Abstract
Zygomycosis is an important granulomatous disease that affects humans and animals, particularly sheep in tropical regions. Rhinofacial and nasopharyngeal zygomycosis were described in sheep in association with Conidiobolus spp. The present study characterized 5 samples of Conidiobolus isolated from 3 herds with clinical disease in Mato Grosso State, Brazil. The clinical and pathological findings were similar to nasopharyngeal zygomycosis. Based on morphological features, isolates were classified as Conidiobolus spp., and molecular phylogenetic analyses based on 18S ribosomal DNA grouped all isolates in a Conidiobolus lamprauges cluster. The current report describes the molecular characterization of ovine nasopharyngeal zygomycosis associated with C. lamprauges.
Conidiobolomycosis (class Zygomycetes, order Entomophthorales) is a zygomycosis caused by fungi that affect humans and animals. Conidiobolus spp. were observed to infect insects, 3 horses, 10 dogs, 1 deer, 14,22 and sheep.2,4,16,20,21 The main Conidiobolus spp. involved in illness and death in humans and animals are Conidiobolus coronatus, Conidiobolus incongruus, and Conidiobolus lamprauges. These species can be found in the soil, decaying vegetation, and insects of tropical and subtropical regions. 4 The majority of these mycotic infections are limited to tropical areas, particularly the African rain forest, 9 but cases were reported worldwide. 5,15,17,19
In Brazil, conidiobolomycosis is common in sheep in the states of Piauí, 21 Paraíba, 20 and Mato Grosso, 2 and cause major economic losses from increased medical costs for the treatment of infected animals and from the loss of animals by late diagnosis. Among the most common symptoms are serous or mucohemorrhagic nasal discharge and dyspnea with noisy and difficult respiration, anorexia, and enlargement of the anterior or posterior nasal cavity. 4,12,16 Mycotic infection may occur through the inhalation of environmental spores that lodge in the nasal cavity or from minor trauma, such as an insect bite. 9 Virulence factors described for these pathogens during the infection are largely unknown; most studies involved insects. 8,24
Disease diagnosis is based on isolation of the agent and the presence of typical clinical and pathological findings, 19 however, mycological culture is slow and may be associated with false-negative test results. Therefore, definitive diagnosis is usually obtained via molecular techniques such as polymerase chain reaction (PCR) assays, which can amplify fungi-specific and highly conserved sequences of multicopy genes (e.g., the 18S ribosomal DNA [rDNA] gene). This technique is clinically useful for the detection of fungal infections in animals 14 and humans. 7,11
In spite of the high frequency of cases and the isolation of Conidiobolus spp. in tropical regions, no molecular data about these genera were determined. 2,4,21,22 The current study was done to provide molecular characterization of ovine zygomycosis associated with Conidiobolus lamprauges in Mato Grosso State, Brazil.
In the current study, 9 sheep with suspected conidiobolomycosis were necropsied at the Veterinary Hospital of the Federal University of Mato Grosso. All sheep had a granulomatous reaction in the nasopharyngeal region that extended to the turbinates, nasal sinuses, orbit, and cribriform plate, with frequent invasion of the brain. Affected areas were characterized by the proliferation of fibrous tissue and infiltrates of lymphocytes, whereas epithelioid macrophages and multinucleated giant cells surrounded areas of necrosis that contained hyphae associated with Splendore–Hoeppli material. Hyphae were 8–30-μm long, rarely septate or ramified, irregular in shape, with a black contoured wall. A bulbous dilatation was sometimes present in the extremities of the organism as revealed by Grocott methenamine silver staining.
Fragments of the nasal lesions were placed on Sabouraud dextrose agar (SDA), with 0.05 g/l of chloramphenicol. These fungal cultures were incubated at 30°C for 7 days and checked daily for growth. The suspected colonies were picked and streaked on new SDA until completely isolated. The genomic DNA of isolated colonies was extracted as described previously. 6 The PCR assay was done by using universal fungal primers to the 18S rDNA gene (forward primer: 5'-ATTGGAGGGCAAGTCTGGTG-3'; reverse primer: 5'-CCGATCCCTAGTCGGCATAG-3') as described previously. 11 The PCR conditions were 10 ng DNA, 50 mM MgCl2, 50 mM of 10x Taq buffer with 50 mM KCl, 1 mM of each deoxynucleotide triphosphate, 20 pmol of each primer, and 1 U of Taq DNA polymerase in a reaction volume of 25 μl. The temperature included an initial denaturing step of 4 min at 94°C; 40 cycles of 30 sec at 94°C for DNA denaturation, 60 sec at 56°C for primer annealing, and 90 sec at 72°C for primer extension; then a final extension of 5 min at 72°C. The PCR product was analyzed on an ethidium bromide–stained 1.0% agarose gel. Amplified products were purified from the gel by using a commercial gel extraction kit, a followed by nucleic acid sequencing. Nucleotide sequences were submitted to GenBank by using BLAST (http://www.ncbi.nlm.nih.gov/blast/Blast.cgi) to search for probable fungal species identification.
Phylogenetic analyses were performed on the 18S rDNA gene to characterize species of Conidiobolus. Multiple sequence alignment was done with C. coronatus (EF392543, AF113417, AF113418, 849042), C. lamprauges (AF113420), and C. incongruus (AF113419) by using ClustalW. 13 A phylogenetic tree was constructed based on the neighbor joining method by using Saccharomyces cerevisiae as an outgroup. 23
Five of 9 clinical samples were positive for mycological isolation on SDA, and all colonies were morphologically classified as Conidiobolus spp. Three samples were obtained from the same property located in the municipality of Nobres, 1 sample from the municipality of Alto Paraguai, and 1 sample from Poconé, Mato Grosso State, Brazil. The macroscopic appearance of the fungus was glabrous, flat, and cream colored, with many small satellite colonies. The colonies grew after 48 hr of incubation, and by the fifth day, covered the whole 100-mm Petri dish. On microscopic examination, the fungus was characterized by wide, ribbon-type, coenocytic hyphae with irregular ramifications. The conidia were globose to obovoid, with a rounded to apiculate basal papilla and thin walls.
After PCR, an amplicon of approximately 540 bp was generated. Double-strand sequencing of the PCR amplicon confirmed the presence of C. lamprauges. The obtained sequences had 97% identity to the 18S rDNA gene of C. lamprauges (GenBank accession nos. GQ221848, GQ221849, GQ21749).
Conidiobolus spp. are important pathogens that cause ovine zygomycosis in tropical areas. 4,16 The northeast 20,21 (Piauí and Paraíba States) and southern (F. Furlan, Santa Catarina State University, personal communication, 2007) regions of Brazil have historically had cases of ovine conidiobolomycosis, but this disease has only recently been reported in the central western region, in the state of Mato Grosso. 2 Meanwhile, in the state of Paraíba, only genera of Conidiobolus (specifically C. coronatus) were described in cases 20 of ovine conidiobolomycosis based on microbiological and histopathological findings without molecular analysis.
Conidiobolus spp. are often difficult to identify microscopically and histologically because of the similarity between the classes of zygomycetes. 11 The current study identified Conidiobolus spp. through microbiological diagnosis; however, PCR and molecular phylogenetic analyses based on the 18S rDNA gene indicated that all isolates belonged to C. lamprauges (Fig. 1).
Conidiobolus coronatus and C. incongruus are frequently isolated from nasopharyngeal lesions in ovine zygomycosis. Conidiobolus lamprauges has only been associated with equine disease, but, in the current study, this pathogen was identified in ovine zygomycosis. According to a previous study, 2 conidiobolomycosis occurs between January and June in Mato Grosso during periods with high rates of rainfall, increased temperature, and high humidity. Large amounts of decomposing plant material may contribute to the growth of the fungus. 18
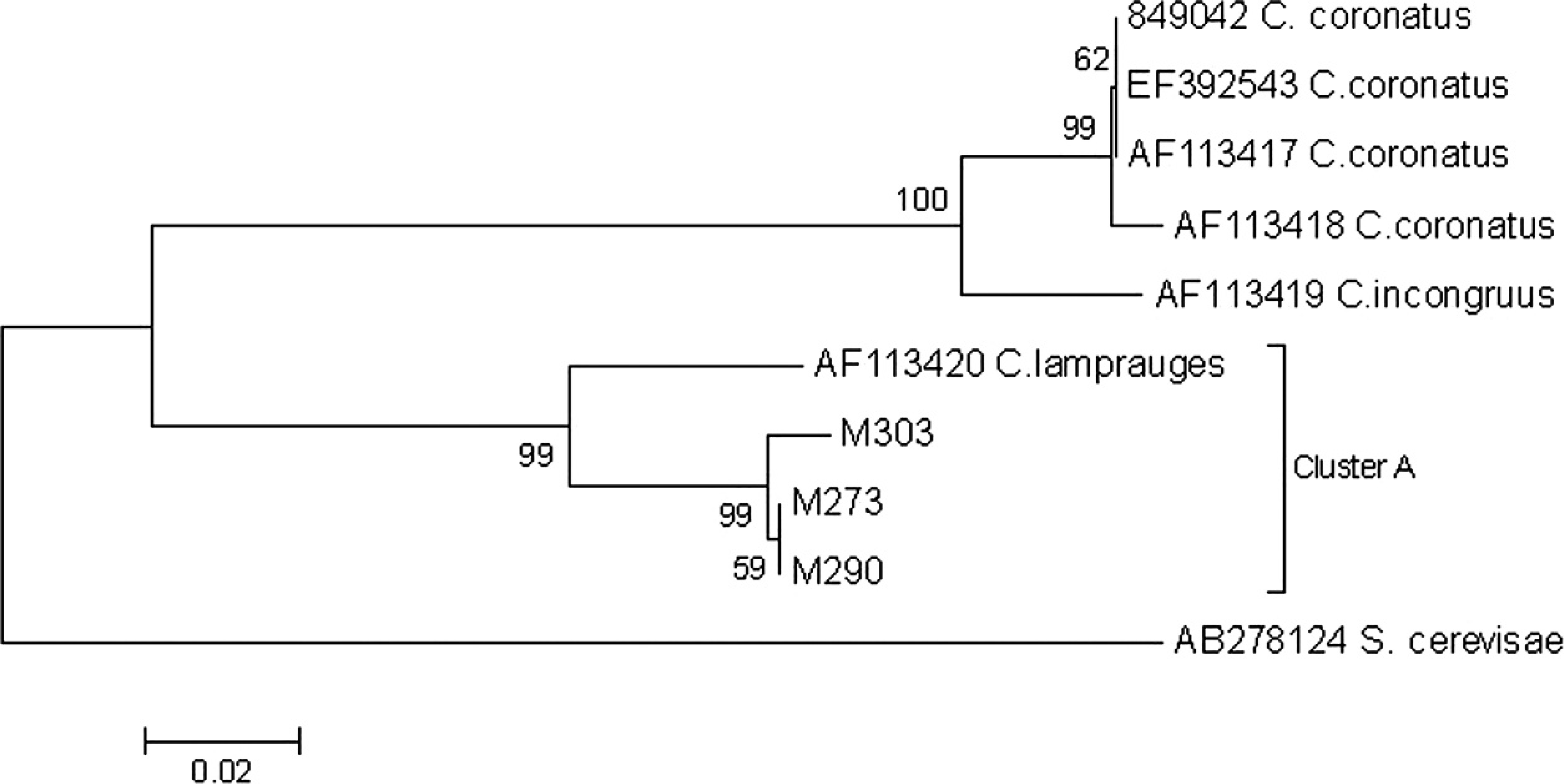
Phylogenetic tree of Conidiobolus spp. from ovine nasopharyngeal zygomycosis. The neighbor joining tree of Conidiobolus genera and 3 isolates of Conidiobolus from Mato Grosso State (MT303, 290, 273) is based on partial sequence data from the 18S ribosomal DNA region. Bootstrap values are shown next to the nodes (1,000 replicates). All isolates in the current study fell within 1 cluster of Conidiobolus lamprauges (cluster A). Saccharomyces cerevisiae was the outgroup.
In summary, the present study identified C. lamprauges in sheep with rhinopharyngeal zygomycosis. This pathogen has not previously been reported to affect sheep. Further studies of this pathogen will be necessary to validate the PCR test for diagnostic use, determine the efficacy of treatment, and identify factors for the prevention of ovine zygomycosis.
Acknowledgements. The authors are grateful to CAPES for financial support through a Masters' scholarship.
Footnotes
a.
Illustra GFXTM PCR DNA and Gel Band Purification Kit, GE Healthcare Life Sciences, Buckinghamshire, UK.
