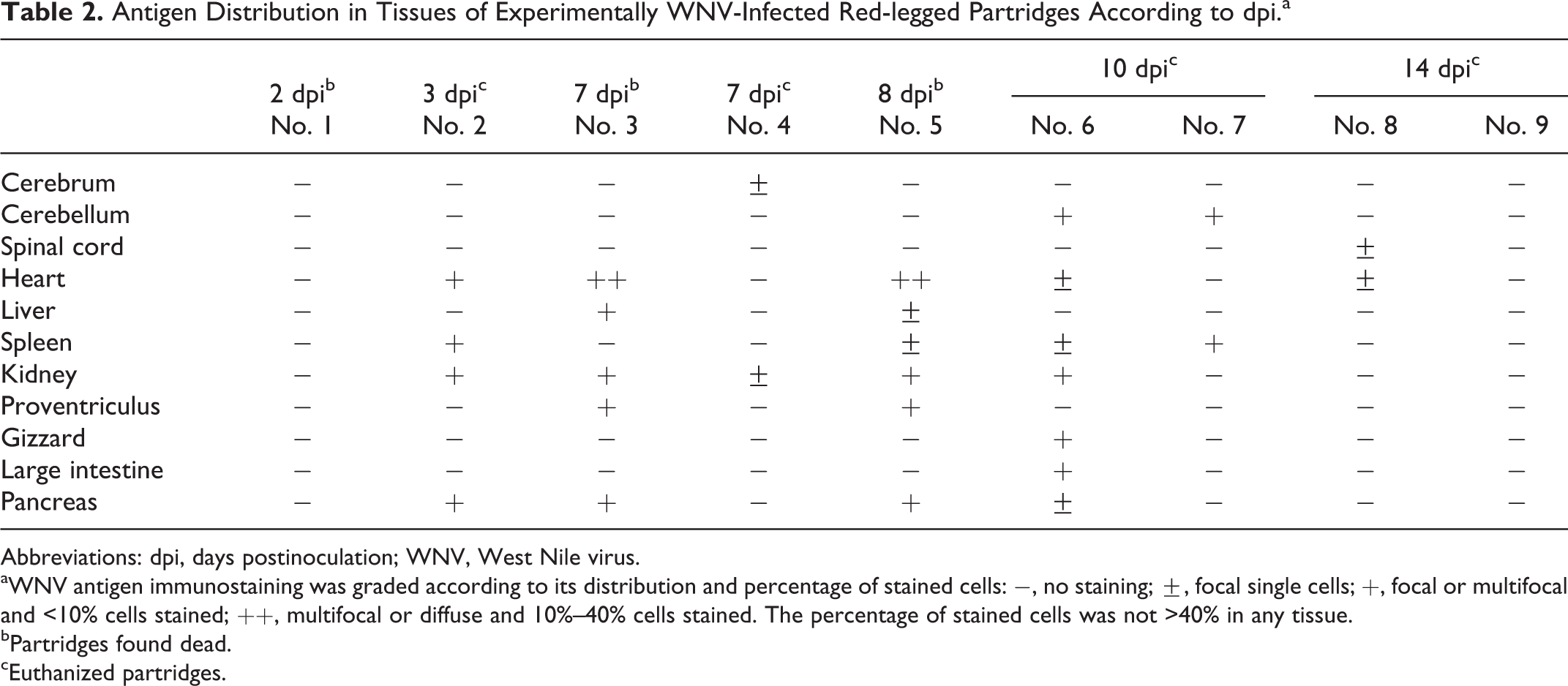
Abbreviations: dpi, days postinoculation; WNV, West Nile virus.
aWNV antigen immunostaining was graded according to its distribution and percentage of stained cells: −, no staining; ±, focal single cells; +, focal or multifocal and <10% cells stained; ++, multifocal or diffuse and 10%–40% cells stained. The percentage of stained cells was not >40% in any tissue.
bPartridges found dead.
cEuthanized partridges.
Inflammatory Cell Activation and Recruitment Into the Brain
Inflammatory cell activation and recruitment into the brain were studied only in euthanized partridges.
Activated microglial cells labeled by their expression of RCA-1 were observed as early as 3 dpi. There were changes in their morphology, distribution, and abundance during the course of the infection and among brain regions (Fig. 7). At 3 dpi, microglial cells displayed a ramified morphology and were found diffusely distributed within the parenchyma. From 7 dpi onward, microglial cells changed to an activated amoeboid morphology (large soma and short thick cellular processes). Numerous microglial/macrophage nodules were present, in some areas surrounding necrotic cells. After 10 dpi, microglia activation and macrophage infiltration slightly decreased in the cerebrum, cerebellum, and optic lobe but not in the pons and the medulla oblongata (Fig. 7).
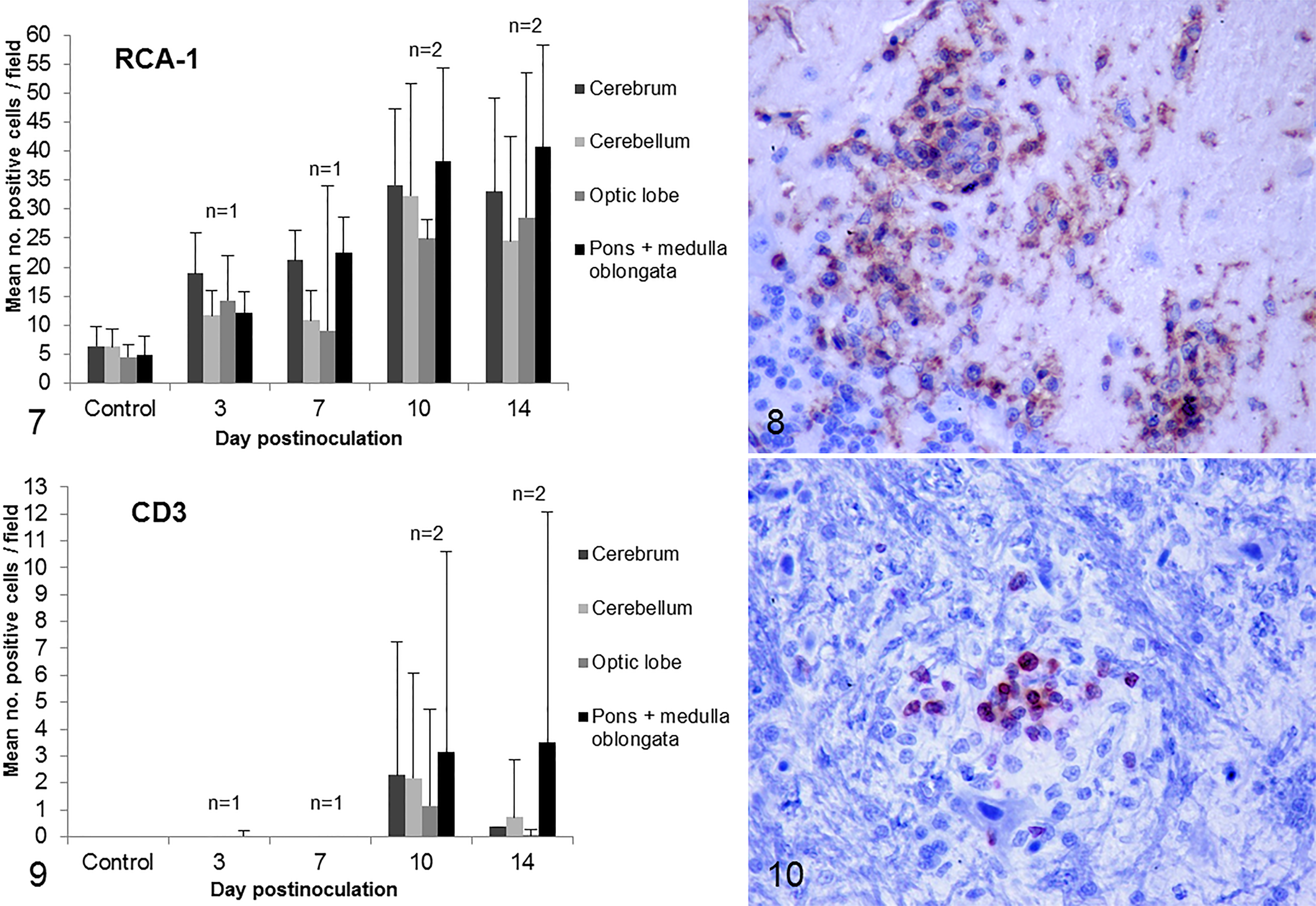
Inflammatory cell recruitment and activation in encephalitis due to experimental West Nile virus infection, red-legged partridges, brain.
In the cerebrum, RCA-1+ cells were more numerous in the peripheral pallium and intensely stained near the lateral ventricle. In the cerebellum, RCA-1+ cells were more numerous in the molecular layer (Fig. 8). In the optic lobe, microglial nodules were mainly located in the stratum griseum et fibrosum superficiale. In the pons and medulla oblongata, there were multiple microglial nodules at 14 dpi, some of them located in nuclei.
Astrocytosis (demonstrated by an increase in the number of GFAP+ cells) was mild at 3 dpi. Between 3 and 10 dpi, no significant increase in number of positive cells was observed. At 14 dpi, astrocytosis was more prominent.
In the cerebrum, multifocal aggregates of GFAP-positive astrocytes were detected in the peripheral pallium as early as 3 dpi. At this time, a mild increase in the length of cellular processes in the astrocytes located in the subpial region was also observed. Numerous star-shaped astrocytes were stained near the lateral ventricle, especially in the 2 partridges euthanized at 14 dpi. In the cerebellum, astrocyte activation was observed as early as 3 dpi in the granular layer, but it was not until 14 dpi when the number of positive cells also increased in the white matter. In the optic lobe, numerous star-shaped astrocytes stained GFAP positive in the stratum opticum (some of them surrounding blood vessels), and at 14 dpi, a mild increase in the number of positive cells was observed in the stratum album centrale. In the pons and medulla oblongata, there was a diffuse meshwork of GFAP+ cells. Multifocal aggregates of astrocytes were detected as early as 3 dpi, but these were more numerous at 14 dpi.
CD3+ T cells infiltrated the brain after 7 dpi. Changes in their distribution and abundance during the course of the infection and differences among brain regions were observed (Fig. 9). T cells were found diffusely distributed but also forming part of inflammatory nodules (surrounding necrotic cells) and perivascular infiltrates. At 14 dpi, with the exception of the pons and medulla oblongata, cellular infiltration decreased markedly (Fig. 9). Similar to RCA-1+ cells, T cells were especially numerous in the peripheral pallium of the cerebrum and in the molecular layer of the cerebellum. In the optic lobe, the stratum opticum and stratum griseum et fibrosum superficiale and, to a lesser extent, the stratum griseum and album centrale showed mild T-cell infiltration. The pons and medulla oblongata were the more markedly infiltrated regions and showed numerous nodules (Fig. 10) corresponding in many cases to microglia activation areas.
Cd79acy+ B cells were very scarce and present only in intravascular spaces. In contrast to the control, they appeared in all examined brain regions, but with no apparent increase in number over time.
Discussion
In this work, we describe the pathogenesis of the experimental infection of juvenile red-legged partridges, a native Euro-Mediterranean gallinaceous bird species, with a North American WNV strain. To our knowledge, this is one of the few experimental WNV infections carried out in a gallinaceous bird species. It is also one of the few studies in which lesions associated with WNV infection were thoroughly reviewed in a native European bird species and one of the few characterizations of the inflammatory response in the CNS of WNV-infected birds.
Most infected partridges developed multiorgan macroscopic and microscopic lesions that, as expected, were more severe and widespread in fatally infected birds and as infection progressed. The partridge that died as early as 2 dpi showed mild microscopic lesions and negative results in the WNV RT-qPCR and IHC. Given this—the fact that the partridge euthanized at 3 dpi had mild acute lesions and that even highly susceptible species (crows or jays) do not usually succumb to WNV infection until 4 to 5 dpi 23,32 —we cannot rule out other underlying causes of death for this individual.
The majority of observed lesions were similar to those described in other birds naturally and experimentally infected with WNV.
22,29,37
The most severely affected tissues were the CNS and heart. Lesions in the CNS have also been described in WNV-infected gallinaceous birds, such as chickens (
The susceptibility of the red-legged partridge to WNV infection had also been demonstrated by Sotelo et al 28 using 2 Mediterranean strains: Morocco/2003 and Spain/2007. Despite the lower dose inoculated (103 PFU) in that case, the virus intensely replicated in the brain, heart, liver, and spleen, and the mortality rate was as high as 70% for Morocco/2003; however, differences in experimental conditions and analytic methods and the low number of birds tested do not allow us to compare the virulence of these strains for the same species.
Based on the high viral inoculation dose (107 PFU), virus detection in tissues by IHC and RT-qPCR was relatively low. Nevertheless, tissues with the most prominent microscopic lesions—such as brain, heart, spleen, and, to a lesser extent, kidney—were those with positive results according to both techniques. The presence of moderate microscopic lesions in tissues such as the brain and heart of birds euthanized at 14 dpi in absence of viral antigen and viral RNA may indicate elimination of the virus by the immune response of the host. The immune response may also contribute to the severity of lesions found in these tissues, as previously suggested for other bird species. 3 Rodent models have demonstrated that despite the importance of inflammatory cells for the elimination of the virus from the CNS, an exacerbated and prolonged activation or recruitment of inflammatory cells can have detrimental effects, contributing to neuron damage. 2,31 The inflammatory response in the brain of our WNV-infected partridges was characterized by an early mild reaction of resident immune cells, although their number increased and morphology changed as infection progressed. Astrocyte activation was strongest at 14 dpi in the partridges of this study. This observation is in agreement with findings in mammals in which astrocyte activation is delayed when compared with microglial activation and frequently peaks at 14 days postinjury. 21 Astrocytes and microglia also participate in the antiflavivirus response in mice and humans. 2,12,13,20 In response to resident immune cell activation and probably to other stimuli, CD3+ T cells infiltrated the brain from 7 dpi onward, being especially numerous at 10 dpi. T lymphocytes are essential to destroy WNV-infected cells and for CNS recovery, in humans and in rodents. 2,26 Detection of CD79αacy+ B cells only in the intravascular space does allow us to draw conclusions about their role in WNV encephalitis. Studies in humans and rodents have indicated that B cells play a limited role in the encephalitis associated to WNV infection and are mainly detected in the perivascular space. 13,31 It is important to highlight that the pons and medulla oblongata were the brain regions with the most significant inflammatory reaction. Similarly, in humans and horses, these regions are the primary sites of lesions of WNV-associated encephalitis. 4,25 In fact, naturally infected birds usually show microscopic lesions in this area. 29,33,36
The red-legged partridge, a native Euro-Mediterranean gallinaceous bird species, develops lesions and moderate mortality when infected with a North American New York/1999 WNV strain. The relatively low mortality observed, despite the high virus inoculation dose used (107 PFU/individual), indicates that this virus strain is of moderate virulence for this bird species. The study of the immune cell activation and recruitment into the brain demonstrates that this avian host responds to WNV infection similarly to experimentally infected mammals. Thus, this bird species could be a suitable model to study West Nile disease and the development of encephalitis in avian hosts.
Footnotes
Acknowledgements
We are grateful to the personnel of the experimental farm of the University of Castilla–La Mancha, La Galiana, for their help during the raising of partridges. We acknowledge the help of Maria de los Angeles Risalde Moya with CD79+ B-lymphocyte immunohistochemistry.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed the following financial support for the research, authorship, and/or publication of this article. This study was supported by grant SAF2008-04232, funded by the Spanish Ministry for Science and Innovation, and grants FAU2008-00006, RTA2011-00036, and RTA2013-0013-C04, from the Instituto Nacional de Investigación y Tecnología Agraria y Alimentaria. V. Gamino (323/09) was a research fellow supported by the Regional Government of Castilla–La Mancha.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
