Abstract
Eighty spontaneously occurring feline vaccine-associated sarcomas (VAS) were evaluated to determine the immunohistochemical expression of the tumor suppressor gene p53. Sixty-five of 80 VAS (81%) exhibited positive immunoreactivity with Mab240, a murine monoclonal antibody that specifically recognizes mutated p53. Only 44 of 81 tumors (55%) were positive with rabbit polyclonal antibody CM-1. CM-1 often yielded nonspecific staining of nonneoplastic tissues. Nonspecific staining was greatly reduced or absent with Mab240. Cytoplasmic staining for p53 was a consistent pattern of VAS, occurring in 44% of tumors evaluated. Cats with tumors that exhibited cytoplasmic p53 had significantly shorter time to tumor recurrence compared to those cats with tumors that exhibited nuclear p53 staining (P = 0.0284), but no significant difference in survival outcome was observed. Immunohistochemical detection of p53 offers a prognostic tool for VAS, and, because abnormal p53 expression appears to be a common feature of feline VAS, molecular targeting of mutant p53, may offer a promising new therapeutic opportunity for this cancer.
Effective treatment of vaccine-associated sarcomas (VAS) in the cat remains a challenging problem. Compared with sarcomas not associated with vaccination, VAS exhibit anaplasia, rapid growth, ulceration, and necrosis. 3, 5, 21 Additionally, VAS are often poorly encapsulated, making complete excision difficult and local recurrences common. 21 Surgery continues to be the mainstay of treatment; however, a recent study suggests that surgery alone is curative in only a small percentage of cases and only where radical excision (i.e., amputation) can be achieved. 15 Radiation therapy and chemotherapy have been used as adjuncts to surgery, and doxorubicin-based chemotherapy has been shown to delay the onset of tumor recurrence but not to extend overall survival time. 29 Thus, novel therapeutic approaches to complement conventional treatment warrant investigation. An understanding of the molecular basis of malignant transformation of these tumors in cats is critical to the development of novel treatment approaches.
Considerable interest has been generated regarding the role of mutation of the tumor suppressor gene p53 in the development of VAS. The tumor suppressor gene p53 encodes for a nuclear protein that plays a key role in the regulation of the cell cycle. In response to DNA damage, wild-type p53 increases, which prevents the cell from progressing through the cell cycle, allowing for DNA repair prior to continued cell replication. 28, 39 Should the damage to the DNA be too great, p53 induces apoptosis, thereby preventing defects in the genetic code from being passed on to subsequent generations. 13, 28, 39 Cells in which p53 is absent or mutated are allowed to proceed unregulated through the cell cycle, giving rise to aberrant clones and potentially to malignant transformation.
Most mutations of p53 occur within several “hot spot” locations of the genome, particularly exons 5–8, which are clearly linked to tumorogenesis. 11 Point mutations of p53, which confer an alteration in the structural conformation of the gene, are most common. This conformational change results in a longer half-life of the protein, allowing accumulation of the mutant p53 within the nucleus of affected cells; this accumulation can then be detected by immunohistochemical methods. 16 In contrast, wild-type gene p53 is not detectable by immunohistochemistry because of its short half-life.
Dysregulation of p53 expression has been demonstrated in a variety of human tumors and may be of prognostic significance. Association between p53 mutations and prognosis has been reported for several human malignancies, including breast cancer, 17, 36, 37 osteosarcoma, 25 thyroid cancer, 2 and soft tissue sarcomas. 19, 28 A recent study by Sagartz et al. was able to demonstrate a correlation between alterations in p53 expression and predicted aggressive tumor behavior in osteogenic tumors of dogs. 31 Expression of mutant p53 has been reported in feline hematopoietic and solid tumors. 22–25 However, these reports in feline tumors have been sporadic, and to date no associations have been made between overexpression of p53 and observed tumor behavior.
This study was initiated to evaluate whether alterations in p53 expression occur in spontaneously occurring VAS in cats, utilizing a panel of anti-p53 antibodies for the immunohistochemical detection of abnormal p53. In humans, studies suggest that a combination of antibodies is a more sensitive method of p53 detection and provides more prognostically relevant information than a single antibody used alone. 4 Correlation between p53 immunoreactivity and clinical outcome was demonstrated in a subset of cats.
Materials and Methods
Tumor acquisition
Eighty formalin-fixed, paraffin-embedded VAS from 80 cats were evaluated. A subset of tissues were from cats that had been part of a previous retrospective study. 15 The remaining tissues were obtained from client-owned cats that presented for surgical excision or incisional biopsy of their tumors at the University of Wisconsin–Madison Veterinary Medical Teaching Hospital. Additionally, three cell culture lines (courtesy of Dr. Greg MacEwen and Jennifer Carew) were evaluated for p53 expression.
Immunohistochemistry
Immunohistochemical detection of p53 expression was performed on deparaffinized sections using the streptavidin–biotin–peroxidase complex method. The initial panel of anti-p53 primary antibodies consisted of rabbit polyclonal antibody CM-1 (Biocare Medical, Walnut Creek, CA) and four monoclonal antibodies: 1801, DO7, DO1, and 240 (Oncogene Research Products, San Diego, CA). Three of the monoclonal antibodies (DO1, DO7, 1801) failed to produce immunoreactivity in any of the feline tissues; this was presumed to be attributable to lack of sufficient sequence homology with feline p53. The final panel of anti-p53 antibodies included the polyclonal antibody CM-1, which recognizes wild-type and mutant p53, and monoclonal antibody 240 (Mab240), a murine monoclonal antibody of the IgG1 isotype that specifically recognizes mutant p53.
Five-micrometer paraffin-embedded sections were placed on Kling-on HIER supercharged slides (Biocare Medical), incubated overnight in a 37°C dry heat oven and then deparaffinized by incubations of 5 minutes each at 25°C in xylene, decreasing concentrations of ethanol, and water. Endogenous peroxidase was blocked in Peroxidazed1 (Biocare Medical) for 5 minutes. The sections were then boiled in a commercial steamer containing either 10 mM sodium citrate buffer at a pH of 6.0 (CM-1) or TUF/Retrievall (Mab240, Signet Laboratories, Dedham, MA) for 35 minutes. Nonspecific staining was blocked using 10% goat serum with Tween 20 in phosphate buffered saline (PBS) (Background Eraser, Biocare Medical) for 5 minutes at room temperature. After pretreatment, the sections were incubated for 1 hour at room temperature with the anti-p53 antibodies at a dilution of 1:200 (CM-1) or 1:150 (Mab240). Dilutions were made in PBS. Following incubation with primary antibody, the sections were successively incubated with anti-rabbit/anti-mouse biotinylated secondary antibody (Biocare Medical) for 10 minutes at room temperature and streptavidin conjugated with peroxidase (Biocare Medical) also for 10 minutes at room temperature in a humidity chamber. The sections were then developed with 3,3′-diaminobenzidine tetrachloride, prepared according to the manufacturer's instructions, for 5 minutes at room temperature. The slides were washed in distilled water and counterstained for 1 minute with Tacha's hematoxylin (Biocare Medical). Slides were washed in tap water, dehydrated in ethanol, and cleared with xylene, and coverslips were mounted with Permount (Fisher Scientific, Pittsburgh, PA)
The positive control was a paraffin-embedded section of a human colon carcinoma cell line known to be positive for abnormal expression of p53 and provided by Biocare Medical. Additional sections of each tumor block were processed with omission of the primary antibody as a negative control.
p53 immunostaining was evaluated by light microscopy using a 40× lens. A positive result was scored if there was nuclear or cytoplasmic staining of the majority of malignant fibroblasts of greater intensity than surrounding nonneoplastic tissues. If there was no stain or stain of equal or lesser intensity than the adjacent nonneoplastic tissues, the result was considered negative.
Results
Eighty sarcomas were assessed for immunoreactivity to p53. Nonspecific staining of hair follicles, epidermis, and muscle was often observed with the CM-1 polyclonal antibody. The presence of nonspecific tissue staining was dramatically decreased or absent with Mab240.
Overall, 81% (n = 65) of the tumors exhibited positive staining with Mab240. By contrast, 55% (n = 44) of tumors were positive with CM-1. Thirty-eight percent (n = 29) of the positive-staining tumors exhibited nuclear immunoreactivity for p53 with both antibodies. Of these tumors, two exhibited cytoplasmic immunoreactivity in addition to nuclear immunoreactivity with CM-1, whereas 14 tumors exhibited both nuclear and cytoplasmic immunoreactivity with Mab240 (Fig. 1). Three tumors that demonstrated positive nuclear staining with CM-1 exhibited positive cytoplasmic staining with Mab240. Sixteen percent (n = 12) of tumors exhibited only cytoplasmic staining with both antibodies. Twenty-six percent (n = 21) of tumors that were considered negative with CM-1 demonstrated convincing positive cytoplasmic staining with Mab240 (Fig. 2). Nineteen percent (n = 15) of tumors stained with Mab420 and 45% (n = 36) stained with CM-1 were considered negative with both antibodies (Table 1). In those tumors demonstrating positive immunoreactivity for p53, nonneoplastic mesenchymal cells peripheral to the tumor cells also exhibited positive staining in many samples, especially with Mab240 (Fig. 3).
Results obtained from immunohistochemical staining of paraffin-embedded vaccine-associated sarcomas in cats.

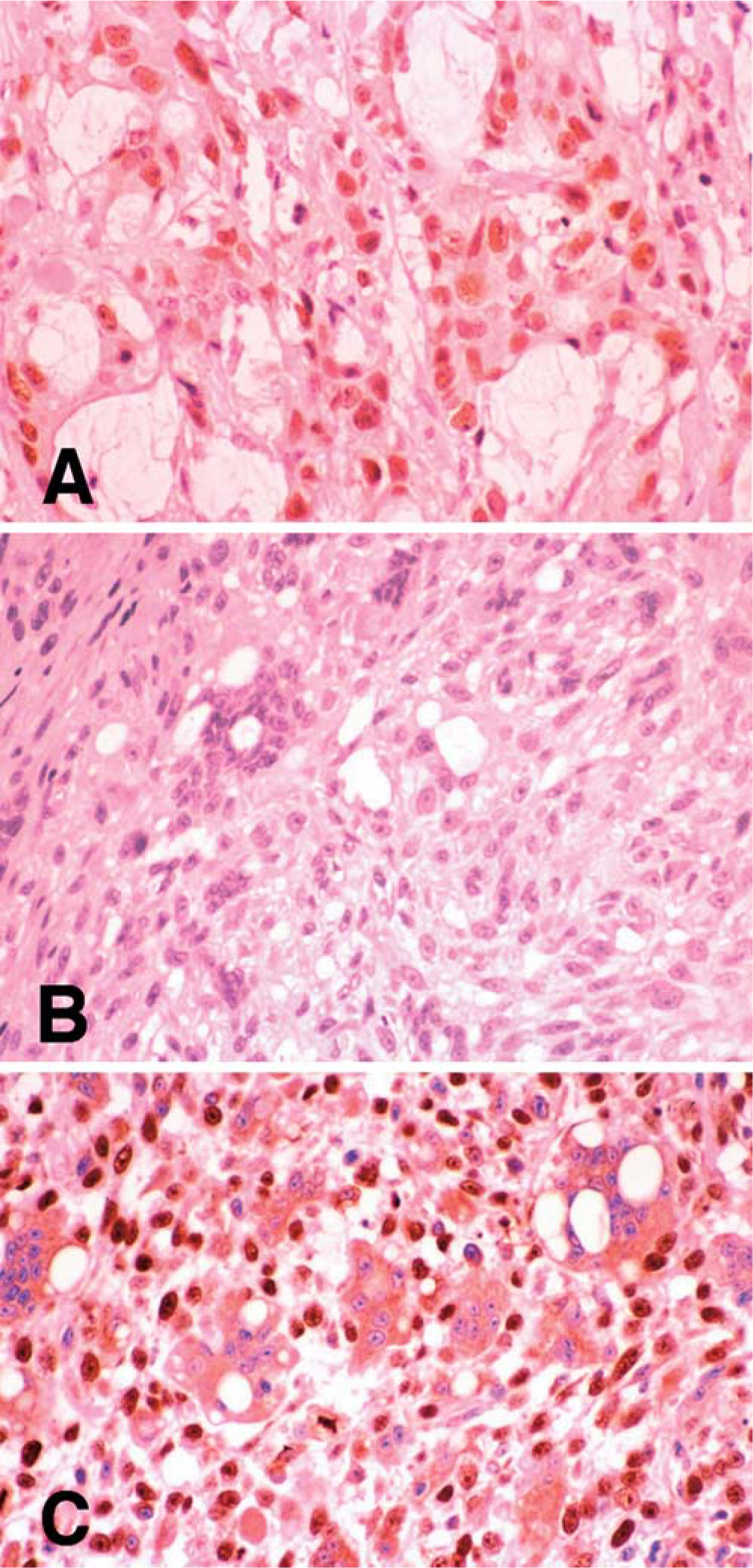
Human colon carcinoma and feline vaccine-associated sarcoma stained with Mab240.
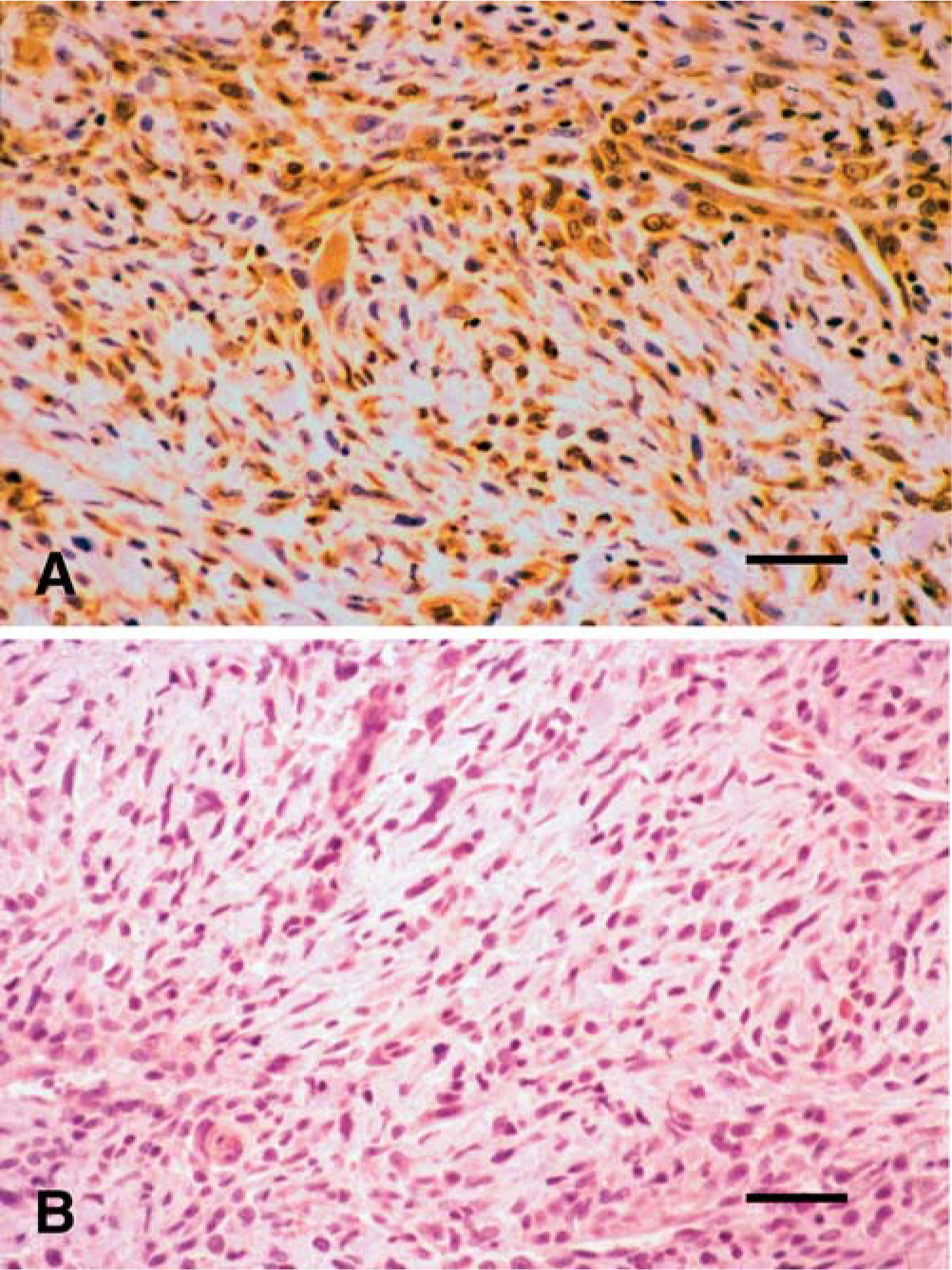
Feline vaccine-associated sarcoma, demonstrating contrasting results between CM-1 and Mab240.
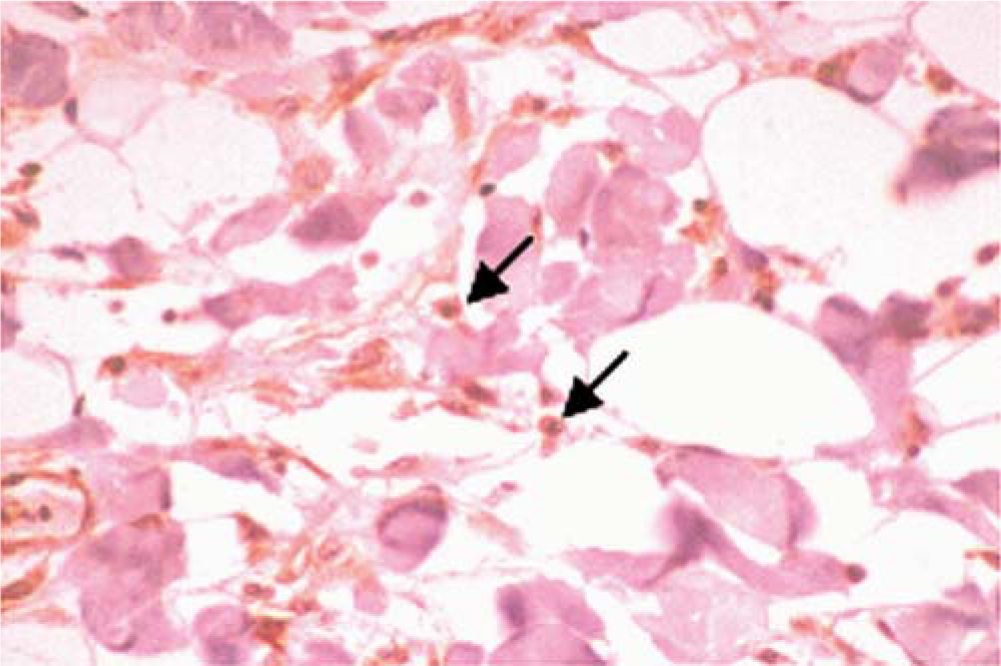
Feline vaccine-associated sarcoma stained with Mab240. Histologically normal mesenchymal cells peripheral to the tumor in Fig. 2 exhibit positive immunoreactivity with Mab240, suggesting the possibility of a “field cancerization” effect in this cat. Immunhistochemistry performed with streptavidin–biotin–peroxidase complex method, developed with 3,3′-diaminobenzidine tetrachloride, and counterstained with Tacha's hematoxylin.
Of the three cell lines evaluated, one demonstrated negative immunoreactivity, one positive cytoplasmic staining, and one positive nuclear plus cytoplasmic staining. The negative sample had previously exhibited positive nuclear immunoreactivity on the paraffin section of the tumor from which the cell line was derived. However, the results for the other two tumors were consistent with those obtained on the paraffin sections. The positive control stained appropriately and the negative controls failed to stain as expected.
In the subset of cats (n = 61) for which the clinical outcome was known, 15 there was no significant difference in time to recurrence or overall survival between p53-positive and p53-negative tumors. However, a significant difference (P = 0.0284) was determined in time to recurrence between cats exhibiting cytoplasmic p53 immunoreactivity versus nuclear p53 immunoreactivity. Those cats with positive cytoplasmic p53 staining had tumors recur earlier postsurgery (135 days) than those cats with tumors exhibiting positive nuclear staining (325 days; Fig. 4). No significant difference was observed in overall survival between cytoplasmic versus nuclear p53 staining. No significant difference was observed in either time to recurrence or overall survival between cats exhibiting positive cytoplasmic p53 staining versus those with p53 negative tumors.
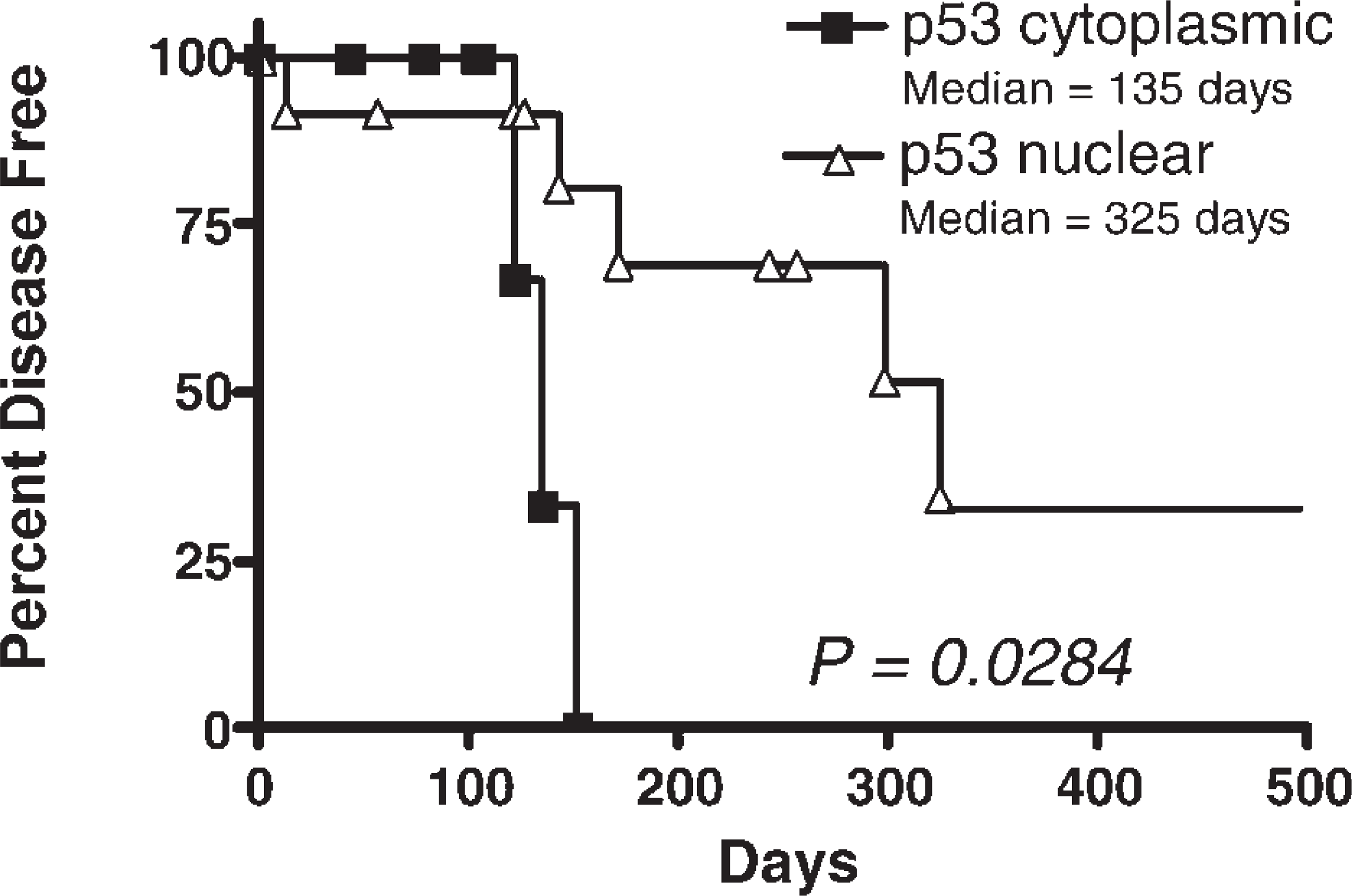
Kaplan-Meier curve, illustrating time to recurrence for cats with cytoplasmic expression of p53 versus cats with nuclear expression of p53. Cats with cytoplasmic p53 positive tumors had significantly earlier (135 days) time to recurrence postsurgery than cats with nuclear p53 positive tumors (325 days, P = 0.0284).
Discussion
Abnormal p53 expression in feline VAS appears to be a common feature of this cancer. Fifty-five percent of VAS stained positive with CM-1, a polyclonal antibody. Eighty-one percent of VAS exhibited positive immunoreactivity with Mab240, a murine monoclonal antibody that specifically recognizes mutant p53 in humans. 9 On the basis of the higher percentage of tumors exhibiting positive immunoreactivity with Mab240 and the reduction in staining of nonneoplastic tissues, Mab240 must be considered superior to CM-1 for the detection of abnormal p53 in feline VAS.
Cytoplasmic staining for p53 is a consistent pattern of VAS stained with Mab240, occurring in 45% of tumors evaluated. It is possible that the large percentage of cytoplasmic staining observed with this antibody is artifactual; however, numerous studies in human tumors have confirmed abnormal cytoplasmic expression of p53 to be commonly seen in cancer. The overexpression of p53 is usually the result of a mutation, which results in an altered molecular conformation, a prolonged protein half-life, and subsequent abnormal accumulation within the cell. Variations in mutations, and thus in conformational effects 1 and biological activity, 12 may alter the ability of p53 to act as a transcription factor 30 or to bind to heat shock protein 70 (hsp70). 12 Heat shock protein 70 is a molecular chaperone molecule capable of binding to mutant p53 and consequently could regulate its accumulation, alter its subcellular localization and distribution, and result in sequestration within the cytoplasm. 10, 27 Cytoplasmic expression of p53 may represent a functional inactivation pattern. 7 Inability of p53 to translocate to the nucleus presumably prevents its transactivating function, and subsequently its ability to act as a suppressor. 26 Cytoplasmic expression of p53 has also been reported in human cancers 20, 33, 34 and has been correlated with a poor prognosis in colorectal cancer 6, 34–36 and breast cancer. 35 Similarly, we have demonstrated that cytoplasmic expression of p53 is associated with a worse prognosis in feline VAS. Cats with tumors that exhibited cytoplasmic p53 had significantly shorter time to tumor recurrence compared to those cats with tumors that exhibited nuclear p53 staining.
An unexpected observation was the staining of normal-appearing mesenchymal cells in the peripheral tissues of tumors demonstrating abnormal p53 expression. VAS are often poorly encapsulated, infiltrating along fascial planes. Thus, these positive-staining peripheral mesenchymal cells may represent microscopic extension from the primary tumor. However, these cells appear histologically benign in comparison to positive-staining neoplastic cells. The most interesting explanation is that these normal-appearing mesenchymal cells may represent the phenomenon of field cancerization, which predicts that precancerous lesions, under the influence of a common carcinogen, exist in histologically normal tissue adjacent to tumors. 38 These lesions may proceed to develop into multiple new primary lesions and do not necessarily represent recurrence of the initial primary tumor. Field cancerization has been demonstrated in various human cancers, particularly squamous cell carcinoma of the head and neck 8 and lung carcinomas. 32 Preliminary work by Kanjilal and colleagues suggests that field cancerization may play an important role in the pathogenesis of VAS. 38 Mutations in p53 have been identified in surrounding histologically normal tissues up to 5 cm distant from the tumor. 18, 38 This may have important implications for prognosis and considerations for adjuvant therapy for VAS. Alternatively, as genetic predisposition likely plays a significant role in the tumorogenesis of VAS, these cells may represent an inherent somatic defect in p53 in certain cats.
This study demonstrates the abnormal expression of p53 to be a highly associated factor of VAS in the cat. Similar to reports of various human cancers, cytoplasmic expression of p53 is associated with a worse prognosis for tumor recurrence. Immunohistochemical detection of p53 is a simple technique that may offer an important prognostic tool for clients. Additionally, because there is a high frequency of abnormal expression in feline VAS, molecular targeting of mutant p53 may offer new therapeutic opportunities for this disease, particularly in those cats with tumors exhibiting cytoplasmic p53 staining.
Footnotes
Acknowledgements
The authors would like to acknowledge Dr. David Vail for statistical assistance and the following laboratories for submission of histologic samples: Marshfield Laboratories, Antech Laboratories, ARUP Laboratories, University of Pennsylvania Veterinary Diagnostic Laboratory, and the University of Wisconsin Veterinary Diagnostic Laboratory. This project was supported by a Companion Animal Research Grant from the University of Wisconsin–Madison School of Veterinary Medicine and by Ms. Virginia Harrington in memory of Scarlet.
