Abstract
Vesicular stomatitis is a viral disease primarily affecting horses and cattle when it occurs in the United States. Outbreaks in the southwestern United States occur sporadically, with initial cases typically occurring in Texas, New Mexico, or Arizona and subsequent cases occurring in a northward progression. The viruses causing vesicular stomatitis can be transmitted by direct contact of lesioned animals with other susceptible animals, but transmission is primarily through arthropod vectors. In 2012, an outbreak of vesicular stomatitis in the United States occurred that was caused by
Vesicular stomatitis (VS) viruses are members of the family
Outbreaks of VS in the southwestern United States are sporadic and typically begin in late spring or early summer and continue through late fall, with a northward progression of disease over this period. Index cases for outbreaks in the United States are usually identified in Texas, New Mexico, or Arizona. Both the temporal and the spatial characteristics of VS outbreaks suggest an association with arthropod abundance. New cases occur as warmer weather induces hatches of insects, which serve as vectors and cease when cold weather predominates, inhibiting vector hatches. In the southwestern United States, vectors are primarily in the family
Although experimental and epidemiologic evidence for the role of arthropods as biologic vectors in the transmission of VS virus is convincing, the mechanisms of virus acquisition by arthropod hosts, particularly considering that viremia in livestock has never been detected, remain unconfirmed. Experiments with domestic cattle showed transmission from infected to noninfected black flies when feeding simultaneously and in close proximity on the same animal. Uninfected flies physically separated from infected flies by up to 11 cm were able to acquire virus. 10 Such experiments are critical in explaining how VS virus may be maintained and transmitted without a viremic reservoir host. Propagation of VS outbreaks may be enhanced by movement of infected animals and spread by direct contact between infected and susceptible animals.
The clinical signs of VS occur in cattle, horses, and rarely in swine and llamas. Signs follow a typical viral incubation period of 3–7 days. An initial febrile period is typically followed by ptyalism and oral lesions in cattle and horses.1,4 Initial lesions of the oral mucosa present as raised, blanched, and occasionally detected, fluid-filled vesicles. The dorsolingual surface is often affected, but the gingival surfaces, palate, and mucocutaneous junctions may also exhibit lesions. 8 Vesicles are very short lived and leave ulcerations and erosions when ruptured. Lesions often coalesce to form large, denuded areas of oral mucosa with epithelial tags. Epithelial tags are important samples to obtain during investigations as they often lead to successful virus isolation. Vesicular and ulcerative lesions outside the oral mucosa occur on the snout of pigs, teats of cattle, and coronary bands of pigs, cattle, and horses. Lesions of the feet typically manifest as coronitis, with edema and inflammation extending from the coronary band proximally up the lower leg. Foot lesions are much less common than oral lesions in outbreaks in the southwestern United States. Crusting or scabbing lesions of the muzzle, ventral abdominal wall, prepuce, and udder may appear in affected horses. These lesions typically start as discrete, small (approximately 1-cm) erosions that quickly coalesce so that large, crusted, or scabbed areas are observed. These lesions are often located on areas of the body favored by hematophagous arthropods. VS viruses have been isolated from these crusting or scabbing lesions.
On April 30, 2012, the USDA, APHIS, Veterinary Services, National Veterinary Services Laboratories (NVSL) in Ames, Iowa, confirmed a finding of VSNJV infection on an equine property in Otero County, New Mexico. The 2 affected horses had clinical signs consistent with VS and positive VS virus serology. One of the 2 horses had a 4-fold increase in virus neutralization test titer on paired sera collected 7 days apart.
APHIS, Veterinary Services provides case definitions for the nation’s index case and index cases in subsequent states. The index case for the country will be based on compatible clinical signs and appropriate laboratory confirmation, which may include 1 or more of the following: virus isolation, 4-fold change in complement fixation test titers in paired sera collected at least 7 days apart, or a 4-fold increase in virus neutralization test titers in paired sera collected at least 7 days apart. Once the index case for the country is confirmed, index cases for additional affected states are determined by the presence of compatible lesions and 1 or more of the following: virus isolation, positive antigen-capture enzyme-linked immunosorbent assay (ELISA), 4-fold change in complement fixation test titers in paired sera collected at least 7 days apart, a 4-fold increase in virus neutralization test titers in paired sera collected at least 7 days apart, or a complement fixation test titer of greater than or equal to 1:40.
On August 1, 2012, the NVSL in Ames, Iowa, confirmed a finding of VSNJV infection on an equine property in Las Animas County, Colorado. One affected horse had clinical signs consistent with VS and a positive VS virus titer at the 1:40 dilution on the complement fixation test meeting the case definition for an index case in a subsequent state. This was the index case of VS for Colorado in 2012. In a case-positive state, subsequent cases are based on the presence of compatible clinical signs and any one or more of the following: virus isolation, positive antigen-capture ELISA, 4-fold increase in virus neutralization test titer in paired sera collected at least 7 days apart, or a complement fixation test titer of greater than or equal to 1:5.
Overall, 36 premises (34 in New Mexico and 2 in Colorado) in 12 different counties met the definition of a case (Fig. 1). The outbreak continued for approximately 8 months, with the last premises detected in October and the last premises released from quarantine on December 24, 2012 (Fig. 2). There were 277 susceptible horses on positive premises, of which 51 were confirmed positive, 49 in New Mexico and 2 in Colorado. There were few other susceptible species on positive premises including 5 sheep and 8 goats, none of which exhibited clinical signs of VS.
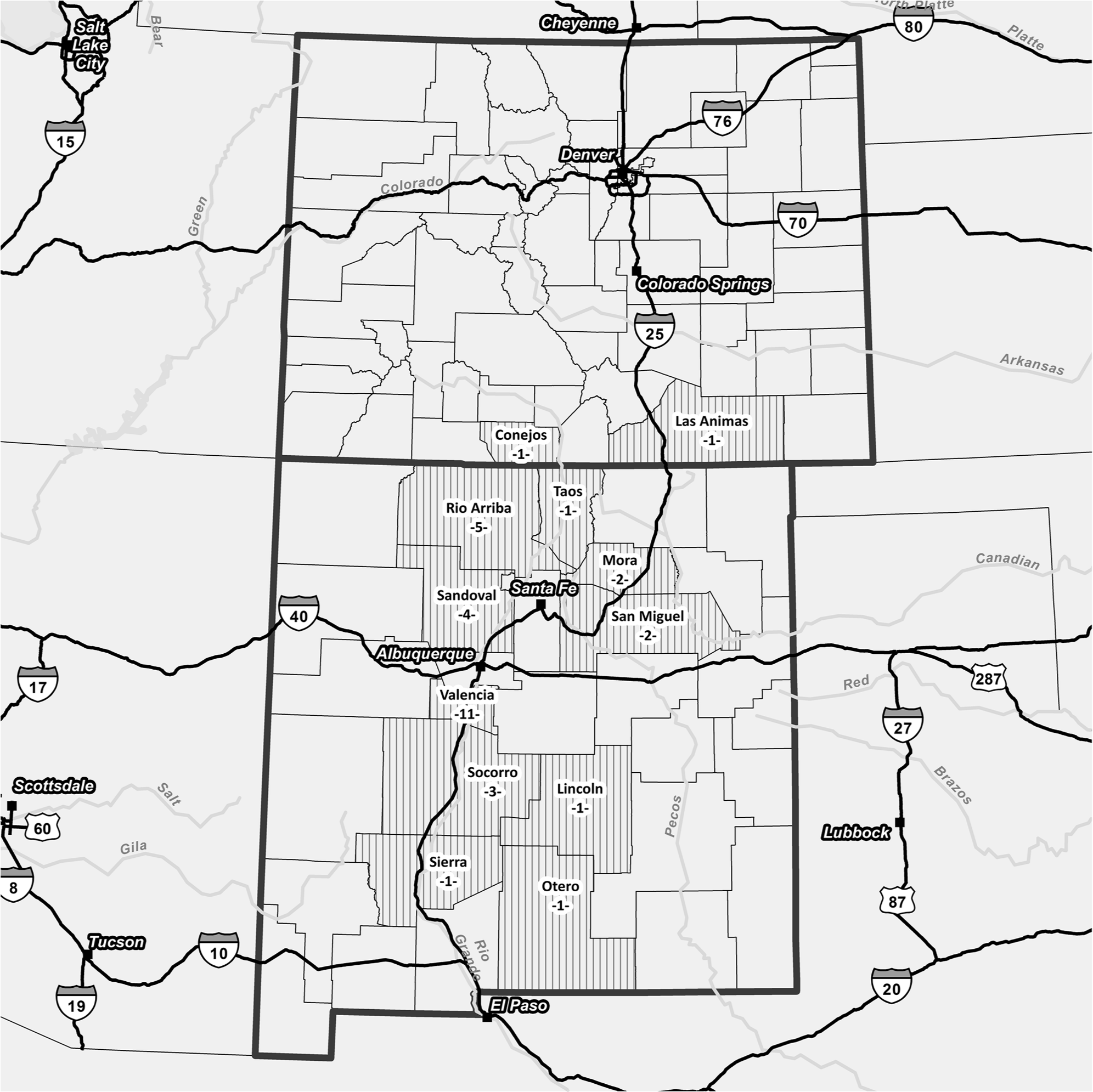
Number of positive premises by U.S. county in which they were detected.
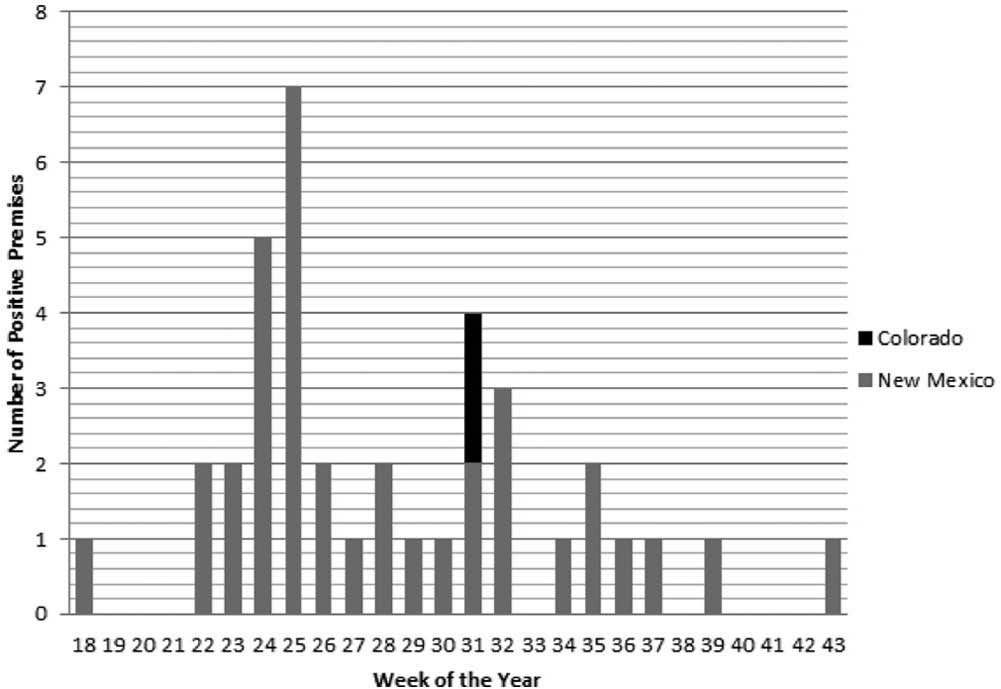
Number of positive premises confirmed by week of the year. Week 18 corresponds to April 29 through May 5, 2012.
Clinical signs observed during this outbreak were oral lesions that included vesiculation and ulceration of the oral mucosal and at the mucocutaneous junction of the mouth. While sloughing of the lingual mucosa has been reported in VS outbreaks, in 2012, severe sloughing of the lingual mucosa was observed in a relatively large number of horses.
A standardized questionnaire designed to collect data on premises characteristics, animal health management, and known risk factors for VS was administered on all affected premises (questionnaire is available from corresponding author). A summary of the frequencies of select questionnaire variables is presented in Table 1.
Frequencies of select farm characteristics, animal health management practices, and known risk factors for vesicular stomatitis collected on positive premises during the 2012 U.S. outbreak.

Virus was isolated from 8 horses located on 5 different premises during this outbreak. An RNA library was prepared for the Ion Torrent using the Ion Total RNA-Seq Library kit. a Prepared libraries were quantitated by quantitative polymerase chain reaction using the Ion Library quantitation kit. a Quantitated libraries were diluted for library amplification using the Ion One Touch and ES systems. Following enrichment, the RNA library was loaded onto an Ion 314 chip and sequenced using the Ion PGM 200 sequencing kit. a
Sequences were assembled using commercial software.
b
Reference-based assemblies were performed using reference sequences generated at NVSL. Single nucleotide polymorphisms and short insertions and deletions were evaluated as potential errors. The number of bases used for reference-based assembly was equal to the
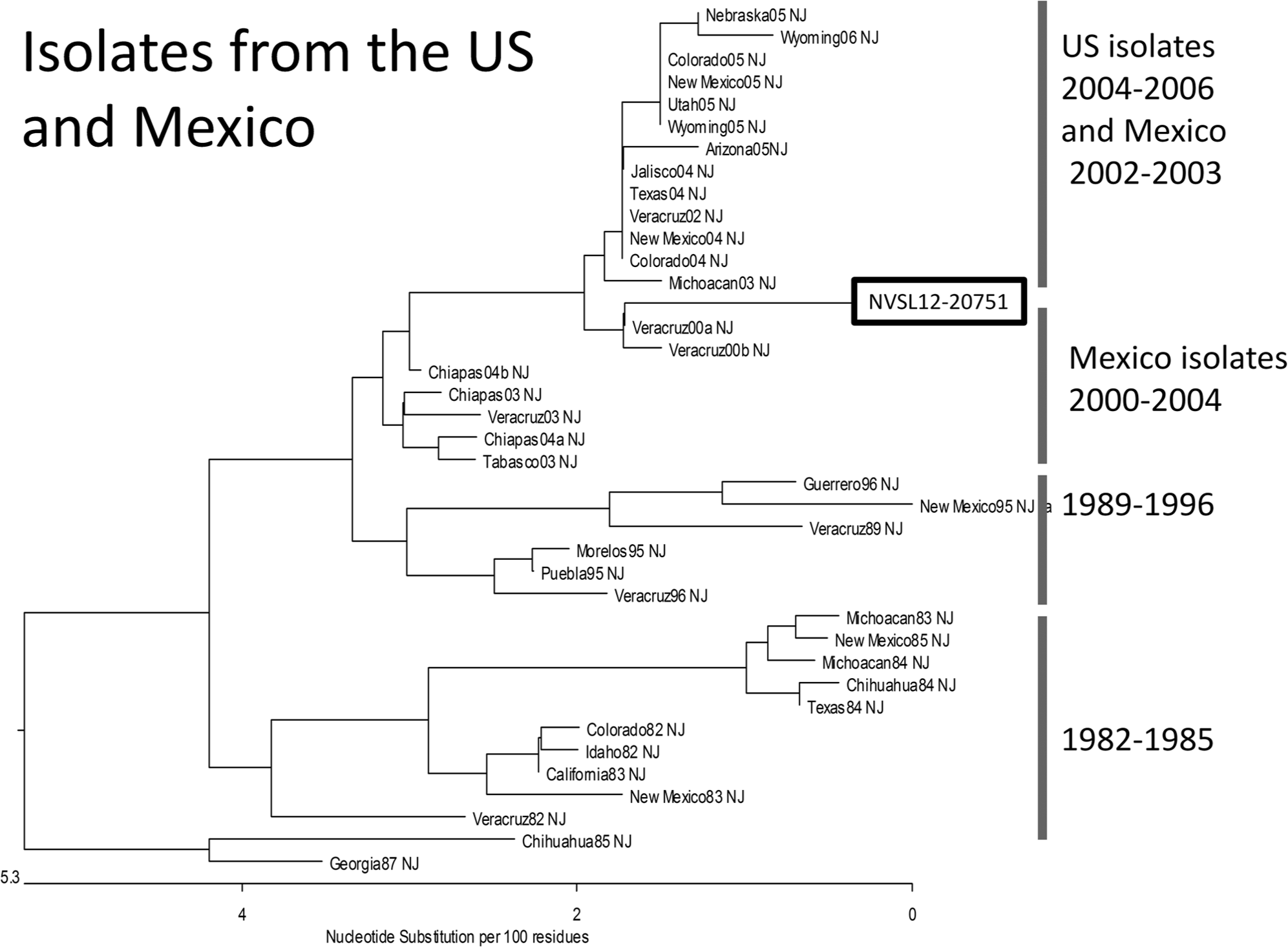
Sequences for the phosphoprotein gene of
Investigations of vesicular lesions in livestock are critically important to facilitate early detection of potentially devastating foreign animal diseases such as foot and mouth disease. In 2012, of the 586 foreign animal disease investigations conducted in the United States by designated Foreign Animal Disease Diagnosticians, 475 were for vesicular complaints with 275 occurring in equids, 152 in bovids (cattle, bison, yaks), 18 in goats, 13 in sheep, 9 in pigs, 5 in alpaca, and 3 in deer (USDA, APHIS: Summary of recent FAD investigations. Available at: http://www.aphis.usda.gov/animal_health/emergency_management/downloads/summary_fad_investigations.pdf. Accessed on March 28, 2013). This number of investigations of vesicular disease was substantially greater than investigated in the previous 3 years (2009: 178 investigations, 2010: 210 investigations, 2011: 194 investigations). This was due in part to the outbreak of VS detected in the southwestern United States, but also to an overlapping outbreak of epizootic hemorrhagic disease affecting cattle and captive cervids occurring across much of the Midwest United States during the summer and fall. Epizootic hemorrhagic disease does not occur in horses, so it can be assumed that the 275 vesicular disease investigations conducted in horses were all investigations of suspected VS. Overall, 36 of 275 investigations (13%) were determined to be positive for VS in 2012.
The 2012 VS outbreak was limited in geographic scope, with cases occurring primarily in New Mexico and with the 2 cases that occurred in Colorado located in counties that border New Mexico. Despite the sporadic occurrence of VS in the United States and associated epidemiologic investigations, the factors that determine the magnitude and distribution of these outbreaks are not well understood. The climatic and environmental factors affecting VS outbreaks have been studied previously but definitive associations have not been found. 5 Transportation of infected animals has been demonstrated in the past to transfer VS virus from one location to another. There was no epidemiological evidence of transportation of horses being a factor in the spread of disease in 2012.
The number of horses on positive premises ranged from 1 to 60 with a median value of 4. Analysis of questionnaire data collected on affected premises was consistent with previous studies investigating farm and animal-level risk factors of VS. Previous studies indicated that animals with access to a shelter or barn had a reduced risk of developing VS. Risk of developing disease was increased where animals had access to pasture. On all premises where owners reported increased insect populations and where animals were housed less than 0.25 miles from a source of running water, odds of developing VS were increased. 3 Large proportions of affected premises in 2012 either had running water (streams, creeks, irrigation canals) on the premises or were located close by them. In addition, most horses (80%) spent 12 or more hours on pasture, and most horses (81%) had access to pasture 7 days a week, making themselves more likely to be contacted by hematophagous insects. Interestingly, a vast majority of premises used chemical insecticides on horses prior to being confirmed positive for VS. While this may suggest that the products used were ineffective, applied after exposure, or misapplied, it is still recommended that owners use insecticides to reduce the risk of VS virus transmission from competent vectors.
Extensive work has been done to describe the molecular epidemiology of VS in the southwestern United States. It has been reported that in the southwest United States, both VSNJV and VSIV were found to have separate viral genetic lineages with each cycle of outbreaks and with each of the lineages also containing viruses from endemic areas of Mexico. 9 It was concluded that VS viruses were not endemic in the United States, but move northward from endemic areas of Mexico. However, overwintering of VS viruses has been demonstrated over at least 2 outbreaks occurring in 1997–1998 and 2005–2006, with phylogenetic and spatial analysis of viruses from the 2005 and 2006 outbreak years indicating that new lineages of VS virus resulted from local evolution of virus following overwintering and not from new virus introduction from Mexico.7,9 The virus isolated in 2012 was genotypically most closely related to viruses isolated in the state of Veracruz in central Mexico in 2000, supporting previous work suggesting the northward movement of virus from endemic areas of Mexico into the United States.
Footnotes
Acknowledgements
The authors would like to acknowledge the contribution of the field veterinary medical officers who conducted investigations and the following laboratory personnel: Melinda Jenkins-Moore, Annette Olson, Tami Beach, Dawn Toms, Mindy Powell, Mary Lea Killian, and Leo Koster.
a.
Life Technologies Corp., Grand Island, NY.
b.
Lasergene DNAStar SeqMan NGen 4.0, DNASTAR Inc., Madison, WI.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
