Abstract
Despite the importance of congenital viral infections in both veterinary and human medicine, only limited experimental work has been carried out to elucidate the mechanisms involved in transplacental virus infections. To further an understanding of fetal infection with pestiviruses, the distribution of bovine pestivirus in the uterine and fetal tissues of ewes in early pregnancy, following a natural route of infection, was investigated. On the 18th day of pregnancy, nine ewes were inoculated by the intranasal route with 1 X 105 50% tissue culture infective doses of an Australian isolate of noncytopathic bovine pestivirus (bovine viral diarrhea virus genotype 1). All ewes were ovariohysterectomized at approximately 100 hours postinfection. Samples from the reproductive tract and conceptus were examined histologically and tested for bovine pestivirus by nested reverse transcription–polymerase chain reaction (RT-PCR) and immunohistochemistry and for interferon-tau mRNA expression by nonnested RT-PCR. Although no histopathologic changes were observed in the maternal or fetal tissues, virus was detected in the reproductive tract of all nine ewes and in all of the conceptuses examined. At the time of surgery, only two of the nine ewes were demonstrably viremic. This study demonstrates that bovine pestivirus can spread from a natural site of infection to the ovine fetus within 4 days in the absence of maternal immunity and despite the presence of interferon expression in the reproductive tract.
Keywords
Virus infection of the nonimmune pregnant animal often results in transplacental infection of the fetus, the outcome of which is determined by the time of fetal infection, properties of the virus, and characteristics specific for the animal species. It is now well established that one of the major consequences of pestivirus infection of ruminants in early pregnancy is the birth of persistently infected offspring, an event with considerable economic implications. 17 The virus seems to be able to readily breach the placental barriers and infect embryos/fetuses. 6,17 In the acutely infected pregnant ewe, placental infection apparently occurs concurrently with fetal infection. 6 Whether fetal infection can be explained by a relatively higher susceptibility of fetal than maternal tissues remains to be elucidated. Various organs may express different levels of a putative receptor for pestivirus, 26 which could lead to preferential infection of either the female reproductive tract or the tissues of the conceptus. However, before such questions can be addressed, the precise time and site(s) of virus establishment in the female reproductive tract and fetus, following a natural route of infection, must be determined. Furthermore, the role of the local uterine immune system, notably the innate defence mechanisms such as locally produced interferons (IFNs), is still relatively unexplored in the context of pestivirus infections. One of the most, or perhaps the most, important immune modulatory cytokine in the ruminant uterine milieu is IFN-τ, a 172–amino acid protein secreted by ovine and bovine preimplantation trophoblasts. 21,22 IFN-τ also has strong antiviral activities, although this is not thought to be its primary role. 22 Other type 1 IFNs (IFN-α and -β) could also be induced locally during an acute infection 20 and exert both direct and indirect effects on virus replication and spread. 4,5
The main objectives of this study were to examine the distribution of pestivirus infection in the female reproductive tract and conceptuses within 4 days of intranasal inoculation, the possible route(s) of pestivirus transplacental infection, and any discernable effect of local IFN-τ on virus infection of the uterus and conceptus. Infection of ewes with bovine pestivirus during early pregnacy was studied. Although there are differences in the definitive synepitheliochorial placental structure between sheep and cattle, the cellular changes involved in implantation, commencing on days 16 and 20 after mating in sheep and cattle, respectively, are very similar. 25 Furthermore, the dam–conceptus interactions in sheep and cattle during early pregnancy are similar, 13 making the early pregnancy ewe a suitable model for the study of the mechanisms of transplacental infection in cattle.
Materials and Methods
Animals and virus challenge
Twenty four mature Merino ewes were initially screened for bovine pestivirus infection. Blood samples were tested for bovine pestivirus antibody and antigen using a virus neutralization test and an antigen-capture enzyme-linked immunosorbent assay respectively, 15 2 weeks before the experiments commenced. The ewes were treated to synchronize estrus and were subsequently artificially inseminated using freshly collected and diluted semen. 9 This process yielded 14 virus- and antibody-negative pregnant ewes. Nine of these ewes were inoculated intranasally with 1 ml of an Australian strain of noncytopathic (ncp) bovine pestivirus (bovine viral diarrhea virus type I, strain No. R1356 17 ) at day 18 of pregnancy. The virus inoculum was made from a single passage virus suspended in pestivirus antibody negative serum and contained 1 × 105 50% tissue culture infective diseases. Five unexposed ewes served as controls.
Tissue sampling
Ovarohysterectomies were performed at approximately 100 ± 9.7 hours after virus inoculation. Multiple samples (n = 4–6) of approximately 200–500 mg/piece from the various anatomic parts of the reproductive tract and fetal membranes were embedded in Tissue-Tek OCT Compound (Miles Inc., USA), snap-frozen in liquid nitrogen, and stored at −80 C until further processing. Each fetus was isolated and frozen in toto. Corresponding samples, including fetuses in the case of twin conceptuses, were fixed in 4% buffered formaldehyde and processed for routine histology. Care was taken not to cross-contaminate samples; separate instruments were used for the various animals and tissues, and samples were stored in separate containers.
Reverse transcription polymerase chain reaction
Nested reverse transcription polymerase chain reaction (RT-PCR) techniques were used to detect virus. RNA was extracted from frozen tissues using Total RNA-Isolation Reagent (Advanced Biotechnologies, UK) according to the manufacturer's protocol. The various tissue samples from each animal were handled separately (often on different days), and the RNA extractions and subsequent RT–PCR steps were carried out in separate precleaned biohazard cabinets. The RT-PCR was performed essentially as previously described 3 using primers designed for the 5′UTR cDNA sequence of the Osloss strain of bovine pestivirus (GenBank accession No. M96687). First-round primers were: 5′-CCATGCCCTTAGTAGGACTAG-3′ (upstream) and 5′-ATGTGCCATGTACAGCAGAGA-3′ (downstream). Nested reactions were performed in a similar manner using 0.1 µl of first-round product and the primers 5′-CAAGGAGGGTAGCAACAGTGG-3′ and 5′- CAAGGAATACAGTGGG-CCTCT-3′. Final products were visualized by ethidium bromide staining of Tris/acetate/ethylenediaminetetraactetic acid–buffered agarose gels. RNA extracted from pestivirus-infected and uninfected tissue cultures served as positive and negative controls, respectively, for the RT-PCR detection of virus.
A similar approach was used for detection of IFN-τ mRNA in the tissues, using the primers 5′-TCTGAATTCTGACGATTTCCCAGGC-3′ and 5′-CCTGTCTGCAGGACAGAAAAGACTT-3′. 18 Other IFN-τ isotype mRNAs 18,23 were also tested for but appeared not to be expressed at this relatively advanced stage of gestation 21 (data not shown). Because a known positive control for IFN-τ was not available to us, we used an IFN-α mRNA signal as a control for the RT-PCR conditions, which were similar for the two primer sets 18 (data not shown). Primers for IFN-α were 5′-GGGAAAAGTGGAGCCTAAGAT-3′ and 5′-GCCACCCTCCACCATCTCCTG-3′.
Immunohistochemistry
Immunohistochemical (IHC) detection of bovine pestivirus antigen in cryosections was carried out as described previously 1 using a mixture of monoclonal antibodies specific for the bovine pestivirus envelope protein gp53/E2 and the nonstructural protein p125/NS2-3. 15 Formaldehyde-fixed, paraffin-embedded tissues were immunolabeled for bovine pestivirus 12 using the monoclonal antibody 15C5. 8 Cytospin preparations of bovine pestivirus–infected and noninfected bovine tracheal cells functioned as positive and negative controls, respectively. Other controls comprised substitution of specific antibodies with nonreactive antibodies (specific for alphaviruses) and omission of reagents. 1
Results
Although the pestivirus challenge was successful in all nine exposed ewes, as evidenced by seroconversion within 14–28 days postchallenge (Table 1), none of the ewes showed clinical signs of infection. No overt macro- or microscopic changes were apparent in the reproductive tract of the ewes or in any of the fetuses recovered, and virus was only detected in the blood of two ewes (Table 2). Pestivirus could not be reisolated from any tissue or fluid samples by conventional isolation protocols. 15 However, bovine pestivirus was detected in most tissue and fluid samples from the reproductive tract of the virus-inoculated ewes by RT-PCR (Fig. 1), IHC (Figs. 2–5), or both methods (Table 2). No virus RNA or antigen was ever detected in any of the five control animals. Apparently only limited virus spread had taken place at the time of tissue sampling, at 100 hours postinfection, based upon the following: 1) nested PCR was required in most cases, notably the maternal tissues, to obtain a cDNA signal detectable by ethidium bromide staining and 2) most pestivirus antigen-positive cells revealed by IHC occurred individually, as scattered cells in the various tissues (Figs. 2–5). Only rarely, and mainly in the fetal membranes, were clusters of antigen-positive cells, indicative of local centrifugal spread, encountered (Figs. 2,5). Most pestivirus antigen–positive cells in the female reproductive tract resembled macrophages in morphology and location, but scattered epithelial cells of the uterine glands were occasionally positive (Fig. 2). No virus antigen was detected in vascular structures. Overall, relatively few virus antigen–positive cells were found in the maternal tissues, corroborating the RT-PCR results, in contrast to the fetal membranes, where individual antigen-positive cells were more readily detected by higher relative cell numbers and greater intensity of the immunolabeling (Fig. 3). In the fetal membranes, virus antigen was localized in cells of the amniotic membrane (Figs. 3,5), whereas in the fetus virus antigen was limited to the developing bulbus cordis (Fig. 4). In cryopreserved tissues, virus antigen was occasionally detected in binucleate cells (BNCs) of the chorionic trophectoderm (Fig. 5), but this finding could not be confirmed in the formaldehyde-fixed tissues. Only two fetuses from pestivirus-inoculated ewes were available for the latter IHC approach, and the different virus proteins detected by the two immunolabeling approaches may have contributed to the apparent discrepancy in detection of virus antigen in BNCs.
Detection of neutralizing pestivirus antibodies in ewes following bovine pestivirus challenge by the nasal route.
Reciprocal virus neutralization titer. NA = not available; ND = not determined.
Ewe died 24 hours postsurgery, but titer on the day of surgery was <2.
Summary of PCR (P) and IHC (I) results for bovine pestivirus in ewes and their fetuses.

One to three samples per tissue were tested by RT-PCR (on separate occasions). A minimum of four sections per tissue sample (three or four samples per tissue per animal) were tested by IHC. Blood and fetal fluid were tested by nestedRT-PCR only.
Both of two fetuses were positive.
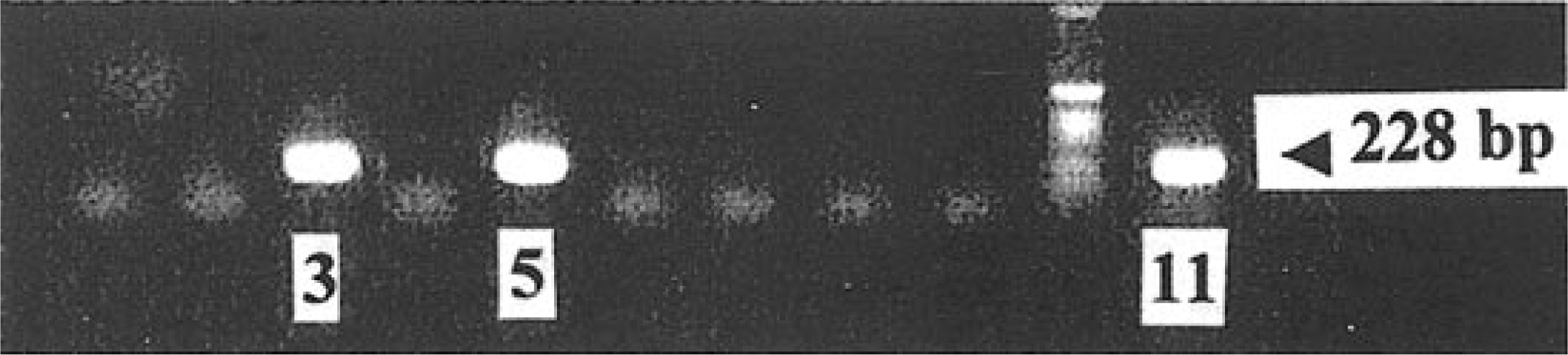
Fetal tissues; sheep. An example of nested RT-PCR results. Lanes 1, 9: negative controls (uninfected bovine cells or tissue from control animal); lanes 2–8: fetal tissues; lane 10: molecular weight marker; lane 11: positive bovine pestivirus control (challenge virus).
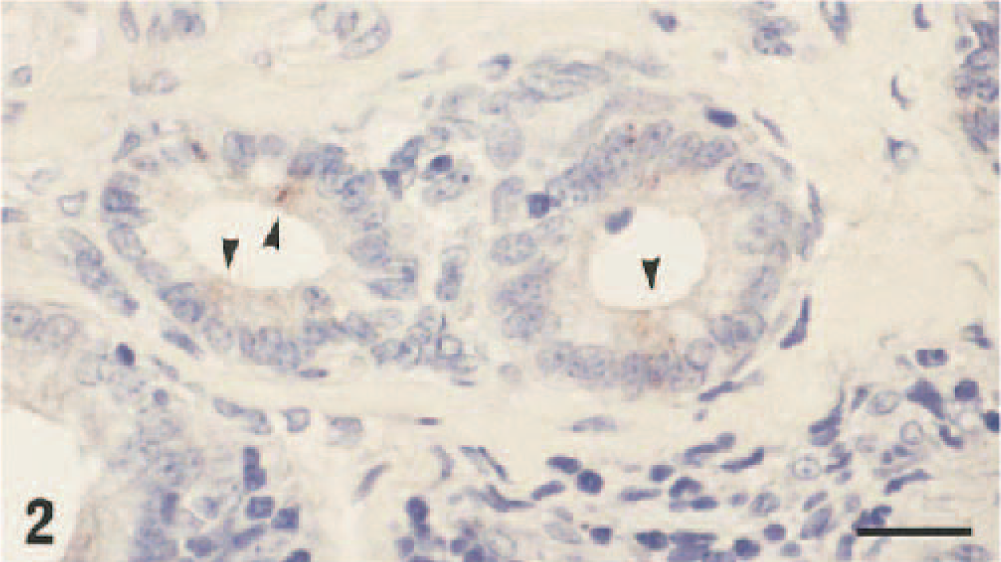
Uterine glands; ewe. Immunohistochemical detection of bovine pestivirus antigen (arrowheads) 4 days after intranasal virus challenge. Formalin-fixed section labeled with the monoclonal antibody 15C5. Hematoxylin and amino-ethyl-carbazole chromagen. Bar = 55 µm.
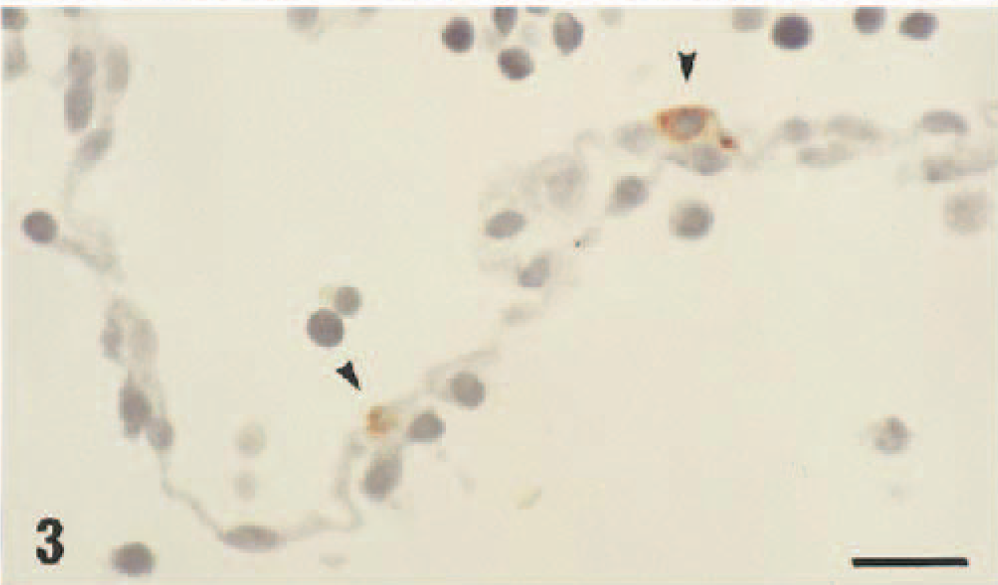
Amnion; ewe. Scattered cells (arrowheads) show intense positive labeling for bovine pestivirus antigen following reaction with the monoclonal antibody 15C5. Formaldehyde fixation Hematoxylin and amino-ethyl-carbazole chromagen. Bar = 55 µm.
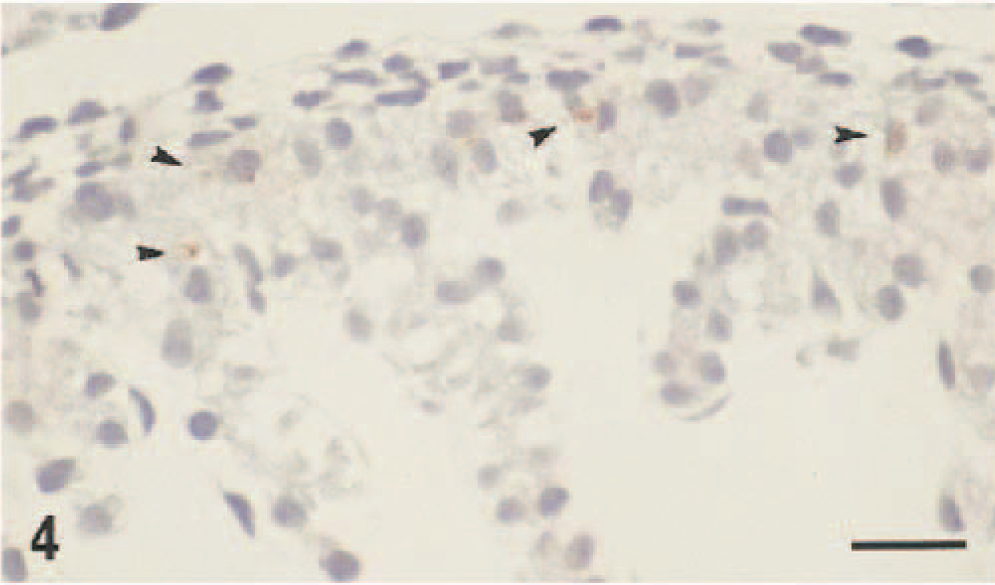
Bulbis cordis; ovine fetus. Weak labeling for bovine pestivirus antigen in scattered cells (arrowhead) 4 days after intranasal challenge of the pregnant ewe with bovine pestivirus. Formaldehyde fixation, monoclonal antibody 15C5. Hematoxylin and amino-ethyl-carbonzole chromagen. Bar = 55 µm.
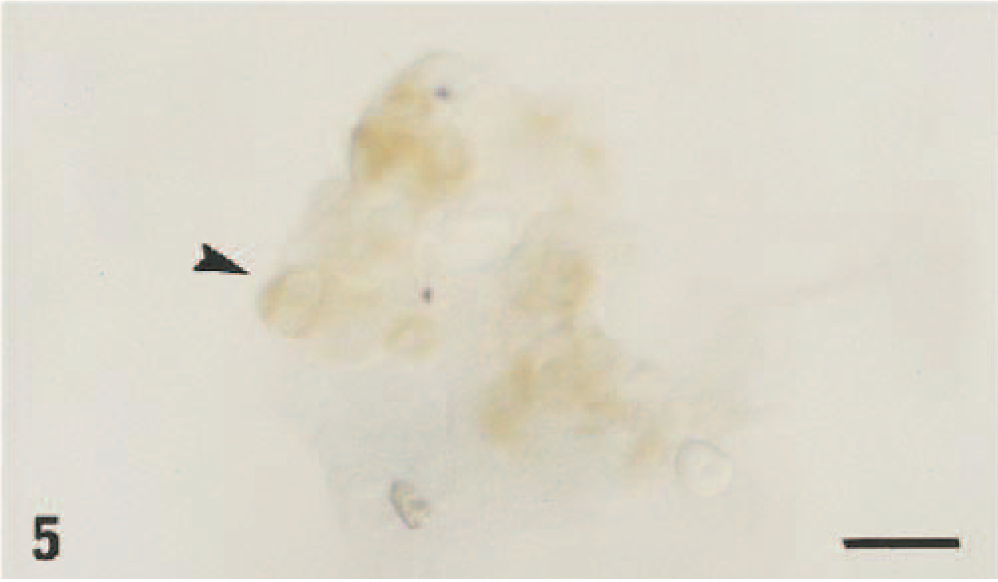
Chorionic trophectoderm; ewe. Demonstration of bovine pestivirus antigen in the binucleated cells (arrowhead) by immunohistochemistry of cryosection using a monoclonal antibody cocktail specific for the bovine pestivirus envelope protein gp53/E2 and the nonstructural protein p125/NS2-3. Methyl green and diaminobenzidine chromagen. Bar = 55 µm.
INF-τ mRNA was detected in all pregnant ewes and in most available fetuses, regardless of pestivirus infection status (Table 3), wheras no signal was present in tissues of the reproductive tract of two nonpregnant ewes included as controls (one nonexposed ewe, No. 860 in Table 3, and one virus-challenged ewe not otherwise used in this study).
Detection of INF-τ in tissues of the female reproductive tract and fetal tissues in noninfected and bovine pestivirus–challenged ewes at day 22 of gestation.
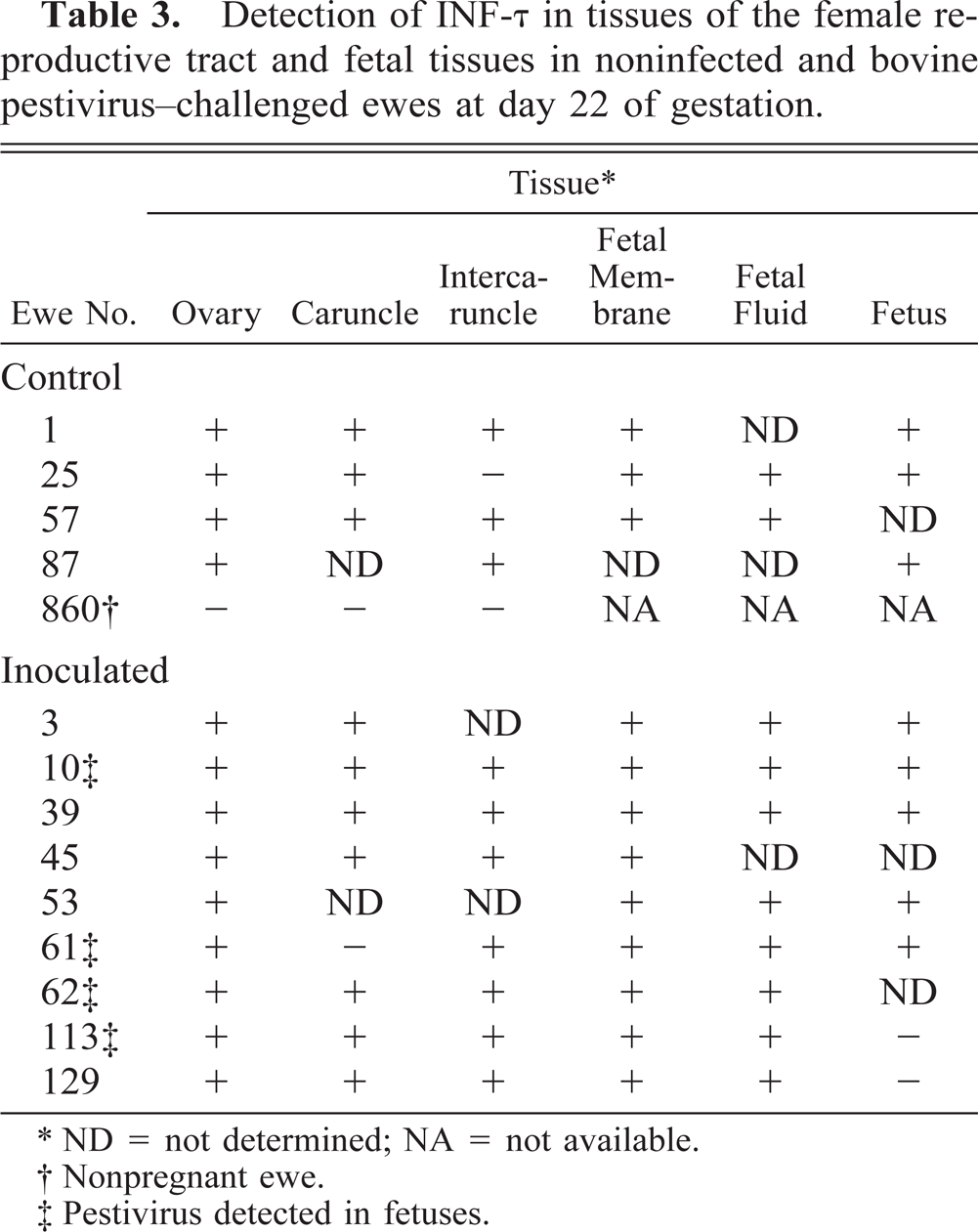
ND = not determined; NA = not available.
Nonpregnant ewe.
Pestivirus detected in fetuses.
Discussion
The pathways of virus infection of the fetus and potential protective mechanisms, notably exerted by the innate immune system, are poorly understood despite the fact that transplacental virus infections account for considerable mortality and morbidity in both animals and humans. Transplacental virus infections may, however, be relatively inefficient, and with some viruses no transplacental transmission is observed even in the case of systemic maternal infection. Thus, either the placenta acts as a barrier to fetal infection or the fetus may be “resistant” to the infection. 14 The placental barrier may be due to efficient innate immunologic surveillance mechanisms in the maternal tissues, 5,10 and fetal resistance may be exerted by antiviral agents secreted by the conceptus 21,22 or the lack of virus permissive cells at any one particular stage of fetal development. 14 However, once fetal infection is established the range of outcomes is vast, with abortion, stillbirth, congenital malformations, and growth retardation described for a variety of viruses. Foremost among those viruses studied are the pestiviruses, which can induce the full range of fetal lesions, depending on gestational stage at time of infection and probably on other virus and host factors. 2 Despite this comprehensive catalogue of pathologies, little is known about the exact mechanism(s) whereby the virus reaches the fetus and exerts adverse effects and/or establishes a persistent infection.
The present study demonstrates for the first time that bovine pestivirus can reach and infect the ruminant fetal membranes and fetus within 4 days of maternal infection by a natural route, in this case the respiratory tract, and before there is a detectable maternal viremia. The virus spread occurred despite expression of at least one isotype of IFN-τ in all the maternal tissues examined and in the absence of detectable vascular endothelial cell infection, suggesting that the virus may initially be transported intracellularly from the site of infection to susceptible target tissues. 2,14 Previous studies have also pointed to a tissue establishment phase, with cyclic appearance and disappearance of viremia. 16 In one study, 19 virus was detected only in the placentomes of ewes (pregnant for 34–45 days) at 4 days postinoculation (d.p.i.), but at 7 d.p.i. virus could be detected in most fetal tissues but only in the fetal fluid at 9 d.p.i., a sequence of virus spread distinct from that found in the present study. Methodologic differences between the two studies, including route of inoculation, inoculum titer, and virus detection protocols, may at least in part explain these different results.
In the present study, tissues were sampled at day 22 of pregnancy, at which time the structural and functional development of the caruncles and intercaruncular tissues has not been completed. 24,25 At this stage, no apparent differences exist between the trophoblastic microvillus protrusion into the intercaruncular and into caruncular tissue. 21 Whether this lack of difference plays a conducive role in the rapid spread of virus to the fetal membranes and the fetus remains unknown. An unambiguous determination of route of virus spread and a better understanding of the role of local specialized cells such as BNCs 21 and the local immune mechanisms, particularly the IFN system, 21,23 in transplacental virus infection are essential for a comprehensive elucidation of the pathogenesis of pestivirus infections. Notably, relatively more virus antigen–positive cells were detected in fetal membranes and the conceptuses than in tissue samples from the maternal reproductive tract, suggesting that 1) cells of the fetal tissues are more readily infected, or 2) these cells better support virus replication, or 3) cells of the female reproductive tract are more sensitive to the antiviral effects of IFNs and other cytokines than are cells of the conceptus and fetal membranes. 4,23 The possibility that the special milieu afforded by the differentiating cells in the fetus may play an important role in the selection of virus variants 7 capable of establishing a persistent infection demands attention. Furthermore, the rapidity with which the virus reached the fetus in the face of local IFN expression suggests that only well-developed preexposure virus-specific maternal immunity may have a chance of preventing fetal infection. In the absence of such protection, the virus will have reached the conceptus and established infection in an immune-privileged site long before the maternal systemic antiviral immune response even becomes measurable. 6
Footnotes
Acknowledgements
We thank Dr. E. Dubovi for the generous gift of antibody and Mr. D. Baker, Mrs. A. Higgins (University of Queensland) and Mr. R. Davis (Elizabeth MacArthur Agricultural Institute) for expert technical support. Funding for this study was received from the Adam Gillespie Memorial Scholarship (Australia), Ubon University (Thailand), the University of Queensland, and the Australian Research Council Small Grants scheme.
