Abstract
Solitary and multiple cutaneous and mucocutaneous masses were identified in 5 of 24 captive African lions (Panthera leo) over a 6-month-period. All masses were surgically excised, and all were histologically similar to equine and feline sarcoids. DNA was extracted from formalin-fixed, paraffin-embedded tissue. Polymerase chain reaction amplified DNA sequences that had been previously detected in feline sarcoids and clinically normal bovine skin. All lions had been fed a diet that included bovine carcasses that had not been skinned. Since the cessation of feeding bovine carcasses with cutaneous lesions, no additional skin lesions have been observed within any of the lions. Herein is described the clinical, gross, and histopathological findings of sarcoids in 5 captive lions. As the causative papillomavirus most likely has a bovine definitive host, it is hypothesized that the lions were exposed to the virus by feeding on bovine carcasses with skin still attached.
Cutaneous sarcoids (fibropapillomas) are well-recognized entities in horses and domestic cats.5,6 Equine sarcoids are thought to be caused by bovine papillomavirus (PV) 1 and 2. 13 A novel PV, designated feline sarcoid associated PV (FeSarPV), was identified in feline sarcoids in North America and New Zealand.10,14 This PV was subsequently detected in bovine fibropapillomas and clinically normal bovine skin, suggesting the PV has a bovine definitive host. 9 A sarcoid was reported in a captive mountain lion, 15 however, to the authors' knowledge, this is the first published report of sarcoids in African lions.
The use of polymerase chain reaction (PCR) revealed FeSarPV sequences within the lion sarcoids. These results suggest FeSarPV can cause disease in multiple felids, and this is the first report of FeSarPV in Australia. As this PV is likely to have a bovine reservoir host, it appears the exposure of the lions to bovine skin may have predisposed to sarcoid development.
Clinical History
Twenty-four African lions (Panthera leo) are held in a collection at a private zoo in Australia. Four separate prides are housed as follows: Pride 1 consists of 15 lions, including 1 adult vasectomised male (14 years old), 7 adult females (5 to 13 years old) and 7 subadults (5 castrated males, 2 entire females, 2.5 years old) in a 10-acre exhibit. Pride 2 consists of 3 adult male lions (5.5 years old) in a 2 acre exhibit. Pride 3 consists of 4 immature adult male lions (4 years old) in a 2-acre exhibit. Pride 4 consists of 2 immature adult lions, 1 male and 1 female (3 and 4 years old, respectively) in a 0.5-acre exhibit.
Prides 1 and 3 and prides 1 and 4 share a common boundary fence, but there is no direct contact between any individuals because there are multiple internal electrified wires along the entire boundary fence of each exhibit. Pride 2 is physically separated from the others by approximately 500 meters. All 4 prides have independent off-limits areas.
All lions were fed fresh whole meat, including skin and bone, from locally sourced cattle, horses, and deer. In the latter half of 2008, husbandry staff noticed intermittent abnormalities in bovine skins, including multiple small cutaneous nodules over the trunk and legs. Protocols were introduced in March 2009 to cease feeding bovine skin with any obvious abnormalities until cleared by veterinary inspection.
Between December 2008 and May 2009, single or multiple (range 1-5) dermal and/or mucocutaneous perioral masses were identified in 5 lions (3 lions from Pride 1 and 2 lions from Pride 3). All masses were grossly similar in appearance ranging from well-circumscribed, firm, pedunculated masses to raised nodules between 2 cm and 5 cm in diameter with intact overlying skin. All masses were surgically excised under general anaesthesia within 24 hours of first being observed. General anaesthesia was induced using 1.1 mg/kg tiletamine-zolazepam (Zoletil 100, Virbac Australia Pty Ltd, Milperra NSW, reconstituted to 200 mg/mL) and 0.02 mg/kg medetomidine (Seda Zoo, Nature Vet Pty Ltd, Glenorie NSW, 10 mg/mL) administered intramuscularly via dart and maintained with isoflurane and oxygen via endotracheal tube. Following completion of surgery the medetomidine was antagonized with intramuscular 0.1 mg/kg atipamezole (Antisedan, Pfizer Animal Health, West Ryde NSW, 5 mg/mL). Masses were fixed in 10% neutral buffered formalin immediately following excision. Individual case details and gross descriptions are summarized in Table 1 .
Signalment, gross appearance and lesion distribution of sarcoids in captive African lions.
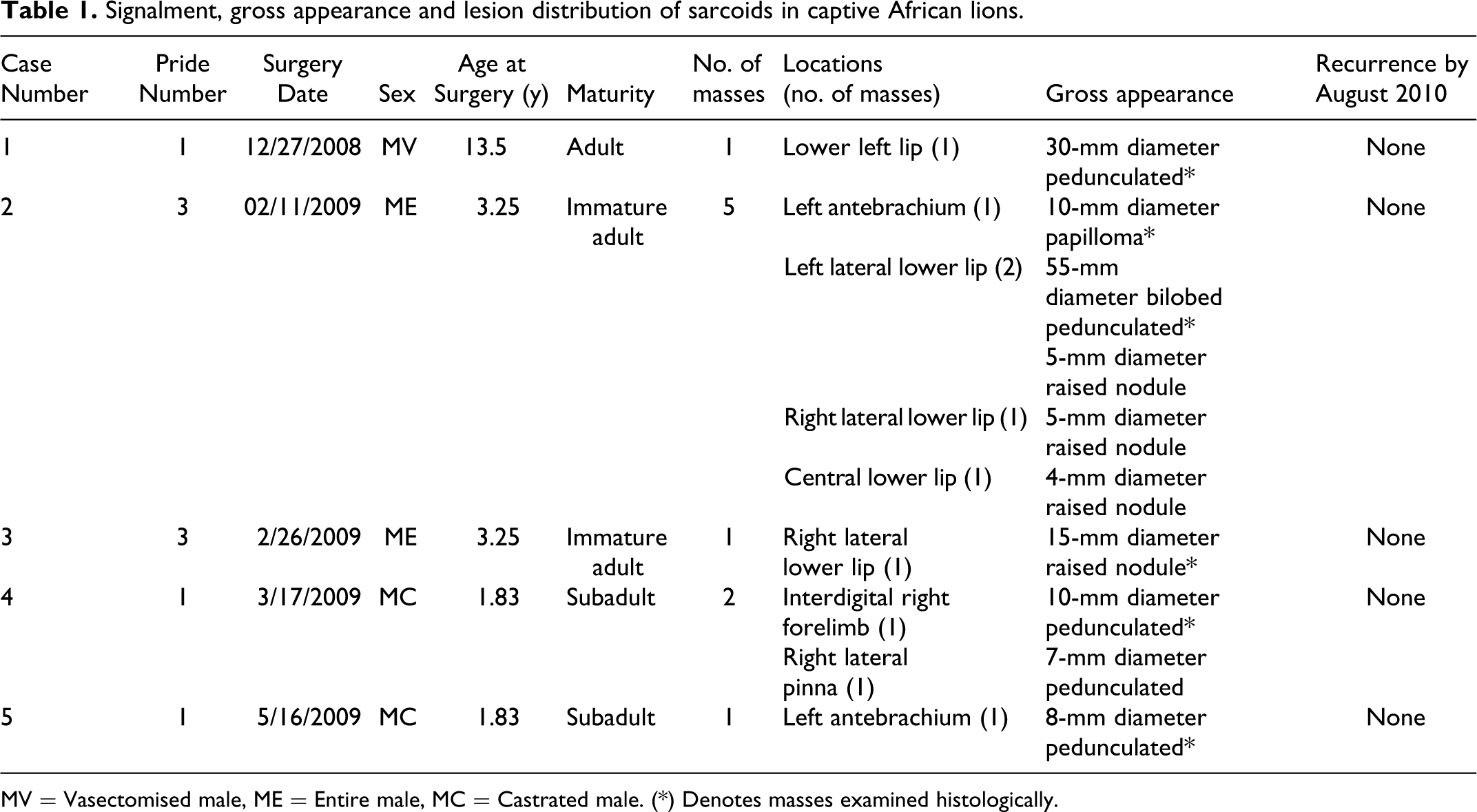
MV = Vasectomised male, ME = Entire male, MC = Castrated male. (*) Denotes masses examined histologically.
Histopathology
Representative sections of 6 excised masses representing all 5 affected lions were fixed in formalin, processed routinely and stained with haematoxylin and eosin. Histological sections were examined by one author (GO). All biopsies consisted of moderately circumscribed, non-encapsulated infiltrative neoplasms consisting of densely packed short bundles and streams underlying a moderately hyperplastic epidermis with rete formation and compact orthokeratotic hyperkeratosis. (Figure 1) Neoplastic cells were spindloid to stellate with indistinct cell borders and scant to moderate amounts of eosinophilic cytoplasm in a collagen matrix. (Figure 2) Nuclei were oval to attenuated with finely clumped chromatin and a solitary variably distinct nucleolus. There was moderate anisocytosis and anisokaryosis with a range of 1-5 mitotic figures per ten 400x fields. A diagnosis of a cutaneous sarcoid (fibropapilloma) was made in each case. Surgical margins were not determined as only a portion of each excised mass was submitted.
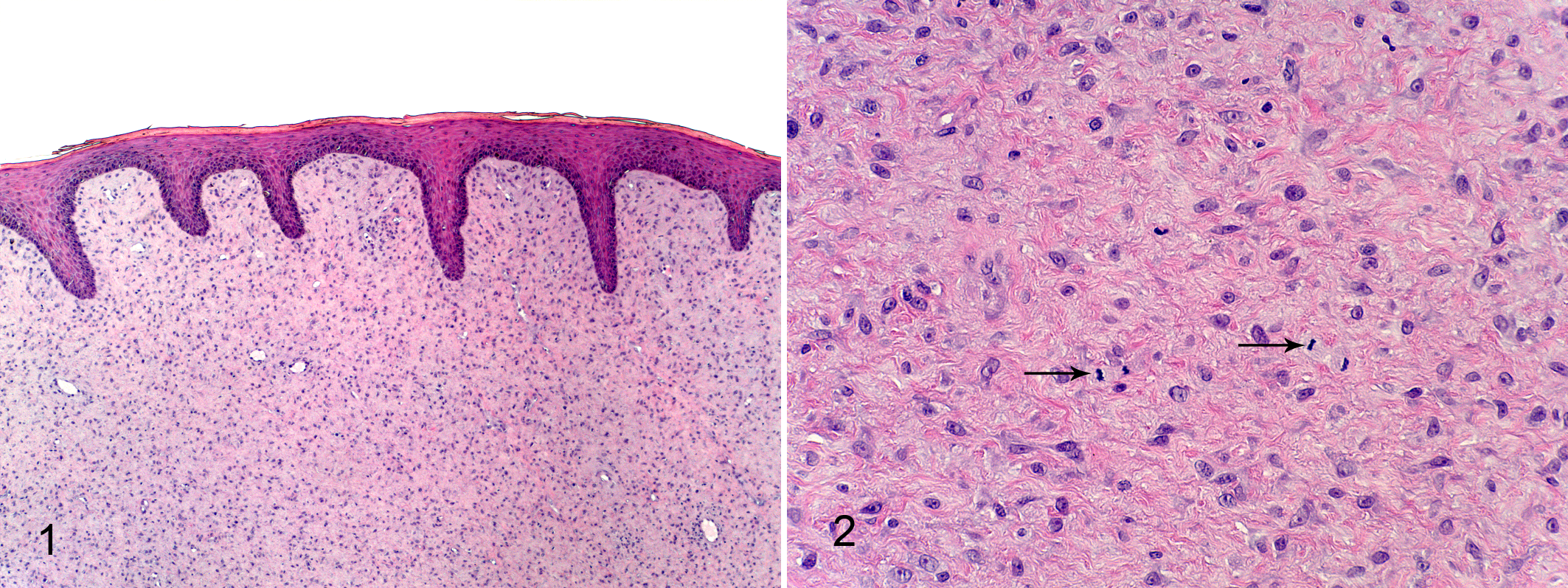
Molecular Diagnostics
Multiple thick 10-mm formalin-fixed, paraffin-embedded sections from 3 sarcoids from 3 different lions (Cases 1-3) were investigated for the presence of PV DNA. DNA was extracted from formalin-fixed, paraffin-embedded tissue as previously described. 8 Three sets of PCR primers were used to amplify sections of the PV L1 gene. FAP59/64 and MY09/11 are both degenerate primer sets that have been shown to amplify the DNA of multiple PV types from many species.1,3,4 In contrast, the jmpSA primers were designed to specifically amplify FeSarPV DNA.9,10 Amplification conditions for all 3 primer sets were as previously reported,3,4,10 and amplified fragments were visualized by electrophoresis through 1% ethidium bromide-stained agarose gels. All PCR reactions were carried out in duplicate. DNA from a feline viral plaque was used as a positive control for amplifications using both consensus primers, 9 and DNA extracted from a feline sarcoid was used as a positive control for the jmpSA primers. All reactions included a nontemplate DNA control, and DNA extracted from 2 equine sarcoids was used as additional negative controls during amplification reactions using the jmpSA primers. All amplicons were sequenced as previously described 8 and compared with known sequences using the basic local alignment search tool (http://www.ncbi.nlm.nih.gov/blast).
The FAP59/64 primers and the MY09/11 primers amplified PV DNA only from the feline viral plaque, but none of the lion sarcoids. In contrast, the jmpSA primers amplified PV DNA from all 3 lion sarcoids and the feline sarcoid, but neither of the equine sarcoids. Direct sequencing of DNA amplified by the jmpSA amplicons from 2 of the lion sarcoids revealed sequences that were 96% and 97% similar to the FeSarPV sequence that had been previously amplified from 6 feline sarcoids (GenBank accession no. FJ977616). 10 The minor variation within the sequences was attributed to errors during sequencing rather than the presence of multiple PV variants.
Discussion
Papillomavirus-associated cutaneous disease in terrestrial mammals has been reported in domestic cats, dogs, horses, rabbits, Western-barred bandicoots, Egyptian fruit bats, Natal multimammate mice, and European harvest mice. 7 In domestic cats, these include preneoplastic and neoplastic lesions such as viral plaques, 11 Bowenoid carcinoma in situ, 8 basal and squamous cell carcinoma, and feline sarcoids. 16
Sarcoids in domestic cats have recently been associated with FeSarPV. 10 The results of the present study demonstrate that African lions are also able to be infected by this PV and that this PV is associated with sarcoids in this species. In addition to feline sarcoids, FeSarPV sequences have previously been detected in bovine fibropapillomas and clinically normal bovine skin. 9 This asymptomatic infection suggests that cattle are the reservoir host of this PV. Feline sarcoids show many histological and clinical similarities to equine sarcoids, and both appear most likely to be the result of cross-species infection by a bovine PV. 7 However, in contrast to equine sarcoids in which recent studies have shown detection of BPV-1 and BPV-2 DNA in 57% of normal equine skin and dermatitis samples, indicating probable host-adapted strains,1,2 FeSarPV was not detected in 120 samples of nonsarcoid feline samples of skin. 10 However, this cannot currently be ruled out in the affected lions, as no nonlesional skin was analyzed for FeSarPV, but it seems unlikely. A previous case report identified a similar, but not identical PV from a sarcoid in a mountain lion, 15 suggesting it is possible that 2 PVs can cause sarcoids in felids, however a different PV gene (E1) was sequenced in this case.
In the present report, affected lions were fed bovine carcases that had skin attached. Some of the carcasses fed to the lions were observed to have small cutaneous masses that may have been fibropapillomas. As FeSarPV appears to have a bovine reservoir host, 9 it seems likely that the lions were infected by FeSarPV by the feeding of bovine skin. The captive mountain lion that was previously reported to have developed a sarcoid was also fed bovine carcasses, as well as deer and chickens. 15
There has been no evidence of recurrence 15 to 20 months after the sarcoids were surgically excised. This finding is in contrast to domestic cats and horses, in which sarcoids frequently recur after surgical excision.6,13 Post-surgical recurrence of sarcoids also occurred in the mountain lion after 12 months. 15 The low rate of sarcoid recurrence suggests that there may be a different host response to FeSarPV infection in lions than in domestic cats. Additionally, as the sarcoids were surgically excised soon after identification, the possibility of future spontaneous regression cannot be ruled out. After cessation of feeding bovine skin with visible cutaneous lesions, no additional sarcoids have developed in any of the lions.
The pathogenesis of feline sarcoids is uncertain, however, trauma has been suggested as a predisposing factor. 16 African lions frequently fight during feeding, commonly resulting in wounds around the oral cavity and face. Contamination of these wounds by FeSarPV from bovine skin during feeding could explain the predominant perioral mucocutaneous location of the sarcoids in these African lions. However, as multiple lions within prides 1 and 3 were affected, transmission between individuals of the same pride by direct contact, for example mutual grooming cannot be ruled out. Additionally, as male lions often feed first in both captive and wild situations, this behavior may explain why only male lions were affected in this case, but lions in Pride 3 were exclusively male. However, there is a similar sex bias with the occurrence of sarcoids in domestic cats, as young male cats in rural areas are predominantly affected. 6 In contrast, the affected mountain lion was female. 15
In conclusion, this is the first report of sarcoids and FeSarPV in African lions. In contrast to sarcoids in domestic cats and horses, none of the sarcoids have recurred after surgical excision in the lions, suggesting these lesions may have a more favorable prognosis in this species. Evidence suggests that the causative PV has a bovine reservoir host, and this report suggests that bovine skin should not be fed to captive felids until the long-term morbidity of these neoplasms is understood in this species.
Footnotes
Acknowledgements
The authors wish to acknowledge K Saint and L Howe for their technical assistance.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
