Abstract
The present report describes a rare case of generalized bovine-type tuberculosis in a slaughtered 4-year-old ewe discovered during routine surveillance at an abattoir. A postmortem examination revealed lesions in the ewe's thoracic and abdominal cavities, ranging from encapsulated, mineralized foci to extensive, soft, caseous tissue. Lesions in the lungs, liver, and lymph nodes were consistent with mycobacterial infection. A histopathological examination detected granulomatous lesions in all tissue samples. The presence of Mycobacterium tuberculosis complex genome was confirmed through a polymerase chain reaction (PCR) analysis of tissues, using IS6110 primers, followed by a nucleotide sequence analysis of PCR products. Acid-fast bacteria, characterized as Mycobacterium bovis, were isolated from lesions following 38 days of incubation.
Mycobacterium bovis, the etiological agent of bovine-type tuberculosis (TB), has an exceptionally wide host range 8 and a complex epidemiological pattern of infection, involving interaction between humans and domestic and wild animals. Information on TB in sheep is scarce, and the general picture that emerges is far from conclusive. In a previous study, postmortem examinations on 70 tuberculin-reacting sheep were carried out. 6 Tuberculous lesions similar to those of cattle were found in 61% of cases. Mycobacterium bovis was also recovered from 32 of 43 sheep with tuberculous lesions in a previous study. 5 These animals belonged to a flock numbering 15,000 head, grazing on a farm with a history of widespread occurrence of TB in both cattle and possums. In a 1988 study, only 2 sheep with histological lesions typical of TB were observed in an abattoir survey of 9.9 million lambs and 1.97 million adult sheep. 1 In another study, an outbreak of M. bovis in a flock of sheep housed in close contact with tuberculous cattle was described. 10 Gross TB lesions were present in 4 of the 6 tuberculin-reacting sheep, and M. bovis was isolated from the lesions. Clinically, however, all affected sheep were healthy. The isolation of M. bovis from a sheep with tuberculous lesions, belonging to a flock of 200 ewes, has also been previously reported. 9
In the current study, a rare case of generalized TB in a slaughtered 4-year-old ewe, discovered during routine surveillance at an abattoir, is described. The ewe in question belonged to a flock of 350 clinically healthy sheep bred on a Sicilian farm where cattle and goats were absent.
Samples for histopathological, molecular, and culture examinations were taken from the ewe's submandibular, mesenteric, and mediastinal lymph nodes, as well as from organs with lesions compatible with TB, namely, diaphragm, lungs, and liver. Tissues were fixed in 10% buffered formalin and processed in 4-mm paraffin-embedded sections for histology. Four-micrometer sections were cut and stained with Ziehl—Neelsen and hematoxylin and eosin for histological examination. Gram and Grocott stains were also performed to identify possible bacteria or fungi.
Samples (about 1 g each) were homogenized, decontaminated with 1 volume of 4% NaOH for 30 min at 37°C, neutralized with 0.067 M phosphate buffered saline (PBS), and centrifuged for 15 min at 3,000 × g. The pellets were suspended in PBS, inoculated into Middlebrook 7H9 medium a with 10% oleic acid-albumin-dextrose-catalase (OADC), b and then incubated in CO2 for 8 weeks at 37°C. Ethylenediamine tetra-acetic acid blood samples (0.5 ml) were mixed with an equal volume of 4% NaOH and processed as above for bacteriological examination.
DNA was extracted using a commercial DNA purification kit. c The polymerase chain reaction (PCR) assay for the detection of Mycobacterium tuberculosis complex (MTC) genome was performed using primers IS6110-F (TGATGTGCTCCTTGAGTTCG) and IS6110-R (GCT-AATTACCCGGTTCATCG), which are able to amplify a 194-bp fragment of the genome in question. The acid-fast isolates were molecularly characterized. DNA was extracted c and typed by PCR—restriction fragment length polymorphism (PCR-RFLP) analysis of the gyrB gene. 7
Amplification of the RD1 region was also performed so as to exclude the presence of the vaccine strain M. bovis BCG. 13
The isolate was tested for susceptibility to the following anti-TB drugs: isoniazid (0.2 μg/ml), ethambutol (7.5 μg/ml), rifampicin (1 μg/ml), streptomycin (2 μg/ml), and kanamycin (6 μg/ml). d The isolate was grown on Middlebrook 7H11 agar a with 10% OADC. b The National Committee for Clinical Laboratory Standards technical guide was followed. 12 Briefly, the turbidity of the inoculum was adjusted to 1 McFarland opacity standard and diluted (1:100). Seventy microliters of the solution was then sealed in 35-mm agar plates. The test was read after 3 weeks of incubation at 37°C in 5% CO2. The isolate was considered resistant if the number of colonies on a medium containing an antimicrobial agent, relative to that observed on a drug-free medium (control), was ≥1%. Strains of M. tuberculosis ATCC 27294 and M. bovis ATCC 19210 e were used as controls.
The sheep flock was tuberculin tested. The skin test was carried out 6 months after the slaughter of the affected ewe. Blood samples were also collected from 48 randomly selected healthy sheep belonging to the same flock and examined by culture and PCR techniques as described above.
Upon postmortem examination, the ewe presented lesions ranging from encapsulated, mineralized foci to extensive, soft, caseous tissue in the thoracic and abdominal cavities. The submandibular and mesenteric lymph nodes were enlarged. One tubercle was detected inside the submandibular lymph node. The mediastinal lymph nodes were enlarged and reddened and contained caseous, gritty nodules. Extensive soft, caseous lesions and encapsulated, calcified tubercles were found in the lungs. The liver and Glisson's capsule were enlarged and contained extensive caseous lesions. Lesions in the lungs (Fig. 1A), liver, and lymph nodes were consistent with infection with mycobacteria.
Histopathological examinations revealed granulomatous lesions in all samples. The liver contained recently formed granulomas characterized by necrotic areas surrounded by mixed inflammatory cells and, occasionally, by multi-nucleated giant cells. Large mineralized necrotic areas surrounded by macrophages, lymphocytes, plasma cells, and peripheral fibroplasias were detected in the lung, lymph nodes (Fig. 1B), and diaphragm. Neither acid-fast bacilli nor other bacteria or fungi were seen in the slides.
Molecular tests, followed by nucleotide sequence analyses of PCR products, confirmed the presence of MTC genome in the tissues (data not shown). Culture results corroborated this finding. Acid-fast bacteria were isolated from all samples after 38 days of incubation. The isolates were characterized as M. bovis by gyrB PCR-RFLP. 7 The RD1 amplification excluded the presence of the vaccine strain M. bovis BCG. All tested bacteria were resistant to ethambutol.
The flock was skin test negative after 72 hr. Both bacteriological and molecular tests for mycobacteria undertaken on blood collected from 48 healthy sheep belonging to the same flock were negative. Tuberculosis is considered rare in sheep, and when occurring, it is often not disseminated. The pathological and histological findings described in the current case of generalized TB in a ewe confirm that ovine TB lesions are similar to those seen in cattle. 6
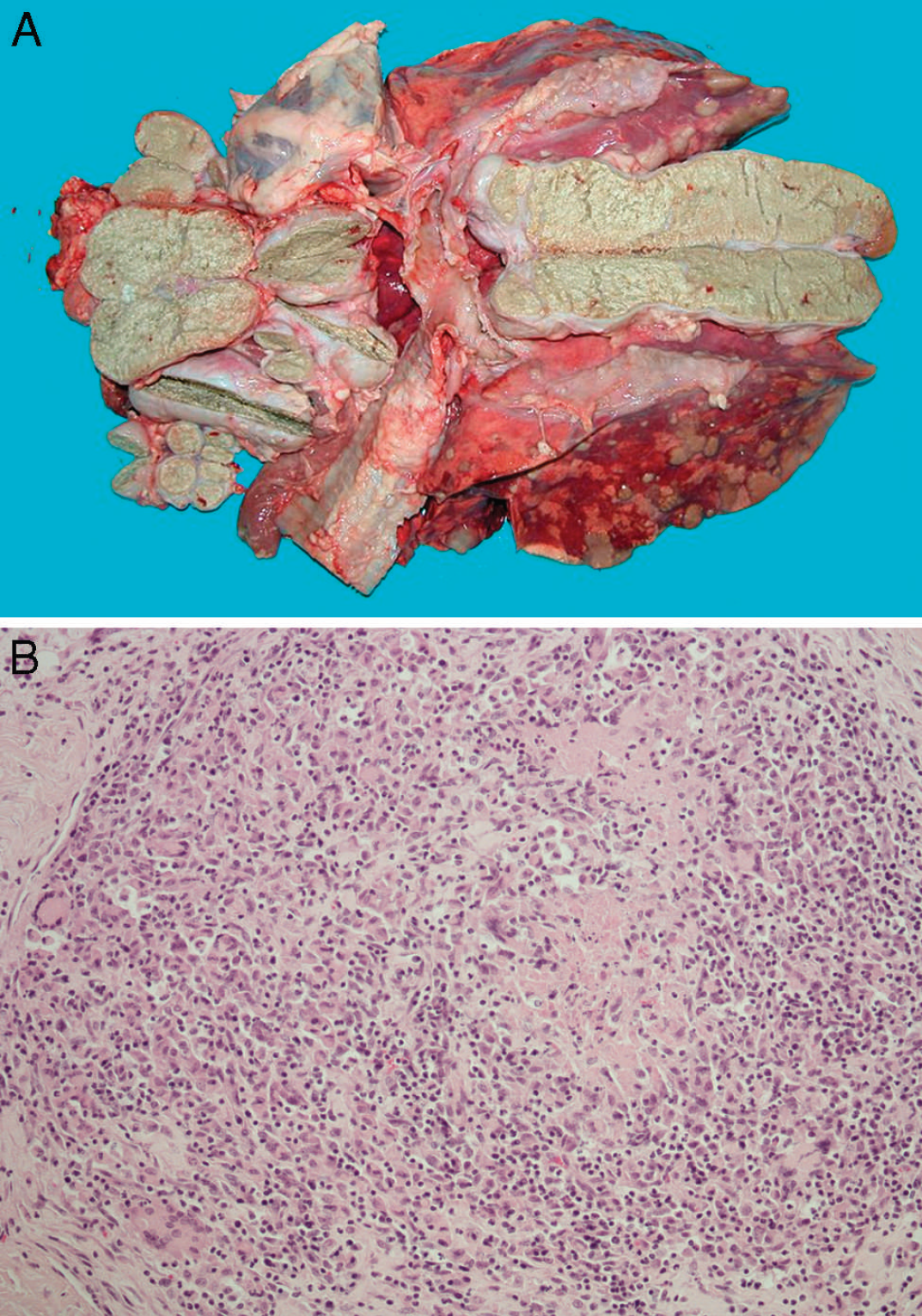
Bovine tuberculosis lesions in a ewe.
Bacteriological examination and molecular tests isolated and characterized the pathogen responsible for these lesions as M. bovis. The pathogen was also characterized as M. bovis spoligotype SB0841 by the National Reference Centre for Mycobacterium bovis tuberculosis at the Istituto Zooprofilattico Sperimentale della Lombardia e dell'Emilia Romagna (Brescia, Italy). SB0841 spoligotype is one of the most common M. bovis spoligotyping profiles isolated and was characterized in 2009 from cattle herds in Italy. 4
Infection with M. bovis followed by TB has been reported mainly in cattle, wild and domestic pigs, primates, and humans. Disease symptoms are indistinguishable from that caused by M. tuberculosis and are therefore generally treated in the same way. Knowledge of resistance of M. bovis toward antibiotics used for therapy of human TB could help avoid cure delay and treatment cost increase when dealing with drug-resistant organisms. Therefore, the susceptibility of the M. bovis isolate toward first-line antibiotics for human TB was evaluated. The strain isolated was resistant to ethambutol. Although limited studies describe the drug-resistance in M. bovis, 3,14 data in the current study highlight the importance of the drug susceptibility evaluation of M. bovis isolates to understand the magnitude of influence of the transmission of drug-resistant or multi—drug-resistant M. bovis strains for treatment of human TB.
Sheep are considered susceptible to M. bovis infection. 5,6 Ovine TB is uncommon, however, and published reports tend to describe individual cases rather than outbreaks. Some authors argue that the low incidence of TB in sheep is a consequence of management and behavioral factors, which tend to reduce their exposure to this pathogen. 11,15 Sheep are extensively managed and grazed predominantly during daylight hours. They also tend to flock together, both when grazing and when resting and ruminating. Therefore, close contact between sheep and potentially infected cattle or wildlife animals is a rare occurrence.
The main trait of M. bovis is its broad host range, the largest of any member of the M. tuberculosis complex. Mycobacterium bovis causes disease in a wide range of domestic, farmed, free-range, and wildlife animals, as well as humans. Only a small proportion of the species that become infected can act as maintenance hosts of these organisms. In maintenance, or reservoir, hosts such as wildlife species, cattle, buffalo, and goats, infection can persist through horizontal transfer in the absence of any other source of M. bovis and may be transmitted to other susceptible hosts as well. Spillover hosts, such as sheep, 2 on the other hand, can become infected with M. bovis when the challenge level is relatively high, although sheep do not seem to maintain infection within the species in the absence of continuing acquisition of infection from maintenance hosts. 11
Infection in a spillover host, as described in the current report, may thus suggest the presence of high levels of M. bovis in the environment, although the possibility that the animal was particularly susceptible to infection due to individual predisposing factors cannot be excluded. The distribution of the lesions in the ewe suggested inhalation as the primary route of infection, probably due to contact with infected animals. In Sicily, most livestock is owned by small farms, where common practices include free breeding and semifree breeding, promiscuity between sheep and cattle, and transhumance. Moreover, there is no complete control over and information regarding the movement of individual animals. These practices increase the possibility of contamination, transmission, and spread of infection.
The infected ewe in question had been bought from a farm in the Catania area where, in 2007, the prevalence and incidence of bovine TB were 29.27% and 24.39%, respectively (unpublished data). Although no cattle or goats were present on the farm of origin, the farmer practiced semifree grazing on neighboring lands, which were also used by others to graze cattle. The farm of origin had 2 TB-infected cattle herds situated within the 10-km radius. Conceivably, in accordance with epidemiological data, the ewe came into close contact with a maintenance host such as an infected cow by sharing pastures or by trespassing on pastures where infected cattle grazed. The ewe was then bought by the current owner, and as shown in the current case, it was unable to spread the infection on the new farm.
The occurrence of TB is linked to both management and ecological factors (population density of various host species, interactions, habitat differences, etc.). 11 Therefore, particular attention should be paid to those farms where free breeding and semifree breeding, promiscuity among different species of livestock, and transhumance are practiced. Moreover, one of the greatest threats to the success of any TB control program in domestic animals is infection among feral maintenance hosts, as such infection is difficult to control and may reintroduce the disease into livestock populations. The study of wildlife TB through appropriate surveillance programs may thus be essential for the effective control of TB in livestock.
Footnotes
a.
Biolife Italiana Srl, Milano, Italy.
b.
BD, Franklin Lakes, NJ.
c.
Gentra® Puregene®, Qiagen SpA, Milan, Italy.
d.
Sigma-Aldrich Company Ltd., Dorset, UK.
e.
Kindly provided by the Department of Infectious, Parasitic and Immune-Mediated Diseases of the Istituto Superiore di Sanità, Rome, Italy.
