Abstract
Sarcomas associated with injection sites are a rare but important problem in cats. Immunohistochemical detection of p53 protein may correlate to mutation of the p53 tumor suppressor gene, a gene known to be important in oncogenesis. The expression of nuclear p53 protein in 40 feline injection site-assocated sarcomas was examined by immunohistochemical staining. In 42.5% (17/40), tumor cell nuclei were stained darkly; in 20% (8/40), tumor cell nuclei were stained palely; and in 37.5% (15/40), tumor cell nuclei were unstained. Immunohistochemical detection of p53 protein in a proportion of injection site-associated sarcomas suggests that mutation of the p53 gene may play a role in the pathogenesis of these tumors.
There is epidemiologic evidence for an association between administration of vaccines and subsequent development of soft tissue sarcomas in cats. 10 12 The low prevalence of these tumors, about 1 or 2/10,000 vaccines administered, 4 suggests that factor(s) within individual cats, other than injection, may be prerequisite for tumorigenesis. Congenital or acquired genetic factors may play a role, especially mutation(s) of oncogenes and tumor suppressor genes. 14 Mutations in the p53 gene have been associated with oncogenesis in people and animals. 5 14 The p53 gene product is a multifunctional transcription factor that regulates induction of apoptosis in cells with irretrievably damaged DNA, thus preventing propagation of damaged DNA. 5 More than 50% of tumors in people, including various sarcomas, have p53 gene mutations. 13 18
Normal/wild-type p53 protein is not generally detected by immunohistochemical staining because of a short half-life of about 15–20 minutes. Missense mutations (mutations resulting in amino acid substitution) may induce conformational changes that stabilize the protein, resulting in accumulations of nuclear p53 that are detectable by immunohistochemical methods. 1 11 A direct correlation has been established between mutations in the p53 gene and detection by immunohistochemistry (IHC). 3 7 8 The purpose of this study was to examine expression of p53 protein in feline injection site-associated sarcomas (ISS) to look for indirect evidence for p53 mutation.
The forty tumors used for this study were fibrosarcomas diagnosed as ISS based on historical and clinical data and histopathological findings consistent with previously described criteria. 9 Formalin-fixed, paraffin-embedded tissues were immunohistochemically stained for p53 protein using a 1:500 dilution of rabbit polyclonal antiserum to p53 protein (CM1, Novacastra Laboratories Ltd., New Castle upon Tyne, UK) and an avidin-biotin complex (Vectastain Elite, Vector Laboratories, Burlingame, CA) immunoperoxidase method adapted to a robotic slide stainer (Fisher Scientific Co., Edmonton, AB, Canada). 6 The sections were counterstained with hematoxylin. Two sections of each tumor were stained with antiserum CM1, and a third section of each tumor was stained replacing CM1 with PBS (omission control). COS7 cells (American Tissue Culture Collection, Bethesda, MD), known to express p53 protein, were injected into normal equine liver, which was formalin-fixed and paraffin-embedded for use as positive controls. Blocks of normal feline tissues, including skin, intestine, lung, urinary bladder, liver, and pancreas, were used as negative controls.
Sections of tumors were classified as darkly staining for p53 protein if there was distinct dark staining of the nuclei of a majority of the tumor cell population similar to the staining of COS7 cells. Other tumors were classified as having either pale staining (staining similar to that seen in normal epithelial cells) or negative (no discernable nuclear staining).
There was dark staining for p53 protein in the nuclei of tumor cells in 17 of 40 (42.5%) ISS (Fig. 1); there was pale staining in 8/40 (20%) tumors and no staining in 15/40 tumors (37.5%) (Fig. 2, Table 1). Of the darkly stained tumors, most cells were similarly stained; in the palely stained tumors, there was staining of at least 50% of tumor cells. In the nonneoplastic epidermis adjacent to both staining and nonstaining tumors and in the skin of the normal cat, there was occasional, pale nuclear staining of keratinocytes in most cases. In one case with dark staining of tumor cell nuclei, there was dark staining in adjacent nonneoplastic epidermal keratinocytes and myocyte nuclei. Pale nuclear staining of lymphocytes was also observed in six tumors (two tumors with dark staining cells, four tumors in which the tumor cells were not stained). There was dark staining of the nuclei of COS7 cells. There was no staining in sections in which the CM1 antiserum was omitted from the staining protocol.
Numbers and percentages of feline injection site associated sarcomas with dark, pale, or no immunohistochemical (IHC) staining of tumor cell nuclei for p53 protein.

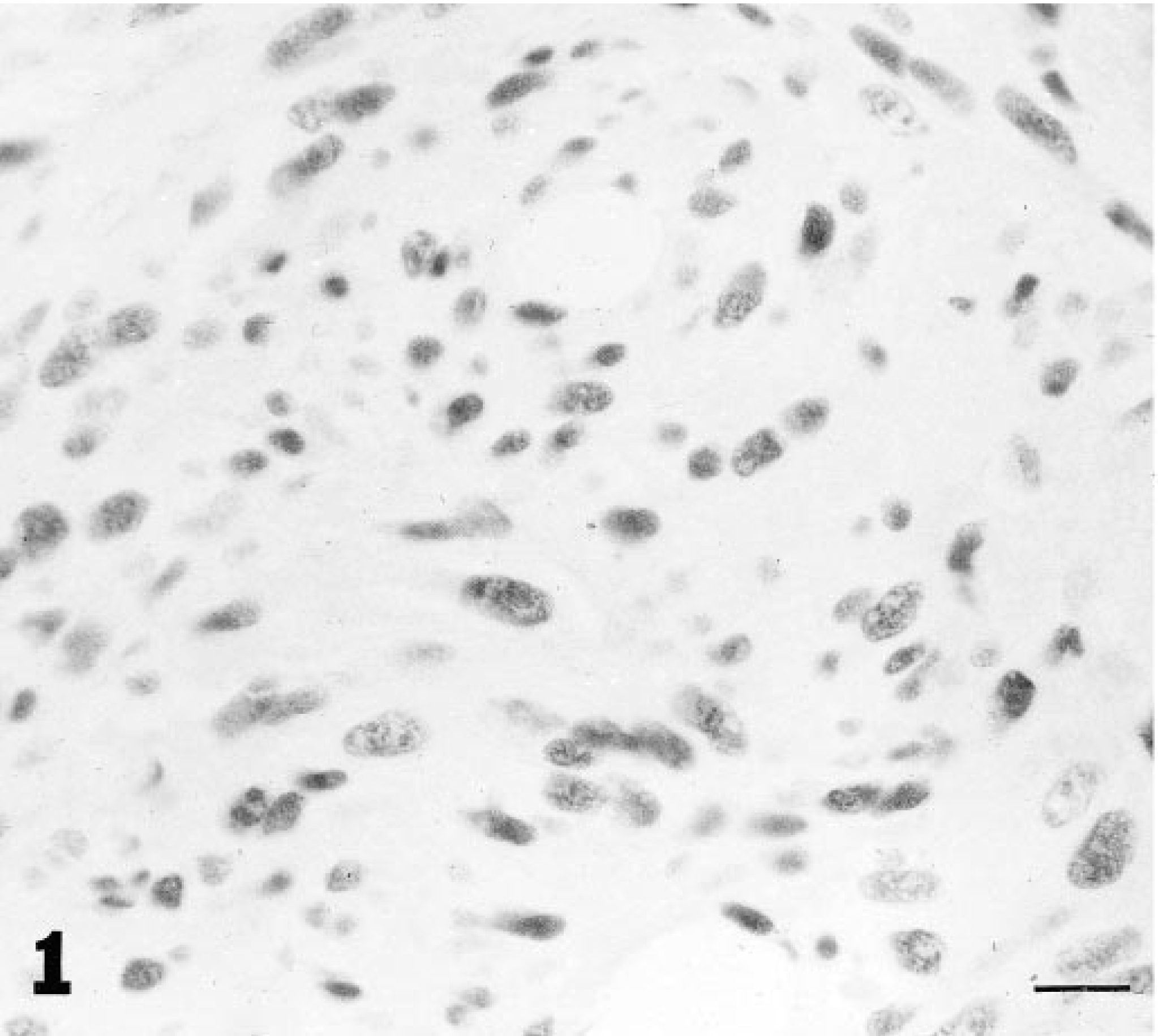
Injection site-associated sarcoma; cat. Dark nuclear staining for p53 antigen in tumor cells. Avidin-biotin immunoperoxidase complex method. CM-1 antibody, Lillie's hematoxylin counterstain. Bar = 30 µm.
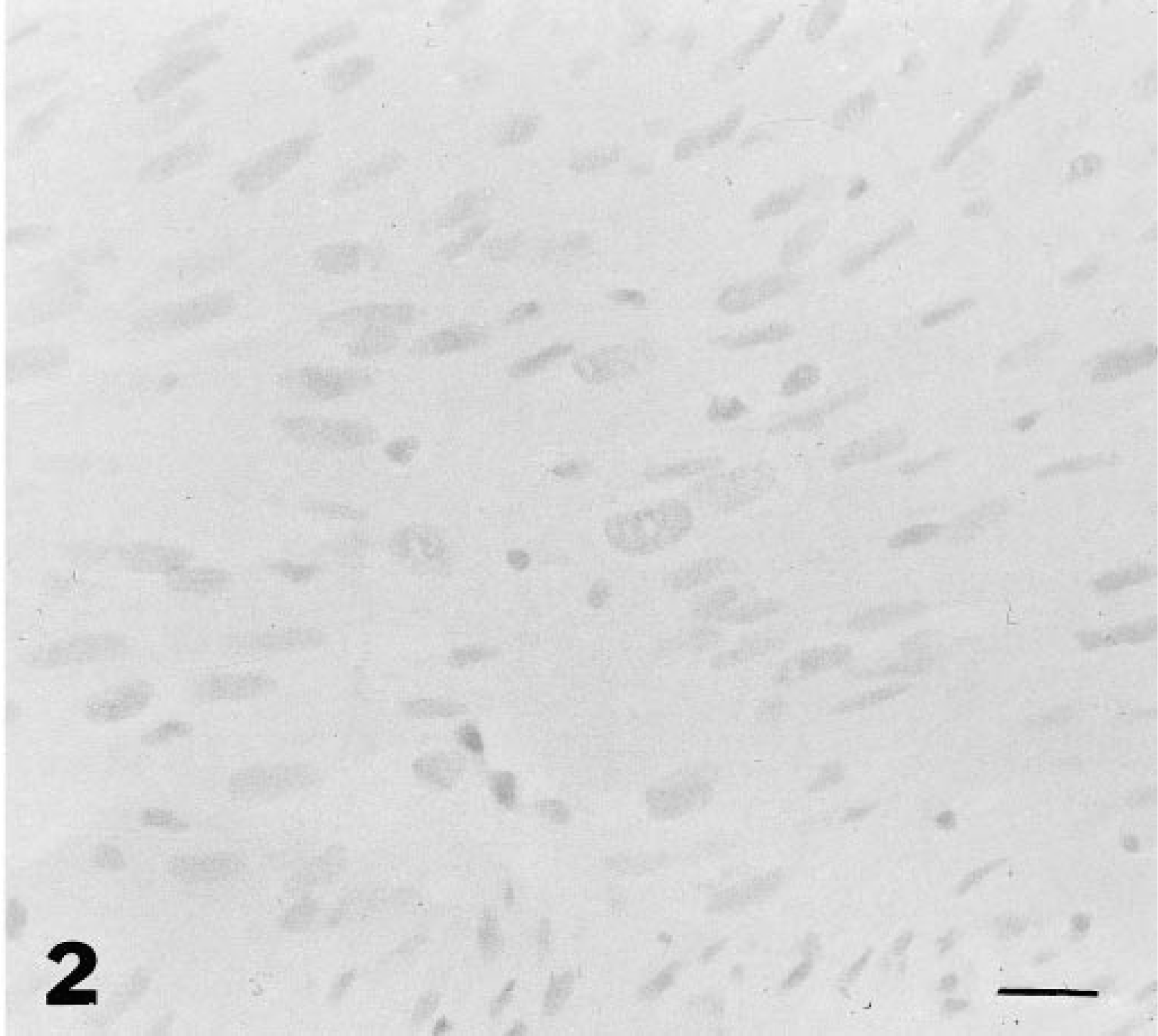
Injection site-associated sarcoma; cat. There is no evidence of nuclear staining for p53 antigen in tumor cells. Avidin-biotin immunoperoxidase complex method. CM-1 antibody, Lillie's hematoxylin counterstain. Bar = 30 µm.
Immunohistochemistry for p53 may be a surrogate test for detection of mutations instead of the more laborious and expensive nucleotide sequencing. 1 The accumulation of p53 protein demonstrated by dark staining of 42.5% ISS suggests that mutation of the p53 gene may play a role in the neoplastic progression of at least this proportion of tumors. The significance of faint staining, seen in 20% (8/40) of ISS, is uncertain. Pale staining occurred only occasionally in nuclei of epidermal epithelial cells in the normal and tumor-bearing cats but in over 50% of the cells in the tumors. The pale staining may correspond to either detection of low levels of abnormal p53 or to detection of wild-type p53. Wild-type p53 is occasionally demonstrated in normal cells due to stabilization of the protein by interaction with certain cellular and viral proteins, 19 from abnormal degradation or sequestration pathways, 2 16 and by increased expression of normal p53 protein in cells that are stressed or subjected to a variety of cellular insults. 2 15 18 DNA sequence analysis is required to confirm if the p53 protein detected in these cases is associated with mutations of the p53 gene. In 15 (37.5%) ISS, there was no staining for p53 protein by IHC, suggesting either that the p53 gene sequences were not mutated or that the mutations in the p53 did not stabilize the protein sufficiently to be detectable by IHC. The findings of this study suggest that mutations of the p53 gene may be involved in the pathogenesis of at least a proportion of ISS and that genetic analyses of ISS for p53 gene alterations are indicated. In an associated study, p53 gene mutations were subsequently found in five of eight ISS with dark staining but not in tumors with pale (n = 3) or no staining (n = 10) or in the adjacent normal tissues in any tumors. 17
Footnotes
Acknowledgements
The authors are grateful to Dr. R. C. Bartsch, Southwest Veterinary Diagnostic Laboratory, Phoenix, Arizona; Dr. L. D. McGill, Animal Reference Pathology, Salt Lake City, Utah; and Dr. S. Lester, Central Laboratory for Veterinarians, Langley, British Columbia, for the provision of tumors. This study was funded with grants from the Companion Animal Health Fund of the Western College of Veterinary Medicine and the Health Services Utilization and Research Commission of the Province of Saskatchewan.
