Abstract
The literature on hemostatic processes in swine is sparse and often fragmentary; hence, we conducted our study to characterize age-related changes in selected parameters of primary and secondary hemostasis in 50 growing pigs between day 2 and week 24 of age. We measured platelet count (PLT), mean platelet volume, platelet-to-large cell ratio, prothrombin time, activated partial thromboplastin time (aPTT), thrombin time (TT), and fibrinogen concentration. Among primary hemostasis parameters, PLT underwent the largest fluctuation with the animals’ age, ranging from 340 to 730 × 109/L. However, statistical significance was only detected for 4-week-old piglets compared to 18-week-old animals. Of the secondary hemostasis parameters measured, TT and aPTT were the most changeable. Activated partial thromboplastin time displayed a characteristic biphasic course, being relatively short before week 5 of age (17.8–19.9 s) and then becoming much longer (28.7–52.5 s). The aPTTs measured in animals 6 weeks of age and older were statistically different (p < 0.01) from those in younger piglets. The 2 main components of hemostasis, platelet hemostasis and plasma coagulation, did not develop at the same time. It took much longer for secondary hemostasis to stabilize, whereas platelet parameters were stable early in life.
Intensification of pig production and, consequently, very rapid growth rates required for efficient weight gain, have a great impact on physiologic functions in swine. An increased pace of the development of internal organs is frequently accompanied by a marked unevenness in their growth and maturation. Some breeds of pigs attain sexual maturity as early as 9–15 weeks after birth. During this time, the animals can increase their body weight ~82 times, whereas the increase of weight of the liver and spleen is reported to be 50 and 117 times, respectively. 7 Such asynchronous development may result in disturbances in some physiologic functions, including mechanisms of hemostasis. Hemostatic processes—a very complex system of blood clotting and fibrinolysis—play an important role in many physiologic and pathologic phenomena, including healing of damaged tissues, inflammatory reactions, and antimicrobial response. 12 Hence, the measurement of clotting parameters in growing pigs may be of practical importance for the monitoring of the animals’ health status.
Interest in hemostasis in swine has also increased because these animals have become a biomedical model for human studies.5,8,20,25 Swine have several advantages that make them promising subjects for biomedical research, including intensive growth and maturation, short gestation time, prolificacy, low rearing costs, and a relatively low risk of transmission of infectious diseases to humans.7–9,20 The swine model has been used in research into pathogenesis of human cardiovascular diseases, disseminated intravascular coagulation, shock, atherosclerosis, diabetes, metabolic disturbances, as well as hereditary disorders.5,8,14,20–22 Because the pig resembles the human in physiology and organ development, intensive studies on the possibility of using porcine organs for xenotransplantation to humans have also been performed.8,25
Considering positive aspects of the swine model, one should not forget the issue of species differences that impose some limitations.9,20,25 A quite important question, for example, is molecular incompatibility of some hemostatic processes in pigs and humans. 4 The direct application of results obtained from a swine model in the therapy of human diseases may lead to serious disturbances of clotting and fibrinolytic processes, including thromboembolic risk, posing a direct threat to the health or even the life of the patient.4,9 This fact imposes the need for thorough understanding of hemostasis mechanisms in swine.
There are several reports concerning both primary (platelet) and secondary (plasma) hemostasis in pigs, based on experiments carried out on pigs of various ages and different genetic lines.5,6,11,13,14,17,18,22,24 Most of the data refer to hematologic and plasma biochemistry parameters in pigs, and often only single parameters of hemostasis have been addressed. Moreover, the current knowledge of swine hemostasis seems to be insufficient for the broader use of a swine model for biomedical research. The aim of our study was therefore to provide information on the development of hemostatic processes in growing pigs. In particular, we focused on the basic parameters of both primary and secondary hemostasis that might be of value for either the monitoring of animal health or comparative assessment of similar processes in humans. All procedures were approved by the Local Ethics Committee at the University of Environmental and Life Sciences, Wroclaw, Poland (permission 58/2011).
The investigation was performed on 50 crossbred (Polish Large White × Polish Landrace) pigs (25 male, 25 female), randomly selected from 14 litters on a commercial farm in south Poland. All the animals were clinically healthy. The pigs were subject to routine husbandry and veterinary treatment performed on the farm (injection of iron dextran on day 1, vaccination against Porcine circovirus 2 on day 28, and deworming with an oral solution of flubendazole on day 60 of life). During the experiment, pigs were fed age-appropriate feed ad libitum. They had free access to water from nipple drinkers. The animals were reared in buildings with optimal humidity and temperature, using a litter-free system. In the farrowing units, piglets were housed with their dams in slatted pens. After weaning, animals were moved to a nursery unit and kept in slatted pens in groups of 10 pigs. In the fattening unit, they were kept in slatted pens in groups of 25 animals. Health status of the animals was checked by clinical examination, performed every 2 weeks, and by determination of total leukocyte count in each blood sample. Additionally, in order to monitor the weight gain in pigs, their body weight was measured on the following days of life (control days): days 2 (litter standardization), 28 (the day of weaning), 70 (transfer to the fattening sector), and 168 (the end of investigation and shipment to a slaughterhouse).
Blood samples were collected on day 2 and in weeks 3, 4, 5, 6, 8, 10, 18, 20, and 24 of life during the morning (8:00–10:00
Blood was drawn from the external jugular vein using 10-mL polypropylene disposable syringes a with 21 gauge × 3.8 cm needles b (in 2-day-old piglets) or 16–19 gauge × 3.8 cm needles b (in older pigs). Blood specimens were immediately divided into 2 portions as described in the literature.1,6
The first portion of blood (1 mL) was transferred into plastic tubes with K2-EDTA (dipotassium salt of ethylenediaminetetra-acetic acid). c The second portion (4.5 mL) was transferred into plastic tubes d containing 3.8% sodium citrate (blood:citrate 9:1 v/v). Samples were promptly mixed with anticoagulants by gently rocking the tubes. Within 3 h of venipuncture, samples were delivered to the laboratory and prepared for analysis. All tests were performed on the day of blood collection. Before analysis, blood samples were visually inspected for possible clotting, and hemolysis-coagulated or hemolyzed samples were rejected. All parameters were measured in duplicate by the same investigator.
Blood with K2-EDTA was used to assess the following platelet parameters: platelet count (PLT), mean platelet volume (MPV), and platelet-to-large cell ratio (P-LCR). Assessments were performed with an automated impedance-based hematology analyzer, e calibrated using a blood control. f Large platelets were defined as those with size ranging between the 15-fL discriminator (set by the analyzer’s manufacturer) and the upper discriminator. The P-LCR (expressed as a percentage) is a ratio of the large platelets to the total number of particles ranging between the lower and upper discriminators.
Citrated blood was centrifuged at 2,500 × g for 10 min. The plasma obtained was transferred with a plastic pipette into plastic 2-mL tubes d and analyzed for the following coagulation parameters: prothrombin time (PT), activated partial thromboplastin time (aPTT), thrombin time (TT), and fibrinogen concentration. All determinations were performed with a coagulometer, g measuring the total change in sample absorbance at a wavelength of 405 nm. The coagulometer was calibrated using pooled pig plasma obtained by mixing plasma aliquots from 40 healthy pigs. All reagents used were reconstituted just before analysis, according to the manufacturer’s instructions.
For determination of PT, 50 µL of plasma were preheated at 37°C for 60 s in the coagulometer heating box. Subsequently, 100 µL of a reagent, containing recombined rabbit brain thromboplastin (International Sensitivity Index = 1.07), synthetic phospholipids, and calcium chloride, g were added. The time (in seconds) from plasma–reagent mixing to clot formation was defined as the PT.
For aPTT determination, 50 µL of plasma with 50 µL of a mixture containing colloidal silicon and synthetic phospholipids g were preheated at 37°C for 3 min. Next, 50 µL of calcium chloride preheated to 37°C were added. The time (in seconds) elapsed from mixing of the preincubated plasma with calcium chloride to clot formation was defined as the aPTT. To measure TT, 100 µL of bovine thrombin (3.8 U/mL) g were added to 100 µL of plasma preheated for 2 min at 37°C. The time (in seconds) between mixing of the plasma with thrombin and clot formation was defined as the TT. The concentration of fibrinogen was read from the PT calibration curve created for pooled pig plasma.
Statistical analysis was performed using statistical software. h All results are expressed as means and standard deviations. For the examination of data distribution, the Shapiro–Wilk test was applied. Differences between means were determined by the Tukey test. P values <0.05 and <0.01 were considered to be statistically significant.
The mean daily gains of body weight in suckling, nursing, and fattening periods were 204, 423, and 718 g, respectively. These normal daily weight gains were consistent with the good health status of the experimental animals.19,23 All total leukocyte counts measured during the experiment were within reference intervals reported for pigs (Table 1). 10
Total leukocyte counts in the pigs examined at consecutive blood-drawing times.*

Values are expressed as means ± standard deviations.
The PLT in piglets of different age groups varied considerably (Fig. 1). It rose from 422 × 109/L in 2-day-old piglets to the maximum of 730 × 109/L in week 4 of life, and then decreased to reach the minimum of 340 × 109/L in week 18. The highest PLT was statistically different from the lowest one (p < 0.05). From week 24 onward, PLT reached values observed in adult pigs (400–450 × 109/L). There were no statistical differences between PLTs in male and female pigs. In contrast, MPV was relatively stable throughout the experiment, with a range of 8.4–9.75 fL.
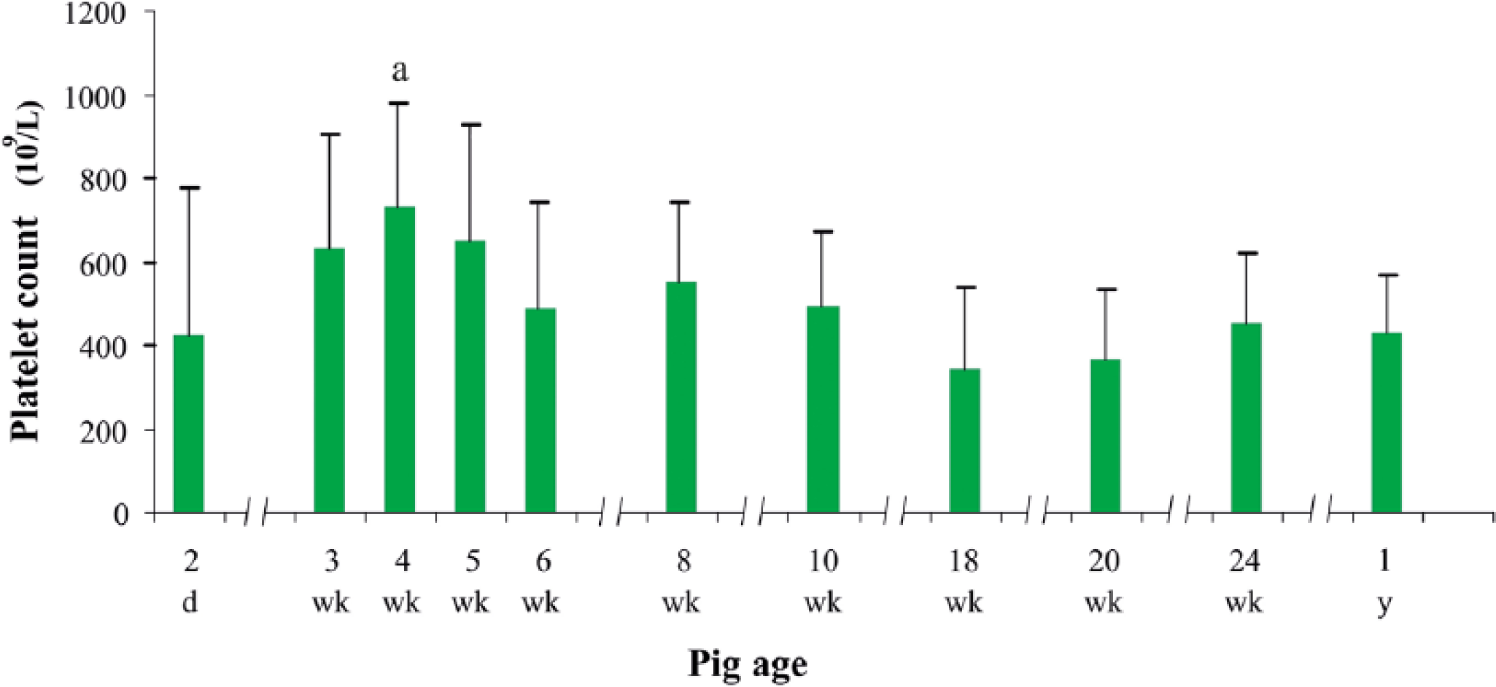
Platelet count in pigs of different ages. Data are expressed as means and standard deviations. a Statistically significant at p < 0.05 compared with 18-week-old piglets.
The P-LCR also displayed some fluctuation with animal age (Fig. 2). Initially, up to week 4 of life, the parameter declined from 19.5% to 12.7%. Then it remained within a range between 11.7% and 15.5%, with the minima in weeks 8 and 20. However, statistical differences were not detected.
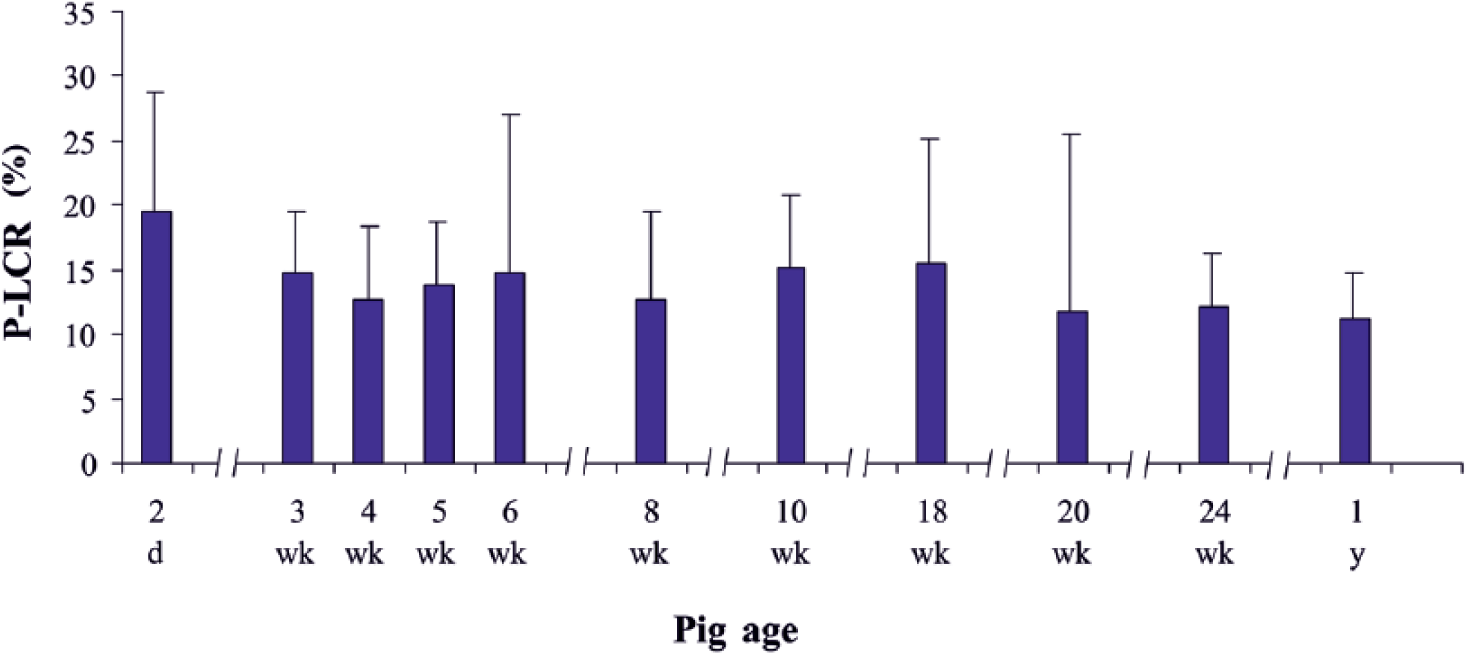
Platelet-to-large cell ratio (P-LCR) in pigs of different ages. Data are expressed as means and standard deviations.
A reverse relation between PLT and P-LCR was noted throughout most of the period analyzed (from day 2 to week 18). At this time, the increase in PLT was accompanied by a decrease in P-LCR. Only in the oldest pigs examined (at 24 weeks of age) did such opposite tendency not occur, and the increase in PLT coincided with an increase of P-LCR.
In our study, PLT differences in piglets of particular groups were accompanied by individual variability of this parameter in each experimental group, which was reflected in the high standard deviation. This observation is in accord with the data of others. 6 However, we detected that only 4-week-old piglets displayed statistically significant differences in PLT. Interestingly, a considerable degree of PLT instability in young pigs is also evident when analyzing results of other investigators. Some authors reported even higher PLT counts (753–785 × 109/L) in piglets between 3 and 8 weeks of age. 13 At the other extreme, much lower PLT counts (210–310 × 109/L) were reported in animals up to week 3. 6 Our results concerning the early stage of pigs’ lives were comparable to those observed in another study of 1–2-week-old piglets (532 × 109/L). 5 This PLT variability in young pigs may be attributable to intensive growth of these animals and maturation of their hematopoietic system. In the older piglets in our study, from week 6 onward, the PLT decreased to its lowest level of 340 × 109/L by week 18. These results were nearly the same as those reported by other authors for farm pigs14,18,22,24 as well as for Göttingen minipigs. 3 Interestingly, the latter report describes a similar rate of decline in PLT in pigs between weeks 8 and 21.
MPVs detected in our study (8.4–9.75 fL) lay within ranges described by other authors for large pigs and minipigs.6,26 It is commonly believed that under normal circumstances there is a reverse relationship between PLT and MPV. When PLT decreases, bone marrow megakaryocytes are activated by thrombopoietin to produce an increased number of large platelets. 2 However, we did not observe this reverse relationship. Fluctuations in the PLT count were accompanied by a relatively stable MPV. Similar observations were also made by a previous group. 6 Assessing PLT in piglets between day 1 and week 3 of life, the authors of the previous study observed that, in animals up to 2 weeks of age, platelets had nearly the same volume despite an increase in their count.
Among plasma coagulation parameters, PT observed in the present study was quite stable throughout most of the period. The longest PT (16.3 s) was observed in 2-day-old piglets and was statistically different (p < 0.01) from those determined in other age groups, except 8-week-old animals (Table 2). From week 3 of life, PT was shorter until the end of the observation period, with a range of 12.2–15.3 s. The shortest PT (12.2 s) was noted in week 6, and this result was statistically different (p < 0.01) compared to other age groups, except 4-week-old piglets. The parameter also displayed statistical differences (p < 0.01) between male and female pigs at weeks 4 and 5 of age.
Coagulation parameters in pigs at different ages.*
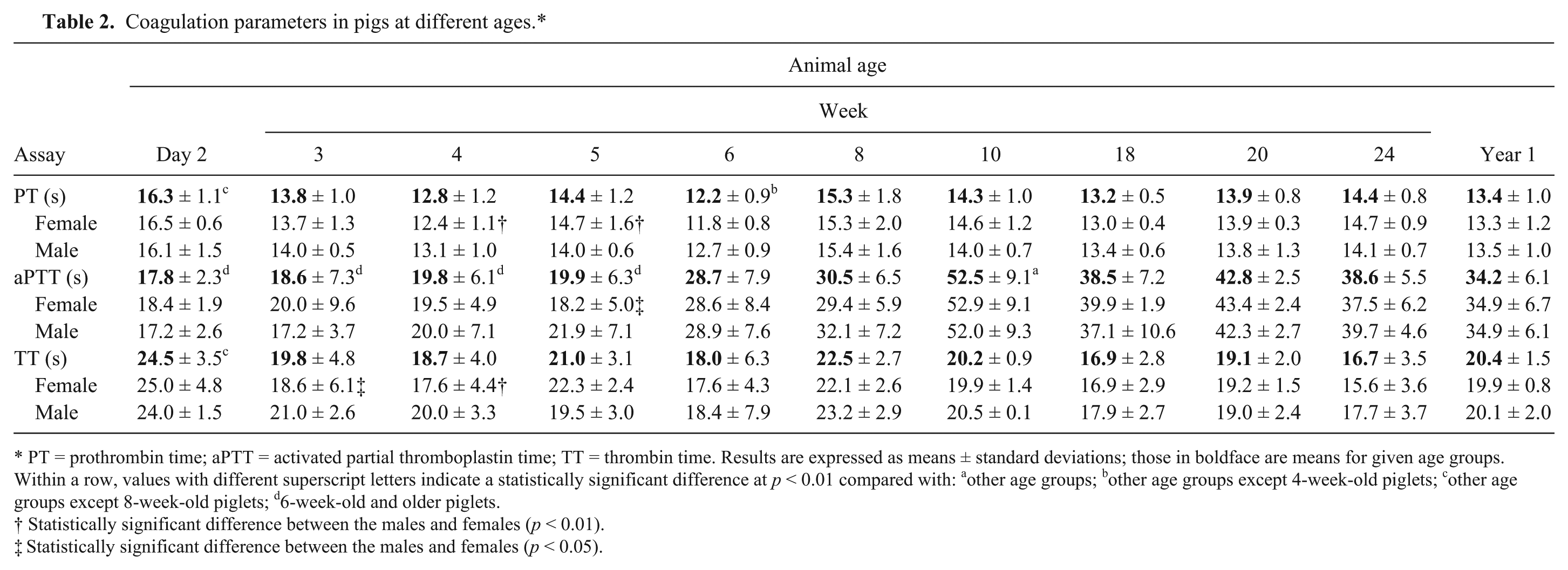
PT = prothrombin time; aPTT = activated partial thromboplastin time; TT = thrombin time. Results are expressed as means ± standard deviations; those in boldface are means for given age groups. Within a row, values with different superscript letters indicate a statistically significant difference at p < 0.01 compared with: a other age groups; b other age groups except 4-week-old piglets; c other age groups except 8-week-old piglets; d 6-week-old and older piglets.
Statistically significant difference between the males and females (p < 0.01).
Statistically significant difference between the males and females (p < 0.05).
Unlike PT, aPTT varied throughout the entire period of animal observation (Table 2). Up to 5 weeks of age, aPTT was very short, with a range of 17.8–19.9 s. Then it became longer, peaking at 52.5 s in week 10 and continuing to be ~40 s in older animals. All of the aPTT results measured in animals 6 weeks old and older were statistically different from those in younger piglets (p < 0.01). In addition, there was a statistical difference between male and female pigs at 5 weeks of age (p < 0.05).
In pigs at different ages, there was also marked fluctuation in the TT (Table 2), with a range of 16.7–24.5 s. In addition, in animals up to week 6, this parameter displayed a great variability in particular individuals. Statistically confirmed differences in TT (p < 0.01) were noted between day 2 of life and other age groups, except 8-week-old piglets, and between the males and females at weeks 3 and 4 of age (p < 0.05 and p < 0.01, respectively).
The plasma fibrinogen concentration also varied with animal age (Fig. 3). It was significantly lower (p < 0.01) on day 2 of life (1.9 g/L), than the values measured in other age groups. In older animals, concentrations of fibrinogen gradually increased to a range of 3.7–6.1 g/L.
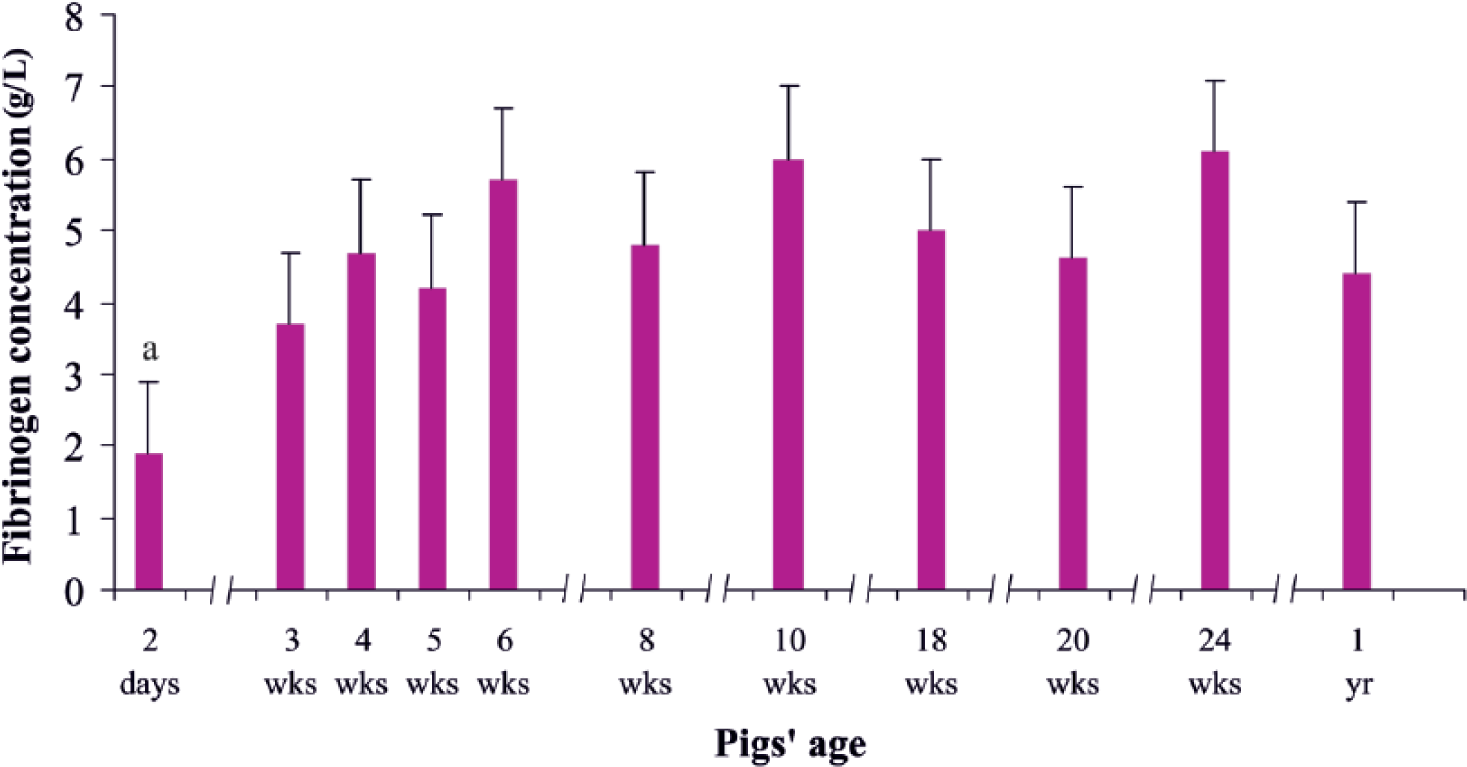
Fibrinogen concentration in pigs of different ages. Data are expressed as means and standard deviations. a Statistically significant at p < 0.01 compared with other age groups.
PT values, obtained in our study for piglets >3 weeks of age, corresponded roughly to results obtained by other authors in pigs of similar age (e.g., in animals weighing 41–45 kg [13.9 s] 18 and in 4- to 5-month-old pigs [13.1 s]). 22 In contrast, miniature Yucatan pigs exhibited shorter PTs of 11.7 s. 21 The initial prolongation of PT that we detected in 2-day-old piglets may have resulted from the low concentration of fibrinogen in newborn animals (1.9 g/L). Subsequent shortening and stabilization of PT values (especially evident from week 8 onward) may argue for the maturation of hemostatic mechanisms in the animals examined. As internal organs (particularly the liver) matured, concentrations of fibrinogen rose gradually to achieve a range of 3.7–6.1 g/L.
However, these fluctuations in fibrinogen concentration did not influence TT, as would be expected considering the prolonged PT in newborns with a decreased fibrinogen level. Instead, even if some age-related differences in TT were observed, they were not correlated with concentration of fibrinogen.
Contrary to TT, there was marked changeability of aPTT observed in growing pigs in our study. Initially, within the first 5 weeks of age, aPTT displayed a stable level (17.8–19.9 s). Then, it soared to very high levels (up to 52.5 s in week 10). The change described was probably caused by alterations in the activity of activators and/or inhibitors of the coagulation process, associated with intensive growth and maturation of pigs. Noteworthy, such high levels of aPTT have not been reported elsewhere. For example, in farm pigs aged ~20 weeks, aPTTs were 19.47 24 and 28.29 s. 22 An even shorter aPTT (15.4 s) was detected in Yucatan minipigs. 21 Interestingly, our observations concerning aPTT in pigs between weeks 10 and 24 are similar to those described for human infants. Many investigators reported a prolonged aPTT during the first months of human life that has been attributed to low levels of contact factors. 16 Prolongation of the aPTT in young pigs was not associated with an increased tendency to bleeding and that is also consistent with observations made in infants. 15
Our investigations showed that platelet parameters (PLT, MPV, P-LCR) in young pigs tended to stabilize in the early period of life, achieving the adult reference values by weeks 4–6 of age. Among plasma coagulation parameters, PT displayed only minor fluctuations in young piglets, attaining the “normal” intervals relatively quickly as well (by week 8). However, other plasma parameters (especially aPTT) showed a much higher extent of fluctuation, and they appeared not to be finally stabilized until weeks 10–18. Although primary hemostasis parameters (PLT, MPV) tend to be similar in farm pigs and miniature pigs, plasma coagulation parameters (especially aPTT) appear to be distinctly shorter in minipigs. 21 This fact may be of practical importance when choosing the best model for porcine experimentation.
Footnotes
Authors’ contributions
A Pliszczak-Król and A Rząsa contributed to conception and design of the study; contributed to acquisition, analysis, and interpretation of data; and drafted the manuscript. J Król contributed to acquisition, analysis, and interpretation of data; and drafted the manuscript. M Gemra, G Łuczak, A Zyzak, D Zalewski, A Iwaszko-Simonik, and S Graczyk contributed to acquisition, analysis, and interpretation of data. All authors critically revised the manuscript, gave final approval, and agreed to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
BD, Franklin Lakes, NJ.
b.
MIFAM, Milanówek, Poland.
c.
PROFILAB, Warszawa, Poland.
d.
FL Medical, Torreglia-Padova, Italy.
e.
PROCAN PE-6800, Procan Electronics Inc., Nanshan, Shenzhen, China.
f.
ABX VetPack Minotrol 16, HORIBA, Montpellier, France.
g.
Coag Chrom 3003, Bio-Ksel, Grudziądz, Poland.
h.
STATISTICA 9.1, StatSoft. Inc., Tulsa, OK.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a Grant from the Ministry of Science and Higher Education of Poland (No. NN311265038).
