Abstract
Reference intervals for coagulation parameters have been rarely determined in dogs for the STA Compact® automated coagulation analyzer, so it is the aim of the current study to validate assays and establish reference ranges for its use in canine specimens. Coagulation parameters were assessed in 56 healthy dogs with a median age of 2 years and evenly distributed sex. The 95% reference intervals were as follows: 1-stage prothrombin time = 5.7–8.0 sec; activated partial thromboplastin time (APTT) = 10.0–14.3 sec; thrombin time (TT) = 11.9–18.3 sec; fibrinogen = 1.3–3.1 g/1; antithrombin (AT) = 107.9–128.0%; D-dimer = 0.023–0.65 μg/ml; anti-factor Xa = 0.04–0.26 IU/1; and activated protein C (APC) ratio = 2.0–3.0. Protein C and S activity was markedly below (<-20%) and factor VIII was 2- to 11-fold above the human calibration standard, so a standard curve had to be prepared from canine pooled plasma. Reference intervals for protein C, protein S, and factor VIII were 75.5–118.9%, 74.4–160.5%, and 70.9–136.4%, respectively, compared with a canine standard curve. Streptokinase-activated plasminogen assay was not suitable for dogs. There was no significant impact of sex on hemostasis test results. Factor VIII activity, AT, protein C, protein S, and APC ratio were overestimated in hemolytic plasma, whereas fibrinogen, TT, and APTT were underestimated. Lipemia resulted only in false-high D-dimers. This study provided useful reference intervals for dogs, but some human tests (i.e., protein C, protein S, factor VIII, and plasminogen) required modification.
Introduction
Bleeding tendencies are caused by defects in the procoagulant aspects of primary and secondary hemostasis, while thrombophilia can be due to a defect in the profibrinolytic tertiary or anticoagulant aspects of the secondary hemostasis system. 2,34 In the past, coagulation analysis focused on patients with a bleeding tendency (i.e., hypocoagulatory conditions). Actual research activity has been directed toward diagnosing patients with a risk of thrombosis (thrombophilia) due to a hypercoagulable state. 2 Thrombophilia may be caused by diseases affecting the fibrinolytic system (e.g., plasminogen) and/or physiologic anticoagulants including antithrombin (AT) and protein C and protein S. Activated protein C (APC) performs an anticoagulant function by inactivating activated factor V (FVa) and activated factor VIII (FVIIIa). 49 In humans, a poor anticoagulant response of APC is known as APC resistance and results in thrombophilia and a hypercoagulable state. 17 In patients, the APC ratio is calculated as follows to characterize APC resistance. 10
An APC ratio <2.1 sec has been considered abnormal in humans. 45 Activated protein C resistance may be hereditary (mutation of factor V) or acquired. 38 As reviewed elsewhere, acquired APC resistance in patients may be associated with pregnancy, acute phase reactants, or cancer. 8 To the authors' knowledge, reference intervals and the diagnostic significance of APC ratio have not been investigated in dogs. In the past 50 years, several risk factors for developing venous thrombosis have been described in humans. 9 In veterinary medicine, knowledge of factors predisposing to thrombophilia is limited and a subject of current investigations.
Reference intervals for routine coagulation parameters, AT, FVIII, and protein C have been established previously in dogs, whereas reference intervals for protein S, plasminogen, APC ratio, and anti-FXa activity have not been previously determined. However, even for routine coagulation parameters, it is generally agreed that analyzer- and method-specific reference intervals should be established. Studies in humans investigating 8 different activated partial thromboplastin time (APTT) assays and modified APTT assays for detection of APC resistance demonstrated that results are highly dependent on both the assay and the coagulation analyzer, so reference intervals were not identical. 23 The STA Compact® is a completely automated bench-top hemostasis analyzer capable of performing clotting, chromogenic, and immunological assays in random access mode. This instrument has been used previously with canine samples to measure routine coagulation parameters, AT, protein C (chromogenic assay), 11,12,46 and fibrin D-dimers. 3,13 The STA-Rotachrom® heparin test, run on the STA coagulation analyzer, has been validated for use with blood plasma of 12 healthy dogs and 10 dogs with immune-mediated hemolytic anemia (IMHA) to determine anti-FXa activity when monitoring heparin therapy. 4 In these previous studies, healthy dogs served as controls and were compared to diseased patients. However, for routine use of STA Compact a coagulation tests in veterinary laboratories, reference intervals should be established. Generally, reference intervals serve as the basis of laboratory testing and are helpful in differentiating healthy from diseased patients. 19 Regarding establishment of reference intervals in humans, the International Federation of Clinical Chemistry and the International Committee for Standardization in Hematology have proposed that at least 40 individuals (and preferably 120 individuals) should be included to provide an accurate calculation of the 0.025 and 0.975 percentiles. 41 These recommendations have been recently updated, and the proposals of the International Organization for Standardization (ISO) Standard 15189 have been included. 7 Even for established reference intervals, ISO Standard 15189 requested that biological reference intervals should be periodically reviewed. 7 Canine reference intervals for a functional protein C chromogenic assay run on an STA coagulation analyzer have been published previously. However, a limitation of the previous study was that statistical methods used to calculate the reference intervals were not given, and only 30 dogs were evaluated. 11
Thus, the aim of the current study was to establish reference intervals for variables reflecting coagulatory states for the STA Compact, which had not been performed previously in a sufficient number of dogs. Moreover, the influences of sex, intra-assay repeatability, and potential sources of interference (i.e., hemolysis and lipemia) on test results were also evaluated. Assays run on the STA Compact included the 1-stage prothrombin time (OSPT), APTT, thrombin time (TT), fibrinogen plasma concentration, AT, protein C, protein S, plasminogen, APC ratio, FVIII activity, and fibrin D-dimers. The anti-FXa activity was also measured to obtain a reference interval for baseline factor Xa inhibition in canine plasma.
Materials and methods
Study design
The prospective investigation was approved by the Ethics Committee for Animal Welfare (Giessen, Germany; no. V54-19c20/15cGi18/17, January 2008). The study was performed in 3 steps. First, each human assay was validated for use with canine samples. Second, assays that were not valid initially were modified and optimized for use with canine specimens. Third, these various tests were evaluated for diagnostic use using the validated, optimized test methods. This process included establishment of a central 95% reference range with 90% confidence intervals (CIs) of the upper and lower reference limits, calculation of the intra-assay coefficient of variation (CV), determination of the mean and range for each analyte, and evaluation of the impact of 3 grades of hemolysis and lipemia on the test results.
Dogs
The study was performed using adult (>1 year old) dogs including staff-owned dogs, blood donors, or healthy dogs presented for routine radiological examination to screen for hereditary hip dysplasia (HD) or elbow dysplasia (ED) at the Clinic for Small Animals, Department of Veterinary Surgery and Clinical Sciences, Faculty of Veterinary Medicine, Justus-Liebig University, Giessen, Germany. Inclusion criteria included a normal physical examination, complete blood count (CBC), and biochemical profile. The biochemical profile included assessment of urea, creatinine, total protein, albumin, globulin, fructosamine, sodium, potassium, ionized calcium, magnesium, and phosphate concentrations as well as the determination of the activities of alkaline phosphatase, alanine aminotransferase, and glutamate dehydrogenase. In addition, there was an absence of any bleeding tendency in the medical history, and the dogs had not been given any medications for at least 2 weeks.
Preparation of canine pooled plasma
Approximately 30 ml of citrated whole blood was obtained from 16 healthy adult dogs (8 females, 6 males, and 2 spayed females) with a median age of 3.5 years (range: 1–8 years). Three Beagles, 2 Belgian Malinois, 2 Labrador Retrievers, 2 French Bulldogs, 2 Maremma Sheepdogs, 2 German Shepherd Dogs, 1 Rottweiler, 1 Staffordshire Bullterrier, and 1 mixed breed dog were included. All dogs were healthy based on the physical examination, CBC, and biochemical profile. The dogs that were used for preparation of the canine pooled plasma were not included in the study population used to establish the reference intervals.
Blood samples
Blood samples were obtained from fasted, resting dogs immediately after placement of an 18-gauge nonheparinized venous catheter in the cephalic vein. In staff-owned dogs and blood donors, the catheter was removed directly after the blood sample was taken. The catheter was left in place for anesthesia administration in patients that were evaluated radiologically for ED and HD.
For measurement of coagulation parameters with the STA Compact, venous blood samples were anticoagulated in siliconized vacutainer tubes containing 3.18% trisodium citrate. A blood to anticoagulant ratio of 9:1 (vol/vol) was collected. The first 2 ml of blood removed from the catheter was used for the CBC and biochemical profile to minimize the presence of contaminating tissue thromboplastin. Blood for hemostasis testing was allowed to drop directly into the citrated tube, or it was aspirated in a plastic syringe. In the latter case, the specimens were gently placed into the citrate-containing tube, and the contents were carefully mixed.
The collection tubes were carefully checked for proper filling, and only specimens with a 9:1 ratio of blood to citrate anticoagulant were included in the study. Sodium-citrated anticoagulated whole blood was centrifuged at 850 × g for 10 min within 1 hr after sampling. Citrated plasma was separated from the blood cells and centrifuged again at 850 × g for 10 min to remove all nonsedimented platelets prior to freezing, as previously recommended. 6 The supernatant was removed and stored at −80°C until analysis. Analysis was performed within 3 weeks of sampling. For all analytes, sample stability was proven to be >12 months by reanalyzing 3 specimens approximately 13 months after storage at −80°C. Directly prior to analysis, plasma samples were thawed at 37°C in a water bath as previously recommended 5 to allow complete dissolution of the cryoprecipitate. Subsequently, the samples were centrifuged at 850 × g for 10 min to remove any nondissolvable material from the plasma.
Default settings and methods of human assays applied to the STA Compact
The OSPT, APTT, and TT were measured automatically as clotting tests using commercial reagents, and the test results were reported in seconds. b–d Default settings for minimal/maximal measuring times for OSPT, APTT, and TT were 6/180 sec, 3/120 sec, and 13/240 sec, respectively. Fibrinogen was detected by the Clauss method c using a human plasma calibration standard provided by the manufacturer. f
Fibrin D-dimers were measured with an immunoturbidimetric D-dimer assay g as described previously. 3 The D-dimer result was reported as mg/ml fibrinogen equivalent units (i.e., the concentration of fibrin degradation products that result from degradation of 1 mg/1 fibrinogen). The assay was precalibrated and allowed a 1-time testing on an automated analyzer. 37 According to the manufacturer, the linearity of the assay ranged from 0.0 to 4.0 μg/ml. Antithrombin activity was detected based on the inhibition of thrombin using a chromogenic substrate kit. h Test results were reported as the percentage of the human plasma calibration standard.
For measurement of protein C, an automated clotting test was performed. i Cephalin (phosphatidylethanolamine, a phospholipid serving as contact activator; 50 μl), human protein C-deficient plasma (50 μl), and 50 μl of a specific protein C activator (Protac®, a snake venom derivative of the Southern Copperhead, Agkistrodon contortrix contortrix) were added to 50 μl of the patient sample prediluted 1:10 with diluent buffer. j This resulted in an activation of protein C within the sample and simultaneously an initiation of the intrinsic clotting system by contact activation. The protein C activity was determined with a modified APTT test using protein C-deficient human plasma. Based on this method, the APTT was solely dependent on the protein C activity in the sample. Activated protein C cleaved FVa and FVIIIa, resulting in an increase in the APTT. Thus, protein C activity was directly proportional to the increase in APTT measured in seconds. The test results could be reported as the percentage of human plasma calibration standard or in seconds. According to the manufacturer, the analytical interval of the test was between 3% and 150%. The default settings for minimal and maximal times of measurement were 10 sec and 180 sec, respectively. In case of results <10%, the test was automatically repeated at a dilution of 1:5.
Protein S activity was also determined with an automated clotting test. k Fifty microliters of human protein S-deficient plasma, 50 μl human protein C-activated with Protac, and 50 μl of bovine FVa were added to 50 μl of the patient sample, which was automatically diluted 1:10 with diluent buffer. As protein S was a cofactor of protein C, the anticoagulatory effect of protein C was increased solely by the protein S activity in the patient sample. For minimal and maximal times of measurement, 10 sec and 250 sec, respectively, were set as a default. In case of results <15%, the test was automatically repeated with at a dilution of 1:5.
The principle of assessment of APC resistance was based on an unusually small prolongation of the clotting time of the tested plasma in the presence of APC and calcified medium. Using a commercial APC test kit, 1 50 μl of patient plasma was diluted 1:10, and coagulation was achieved in the presence of 50 μl of factor V-deficient plasma and 50 μl of Crotalus viridis helleri venom, which acted as an activator of factor X. Thus, the coagulation cascade was triggered downstream from factor X so that an influence of all coagulation factors acting upstream could be excluded. The prolongation of the clotting time of normal plasma in the presence of APC resulted from the capacity of 50 μl of APC derived from the test kit to inactivate FVa in the test sample. The result was reported in seconds. Based on information provided by the manufacturer, the analytical interval of the test was between 20 sec and 300 sec.
Plasminogen activity was determined photometrically with a chromogenic assay. m An excess of streptokinase (100 μl of reagent 1 of the STA plasminogen test kit containing approximately 5,000 IU streptokinase/ml) was added to 100 μl of the patient sample, which was diluted 1:20 with diluent buffer. A complex with plasmin-like activity was formed between the streptokinase and plasminogen. The quantity of streptokinase-plasminogen complex was assessed by its action on 100 μl of a chromogenic synthetic substrate (H-D-But-CHA-Lys-pNA × 2 AcOH) at a concentration of 5.5 mol/ml. Quantification was based on the amount of p-nitroaniline released, which was photometrically detected at 405 nm. Results were reported as the percentage of human plasma calibration standard. A measuring range between 11% and 160% plasminogen activity compared to the human standard plasma was reported by the manufacturer.
Measurement of FVIII was performed in a modified 1-stage APTT with a human FVIII-deficient substrate plasma. n The default setting for dilution of patient samples was 1:10 with diluent buffer. If FVIII activity was >150%, the measurement was automatically repeated at a dilution of 1:40. The default measuring range was between 1% and 600%.
For measurement of anti-FXa activity, an automatic chromogenic assay was applied, o as described previously. 4 The test principle was based on a 1-step reaction wherein 100 μl of bovine factor Xa (0.23 IU/ml) was added to a mixture of 50 μl of patient plasma diluted 1:2 with diluent buffer and a chromogenic substrate (MAPA-Gly-Arg-pNA × AcOH, 1.5 μmol/ml). This resulted in an initiation of 2 reactions, including the hydrolysis of the substrate by factor Xa and inhibition of factor Xa by the heparin-AT complex, which consisted of the heparin and AT specific to the patient. After an incubation of 240 sec, the quantity of pNA released was measured photometrically and was inversely proportional to the heparin concentration in the test medium. Results were reported in UI/ml. According to the manufacturer, the analytical range of the test was between 0.00 UI/ml and 0.70 UI/ml.
For all variables, internal quality control material (normal and abnormal) provided by the manufacturer was run each day. STA PreciClot Plus p I and II was used for quality assurance of most variables including OSPT, APTT, TT, fibrinogen, AT, protein C, protein S, FVIII, and plasminogen. A third-level control (STA PreciClot Plus III, also abnormal) was run in addition for OSPT, APTT, fibrinogen, and AT. For internal quality control of fibrin D-dimer measurements, Liquicheck™ D-dimer control levels I and II q were used. For the anti-FXa activity and APC response tests, materials for internal quality control were included in each reagent package. r,s
Assessment of interferences
To assess the influence of lipemia on coagulation parameters, samples with 3 grades of lipemia were prepared by adding a commercially available fat emulsion t in different dilutions (solutions A, B, and C) to the plasma samples of 3 healthy dogs. Solution A was obtained by adding the fat emulsion to an equal volume of NaCl (1:1). Solutions B and C were consistent with a 1:4 and 1:16 dilution of fat emulsion and NaCl. For preparation of samples with grades 1 to 3 lipemia, 20 μl of solutions C, B, and A, respectively, were added to 1,480 μl of citrated plasma, resulting in final dilutions of 1:74 (grade 3), 1:296 (grade 2), and 1:1,184 (grade 1). In grade 1 lipemia, letters behind the tube could be read easily. In grade 2 lipemia, letters could be recognized, whereas in grade 3 lipemia, recognition of letters behind the tube was impossible. Citrated plasma of the same dog to which 20 μl of NaCl was added served as a control.
Hemolysis was achieved by subjecting whole blood to a freeze-thaw cycle. Approximately 20 ml of citrated whole blood was obtained from 3 healthy dogs and was divided into 2 aliquots. One aliquot was frozen at −80°C to induce hemolysis; from the other aliquot, citrated plasma was prepared as described above and was also stored at −80°C until analysis. After thawing at 37°C, hemolyzed and nonhemolyzed aliquots were centrifuged at 850 × g for 10 min, and specimens with 3 grades of hemolysis were prepared. In samples with grade 1 hemolysis (ratio of hemolyzed to nonhemolyzed plasma 1:9), letters behind the tube could be read easily. In grade 2 (ratio of hemolyzed to nonhemolyzed plasma 1:4), letters could be recognized, whereas in grade 3 hemolysis (undiluted hemolyzed plasma), recognition of letters behind the tube was impossible. Hemoglobin concentration was assessed colorimetrically using a species-specific hematology analyzer u and a cyanide-free reagent as described previously. 1
Statistical analysis
Results were analyzed with the Graph Pad Prism v and Analyse-it Method Evaluation statistical software. w An Anderson-Darling test was performed to verify the assumption of normality.
In the case of normal and log-normal distribution of data, double-sided reference intervals were obtained by calculating the mean ± 1.96 standard deviation (SD) so that 95% of the reference population was included. If nonnormal distribution of data was present, the nonparametric percentile method was applied. The 2.5 and 97.5 percentiles were calculated to obtain the 95% double-sided reference interval. Data were depicted as histograms with the reference interval as well as the 90% CIs of the upper and lower reference limits.
The influence of sex was assessed by using an unpaired t-test or a comparable nonparametric test (Mann-Whitney U-test). Fifteen-run intra-assay repeatability for normal values was calculated using a sample from a healthy dog. A paired t-test was applied to investigate the effect of interferences (hemolysis, lipemia) on coagulation parameters. Results were shown as percentage bias compared with the control. After Bonferroni correction, significance was set at P < 0.005 for comparison of the influence of sex and at P < 0.0015 for assessment of the effect of interferences.
Results
Validation of human assays
Most human assays (OSPT, APTT, fibrinogen, AT, APC ratio, fibrin D-dimer) were valid for canine specimens (Table 1). In 11 of 57 (19%) dogs, the TT was below the minimal measuring time of 13 sec, which was set as a default for the human assay. The most striking findings were that the protein C, protein S, and plasminogen activities were markedly lower than the concentrations in the human standard plasma (100% per definition). The protein C and protein S reference intervals determined with the clotting assay were markedly below the measurement range of the human assay, whereas the canine reference interval for plasminogen activity was slightly lower than the measurement range of the commercial plasminogen test. m In contrast, the canine reference interval for FVIII activity was significantly higher than the concentrations in the human standard plasma and exceeded the upper limit of the measuring range of the FVIII assay. Based on the results for FVIII, protein C, protein S, and plasminogen activity, an adaptation of the human test protocols was required for canine specimens.
Reference intervals for variables reflecting secondary hemostasis, fibrinolysis, and physiological anticoagulants (n = 56 dogs).*
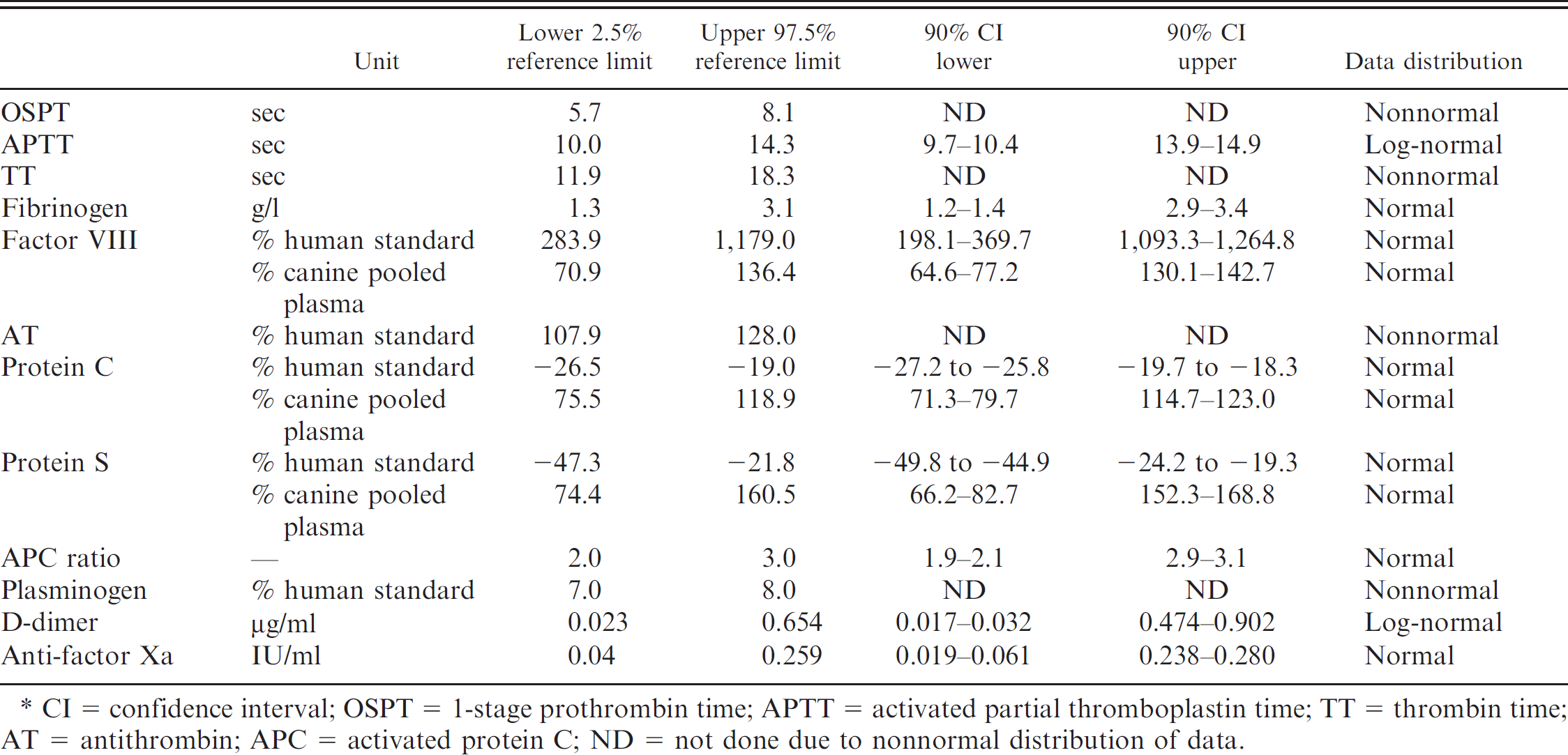
CI = confidence interval; OSPT = 1-stage prothrombin time; APTT = activated partial thromboplastin time; TT = thrombin time; AT = antithrombin; APC = activated protein C; ND = not done due to nonnormal distribution of data.
Modification of test methods applied to the STA Compact
Based on the results of the present study, assays for the measurement of TT, FVIII, protein C, protein S, and plasminogen had to be optimized for use with canine blood plasma. For the TT, the minimal measuring time was defined as 8 sec instead of 13 sec set previously as the default. The 11 samples for which the TT was <13 sec were reanalyzed; the new settings for the measuring time yielded a result in all cases. The tests for FVIII, protein C, protein S, and plasminogen were performed as described before, but canine pooled plasma was used in serial dilutions (FVIII = 1:40, 1:60, 1:80; protein C and S = 1:5, 1:10, 1:20, 1:40; plasminogen = 1:100, 1:40, 1:20) instead of using a human plasma calibration standard to obtain a species-specific reference curve. Dilutions of patient plasma were 1:40 for FVIII, 1:5 for proteins C and S, and 1:20 for plasminogen.
Internal quality control was performed at 2 levels (normal and low) with aliquots of citrated plasma obtained from one of the dogs included in the reference population. Plasma was stored at −80°C until analysis. Undiluted citrated plasma and plasma diluted 1:1 with saline served as normal and low controls, respectively.
Reference intervals
Fifty-six dogs (19 German Shepherd Dogs, 15 Beagles, 8 Golden Retrievers, 4 Labrador Retrievers, 2 mixed breeds, 2 Great Danes, 1 Hovawart, 1 Saint Bernard, 1 English Springer Spaniel, 1 Bernese Mountain Dog, 1 Boxer, and 1 Giant Schnauzer) with a median age of 2 years (range, 1–6 years) were included in the study. Sex was equally distributed with 24 males, 20 females, 7 neutered males, and 5 spayed females. The dogs were healthy based on physical examination, CBC, biochemical profile, and routine coagulation profiles.
Most variables were normally distributed, including the fibrinogen concentration, the APC ratio, FVIII, proteins C and S, and anti-FXa (Table 1; Fig. 1). Results of APTT and fibrin D-dimer measurements were normally distributed after logarithmic transformation. A nonnormal distribution was observed for OSPT, TT, AT, and plasminogen activity. Reference intervals are displayed in Table 1. There were no significant differences between the results obtained for male and female dogs, with P values being >0.1 for all variables.
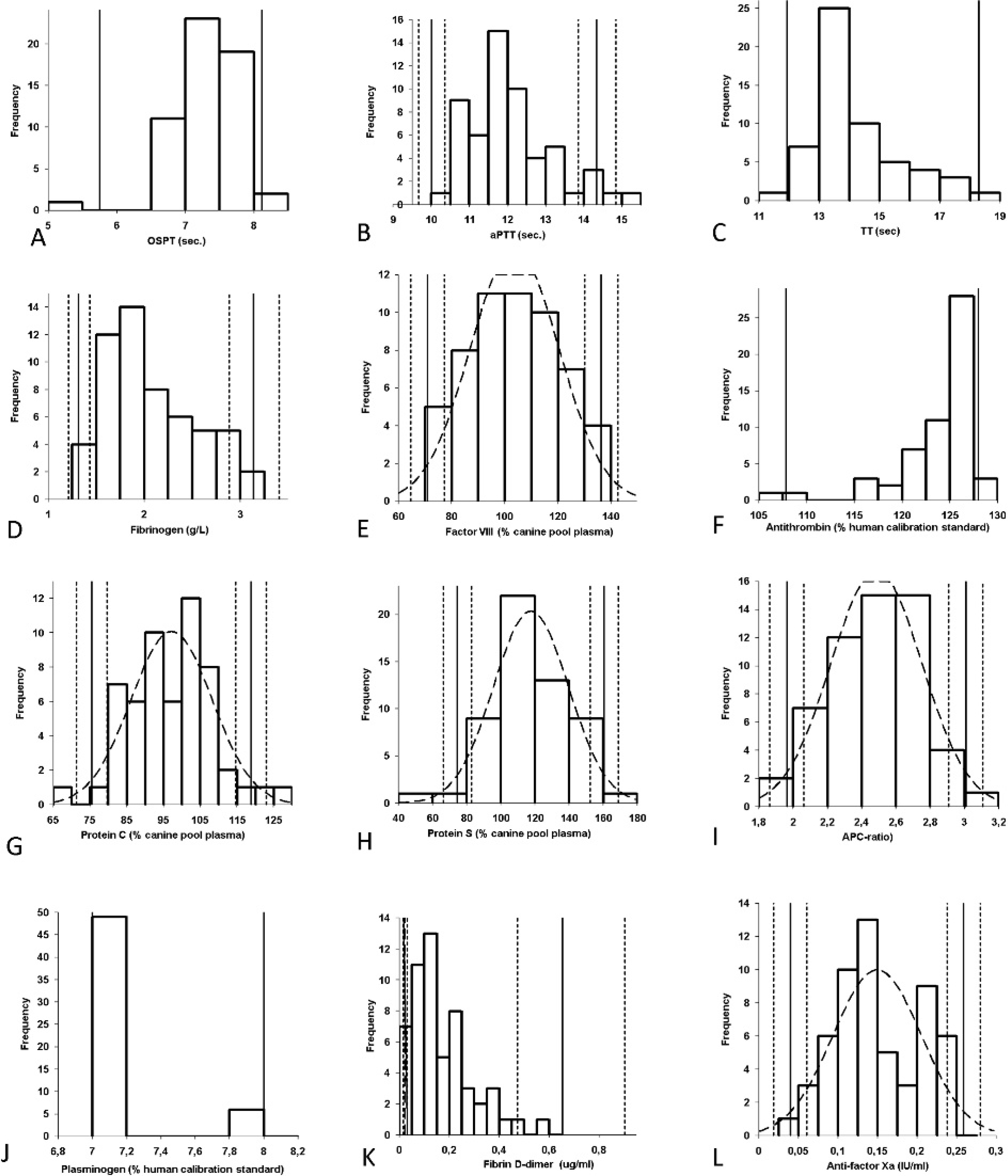
Frequency histograms demonstrating the distribution of data obtained for calculation of reference intervals (n = 56 healthy dogs). The solid vertical line indicates the reference interval. The dotted lines are consistent with the 90% confidence interval of the upper and lower reference limits. For normally distributed data, frequency histogram and superimposed Gaussian curve (dotted lines) are shown. For nonnormal distribution of data, only the upper and lower reference limits are given. APC = activated protein C; APTT = activated partial thromboplastin time; OSPT = prothrombin time; sec = seconds; TT = thrombin time.
Intra-assay repeatability
Fifteen-run, intra-assay repeatability is shown in Table 2. For the routine coagulation parameters (APTT, OSPT, and TT), a low CV was obtained, ranging from 1.02% to 1.64%. Factor VIII activity, AT, protein C, plasminogen, and APC ratio had intra-assay CVs ranging from 1.45% to 4.68% (Table 2). For fibrinogen concentration and protein S activity, 1 outlier exceeding the 3-fold SD from the mean was present in the series of measurements. Removing the outlier resulted in an intra-assay CV of 5.6% for fibrinogen and 1.4% for protein S. Variables with low plasma concentrations in healthy dogs such as fibrin D-dimer plasma concentration and anti-FXa activity demonstrated high intra-assay CVs.
Assessment of interferences
Hemoglobin concentration induced by freezingthawing ranged from 0.6 ± 0 mmol/l (grade 1 hemolysis) to 1.3 ± 0.06 mmol/l (grade 2 hemolysis) to 6.6 ± 0.06 mmol/l (grade 3 hemolysis), respectively. As depicted in Table 3, hemolysis had a major impact on results obtained for most coagulation tests. Anti-FXa activity was most susceptible to hemolysis, demonstrating a highly significant increase of approximately 600% compared with the control (P < 0.001 for grades 1–3 hemolysis). Grade 1 hemolysis resulted in a false-high measurement of fibrin D-dimers of approximately 100%. In plasma with a higher degree of hemolysis, measurement of D-dimers was not possible because of technical errors of the analyzer. Factor VIII activity, AT, protein C, protein S, and APC ratio were overestimated in hemolyzed plasma, whereas fibrinogen, TT, and APTT tended to be underestimated compared with the control.
Fifteen-run intra-assay repeatability for hemostasis variables using a specimen from 1 dog.*
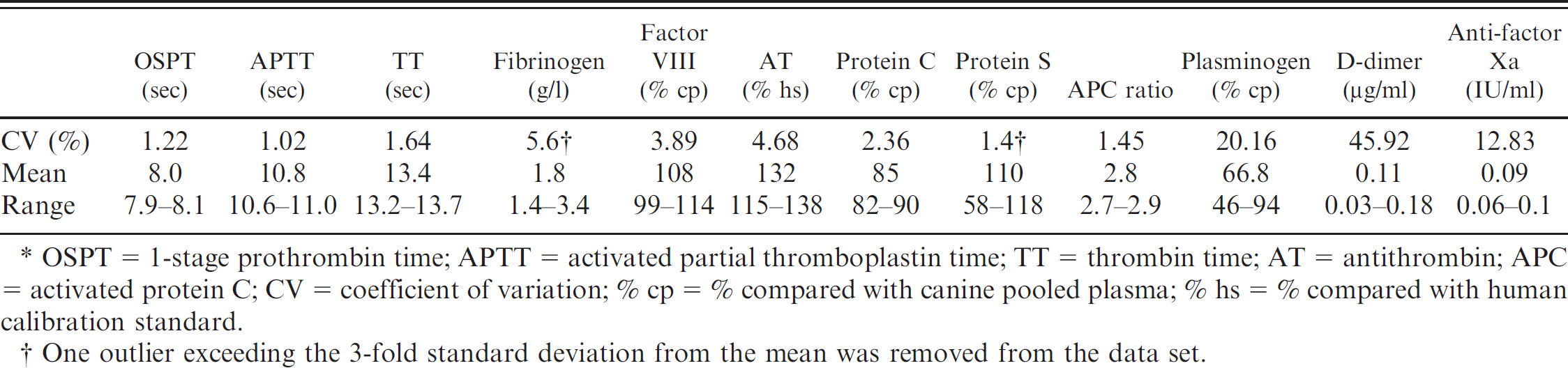
OSPT = 1-stage prothrombin time; APTT = activated partial thromboplastin time; TT = thrombin time; AT = antithrombin; APC = activated protein C; CV = coefficient of variation; % cp = % compared with canine pooled plasma; % hs = % compared with human calibration standard.
One outlier exceeding the 3-fold standard deviation from the mean was removed from the data set.
In contrast to hemolysis, lipemia generally did not significantly influence measurement of coagulation parameters except for the immunoturbidimetric D-dimer assay g (Table 4). In the presence of lipemia, D-dimer test results were markedly overestimated, ranging from a bias of 32% (grade 1 lipemia) to 160% (grade 3 lipemia). Because of the lack of a valid test for canine specimens, the effect of hemolysis and lipemia was not assessed for plasminogen activity.
Discussion
Canine reference intervals for OSPT, APTT, and fibrinogen (Clauss method) were comparable with those described previously for manual tests applied to canine specimens. 34 Using a commercial coagulometric test, FVIII activity in canine plasma was reported to be 7.9 times higher in canine plasma compared with activities in human standard plasma. 33 This finding was in accordance with results for the automated coagulation analyzer in the current study and was also demonstrated in less recent studies and case reports evaluating canine FVIII activity using coagulometry. 31,36 It has been shown that all coagulation tests using human deficient plasma for measurement of coagulation factors in canine plasma were influenced by the very high activities of FVIII (9.3-fold higher than in human plasma) and factor V, which are typical for dogs. 33 This was particularly the case when a low dilution of plasma was applied. Therefore, the use of higher sample dilutions (>1:20; e.g., 1:40) was recommended to reduce this effect. 33 A similar finding has been reported in specimens from cats 30 and cattle. 29
Reference intervals for a functional protein C chromogenic assay have been previously reported in dogs to be 75–135% compared with canine pooled plasma. 11 This was comparable with the results obtained in the present study, although the upper reference limit was slightly lower (118%). Differences might be due to a different methodology (chromogenic vs. clotting assay) and use of different pooled plasma. A limitation of the previous study was that statistical methods to calculate the reference intervals were not given and only 30 dogs were evaluated. 11 To the authors' knowledge, the current investigation is the first study to evaluate protein C and protein S activity in dogs with a functional clotting assay using Protac as an activator of protein C. Protein C activity has been determined previously in canine plasma with a Protac-activated chromogenic assay; however, the results were compared with canine pooled plasma rather than to the human standard. 11 The current study clearly demonstrated that protein C and protein S activities determined with the clotting test were markedly lower than in the human calibration standard. Theoretically, this might have been due to the plasma concentration being lower in dogs than in humans. As the clotting assay is a functional test, it might also be hypothesized that a decreased biological activity of proteins C and S in dogs was the reason for different activities observed in human and canine specimens. However, given the physiology of the coagulation process, it is unlikely that dogs are lacking the natural anticoagulant protein C. Moreover, an impaired activation of protein C and protein S in the clotting assay could have been responsible for different results obtained with human and canine specimens. A previous investigation demonstrated that canine protein C was unresponsive to activation with Protac even when an activating dose 3 times higher than that used with human specimens was employed. 22 Although canine protein C was reported to have 72% homology with the human protein C molecule, the existing variations may account for the differences in the ability of Protac to activate the human and canine proteins. Surprisingly, dilution of the concentration of the contact activator resulted in a shortening of the APTT compared with the control (untreated canine blood plasma), whereas a minimal prolongation of APTT was observed after dilution of both plasma and activator. 22 A similar finding was reported in porcine samples, and it was hypothesized that this effect could be attributed to the presence of protein C inhibitors or anti-activators since Protac did induce protein C-related amidolytic activity when the sample was diluted. 43,44 In contrast, protein C-like amidolytic activity was not observed in canine plasma after activation with Protac, so it was considered more likely that the snake venom extract, aPTT reagents, and canine plasma failed to produce a stoichiometric complex with strong protein C activity. 22
Percentage bias after induction of 3 grades of hemolysis (n = 3 dogs).*
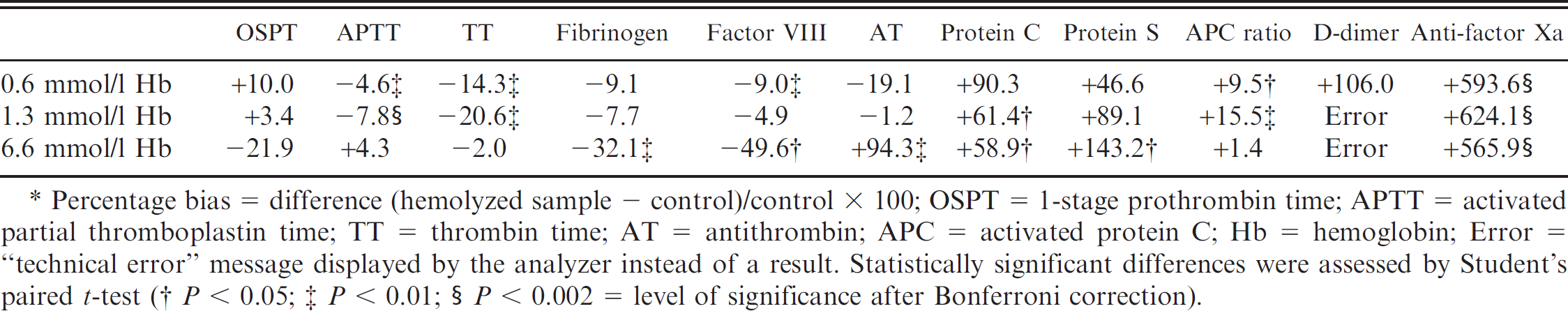
Percentage bias = difference (hemolyzed sample − control)/control × 100; OSPT = 1-stage prothrombin time; APTT = activated partial thromboplastin time; TT = thrombin time; AT = antithrombin; APC = activated protein C; Hb = hemoglobin; Error = “technical error” message displayed by the analyzer instead of a result. Statistically significant differences were assessed by Student's paired t-test.
P < 0.05;
P < 0.01;
P < 0.002 = level of significance after Bonferroni correction.
Percentage bias after induction of 3 grades of lipemia (n = 3 dogs).*
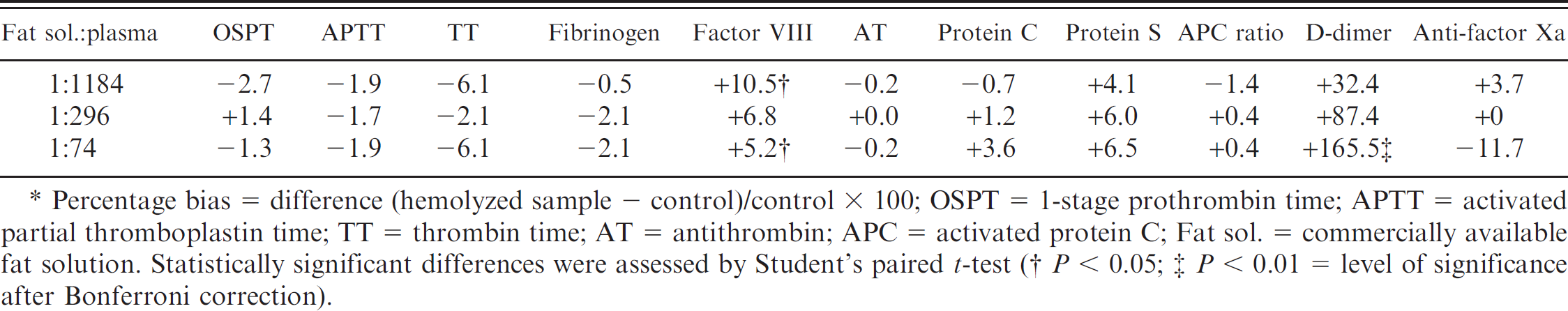
Percentage bias = difference (hemolyzed sample − control)/control × 100; OSPT = 1-stage prothrombin time; APTT = activated partial thromboplastin time; TT = thrombin time; AT = antithrombin; APC = activated protein C; Fat sol. = commercially available fat solution. Statistically significant differences were assessed by Student's paired t-test.
P < 0.05;
P < 0.01 = level of significance after Bonferroni correction.
Despite these reports, evidence in the veterinary literature indicates that at least a minimal activation of canine protein C occurs by Protac. In the current study and in another investigation, linearity of the test was demonstrated from 0% to 100% when using a serial dilution of canine pooled plasma. 11 A functional chromogenic assay with activation of protein C using venom of the copperhead snake was used previously, and the results were compared with a canine pooled plasma sample obtained from 15 healthy dogs. 11 Previous investigations in dogs have proven that functional protein C assays will detect significant differences between protein C concentrations in patients with various diseases including sepsis 11,12 and portosystemic shunts. 46
Regarding the resistance against activated protein C, a reference interval for APC ratio has not been established in dogs. In humans, an APC ratio <2.1 sec has been considered abnormal, 45 which was comparable with the results in dogs of the present study. However, the clinical significance of this finding must still be elucidated for animals.
The results of the current study clearly demonstrate that plasminogen activity was markedly lower in canine plasma than the human standard. Previous investigators also found lower plasminogen concentrations in dogs, which was approximately 50% of the activity present in human control plasma. These investigators used a chromogenic assay that employed an excess streptokinase concentration (8,000 IU/ml) to activate plasminogen x (Wilke K: 2008, Untersuchungen zur Beurteilung der fibrinolytischen Aktivität im Plasma von verschiedenen Spezies (inklusive Mensch) und Korrelation mit der in vivo fibrinolytischen Wirkung von therapeutisch wirksamen Prinzipien [Evaluation of fibrinolytic activity in the plasma of different species (including man) and correlation with the in vivo fibrinolytic effect of therapeutic principles]. Thesis, Justus-Liebig University Giessen. In German. Abstract in English). However, the test results in the previous study were still higher than the reference interval obtained in the current study. A possible reason for the discrepancy among different assays may be the fact that the concentration of streptokinase required for activation of plasminogen is species specific. It was reported previously that markedly higher concentrations of streptokinase were necessary to activate specimens from rats, rabbits, and dogs than from humans. 18 For this reason, different concentrations of streptokinase used in the test kits (8,000 IU/ml in the HemosIL plasminogen assay [Wilke K: 2008, Evaluation of fibrinolytic activity] vs. 5,000 IU/ml in the STA Plasminogen assay applied in the current study) may be responsible for intralaboratory differences in plasminogen activity. Based on these findings, it was evident that human assays used for measurement of canine plasminogen activity reflected the capability of the fibrinolytic system to be activated by streptokinase rather than the plasminogen concentration itself. 24 This is in accordance with a previous study in dogs reporting low capability of streptokinase (4,000 IU/ml) to activate canine plasminogen in a commercial assay, which was adapted to a microtiter plate. 26 Urokinase (20,000 IU/ml) was demonstrated to be a more potent activator of plasminogen than streptokinase in canine specimens. 26 Although commercial plasminogen test kits containing comparatively high streptokinase concentrations (e.g., Hemos-IL Plasminogen assay) showed a certain capability to activate canine plasminogen (Wilke K: 2008, Evaluation of fibrinolytic activity), substitution of streptokinase by urokinase appears to be preferable to achieve an expansion of the standard curve. In the current study, additional analysis of samples using urokinase or streptokinase in higher concentrations as activator was not performed because of insufficient sample volume.
Anti-FXa activity was previously evaluated in 12 healthy dogs and 10 patients with IMHA using the same assay, analyzer, and test protocol. 4 Baseline anti-FXa activity ranged from 0.06 to 0.17 IU/l and was slightly lower than that of the current study. This finding indicates that canine plasma contains endogenous factor Xa inhibitors at higher concentration or increased activity compared with human plasma. 4 Therefore, a higher dilution of canine plasma (3:8 instead of the default setting of 1:2 for human samples) was recommended to monitor heparin therapy in dogs and to achieve a baseline activity close to zero.
Previous investigations in dogs revealed significantly increased concentrations of fibrinogen and fibrin and/or fibrinogen degradation products when progesterone concentrations were high. In contrast, plasma AT was decreased in the presence of high progesterone concentrations. 16 The current investigation failed to demonstrate any significant impact of sex on coagulation parameters, but the phase of the estrus cycle was not determined. Therefore, specific effects of certain reproductive cycle phases might have been missed. In humans, several sex-specific findings were demonstrated for hemostasis parameters. Men were reported to have lower prothrombin activity 40 as well as higher plasma AT and protein C concentrations. 14 Women were reported to have higher plasma D-dimer concentrations. 20 The lower prothrombin activity in men may explain the increased frequency of major hemorrhage in elderly male patients receiving anticoagulant therapy. 40 In contrast, a higher risk for arterial fibrillation-related thromboembolism was reported in female patients, 15 which further supports increased coagulation activity in women. Moreover, pregnancy and oral contraceptive use were reported to be associated with hypercoagulable states 21 and APC resistance in women. 8 In the current study, pregnant dogs were not included, but the injection of depot preparations of contraceptives by the referring veterinarians could not be excluded. However, contraceptive use is unlikely because of the absence of drug administration in the medical history and lack of sex-related differences in the hemostasis results of the current study. Interestingly, most human epidemiological studies demonstrated that women have a 40–400% lower risk of recurrent venous thromboembolism than men, 25 but probable reasons for this finding were not addressed. Comparable epidemiological investigations have not been performed in dogs.
A limitation of the current study was that most samples were obtained from Beagles and German Shepherd Dogs. Most healthy dogs available at the university clinic are either blood donors (mainly medium-sized to large breed dogs and Beagles) or healthy dogs presented for routine radiological examination for HD or ED, which is known to affect large breed dogs.
Because of the predominance of Beagles and German Shepherd Dogs in the reference population, the reference intervals constructed in the current study might not be applicable for all breeds of dogs. Breed-specific characteristics of the coagulation system have been rarely investigated in dogs. Significant differences between different breeds were reported for factor VII, 32 which was not investigated in the present study. Greyhounds are reported to have a clinical bleeding tendency despite a normal routine coagulation profile. 48 A recent investigation revealed significantly slower clotting kinetics and reduced clot strength during kaolin-activated thromboelastography in Greyhounds 48 ; however, this breed was not included in the current study.
In summary, it can be concluded that some human assays for the analysis of OSPT, APTT, fibrinogen, AT, and D-dimer concentrations are suitable for use with canine specimens without any further adaptation of the human test protocols. Other assays, such as protein C and protein S evaluation, can be used only when the test results are compared with canine pooled plasma because of species-specific characteristics of the coagulation process. With modifications, the commercial human protein C, protein S, and FVIII test kits may detect canine diseases associated with thrombophilia such as disseminated intravascular coagulation. However, the high cost of these assays limits their application in routine practice. Based on the results of the current study, the STA plasminogen assay is not suitable to evaluate plasminogen activity in canine samples, most likely because of the low streptokinase concentration used in the test kit.
Intra-assay CVs for OSPT, aPTT, TT, FVIII, AT, and proteins C and S were comparable with or even slightly lower than (Table 2) those reported by the manufacturer for human plasma samples. y The 21-run intra-assay repeatability for human specimens was as follows: OSPT = 2.8%; APTT = 0.6%; TT = 2.8%; FVIII = 6.5%; AT = 2.6%; protein C = 4.6%; and protein S = 2.75%. y The functional chromogenic protein C assay had similar intra-assay CVs in humans (2.9%) 11 when compared with the clotting assay used in the current study.
Intra-assay CV was slightly higher for fibrinogen concentration even after removal of an outlier (5.6% in dogs vs. 2.3% in humans). Variables with low plasma concentrations in healthy dogs such as plasma fibrin D-dimer concentration and anti-FXa activity also demonstrated high intra-assay CVs, which were higher than those reported for human plasma. y Intra-assay CVs for fibrin D-dimers and anti-FXa were 45.9% and 12.8% (Table 1) in canine plasma compared with 13.7% and 4.4.% in human specimens, respectively. y However, concentrations of fibrin D-dimers and anti-FXa activities are extremely low in healthy dogs and result in high intra-assay CVs. 39 The 6-run intra-assay repeatability for fibrin D-dimers was reported to be 6.1% (range: 2–10%) for the same method and analyzer when determined using canine plasma with increased fibrin D-dimer plasma concentrations ranging from 0.94 μg/ml to 3.08 μg/ml (n = 6 dogs). 3 In the previous investigation evaluating the STA Rotachrom heparin assay for measurement of activity in canine blood plasma, the 15-run intra-assay repeatability was 13% at low anti-FXa activities (mean: 0.268 IU/ml) and comparable with the results obtained in the present study. 4
Previous studies in human specimens have shown that hemolysis causes activation or significant alteration of coagulation. 28,42 Comparable with the current study, hemolysis was induced by freezing whole-blood specimens and separating hemolyzed plasma by centrifugation, so that serial dilutions with nonhemolyzed plasma of the same subject could be prepared. 28 In humans, hemolysis does not cause interference solely by the presence of hemoglobin, as many substances may be released from lysed erythrocytes that might influence coagulation. 28 The release of intracellular and thromboplastic substances from leukocytes and platelets was considered to be responsible for the shortened APTT observed previously in human specimens 27 and in the current study. In humans, the literature regarding this topic is controversial. 28 Measurement of OSPT, APTT, fibrinogen, and D-dimer concentrations in human patients is more susceptible to preanalytic errors. 50 A recent investigation evaluating 12 degrees of hemolysis on routine coagulation parameters in human specimens reported a significant increase in OSPT and D-dimers, whereas a decrease in PT and fibrinogen was noted in samples containing more than 9% hemolysate. 28 The impact of hemolysis on AT has been rarely investigated in humans and dogs. Increasing concentrations of hemoglobin resulted in a linear decrease in AT activity in canine plasma, 47 which is in accordance with the results obtained in the current study.
To assess the influence of hemolysis on laboratory analytes, hemolyzed specimens are commonly achieved by freezing-thawing cycles, as performed in the canine specimens and in previous human specimens. 28 A drawback of this method is that naturally occurring hemolysis due to preanalytic factors or diseases is associated with destruction of platelets and leukocytes, as demonstrated in human samples. 28 Thus, the results reported in the present study may not be representative for all instances of hemolyzed specimens evaluated in clinical laboratories.
According to the manufacturer, hemoglobin concentrations up to 7.6 g/l (4.7 mmol/l) for AT, 5 g/l (3.1 mmol/l) for fibrin D-dimers, and 2 g/l (1.2 mmol/l) for anti-FXa activity will not interfere with the assay. y For the APC assay, resampling was recommended for hemolytic human samples. y Regarding the remaining variables, information about the effect of hemolysis was not provided by the manufacturer.
The effect of lipemia on routine coagulation parameters (OSPT, APTT, TT, and fibrinogen) has rarely been investigated in canine specimens. 35 Studies using a ball coagulometer indicated that severe lipemia (triglyceride concentration of >1.25 g/l) resulted in a slight but clinically insignificant shortening of OSPT and TT in canine blood plasma. 35 Lipemia also results in a 15% decrease in fibrinogen concentrations of human specimens. Significantly false-low plasma fibrinogen concentrations were observed in canine samples of the present study and in a previous investigation studying the effects of lipemia on canine plasma. 35
According to the manufacturer, assays are insensitive to triglyceride concentrations up to 7.6 g/l for AT and 3.6 g/l for anti-FXa activity that have been added to human blood plasma. y In humans, lipemia was reported to result in false-low D-dimer measurements, y which was not in accordance with the current findings in canine specimens.
Based on the results of the current study, it can be concluded that most human coagulation tests can be used with the default settings for human specimens; however, some test protocols (i.e., protein C, protein S, FVIII, and plasminogen) have to be modified to work reliably with canine plasma specimens. The reference intervals for coagulation parameters derived with the STA Compact coagulation analyzer are equally useful for plasma specimens from male and female dogs. Lipemic plasma may be used for analysis of all coagulation parameters except D-dimer concentration. In contrast, sample recollection is recommended if hemolysis is present due to preanalytic causes.
Acknowledgements
The authors wish to thank Robert Siranovic, Roche Diagnostics GmbH, Mannheim, Germany, and Eberhard Hock, Scil animal care company, Viernheim, Germany, for their kind support of this study.
Footnotes
a.
STA Compact®, Roche Diagnostics GmbH, Mannheim, Germany.
b.
STA Neoplastin Plus, Roche Diagnostics GmbH, Mannheim, Germany.
c.
STA APTT Kaolin, Roche Diagnostics GmbH, Mannheim, Germany.
d.
STA Thrombin Reagenz, Roche Diagnostics GmbH, Mannheim, Germany.
e.
STA Fibrinogen, Roche Diagnostics GmbH, Mannheim, Germany.
f.
STA Unicalibrator, Roche Diagnostics GmbH, Mannheim, Germany.
g.
STA Liatest D-Dimer, Roche Diagnostics GmbH, Mannheim, Germany.
h.
STA Antithrombin III, Roche Diagnostics GmbH, Mannheim, Germany.
i.
STA Protein C Clotting, Roche Diagnostics GmbH, Mannheim, Germany.
j.
STA diluent buffer, Roche Diagnostics GmbH, Mannheim, Germany.
k.
STA Protein S Clotting, Roche Diagnostics GmbH, Mannheim, Germany.
l.
STA-Staclot APC-R, Roche Diagnostics GmbH, Mannheim, Germany.
m.
STA Plasminogen, Roche Diagnostics GmbH, Mannheim, Germany.
n.
STA Factor VIII, Roche Diagnostics GmbH, Mannheim, Germany.
o.
STA-Rotachrom Heparin, Roche Diagnostics GmbH, Mannheim, Germany.
p.
STA PreciClot Plus I, II, III; Roche Diagnostics GmbH, Mannheim, Germany.
q.
Liquicheck D-dimer control Level I and II, Roche Diagnostics GmbH, Mannheim, Germany.
r.
STA-Staclot APC-R Control N and P, Roche Diagnostics GmbH, Mannheim, Germany.
s.
STA Heparin Control I and II, Roche Diagnostics GmbH, Mannheim, Germany.
t.
Liquigen, Pfrimmer Nutricia GmbH, Erlangen, Germany. This fat emulsion consists of medium-chain triglyceride from fractionated coconut oil and water in equal parts.
u.
ADVIA 2120, Siemens Healthcare Diagnostics, Deerfield, IL.
v.
GraphPad Software Inc., La Jolla, CA.
w.
Analyse-it Method validation version 2.12, Analyse-it Software Ltd., Leeds, UK.
x.
HemosIL plasminogen assay, Instrumentation Laboratory GmBH, Kirchheim, Germany.
y.
Package insert of STA test kits (see footnotes b-s; information in English).

