Abstract
Twenty-three feline cutaneous fibropapillomas with histologic features similar to equine sarcoids were diagnosed. They were characterized by dermal fibroblastic proliferation with overlying, often ulcerated hyperplastic epidermis. Electron microscopic findings supported the fibroblastic nature of the neoplastic cells. The 23 tumors came from 20 cats and were submitted from veterinary clinics in Wisconsin and Minnesota. These tumors occurred most commonly in young cats and were found primarily on the head, neck, and digits. Fifteen of the 17 cats for which breed was reported were domestic shorthair cats. In 11/20 cases, there was confirmed exposure to cattle. Local recurrence of the tumor following surgical excision was reported in 7 of the 18 cats for which follow-up information was available. Metastasis was not documented in any of the cases. Two of the 19 tumors tested by polymerase chain reaction (PCR) had no amplifiable DNA. The remaining 17 were positive for papillomavirus by PCR. No papillomavirus DNA was detected in three other feline skin tumors (cutaneous mast cell tumor, malignant lymphoma, and fibrosarcoma) that served as controls. This is the first report of detection of papillomavirus in feline tumors that have clinicopathologic features similar to equine sarcoids.
In veterinary medicine, the term fibropapilloma is used to refer to a benign fibroblastic proliferation with overlying acanthosis, hyperkeratosis, and a down-growth of rete ridges. 20,21 These tumors are generally attributed to papillomavirus infections and are most commonly seen in cattle and horses. 20 In cattle, at least three different bovine papillomaviruses (BPV1, BPV2, and BPV5) cause fibropapillomas. 20
Equine fibropapillomas are termed sarcoids and were first described in 1936. 6 A bovine papillomavirus etiology for equine sarcoids was first proposed in 1951. 12 Since then, a number of studies have provided evidence that bovine papillomaviruses play a role in the development of equine sarcoids. 1,13–15,19 Bovine papillomaviruses 1 and 2 have been implicated. 13,15,19
To the best of our knowledge, fibropapillomas with papillomaviral associations have only been reported in cattle, horses, donkeys, mules, cervids, and a pronghorn antelope. 8,15,17,20 Feline cutaneous fibropapillomas with histologic features similar to that of bovine fibropapilloma and equine sarcoid are occasionally seen in veterinary diagnostic services. 5,21 This report describes the microscopic and clinical features of these lesions as well as evidence for a papillomavirus etiology.
Materials and Methods
Tissue acquisition and preparation for histology
Feline cutaneous fibropapillomas were collected from biopsy specimens submitted to Marshfield Laboratories, Veterinary Division, Marshfield, Wisconsin. Twenty-three tumors from 20 cats were collected from August 1995 through December 1999. One author (FYS) collected 19 of the 23 tumors, and other pathologists at the laboratory brought 4 others to her attention. The prevalence of this lesion cannot be determined since the number of cat lesions and cat tumors examined over the collection period are not known.
Three other feline skin tumors, including a mast cell tumor, a high-grade malignant lymphoma, and a fibrosarcoma, served as controls.
Tissue sections were processed routinely, paraffin embedded, sectioned at 5 µm, and stained with hematoxylin and eosin (HE).
Electron microscopy
For ultrastructural studies, tissue from two tumors (case Nos. 12 and 20) were deparaffinized, hydrated, and postfixed in 1% osmium tetroxide, then dehydrated, cleared, and embedded in epoxy resin. One-micron sections were cut and stained with toluidine blue for preliminary light microscopic examination. Thin sections (80 to 90 nm) were cut and stained with uranyl acetate and lead citrate and were examined with a Zeiss EM10 or a Zeiss EM900 transmission electron microscope.
Polymerase chain reaction testing
Polymerase chain reaction (PCR) testing was conducted on the 19 tumors for which paraffin-embedded tissue was available and on all control tumors. To prepare DNA lysates, three 12-µm sections were cut from formalin-fixed, paraffin-embedded (FFPE) blocks. Sections were deparaffinized, and nucleic acid was released by proteinase K digestion in nonionic buffer (50 mM KCl, 10 mM Tris, pH 8, 0.1 mM EDTA, 0.5% Tween 20) (60 µg/100 µl) overnight at 55 C, followed by heating at 95 C for 10 minutes. The digest was centrifuged at 13,000 rpm for 5 minutes and the supernatant lysates were stored at −20 C.
To design the consensus papillomavirus (PV) assay, the complete nucleotide sequences for bovine, canine oral, chimp, deer, European elk, hamster, ovine, rat, two rabbits, and one human papillomavirus were aligned pairwise using the DNA Star, Inc., software, searching for conserved regions. Two 21 base pair (bp) regions of the E1 gene were identified as the most highly conserved sequences among all PV species examined and were used to design degenerate primers to give a 176 bp PCR product. The consensus primers (with IUPAC mixed base codes) were as follows: PV5-5′-TATGTDTCAAADTABYTCCAK-3′ and PV3-5′-GGBCCTCCAAAYASWGGVAAD-3′. Positive controls included FFPE equine sarcoid tissue containing bovine papillomavirus (BPV) and plasmids containing cloned BPV1, deer PV, cottontail rabbit PV (kindly provided by Carl Baker), canine oral PV (kindly provided by Bennett Jenson), and human PV type 31 (ATCC 65446, Manassas, VA).
A single-round hot start procedure with 32P-dATP incorporation was used for PCR amplification. Radioactive nucleotide incorporation assay with product separation on denaturing polyacrylamide gels was used because it offers greatly enhanced sensitivity over ordinary ethidium bromide-stained gels for PCR assays that use highly degenerate primers. 7,18 In this consensus PV assay, the upstream primers are 315-fold degenerate while the downstream primers are 210-fold degenerate. Therefore, with so many mismatched primers in the reaction, the primers complementary to the new feline PV sequences are present at very low effective concentrations and the PCR reaction is not very efficient. The 50-µl reactions contained 1× PCR Buffer II (Perkin Elmer), 3.5 mM MgCl2, 100 µM dGTP, dCTP, dTTP, 25µM dATP, 2.5 uM of each primer (Integrated DNA Technologies, Inc., Coralville, IA), 1.6 unit AmpliTaq Gold DNA Polymerase (Perkin Elmer), and 2.5 µCi 32P-dATP, specific activity 3,000 Ci/mmol (Amersham). Reactions were heated at 95 C for 10 minutes, followed by 40 cycles of 95 C for 1 minute, 42 C for 1 minute, and 72 C for 1 minute, and a final 72 C extension step for 7 minutes in a Perkin Elmer 9700 Thermal Cycler.
Products were separated on a 6% denaturing polyacrylamide gel and exposed to Kodak XAR-5 film for 24 hours at −70 C for autoradiography. A single band of 176 bp was excised from the gel, electroeluted, ethanol precipitated, and cloned into the pCR 4-TOPO® vector (Invitrogen, Carlsbad, CA) according to the manufacturer's instructions. 18 Selected colonies were sequenced using M13 primers and cycle sequencing on an ABI PRISM model 377. The nucleotide sequences obtained in forward and reverse orientations were compared to GenBank sequences by the National Library of Medicine BLAST sequence similarity search program at http://www.ncbi.nlm.nih.gov.
Follow-up information
The submitting clinicians and/or owners were contacted by telephone for follow-up information. In addition to trying to obtain missing demographic information, three basic questions were asked: 1) Did the tumor recur (if so, how often and when)? 2) Was surgery the only treatment (if not, what other treatment)? 3) Does the cat have known exposure to cattle?
Results
Histologically, the tumors were characterized by spindloid to stellate cells that had infiltrated and expanded the dermis and subcutis and surrounded and widely separated adnexa (Fig. 1). Neoplastic cells were arranged haphazardly and in vague fascicles and were separated by variable amounts of collagenous matrix. The cellularity was often higher around adnexa and adjacent to the epidermis. The neoplastic cells had oval to elongate nuclei with finely stippled chromatin, usually single small nucleoli, small amounts of eosinophilic cytoplasm, and indistinct cell margins (Fig. 2). The mitotic rate varied in different areas of the tumor, being higher in the more cellular areas. The range of mitotic index was from one to five per 10 high power fields. The overlying epidermis was mildly to moderately hyperplastic with rete ridges (Fig. 3). Multifocally, neoplastic cells were arranged perpendicularly to the dermal–epidermal junction. Ten of the tumors were ulcerated. There were moderate numbers of mast cells scattered individually and in small clusters throughout all of the tumors.
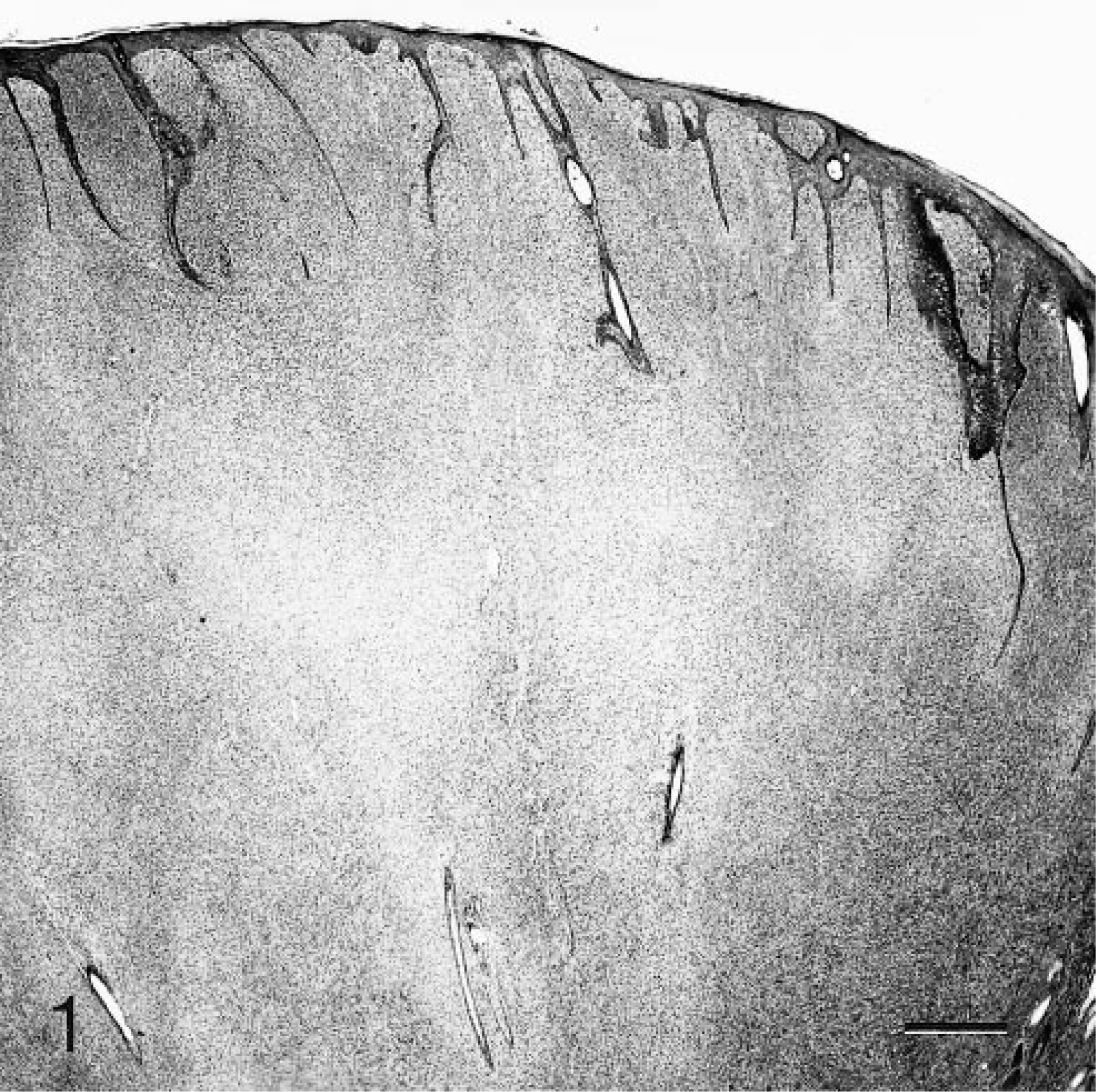
Skin; case No. 6. Feline fibropapilloma that is composed of a dermal fibroblastic proliferation that surrounds and widely separates adnexa and is covered by hyperplastic epithelium with rete ridges. HE. Bar = 250 µm.
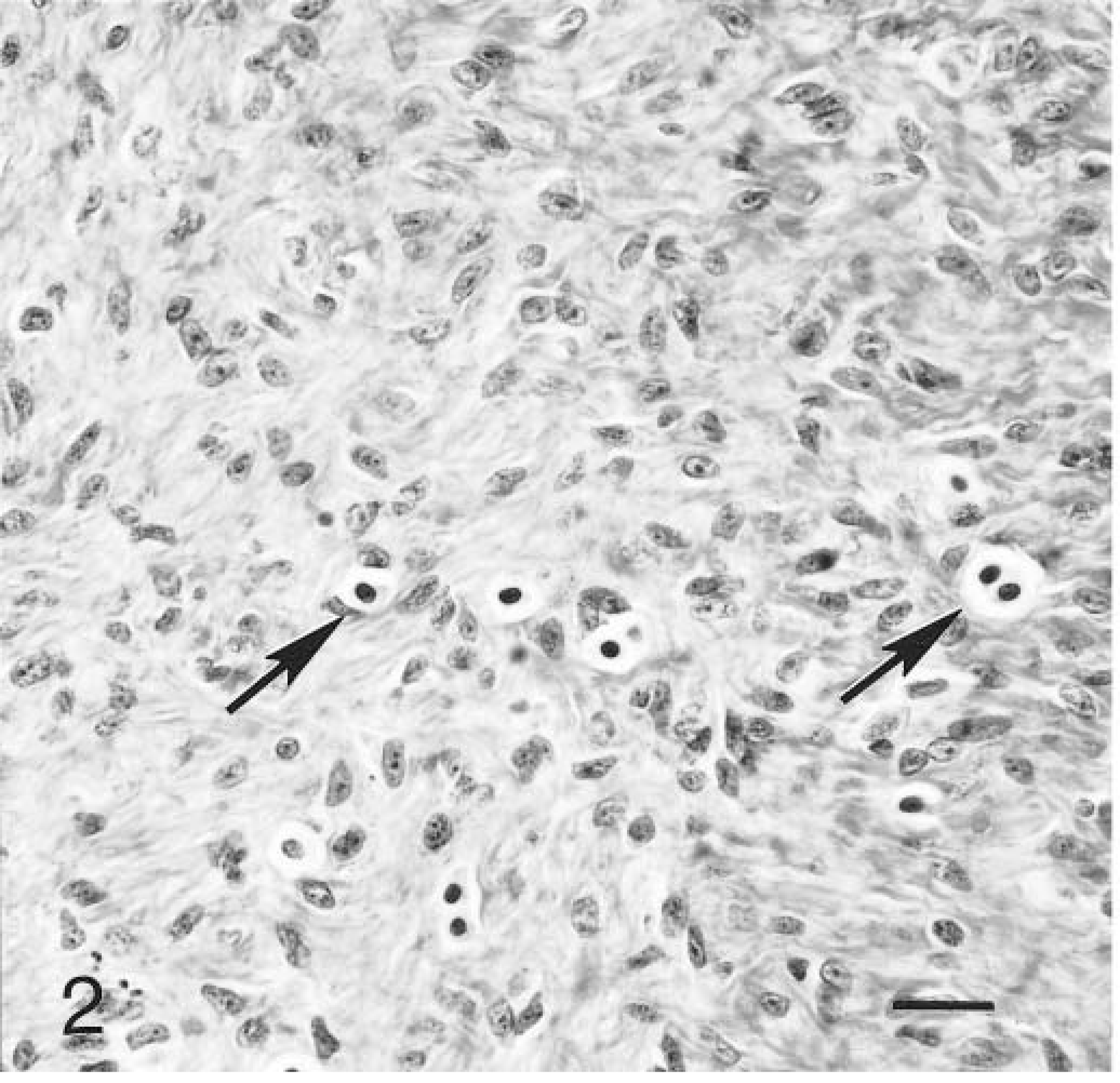
Skin; case No. 6. Fibroblastic proliferation of feline fibropapilloma with mast cells (arrows) scattered throughout. HE. Bar = 25 µm.
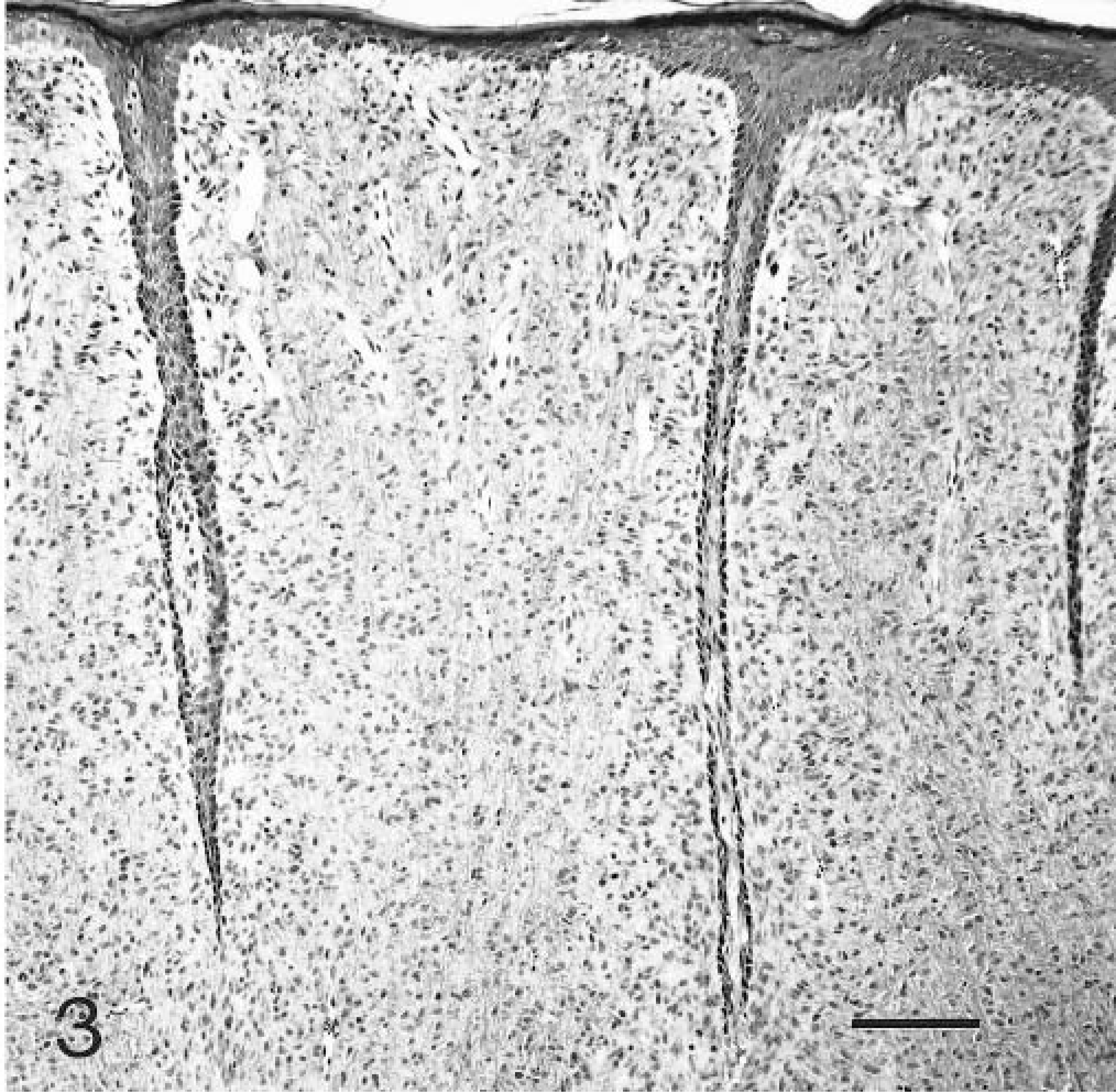
Skin; case No. 6. Superficial aspect of feline fibropapilloma with epidermal hyperplasia and long thin rete ridges. HE. Bar = 125 µm.
Ultrastructurally, neoplastic cells had elongate nuclei containing peripheralized heterochromatin and central euchromatin, few intracytoplasmic organelles comprised primarily of rough endoplasmic reticulum and mitochondria, and few cytoplasmic processes. Neoplastic cells were separated by many collagen fibrils. There were no external laminae or cellular junctions.
Polymerase chain reaction (PCR) testing results are summarized in Table 1. Of the 19 tumors examined, 2 had no amplifiable DNA. The papillomavirus PCR assay gave a product of 176 base pair (bp) in the other 17 feline cutaneous fibropapilloma samples that was identical in size to the products from the equine sarcoid tissue and from the plasmids containing BPV1, deer PV, cottontail rabbit PV, canine oral PV, and human PV type 31 (Fig. 4). The primers detected BPV1, deer PV, and cottontail rabbit PV reproducibly and canine oral PV and human PV type 31 inconsistently, reflecting the variable PCR efficiency on different runs. The band intensity from the FFPE equine sarcoid sample was similar to that obtained with many of the cat samples. The weak band found in one feline case (case No. 14) was cloned, sequenced, and compared with known PV sequences. BLAST analysis showed that the 176 bp feline sequence was most closely related to the E1 gene sequences of BPV1 and BPV2, with 75 and 64% homology, respectively (Fig. 5). The feline sequence had very limited homology with other known papillomavirus sequences, including deer PV, European elk PV, ovine PV, BPV4, and canine oral PV. No papillomavirus DNA was identified in the three control tumors, all of which had amplifiable DNA.
Papillomavirus PCR results for 23 feline cutaneous fibropapillomas.

NA = tissue not available.
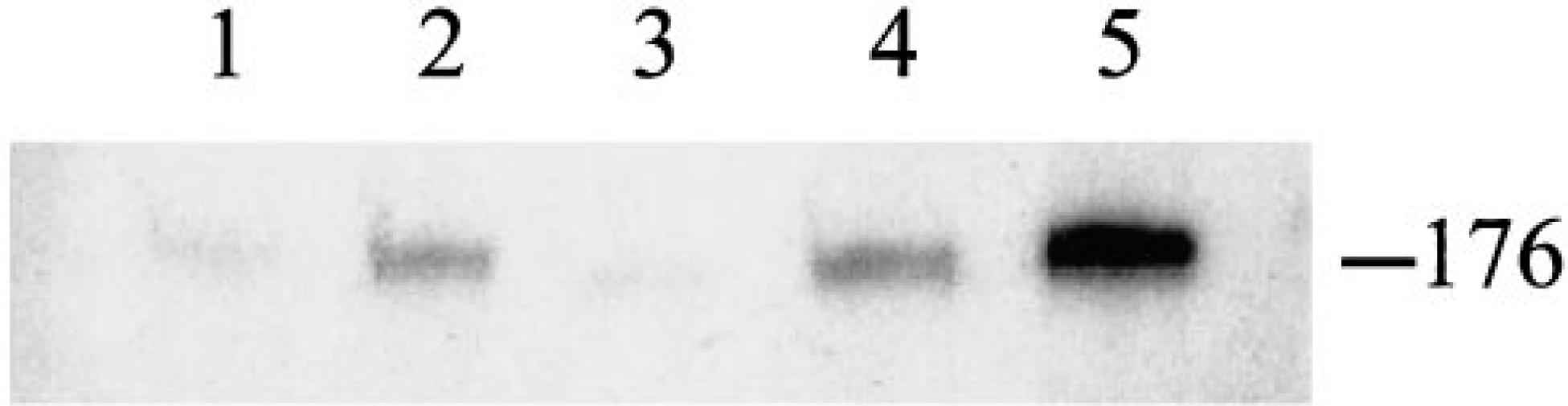
Autoradiogram of 176 bp PCR product from P-32 incorporation assay for papillomavirus with feline samples. Lanes 1 and 3 are weak positive; lanes 2, 4, and 5 are positive.
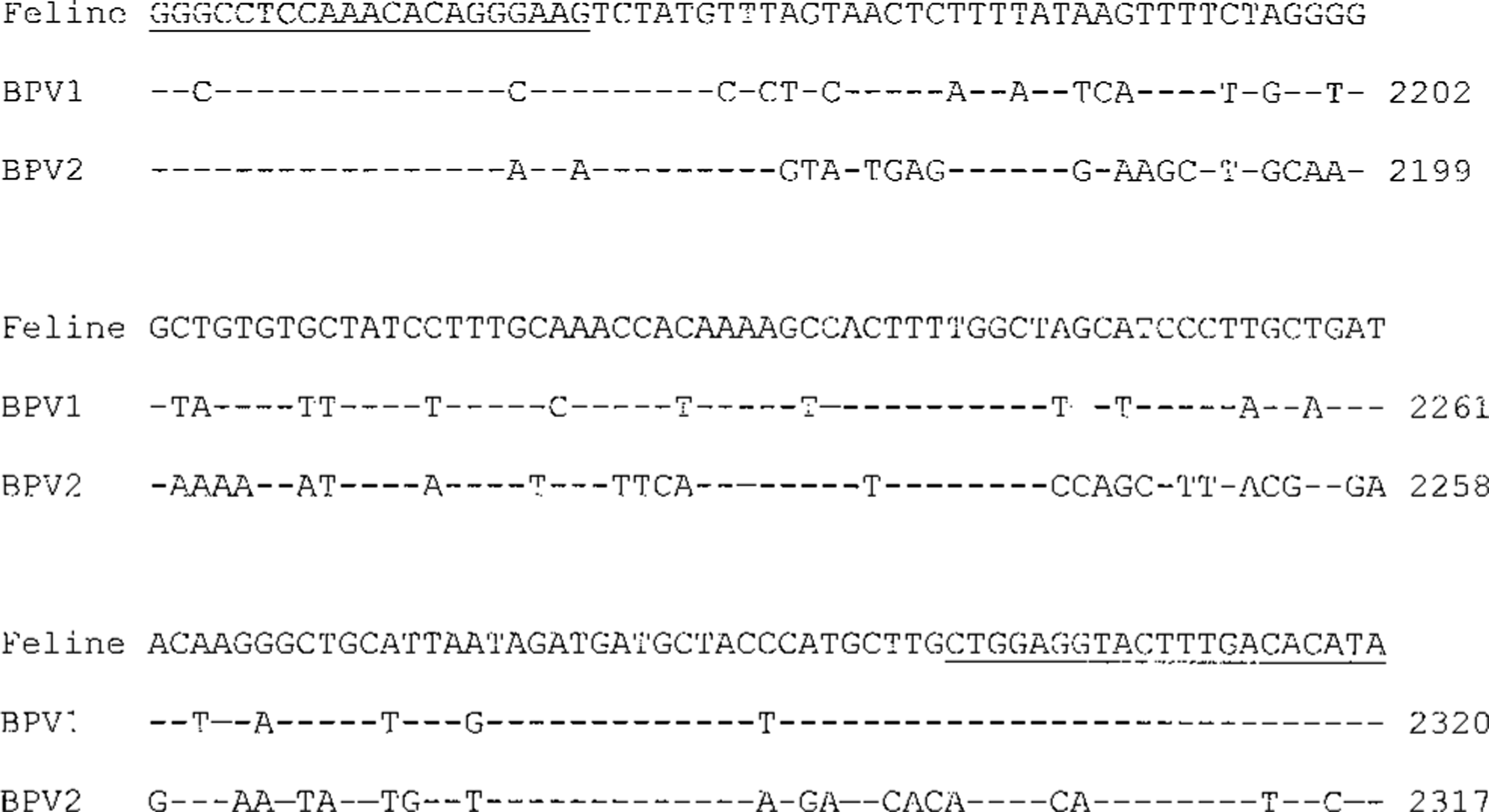
Comparison of 176 bp feline sequence to E1 gene sequences of bovine papillomaviruses type 1 (BPV1, GenBank X02346.1) and type 2 (BPV2, GenBank M20219). A dash indicates an identical nucleotide in BPV aligned with the feline sequence. The numbering at the ends refers to the nucleotide position within the complete PV genome. Underlined sequences are the binding sites for PV consensus PCR primers.
Follow-up information was available on 18 of the 20 cats and was obtained 5 months to 5 years after histologic diagnosis. Most of the clinical information is presented in Table 2. Additionally, all but one case was submitted from veterinary clinics in Wisconsin. The other came from Minnesota.
Summary of clinical information for 20 cats with cutaneous fibropapillomas.∗
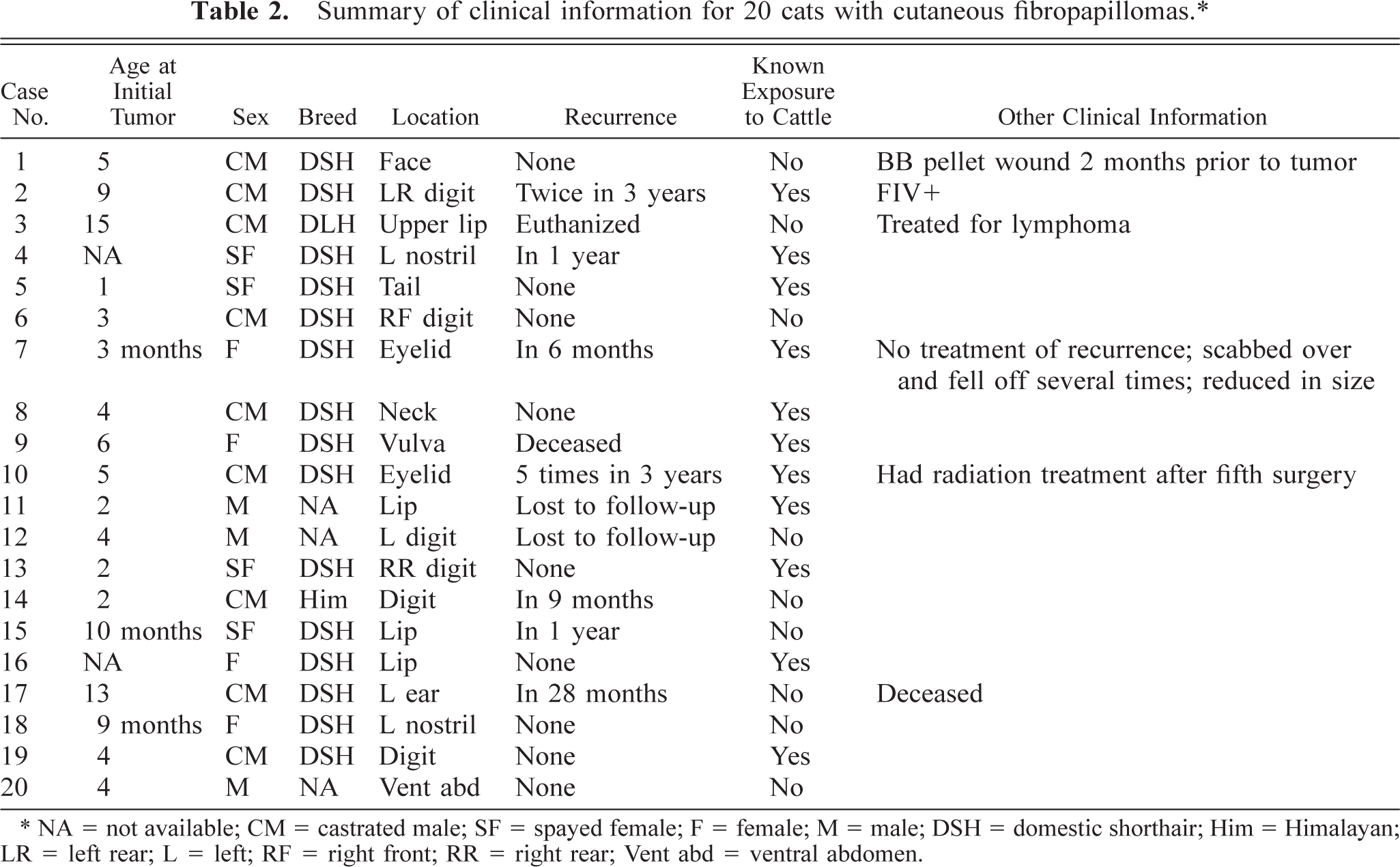
NA = not available; CM = castrated male; SF = spayed female; F = female; M = male; DSH = domestic shorthair; Him = Himalayan; LR = left rear; L = left; RF = right front; RR = right rear; Vent abd = ventral abdomen.
Discussion
Feline fibropapillomas have many similarities to equine sarcoids. The histology of feline fibropapillomas and equine sarcoids is virtually identical, characterized by a fibroblastic proliferation with overlying epithelial hyperplasia and rete ridges. 4,20,21 Electron microscopy confirmed the fibroblastic nature of the neoplastic cells. In horses, sarcoids are usually first observed in young animals between 3 and 6 years of age. 10 In this study, 12 of the 18 cats of known age were less than 5 years old. In horses, sarcoids most commonly occur on the head, neck, ventral abdomen, and limbs. 6 All but two of these cat tumors were on the head, neck, ventral abdomen, and limbs. Equine sarcoids often recur following surgical excision, 4 and recurrence was reported in 7 of the 18 cats for which follow-up information was available. Equine sarcoids do not metastasize, and metastasis was not documented in any of the feline cases.
Equine sarcoids are known to have breed predispositions and have been associated with certain major histocompatibility antigens, suggesting a genetic basis for sarcoid susceptibility. 4,9,11 In this study, 15 of the 17 cats for which breed was reported were domestic shorthair cats, but this is more likely a reflection of the demographics of the cat population sampled than a true breed predisposition.
Both tumor types have a strong association with papillomaviruses. Equine sarcoids have been associated with bovine papillomaviruses 1 and 2. 1 In all of the feline tumors for which paraffin-embedded material was available and that had amplifiable DNA, papillomavirus DNA was identified by PCR. The nucleotide sequence from the feline tumor was distinct from, but most similar to, bovine papillomavirus type 1. Eleven of the 20 cats had known exposure to cattle and all but 1 were submitted from veterinary clinics in Wisconsin, a state known for its dairy farms. The remaining nine cats did not have confirmed exposure to cattle but may have been exposed unbeknownst to the clinician or owner, i.e., prior to living with the present owner.
While sarcoids are widely recognized and there is a plethora of information concerning their clinical behavior and association with papillomaviruses, there are no peer-reviewed articles on fibropapillomas in cats. There is one published letter 5 reporting solitary dermal fibropapillomas in two cats, one in a 2-year-old cat on the lip and one in a 13-month-old cat on a hind foot. The lip fibropapilloma recurred once in 3 months, but repeat surgery was reportedly curative. The other tumor did not recur. In their textbook, Yager and Wilcock also reported 14 cases of fibropapilloma in dogs and cats collected over 3 years, but the number of feline cases and other specifics are not given. 21 While papillomavirus etiology was suspected in both reports, no direct evidence of a viral infection was provided.
The reasons for the limited information on fibropapillomas in cats are not known. These tumors may not always be recognized as fibropapillomas. If only a portion of the tumor is submitted, especially if there is no epidermis included in the biopsy, the differential diagnosis may include fibrosarcoma, fibroma, amelanotic melanocytoma, and granulation tissue. In general, fibrosarcomas are more pleomorphic than fibropapillomas and have cytological evidence of malignancy. Fibromas are well circumscribed and less cellular than fibropapillomas. Both fibrosarcomas and fibromas lack epidermal participation. The clinical history, immunohistochemistry, and electron microscopy may be used to differentiate fibropapilloma from amelanotic melanocytoma. Granulation tissue is characterized by fibrous tissue oriented perpendicular to capillaries and not by haphazardly arranged fibroblasts, as are seen in fibropapillomas. If definitive diagnosis cannot be made from an incisional biopsy, excisional biopsy is recommended.
In addition to misdiagnosis, there are other possible reasons for the apparent low incidence of feline cutaneous fibropapillomas. Cats may have a low susceptibility to papillomavirus infection and/or cellular transformation. If the papillomavirus is of bovine origin, cats may have less exposure to cattle than horses except in areas of concentrated dairy farming, such as Wisconsin. Cats that have greater contact with cattle, i.e., farm cats, may not be biopsied often. In addition, the molecular techniques for identifying viral DNA in these tumors have only recently become available. Any or all of these factors may contribute to the paucity of reports of feline fibropapillomas.
A few cases of papillomavirus-associated cutaneous squamous papillomas in cats have been reported. 2,3,16 Unlike the fibropapillomas, feline squamous papillomas are plaquelike and are characterized histologically by epidermal and follicular hyperplasia with hypergranulosis and swollen cells containing clear cytoplasm and vesicular nuclei (koilocytes). Based on immunohistochemistry, electron microscopy, and molecular tests, the papillomavirus in feline cutaneous squamous papillomas has been considered to represent Feline domesticus papillomavirus-type 1; however, results of DNA sequencing have not been reported. Squamous papillomas appear to affect immunodeficient cats. Two of the older cats with fibropapillomas had concomitant disease (lymphoma in one and feline immunodeficiency virus infection in another) that may well have caused immunosuppression. It seems logical that immunosuppression might play a role in viral oncogenesis and apply to both manifestations of cutaneous papillomavirus infection, i.e., squamous papilloma and fibropapilloma.
In summary, these feline tumors share many clinicopathologic features with equine sarcoids and have a strong association with papillomavirus, suggesting a causal relationship. Additional studies of these tumors may help in the understanding of viral oncogenesis.
Footnotes
Acknowledgements
The authors thank Ms. V. Ferris, Mr. B. Jones, and Mr. J. Jenkins of the Armed Forces Institute of Pathology and Mr. D. Stoiber of Marshfield Laboratories for technical assistance. We also thank all the clinicians and owners for their willingness to contribute to this study. Ms. S. Molter, Ms. M. Gregorich, and Ms. L. Zimmermann of Marshfield Laboratories provided invaluable logistic support. Drs. J. L. Carpenter, T. G. Taylor, M. J. Tomlinson, and D. A. Belote helped with the collection and characterization of the tumors. The opinions and assertions contained herein are the private views of the authors and are not to be construed as official or as reflecting the views of the Department of the Army or Department of Defense.
