Abstract
The study of mutant mice with altered or deficient hematopoietic or hemostatic gene products provides a challenge to the researcher, particularly when genetic alterations lead to lethal phenotypes. The following review provides a framework for understanding murine hematopoiesis, based on work with mutant mice, and details experimental approaches used to evaluate these animals. Mice with deficiencies in hemostatic and fibrinolytic system proteins are discussed, and the investigation of their phenotypes is reviewed.
Embryonic stem (ES) cell technology, facilitating targeted murine gene deletion, replacement, or insertion, has permitted the generation of many mutant mice in the study of hematopoiesis, leukemia, the immune system, and hemostasis. Dissection of the phenotypes of such mutant mice requires the application of all instrumentation currently employed in the clinical pathology laboratory, including hematology, chemistry, and coagulation analyzers, and the histology laboratory (see Brayton and Montgomery, 11 this issue). This review details approaches to the study of murine hematology and coagulation in addition to those typically employed in the veterinary clinical laboratory setting. Virtually all studies relating to the phenotyping of mice appear outside the veterinary literature and are undertaken in a wide variety of laboratories. To better serve the scientific community as veterinarians, it is important to acquire expertise with these technologies and suggest appropriate approaches based on a thorough understanding of the scientific questions to be addressed by analysis of mutant murine phenotypes. The ability to reproduce diseases by recapitulating human genetic alterations in mice has resulted in the creation of numerous animal models of human disease. Previously, the association of the Philadelphia chromosome with human chronic myelogenous leukemia (CML) was of academic interest in veterinary pathology. The reproduction of this 1 43 45 50 116 and other diseases in mice has placed an onus on comparative pathologists to be cognizant of the pathogenesis of important human conditions.
Murine Hematopoiesis
Collectively, numerous studies of gene-deleted mice have provided a model for the ontogeny of hematopoiesis. Primitive hematopoiesis is initiated in the yolk sac around gestation day 7 (E7), with aggregates of mesodermal cells forming the extraembryonic blood islands. Such islands are comprised of internal erythropoietic elements and surrounded by cells that differentiate into endothelial cells. 2 Coexpression of proteins (Flk-1) by endothelial cells and hematopoetic pluripotential stem cells led to the hypothesis that a pluripotent “hemangioblast” exists with potential to differentiate into hematopoietic and endothelial cells. 21 101 In the Flk-1−/− mouse, primitive hematopoiesis and blood vasculature fail to develop, supporting this premise. 101 127 During organogenesis, definitive hematopoietic cells arise and proliferate in the anterior part of the aorta–gonad–mesonephros (AGM) region around E9 until E11/12, gradually replacing yolk sac–derived cells. 76 Lymphopoiesis arises in the closely associated intraembrionic paraortic splanchnopleura (PAS). 26 33 Extensive interchange of hematopoietic stem cells occurs through the blood vasculature, with secondary population of yolk sac and liver. The fetal liver takes over as the principal site of hematopoiesis around E12–14 until term, when the bone marrow and spleen become the quantitatively most important sites of hematopoiesis and the thymus and bone marrow the most important sites of lymphopoiesis. 26 33 A partial list of the phenotypes of gene-deleted mice employed in the study of hematopoiesis is provided (Table 1). Since most nonredundant genetic deletions with pronounced hematologic phenotype are either lethal or have reduced viability, novel approaches have been developed to understand the mechanisms of these phenotypes. Subtle phenotypic alterations may also be detected by sensitive methodologies in mice with apparently normal peripheral blood and marrow cytology.
Selected phenotypes of mutant mice important in hematology and hematopoiesis.∗
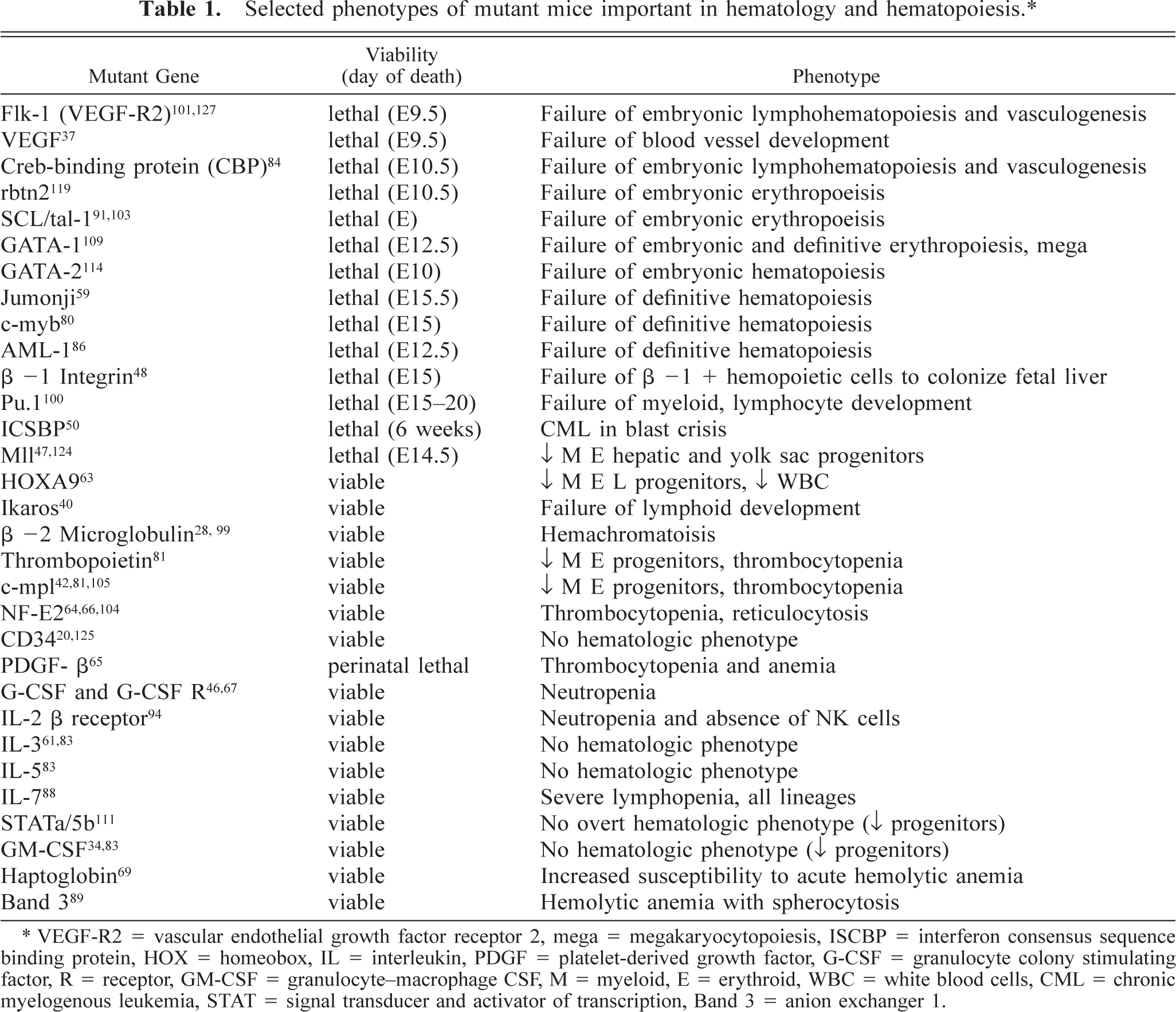


VEGF-R2 = vascular endothelial growth factor receptor 2, mega = megakaryocytopoiesis, ISCBP = interferon consensus sequence binding protein, HOX = homeobox, IL = interleukin, PDGF = platelet-derived growth factor, G-CSF = granulocyte colony stimulating factor, R = receptor, GM-CSF = granulocyte-macrophage CSF, M = myeloid, E = erythroid, WBC = white blood cells, CML = chronic myelogenous leukemia, STAT = signal transducer and activator of transcription, Band 3 = anion exchanger 1.
Approaches to Understanding Lethal Phenotypes
Once inbred to homozygosity at the target gene, murine hematopoietic gene mutations frequently prove to be lethal phenotypes. Creative alternatives have been developed to understand the nature of the defect leading to embryonic or fetal death. Genetic approaches involving deletion or reinsertion of whole or mutated genes into mice under tissue-specific promoters (Cre-loxP system) are used to study normal and aberrant hematopoiesis. 8 54 86 Approaches to the knocking in and out of genes and transgenes involved in murine hematopoiesis has recently been reviewed in detail. 87
Examination of Fetal Blood and Liver
Blood smears prepared with 2 µl of heart blood and Wright-Giemsa–stained liver impressions made from E12+ fetuses are used to evaluate fetal blood and bone marrow morphologies, respectively. Mutant mice with defects resulting in impaired definitive hematopoiesis contain far more nucleated red cells in peripheral blood after E12 than wild-type mice. Embryonic (yolk sac–derived) hematopoiesis does not yield mature erythrocytes. By E12, enucleated discocytes appear in peripheral blood. By E14.5, approximately 80% of erythrocytes in peripheral blood are enucleated. In mutant mice with defects affecting primitive hematopoiesis, only 20% of the circulating erythron may be enucleated at E14.5. 59 Primitive erythropoiesis is further distinguished in mice by the specific expression of an embryonic type of globin in nucleated erythrocytes.
Bone Marrow Transplantation
To evaluate the long-term repopulating hematopoietic stem cells (LTR-HSC) and multipotent hematopoietic progenitors (colony-forming units—spleen, CFU-S) of lethal mutants, cell suspensions of yolk sac, AGM region, or liver are cultured for 2 to 3 days followed by dispersion. These cells are injected intravenously into lethally irradiated mice exposed to 1,000 rads, or approximately 9 Gy of a 60C source. Generally, male cells are injected into female recipients. Following a period of recovery in isolators during which antibiotics are provided in the drinking water, mice are evaluated at 11 to 12 days postinjection for CFU-S and after several months for HSC. Hematopoietic clones form nodules readily enumerated on the surface of Bouin's fixed spleens. Splenic weight correlates highly with CFU-S. Long-term survival of lethally irradiated, transfused mice requires adequate HSC to be present. Periodic blood collections provide leukocyte DNA for PCR quantification of male determinants (YMT2/B) and construct inserts (typically the neomycin gene) or cytoplasmic protein for glucose 6-phosphate isomerase (GPI) isozyme patterning. 76 These markers allow the determination of the relative contribution of engrafted HSC to marrow lineages. Routine hematology and bone marrow cytology are also evaluated. In addition to stem cell analyses, bone marrow transplantation allows defects in marrow stroma to be differentiated from effects on hematopoietic elements, such as the stromal cell defect (defective c-kit or stem cell factor receptor) in Sl/Sld (Steel anemia) and the complementary defect in W/Wv (white spotted) mice (kit ligand or stem cell factor [SCF] deficiency). 32 51 The sites of definitive lymphohematopoiesis and, therefore, of LTR-HSC generation (AGM and PAS regions) were identified by transplantation into lethally irradiated mice. 26 33 63
Bone Marrow Cell Culture
Three main bone marrow cell culture methodologies are used to evaluate hematopoiesis in vitro.
ES cells may be differentiated into hematopoietic lineages when cultured in the absence of fibroblast feeder cells or leukemia inhibitory factor (LIF) with inclusion of hematopoietic cytokines. Embryoid bodies develop in such cultures after 9 to 14 days. 9 36 57 These are dispersed by collagenase and subsequently cultured in semisolid methylcellulose-based media as described below. 85 86
Numbers of committed progenitor cells (those cells not differentiated by conventional cytology) in the hematopoietic tissues of yolk sac, AGM region, liver, or dispersed embryoid bodies from lethal mutants are best assayed in semisolid, methylcellulose-based tissue culture media. 126 Cells are plated with hematopoietic cytokines (erythropoetin, G-CSF, GM-CSF, IL-1, IL-3, IL-11, SCF, etc.) for 7 to 9 days. Committed progenitor cells named for their morphology in tissue culture are visually enumerated based on appearance in phase contrast or direct illumination microscopy. These colonies carry abbreviated designations such as CFU-E (erythroid), BFU (blast-forming unit)-E, CFU-GM (granulocyte macrophage), CFU-Emega (erythroid megakaryocytic), CFU-GEMM (multilineage), etc. The colony designation is periodically checked by selecting and smearing cell clusters, then evaluating their morphology after staining with Wright-Giemsa.
Long-term bone marrow cultures more closely recapitulate the hematopoietic microenvironment and may be used to assay for the presence of stem cells with potential for self-renewal. 32 52 93 The Dexter culture system is commonly employed. This system utilizes an irradiated layer of fibroblasts, upon which bone marrow is seeded and cultured together with a complete media containing hematopoietic cytokines. 93
First Generation Chimeras
Murine offspring generated by blastocyst injection of homozygous negative ES cells that have the brindled coat color combination of both parent (C57BL/6, black) and ES cell (SV129, agouti) strains are usually viable chimeras. The contribution of mutant cells to different tissues is a random process. The complete absence of mutant cells from a tissue of several chimeric mice suggests that the targeted gene is required for normal differentiation of that tissue. The isoenzyme differences between C57BL/6 (GPI-1B) and SV129 (GPI-1A) mice for the GPI gene are readily exploited to establish mutant versus parent cell contribution in hematopoietic and lymphoid tissues. After electrophoretic separation of tissue homogenate on cellulose acetate, membranes are incubated with substrate and color reagent to reveal lighter (1A) and heavier (1B) bands of GPI. 5 85 92 101
Complementation Chimeras
Mice with severe combined immunodeficiency (SCID) are deficient in functional T and B cells and are therefore unable to reject xenogeneic organ grafts. Acute lymphoblastic (ALL), myeloblastic (AML), CML, or chronic lymphocytic leukemic (CLL) cells induce overt disease when injected into SCID or NOD (nonobese diabetic)-SCID mice, reproducing patterns reminiscent of the human disease. 1 45 116 The absence of lymphocytes in SCID and Rag (recombinase activating gene)-deficient mice may be exploited in complementation assays. ES cells with mutations that produce embryonic or fetal lethality may be inserted into SCID or Rag−/− blastocysts. Lymphocyte populations in resultant chimeras will derive solely from injected ES cells. Complementation chimeras may also be created by injecting the blastocyst of a lethal mutant with modified ES cells used to generate the same mutant. 85 Gene insertions in these ES cells are aimed at restoring normal hematopoietic function. The insertion of complementation genes together with the gene for bacterial β-galactosidase (lacZ) enables ready localization of lacZ-containing cells based on the blue color reaction obtained when lacZ is incubated with substrate. 101 This approach was used to analyze the angiogenic and hematopoietic functions of Flk-1. 101
In Vivo Stimulation of Hematologic Alterations
Commonly used approaches to study accelerated hematopoiesis in mice include treatment with 5-fluorouracil (5FU, 150 mg/kg/day IP) or phenylhydazine (60 mg/kg IP). 27 71 The former treatment effaces all dividing marrow elements, and cytopenias result from acute marrow ablation. Phenylhydrazine causes an acute Heinz body hemolytic anemia; recovery occurs with intact bone marrow. 76 110 Alternatively, a fixed volume of blood (to 3% of body weight) may be removed from a rodent and replaced with intraperitoneal saline, stimulating erythropoiesis. Many mice with deficiencies of leukocyte adhesion molecules or chemotaxis receptors are leukocytotic. Due to extensive redundancy among adhesion molecules, the expected leukocytotic reactions may not manifest (Table 2). To assess the mobilization of granulocytes and accelerated granulopoiesis, Bacto-tryptone (a potent chemotactic casein digest), thioglycollate, or specific chemotactic agents may be administered interperitoneally 39 75 with subsequent enumeration of peritoneal lavage cells. By intravenous dosing of G-CSF at a dose of 5 µg/kg, 5 recruitment of neutrophils from the marrow may be assessed. To determine if leukocytosis relates to increased production or decreased margination, unanesthetized mutant and wildtype mice are injected intravenously with epinephrine (0.25 mg/kg) to demarginate cells. Blood is collected 35 minutes postinjection from the tail vein, and leukocytes are compared to baseline values. 56 60
Mutant mice with potential leukon alterations.∗

ICAM = intracellular adhesion molecule.
Murine Erythrokinetics
When interpreting changes in the murine erythron, consideration must be given to the short lifespan of the mouse erythrocyte (approximately 20 to 30 days), relative to larger domestic species and humans. 44 Reduction in progenitor or precursor pools or increased turnover of mature red cells are, therefore, more rapidly reflected in anemia in mice.
Uptake of iron into hemoglobin is determined from the half-life of intravenously injected 59Fe. 10 Plasma samples are taken at regular intervals up to 50 minutes after injection and counted in a scintillation spectrometer. Radioactivity is also measured on 16-µm-thick cryosections using a ß-particle imaging instrument. To determine erythrocyte life span, approximately 1 × 109 cells are fluorescently labeled with 5-chloromethylfluorescein diacetate (CMFDA) and injected into the tail veins of recipient mice. Blood samples are collected at various times up to 30 days postinjection and analyzed by flow cytometry for the percentage of labeled erythrocytes. Red blood cell (RBC) survival curves are constructed by plotting the circulating labeled cells as the percentage of the number of circulating erythrocytes at 2 hours postinjection of CMFDA. 66
Flow Cytometry
The flow cytometric evaluation of murine bone marrow and fetal liver has become standardized (Table 3). 98 126 From the differential count obtained by flow cytometry or cytologic examination and femoral bone marrow cellularity (approximately 15 to 30 million cells per normal mouse femur), the absolute numbers of identifiable femoral precursor cells may be readily quantified. Since the cells of a single femur contain approximately 6% of the total murine marrow elements, an estimate of absolute numbers of precursors per mouse may also be calculated (approximately 250 to 500 million hematopoietic cells per mouse). 120 Except for rare reports, 54 basophils are generally considered to be absent from the peripheral blood of mice and are best evaluated by bone marrow cytology or flow cytometry. 7 To reduce background fluorescence related to receptor binding of the Fc portion of immunoglobulins, bone marrow cells are commonly blocked with antibodies to the two Fcγ receptors, CD16 and CD32.
Cellular markers routinely used in flow cytometry.∗
BM indicates this antigen is preferred for use with bone marrow cells.
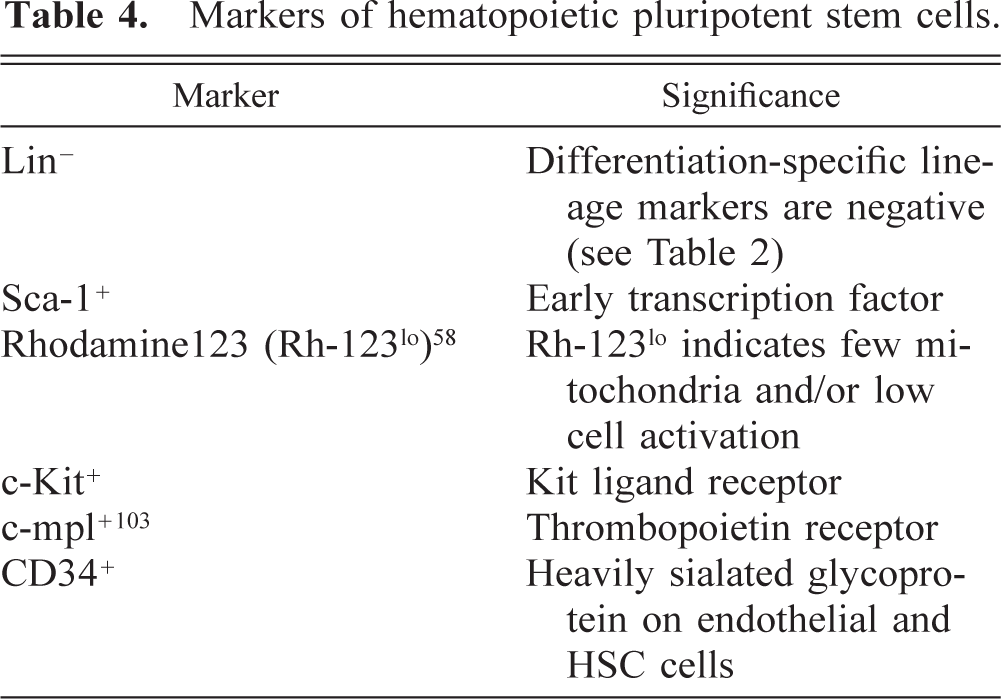
Pluripotent hematopoietic stem cells are frequently studied in mutant mice. The identification of markers for these cells will ultimately provide tools for transplantation of these cells with minimal contamination by nonpluripotent cells. HSCs are currently defined by their ability to repopulate the bone marrow of lethally irradiated mice (see above) and by a series of antigens or properties of cells revealed by flow cytometric evaluation (Table 4).
Markers of hematopoietic pluripotent stem cells.
Murine Hemostasis
Megakaryocytes and platelets
Megakaryocytopoiesis, thrombopoiesis, and platelet function have been studied extensively in mutant mice. Alterations in megakaryocyte and/or platelet function commonly manifest as thrombocytosis or thrombocytopenia.
Thrombocytopenia in mutant mice results from increased consumption or decreased production of platelets. The assessment of decreased production is best ascertained by cytologic or histologic examination of marrow or other hematopoietic tissue or, alternatively, culture of hematopoietic tissue for megakaryocytic progenitors. Increased consumption is generally linked to increased production, for which mean platelet volume (MPV) is a sensitive indicator in mice. Following experimental acute induction of thrombocytopenia in mice or dosing with thrombopoietin, the MPV increases from 4.7 to 8.2 fl, peaking at 36 hours and returning to normal by approximately 3 days. 81 117 A variety of methods are utilized to establish the cause of thrombocytopenia. Splenectomy allows the assessment of the effects of hypersplenism, altered monocyte–macrophage phagocytic activity, or immune-mediated thrombocytopenia on platelet turnover. Platelet life span is readily assessed in mice. Platelets are fluorescently labeled with CMFDA as described for red blood cells 4 66 and injected into the tail veins of recipient mice. Blood samples are collected periodically for 4 days and analyzed by flow cytometry to determine the proportion of labeled platelets present at each time point. Alternatively, platelets may be biotinylated by in vivo injection of sulfo-NHS-biotin (35 mg/kg), with platelet disappearance studied periodically as for CMFDA. 7 24 The life span of platelets in mice relative to other species is short (∼5 days). 3 Induction of thrombocytopenia is used to study accelerated megakaryocytopoiesis and thrombopoiesis in mice. To this end, antiplatelet antiserum or 5FU is administered intraperitoneally, and recovery of platelet numbers to pretreatment values is evaluated. 106
To assess thrombopoiesis cytologically, blood is collected by cardiac puncture, and the morphology of platelets (discoid, elongated, beaded) in platelet-rich plasma 19 is evaluated by phase contrast microscopy at 400× magnification. 65 The total percent of nondiscoid platelets is considered the proplatelet fraction.
Megakaryocyte ploidy is commonly assessed in targeted mutants with altered megakaryocytopoiesis or mice treated with thrombopoietic cytokines. The ploidy distribution of cells is determined by flow cytometry following prolonged incubation (∼24 hours) with propidium iodide. 4 109 Consistent with most species examined, the mean ploidy of murine megakaryocytes is 16N. This increases with acute thrombocytopenia and is higher in certain mouse strains such as the C3H. 53 73 74 Given the large capacity for increasing production, increased megakaryocyte ploidy 53 or mean platelet volume 23 may be the only indicators belying compensated, increased platelet turnover in mice.
Intravascular activation of platelets may cause increased platelet clearance and result in thrombocytopenia. The activation state of platelets may be assessed by flow cytometric evaluation of the expression of surface P-selectin and fibrinogen binding by GpIIbIIIa. 78 Flow cytometric approaches for evaluation of platelets utilize low volumes (µl) of whole blood and are ideal for use with mice. This approach also will readily detect changes in MPV or reveal subpopulations of large platelets observed during states of accelerated thrombopoiesis. Urinary assays for metabolites of thromboxane-A2 (TXA2), such as TXB2, and plasma assays for β-thromboglobulin provide an indirect assessment of in vivo platelet activation.
Platelet aggregation may be evaluated in platelet-rich plasma or whole blood from mice. Adenosine diphosphate (ADP), collagen, arachidonate, and thrombin are potent agonists of platelet aggregation in the mouse and are potentiated by heparinized versus citrated plasma. In contrast to their effects in other species, epinephrine, serotonin and platelet activating factor (PAF) do not cause aggregation of mouse platelets. 77 97 115
Bleeding time assays, as for higher species, are also used to assess platelet function in mice. However, unlike higher species, bleeding times in mice are also markedly prolonged by factor deficiencies that decrease thrombin generation (Table 4). 115
Murine blood coagulation and fibrinolysis
In general, mechanisms of blood coagulation and fibrinolysis are common to domestic animal species and laboratory rodents, with limited differences. Mice deficient in most proteins known to be directly involved in human coagulation and fibrinolysis have been generated (Table 5). The human and domestic animal phenotypes of deficiencies of proteins involved in blood coagulation and fibrinolysis are among the best understood of inborn errors, lending the comparison to the phenotypes of orthologous murine mutants of particular interest. Murine hemostasis has recently been extensively reviewed. 6 115
Selected phenotypes of mutant mice important in hemostasis and fibrinolysis.∗
t-PA = tissue plasminogen activator, u-PA = urokinase plasminogen activator, PAI-1 = plasminogen activator inhibitor-1.
Most important in the evaluation of mutants with suspected or known hemostatic abnormalities is the careful assessment of fetuses or mice for macroscopic and microscopic evidence of hemorrhage or tissue infarction. In addition to tests of platelet function described above, standard tests for coagulation factors (prothrombin time, activated partial thromboplastin time, factor assays) may be readily applied to murine plasma samples. Murine fibrin degradation products and
General Technical Considerations
The application of typical laboratory instrumentation to the assessment of mutant mouse hematology and hemostasis is valuable but must be undertaken cautiously. Where specific software settings are available for mice, they should be used. Frequently, mutations causing altered cell size or differentiation (e.g., murine GpIb deficiency 118 ) result in incorrect or overlapping gating of cell populations by analyzers—blood smears should always be evaluated during initial phenotyping to confirm instrument interpretations. The volume of blood or other samples collected from mice will often fall below the dead volume of laboratory instrumentation and require dilution. Potential artifacts of linearity with dilutions of small quantities can be overcome by comparing results from treated or genetically altered mice with similarly diluted volumes from control or wild-type mice, permitting valid conclusions.
The technology that permits the generation of mice deficient in single genes has provided the tools necessary to probe hematopoiesis and hemostasis in great detail. Given the small size of mice and the frequency with which embryonic lethal or unexpected phenotypes manifest, experimental pathologists must be knowledgeable and creative in exploring the pathophysiology of observed effects.
Footnotes
Acknowledgements
E. Calvert is gratefully acknowledged for preparation of citations.
