Abstract
This article describes 10 cases of paranasal sinus masses in Rocky Mountain bighorn sheep (Ovis canadensis canadensis). Among 21 bighorns that were examined from 11 herds in Colorado, 10 individuals (48%) from 4 herds (36%) had masses arising from the paranasal sinuses. Affected animals included 9 of 17 females (53%) and 1 of 4 males (25%), ranging in age from approximately 2 years to greater than 10 years. Defining gross features of these masses included unilateral or bilateral diffuse thickening of the respiratory lining of the maxillary and/or frontal sinuses, with abundant seromucinous exudate in the affected sinus cavities. Defining histologic features of these masses included chronic inflammation and proliferation of mesenchymal and epithelial cells of the mucosa and submucosa. Epithelial changes included hyperplasia of mucosal epithelium, hyperplasia of submucosal glands and ducts, and neoplasia (adenocarcinoma). Mesenchymal changes included submucosal myxedema, submucosal fibroplasia/fibrosis, bone destruction, and neoplasia (myxomatous fibroma). Specific immunohistochemistry and polymerase chain reaction for Jaagsiekte sheep retrovirus and enzootic nasal tumor virus were performed with negative results.
Keywords
Respiratory disease has been identified as a leading cause of population declines in bighorn sheep since the late 19th century and has been well described elsewhere. 16,21,29 The syndrome of respiratory disease is characterized by sporadic outbreaks of bronchopneumonia in all ages of bighorn sheep, commonly followed by multiple years of poor neonatal survival. 16,29 The cause of this disease is likely multifactorial, including bacterial, viral, parasitic, and environmental (stress) factors, with poorly understood interactions and predisposing conditions. 16,29
Currently, reports of neoplasms in wild sheep are limited to single-case reports of sporadically occurring tumors in individual animals 15,18,19,22,30 with no previous reports of primary neoplasia affecting the respiratory tract of free-ranging or captive wild sheep. This is in contrast to respiratory neoplasia in domestic sheep and goats, which is associated with the oncogenic retroviruses Jaagsiekte sheep retrovirus (JSRV) and enzootic nasal tumor virus (ENTV). Neoplasms caused by these viruses occur in the lungs of sheep in the case of JSRV 6,10,17,25,33,34 and the ethmoid turbinates of sheep and goats in the case of ENTV-1 3,4,7,8 and ENTV-2, 4,5,7,9,24 respectively. For all entities, neoplasms are generally classified as low-grade adenocarcinomas and rarely metastasize. 6,7 Occurrence of these diseases in bighorn sheep has not been reported.
This report describes the gross pathology, histopathology, and preliminary molecular diagnostic findings for 10 cases of paranasal sinus masses in bighorn sheep. To our knowledge, this is the first report describing such lesions in bighorn sheep.
Materials and Methods
Animals
In February 2009, as part of ongoing respiratory disease investigation and management activities, a remnant free-ranging herd of female bighorn sheep (n = 7) from the Poudre River canyon in northern Colorado was culled for population management purposes. Postmortem examination was performed on each carcass. In addition to variable lesions of chronic bronchopneumonia, paranasal sinus masses were identified in all 7 animals. Following the discovery of this novel lesion, all bighorn sheep submitted for necropsy to the Colorado Division of Wildlife were screened for the occurrence of similar masses. From February 2009 through September 2009, a total of 21 bighorn sheep 1 year of age or older were examined, including 17 females and 4 males, ranging in age from approximately 2 years to greater than 10 years. For the majority of cases, the postmortem interval was less than 6 hours, although some carcasses had been frozen before examination and postmortem interval could not be determined.
Histopathology
For all carcass submissions, representative sections of all present major organ systems—including paranasal sinus masses—were fixed in 10% neutral buffered formalin. Selected sections were embedded in paraffin blocks, sectioned at 4 to 6 μm, and stained with hematoxylin and eosin for examination by light microscopy. Selected sections were additionally stained with periodic acid–Schiff (PAS) reaction, Alcian blue stain (pH, 2.5), and Masson’s trichrome stain. Samples of all present masses and exudates within the paranasal sinuses were aseptically collected and stored at –80°C for additional diagnostics.
Immunohistochemistry
To detect specific viral antigens of known sheep retroviruses, immunohistochemistry (IHC) was performed on paranasal sinus masses (n = 7) with monoclonal antibody (mAb) with reactivity for the envelope protein of ENTV and JSRV as previously described. 31 Briefly, samples were deparaffinized, and antigen retrieval was performed in a pressure cooker (heated to 120°C, held for 3 minutes, allowed to cool to 90°C, held for 3 minutes) with Antigen Unmasking Solution (pH, 6; Vector Laboratories, Burlingame, CA). After cooling, endogenous peroxide was quenched with 3% hydrogen peroxide for 5 minutes. Slides were washed 2 times for 10 minutes each with phosphate buffered saline. The slides were incubated with a 1:50 dilution of anti-JSRV envelope mAb (from hybridoma cells) for 1 hour at room temperature. This anti-JSRV envelope antibody cross-reacts with the ENTV envelope. 31 Slides were washed and incubated with a 1:300 dilution of biotinylated horse–anti-mouse immunoglobulin G (Vector Laboratories) for 30 minutes at room temperature. Slides were washed again and incubated with avidin:biotinylated enzyme complex (Vectastain Elite ABC Kit, Vector Laboratories). 3,3′-Diaminobenzidine tetrahydrochloride with nickel chloride enhancement was used as a peroxidase substrate, and the sections were counterstained with hematoxylin. Positive and negative controls were included and examined for each IHC run, using affected and unaffected ovine lung for JSRV and affected and unaffected ovine nasal turbinates for ENTV. An isotype control was also used.
To further characterize proliferative and neoplastic cells, IHC for vimentin and pancytokeratin was performed with an indirect biotin-free system (ultraView Universal Alkaline Phosphatase Red Detection Kit, Ventana Medical Systems, Tucson, AZ) designed for use with an automated immunostainer (NexES IHC module, Ventana Medical Systems). Primary antibodies used were pan-keratin (Ventana Medical System) and vimentin (Ventana Medical Systems). Positive and negative controls were included and examined for each IHC run.
Polymerase Chain Reaction
For polymerase chain reaction (PCR), genomic DNA was extracted from tissue homogenates—sample (n = 9), positive control (n = 3), and negative control (n = 1)—using phenol/chloroform extractions. 28 All tissues had been maintained at –80°C following removal at necropsy. Positive-control genomic DNA for JSRV was extracted from lung tumor tissue of a 3-month-old male domestic sheep (Ovis aries) with experimentally induced pulmonary adenocarcinoma. Positive-control genomic DNA for ENTV-1 was extracted from nasal tumor tissue of a 4.5-year-old female domestic sheep (Dorset breed) with naturally occurring nasal adenocarcinoma. Positive-control genomic DNA for ENTV-2 was obtained from an approximately 3-year-old domestic goat (Capra hircus) with naturally occurring disease. Negative-control genomic DNA for ENTV and JSRV PCR was obtained from lung tissue of a 3-year-old female domestic sheep (Suffolk breed) lacking clinical signs of ENTV or JSRV. PCR was performed with specific primers for JSRV, ENTV-1, and ENTV-2.
PCR for ENTV-1 was performed with specific primers to amplify a 1400–base pair product from the gag gene: forward, 5′-ATCCGTCCCTACATTCGTC-3′; reverse, 5′-CCTTGAACATCTGTTTTGGACC-3′. Each 50-μl PCR reaction contained 45 μl of Platinum PCR Supermix (Invitrogen, Carlsbad, CA), 40 pmol of each primer, and 100 ng of genomic DNA. Thermocycling parameters included initial denaturation at 95°C for 2 minutes, followed by 30 cycles of denaturation at 95°C for 30 seconds, annealing at 56°C for 30 seconds, and extension at 68°C for 45 seconds. Final extension was at 68°C for 3 minutes. PCR for ENTV-2 was performed with specific primers to amplify a 180–base pair product from the U3 region: 24 forward, 5′-GCAAAATGCCAGGACCTTGG-3′ and reverse, 5′-GATCTTATCTGCTTATTTTCAG-3′. Each 25-μl PCR reaction contained 22 μl of Platinum PCR Supermix (Invitrogen), 20 pmol of each primer, and 500 ng of genomic DNA. Thermocycling conditions included initial denaturation at 95°C for 3 minutes, followed by 35 cycles of denaturation at 94°C for 1 minute, annealing at 55°C for 1 minute, and extension at 72°C for 30 seconds. Final extension was at 72°C for 3 minutes. PCR for JSRV was performed with specific primers to amplify a 300–base pair product from the gag gene: forward, 5′-CCCCATCTCTGAAAATGCAC-3′; reverse, 5′-TGTTTAGACGGTGGAGGAAA-3′. Each 50-μl PCR reaction contained 45 μl of Platinum PCR Supermix (Invitrogen), 40 pmol of each primer, and 200 ng of genomic DNA. Thermocycling conditions included initial denaturation at 95°C for 2 minutes, followed by 40 cycles of denaturation at 95°C for 30 seconds, annealing at 56°C for 30 seconds, and extension at 68°C for 45 seconds. Final extension was at 68°C for 3 minutes.
Results
Gross Pathology
Of the 21 carcasses examined, 10 animals had masses arising from the lining of the paranasal sinuses (maxillary and/or frontal), including 9 of 17 females and 1 of 4 males from 4 of 11 sampled herds. In the index herd where all 7 individuals had paranasal sinus masses, all the animals were greater than 10 years of age. Sample sizes for all other herds were small (3 or fewer animals); therefore, prevalence for these herds was not calculated.
Grossly, masses ranged from moderate and diffuse thickening of the sinus lining to solid masses filling the sinus cavity. No discrete polypoid masses were identified, and in only one case did the mass extend to involve the palatine sinus and caudal nasal turbinates (case No. 10). Lesions were bilateral in 8 of 10 cases and unilateral in 2 cases. In all cases, the bone underlying the mass was either grossly invaded or had an irregular pitted surface, suggesting bone remodeling. The thickened sinus lining was easily separated from the underlying bone except in one case (case No. 8), for which the mass severely invaded the bone surrounding the cornual diverticulum of the frontal sinus, causing sloughing of the horn and protrusion of the mass from the top of the skull (Fig. 1 ).
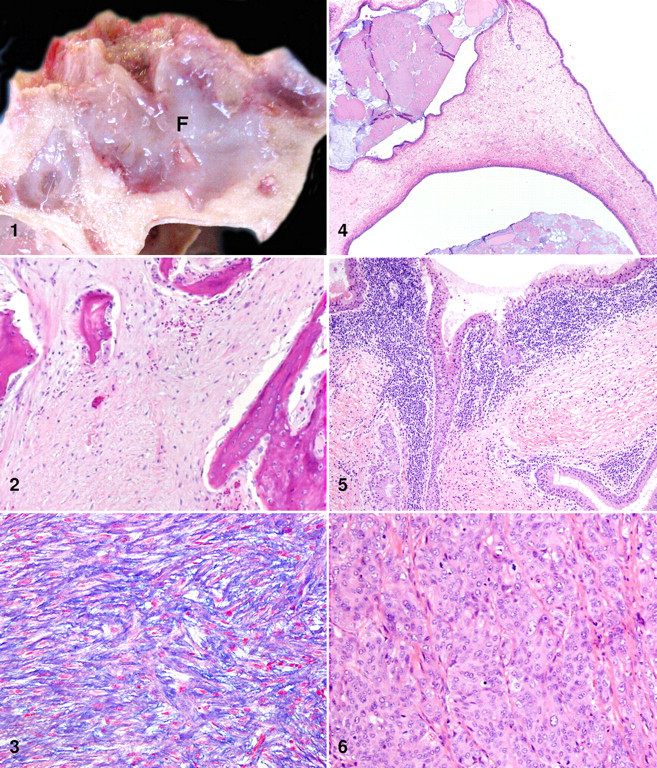
All masses were homogeneous, white, shiny, and soft to gelatinous, and all frequently contained mucinous cysts. The affected sinus cavities were often filled with seromucinous to mucopurulent exudate. In at least 2 cases, similar mucinous material had been noted antemortem as nasal discharge.
Additional necropsy findings of the respiratory tract included moderate numbers of lungworm nodules in 9 of 10 animals, bronchopneumonia in 3 of 10 animals, fibrous pleural adhesions suggesting previous bronchopneumonia in 2 of 10 animals, and a tooth root abscess into the maxillary sinus of 1 animal. No Oestrus ovis larvae were seen in any of the carcasses examined. Cause of death in all animals was considered unrelated to the paranasal sinus masses or attributed to other respiratory disease.
Histopathology
All paranasal sinus masses examined had components of epithelial and mesenchymal proliferation within the mucosa and submucosa of the sinus lining, and lesions varied from hyperplasia to neoplasia (Table 1 ). Epithelial and mesenchymal origin of cells was confirmed with IHC for pancytokeratin and vimentin, respectively.
Histologic Features for 10 Cases of Paranasal Sinus Masses in Rocky Mountain Bighorn Sheep a

a Case numbers were not assigned in chronological order. A plus sign (+) represents presence of the listed histologic feature.
The majority of the examined masses were predominated by mesenchymal proliferation within the submucosa. These masses contained a population of well-differentiated spindle to stellate cells forming a well-vascularized loose edematous or myxomatous stroma to dense fibroplasia and fibrosis (Table 1). In selected cases, the presence of a myxomatous matrix composed of acid mucopolysaccharides was confirmed by staining with Alcian blue and a lack of magenta staining with the PAS reaction. The presence of collagen fibers was demonstrated by blue staining with Masson’s trichrome. In all cases, spindle cells were well differentiated despite frequent invasion of the underlying bone.
A single mass (case No. 8) was diagnosed as a myxomatous fibroma based on cellular features, including massive proliferation of well-differentiated fibroblasts and collagen bundles (Fig. 2 ) and a variably loose myxomatous background (Fig. 3 ). Although the cells were well differentiated with minimal anisocytosis and anisokaryosis and rare mitotic figures, the diagnosis of neoplasia was based on marked invasion and destruction of the surrounding bone in the absence of significant inflammation. The remnant bone spicules were often rimmed by osteoblasts and fewer osteoclasts (Fig. 2), suggesting either bone remodeling or bone production by the tumor. Additional differentials considered for this mass included myxoma, myxosarcoma, ossifying fibroma, fibrous dysplasia, and periosteal fibrosarcoma.
Although typically less prominent than the mesenchymal proliferation, all masses were characterized by hyperplasia of epithelial components, including the pseudostratified ciliated surface epithelium, submucosal serous and mucous glands, and submucosal simple cuboidal ductular epithelium (Table 1). Masses frequently contained clusters of well-differentiated acini deep within the submucosa, as well as many large cystic structures containing abundant intraluminal mucin (PAS positive), lined by well-differentiated epithelial cells (Fig. 4 ). Hyperplastic epithelial cells occasionally demonstrated multifocal dysplasia but lacked prominent features of neoplasia (Fig. 5 ).
Two masses were diagnosed as adenocarcinoma based on the presence of poorly differentiated epithelial cells forming sheets and solid nests of cells deep within the submucosa, in addition to more differentiated tubuloacinar structures. One of these cases (case No. 10) additionally had frequent mitotic figures and moderate anisocytosis and anisokaryosis (Fig. 6 ).
Both hyperplastic and neoplastic masses frequently contained a significant population of inflammatory cells. Lymphoplasmacytic sinusitis was diagnosed in 10 of 10 cases, characterized by dense infiltrates of well-differentiated plasma cells and fewer lymphocytes, associated with proliferating epithelial cells (Fig. 5), and occasionally located within perivascular spaces. Additionally, cystic masses occasionally contained intraluminal suppurative exudate suggesting secondary bacterial infection. Aerobic culture of sinus exudate from one case (case No. 10) yielded heavy growth of Pasteurellaceae. Similar inflammatory lesions were not present in the sinus linings of unaffected animals.
Histopathology of retropharyngeal lymph node was performed for all cases, with no evidence of neoplastic metastasis. Moderate to marked lymphoid hyperplasia and increased numbers of plasma cells in lymph node sinuses were noted in all cases.
Molecular Diagnostics
Genomic DNA was extracted from 9 of 10 masses as well as positive and negative controls. None of the 9 samples nor negative controls were positive by PCR using specific primers for JSRV and ENTV. IHC for envelope protein, with demonstrated reactivity for JSRV and ENTV, 31 was performed for 7 of 10 cases, and results were negative for all cases examined. The single case not evaluated by PCR was evaluated by IHC.
Discussion
Following the discovery of paranasal sinus masses in 7 of 7 Rocky Mountain bighorn sheep from a single herd in Colorado, we hypothesized that these masses were a symptom of an infectious disease. To further investigate this hypothesis, we sampled additional animals to document the occurrence of this disease, further characterize the disease, and test for viral agents known to cause similar diseases in domestic sheep and goats.
Although the gross pathology and histopathology of these masses varied, defining gross and histologic features were present in all cases. Defining gross features of all masses included diffuse thickening of the respiratory lining of the maxillary and/or frontal sinuses, as well as abundant seromucinous exudate in the affected sinus cavities. This gross lesion varied in severity, with the most severe cases characterized by masses that filled the sinus cavity and markedly invaded and destroyed the underlying bone.
The defining histopathologic feature of all masses was proliferation of mesenchymal and epithelial cells, with neoplasia (mesenchymal or epithelial) at the most extreme end of a presumed continuum of changes.
These features are not unlike those of the recognized disease entity enzootic nasal tumor (ENT) of domestic sheep and goats. Clinically, domestic sheep and goats affected by ENT have abundant seromucinous nasal discharge. 5,11,13,20,23,27,32,35 Grossly, masses originate from the ethmoid turbinates and expand to fill the nasal cavity, with frequent invasion of the surrounding paranasal sinuses associated with tumor expansion. 5,13,20,32 Tumors vary from soft, shiny, white gelatinous masses to firm, meaty, or granular grey-red masses. 11,13,20,32,35 Histologically, these tumors are classified as adenomas, 11,20,27,32 adenopapillomas, 11,23,35 or low-grade adenocarcinomas. 5,11,13,20,27,32,35 Nonneoplastic, hyperplastic inflammatory polyps are occasionally found adjacent to neoplastic masses. 5,11,27
Inflammatory polyps are focal, raised, often pedunculated masses with an edematous, often chronically inflamed fibrovascular core lined by mucosal epithelium. 14 When associated with ENT, these hyperplastic masses have been proposed to be preneoplastic lesions, but this association has not been proven. 27 Histologically, polyps of affected domestic sheep and goats have a highly edematous stroma and marked lymphoplasmacytic infiltrates. 5,27 The overlying epithelium is often hyperplastic. 5,27
Similarities between ENT and paranasal sinus masses in bighorn sheep include the presence of seromucinous nasal discharge clinically, the gross finding of a soft white mass in the sinus cavity, and classification of some masses as adenocarcinoma. Additionally, the inflammatory nasal polyps often associated with ENT share characteristics with the hyperplastic masses described here for bighorn sheep, although in bighorn sheep the mass is a diffuse thickening of the sinus lining and not a discrete polypoid mass.
Prominent differences between ENT and the masses described here are location (paranasal in bighorn sheep and nasal in domestic sheep and goats) and malignancy (predominantly, hyperplastic masses in bighorn sheep and neoplastic masses in domestic sheep and goats). Interestingly, in one bighorn sheep case, the mass did extend to involve the nasal turbinates (case No. 10), and this mass was classified as an adenocarcinoma.
Other prominent differences between the 2 entities include the papillary appearance and often grey-red color of ENT tumors not characteristic of bighorn sheep masses, as well as the prominent mesenchymal population histologically present in bighorn sheep masses but infrequently described for ENT.
Because the masses discovered in bighorn sheep were somewhat reminiscent of ENT, we considered ENTV or a closely related retrovirus as a possible etiology. Due to the close relatedness of ENTV-1 (the causative agent of ENT in domestic sheep), ENTV-2 (the causative agent of ENT in domestic goats), and JSRV, we screened samples for all 3 viruses. Our negative findings indicate that these specific retroviruses are unlikely to be involved in the pathogenesis of the lesion described here. Alternative hypotheses for the cause of these masses include infection by other viral agents, genetic predisposition, toxins, and chronic inflammation, including chronic bacterial infections.
Chronic inflammation associated with the nasal bot O ovis has been suggested as a possible associated condition occurring with ENT, 12,23 although no association has been proven. O ovis infection is relatively common in bighorn sheep and may thus be a source of chronic inflammation in the sinus cavities. However, no O ovis larvae or eosinophilic infiltrates were present in the cases described here. A syndrome of chronic sinusitis has been reported in desert bighorn sheep (Ovis canadensis nelsoni) and is attributed to aberrant migration of O ovis larvae. 1,2,26 These lesions are characterized by extensive bone destruction in the maxillary and frontal sinuses and are predominantly described as osteonecrosis and osteolysis. 1,2,26 However, some cases of chronic sinusitis with osteolysis have been diagnosed after decomposition of soft tissues; 2 therefore, the diagnosis of either chronic sinusitis or paranasal masses should be made with caution for desiccated specimens.
Although the cause of bighorn sheep paranasal sinus masses remains uncertain, the continuum of lesions among cases suggests a shared etiology. However, the examination of additional cases and more extensive diagnostics will be necessary to further define this disease and investigate possible infectious etiologies.
Regardless of etiology, the changes to the normal sinus respiratory mucosa, the space-occupying nature, the abundant mucus production, and the bone invasion of these masses may affect normal upper respiratory function and are factors to consider when investigating bighorn sheep respiratory disease. Therefore, continued surveillance for paranasal sinus masses, in addition to continued attempts to identify an inciting cause, will be important for future management of affected herds.
Footnotes
Acknowledgements
This work was funded by the Colorado Division of Wildlife. We would like to acknowledge the following individuals for their contributions to this project: James DeMartini, professor emeritus, Colorado State University, for mentorship, provision of Jaagsiekte sheep retrovirus–positive control tissue, and comments on the manuscript; Christina Cousens, Moredon Research Institute, Edinburgh, United Kingdom, for provision of enzootic nasal tumor virus 2–positive control tissue; Joel Rovnak and Connie Brewster, Colorado State University, for technical support and assistance; Mark Vieira, Shane Craig, Sherri Huwer, and Dan Walsh, Colorado Division of Wildlife, for submission of cases; and Kate Huyvaert, Dan Tripp, and Michael Sirochman, for comments on the manuscript.
The authors declared that they had no conflicts of interests with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
