Abstract
Tissue factor (TF)- and kaolin-activated thromboelastography (TEG) have been performed in a small number of healthy dogs, but reference intervals have not been assessed in a larger number of dogs. The goal of the current study was to establish reference intervals and assess intra-assay repeatability for kaolin-activated TEG in dogs. Additionally, the impact of sex and the influence of anticoagulant (native blood vs. recalcified citrate anticoagulated blood) were evaluated. Thromboelastography analyses were performed in 56 healthy dogs including German Shepherd Dogs (n = 19), Beagles (n = 15), and others (n = 22). Median age was 2 years (range: 1–6 years) and sex was evenly distributed (31 males and 25 females). To establish reference intervals, citrated whole-blood samples were collected, and TEG was performed 1 hr after sampling. Five TEG variables (R = reaction time; K = clot formation time; α = angle α; MA = maximal amplitude; G-value reflecting clot stability) were evaluated, and reference intervals were defined as the mean + 1.96-fold standard deviation. Intra-assay repeatability was assessed by calculating the pooled variance estimate in duplicate measurements of 6 healthy dogs. The effect of anticoagulant was assessed in 17 specimens. Reference intervals were as follows: R = 1.8–8.6 min; angle α = 36.9–74.6 degrees; K = 1.3–5.7 min; MA = 42.9–67.9 mm, and G = 3.2–9.6 Kdyn/cm 2 . Coefficients of variation for R, K, angle α, MA, and G were 7.6%, 17.7%, 7.4%, 2.9%, and 6.6%, respectively. There was no significant impact of sex or anticoagulant on results. Interindividual variation was higher in native samples than in citrated whole blood. A limitation of the current study was that most of the samples were obtained from Beagles and German Shepherd Dogs. This study provides useful reference intervals for kaolin-activated TEG.
Introduction
Traditionally, the hemostatic system has been divided into 3 phases, including primary hemostasis, secondary hemostasis, and inhibition of coagulation, including fibrinolysis and natural anticoagulants. 2,18 Conventional coagulation tests usually evaluate 1 isolated part of the coagulation system, whereas thromboelastography (TEG) provides information about all phases of clot formation and fibrinolysis. 17 TEG has been utilized worldwide to evaluate hemostasis in people and animals. 10 In canine patients, a variety of studies have been performed to assess their state of coagulation with TEG. A recent study using computerized TEG has shown that the tissue factor (TF)-activated TEG G-value correctly identified dogs with clinical signs of bleeding with high positive and negative predictive values. 29 Grading dogs with disseminated intravascular coagulation (DIC) in a hyper-, normo-, or hypocoagulable state based on the TF-activated TEG was reported to be of prognostic value. 26 In 49 dogs with neoplasia, hypercoagulability was the most common finding, while hypocoagulability was consistently present in dogs with metastasis. 16
In the majority of studies, 5 TEG variables (R, K, angle α, MA, and G) have been evaluated. Reaction (or gelation) time (R) is measured from the commencement of the test until preset fibrin formation occurs (i.e., until the trace is 2 mm wide). 1 Reaction time is primarily influenced by plasma clotting factors and natural anticoagulants or inhibitors of coagulation such as antithrombin, protein C, and protein S activity. The R-value is prolonged by anticoagulants and clotting factor deficiencies and shortened by hypercoagulable states. 1 As fibrin polymerization proceeds, clot stability increases, which is indicated by an increasing distance of leaflets constituting TEG tracings. The coagulation time (K) is the time required from initial clot formation until a predetermined clot strength is achieved and is measured from the end of R until tracings reach a diversion of 20 mm. Coagulation time is influenced by coagulation factors, plasma fibrinogen concentration, and platelet count. 1 Hyperfibrinogenemia, and to a lesser extent platelet hyperfunction, result in a shortened K-value, whereas the K-value is increased by anticoagulants affecting fibrin polymerization and platelet function. 1 Angle α is closely related to the K-value and indicates the rapidity of fibrin cross-linking, depending on plasma fibrinogen concentration and to a lesser degree on platelet count. 1 Similar to the K-value, angle α is increased by hyperfibrinogenemia and increased platelet function. Maximal amplitude (MA) is a measure of the peak rigidity manifested by the clot. About 80% of MA is dependent on platelet number and function. 28 Fibrin contributes to a lesser extent to clot strength. 1 For computerized TEG, a measure of clot rigidity (G) and thus the overall coagulation state (in dynes per square mm) is calculated as follows: G = (5000–MA)/(96–MA). 2 As G is an exponential reflection of MA, it is more indicative of small changes in clot rigidity or fibrinolysis than the amplitude in millimeters. 1
In all previous studies, diseased patients were compared with healthy controls. However, for routine use of TEG in veterinary practices, reference intervals should be established. Reference intervals are integral to laboratory testing and are helpful for differentiating between healthy and diseased patients. 14 Mean, standard deviation, 28 and range 25 of several TEG variables have been reported for both TF-activated TEG 28 and TEG performed with native recalcified citrated whole blood 25 in 18 and 15 healthy dogs, respectively. In a further investigation, an index of individuality and biological variation of TEG variables have been assessed for recalcified citrated plasma samples in 8 dogs and, thus, provided useful information for interpretation of population-based reference intervals. 27 To the authors’ knowledge, however, TEG variables and reference intervals have not been assessed in a larger number of healthy dogs.
Regarding establishment of reference intervals in humans, the International Federation of Clinical Chemistry (IFCC) has proposed that at least 40 individuals should be included in the reference population to provide an accurate calculation of the 0.025 and 0.975 percentiles. 23 For interpretation of TEG test results, the interindividual variation has to be considered. One investigation demonstrated substantial individual variation of coagulation parameters, including clotting time, activated partial thromboplastin time (aPTT), prothrombin time (PT), thrombin time (TT), fibrinogen, antithrombin (AT), and fibrin D-dimer. These test results, when compared with population-based reference ranges, provide insensitive criteria for the interpretation of patient data. 27 In contrast, population-based reference intervals appeared to be more sensitive for interpreting all patient-based TEG parameters because of a low intra-individual variation. 27
In humans, TEG has been considered a useful measure of global hemostasis, but its use has been limited by the fact that fresh native blood must be used within 6 min after venipuncture. 6 If transport and handling time of native blood exceeds 6 min, citrate anticoagulation with subsequent recalcification immediately prior to analysis has been recommended. 6 Studies in healthy adult volunteers 4,30 and healthy children 5 undergoing minor surgical procedures have demonstrated that TEG variables were significantly different when measured in recalcified citrated whole blood (analyzed 30 min after sampling) compared with native whole blood. To the authors’ knowledge, the effect of the anticoagulant has not been evaluated in dogs. In addition to the anticoagulant, the activator of the coagulation process has to be considered. Results of TF-activated TEG are not necessarily comparable with kaolin-activated TEG, as the first represents activation of the extrinsic coagulation pathway, whereas the latter represents activation of the intrinsic pathway of the coagulation cascade.
The aims of the current study were to establish reference intervals for TEG variables in dogs using kaolin-activated TEG analysis and to investigate the influence of sex on the TEG test results. Furthermore, the effect of the use of an anticoagulant was compared with the use of native whole blood in the performance of TEG.
Materials and methods
Study design
This prospective investigation was approved by the Ethics Committee for animal welfare, Giessen, Germany (no. V54–19c20/15cGi18/17; January 2008). The aim of the current investigation was to establish reference intervals for 5 variables assessed with kaolin-activated TEG and to evaluate intra-assay repeatability. The impact of sex and anticoagulant use on TEG performance was also evaluated. To minimize technical variables, TEG analysis was performed by one of the authors (Eralp).
Dogs
This study was conducted in adult (>1 year old, <6 years old) dogs, including staff-owned dogs, blood donors, or healthy dogs presented at the Clinic for Small Animal Surgery, Department of Veterinary Clinical Sciences, Faculty of Veterinary Medicine, Justus-Liebig University (Giessen, Germany) for routine radiological examination to screen for hereditary hip (HD) or elbow dysplasia (ED). Inclusion criteria were a normal physical examination, complete blood cell count (CBC), biochemical profile, and coagulation profile. The plasma biochemical profile included urea, creatinine, total protein, albumin, globulin, fructosamine, sodium, potassium, ionized calcium, magnesium, and phosphate concentrations as well as the determination of the activities of alkaline phosphatase, alanine aminotransferase, and glutamate dehydrogenase. In addition, none of the dogs in the study population had a medical history of bleeding tendencies, and none of the dogs had been medicated for 2 weeks prior to presentation at the clinic.
The standard plasma coagulation profile consisted of PT, aPTT, TT, fibrinogen concentration, and factor VIII (FVIII) activity. These parameters were determined with an automated coagulation analyzer. a Prothrombin time, aPTT, and TT were measured automatically in clotting tests using commercial reagents. b , c , d Fibrinogen concentration was measured by the Clauss method using a human plasma calibration standard e provided by the manufacturer. Measurement of FVIII activity was performed in a modified 1-stage aPTT using a human FVIII-deficient substrate plasma. f Patient samples were routinely diluted 1:40 with buffer. Factor VIII activity was calculated by comparing the results with a canine standard curve prepared by serial dilution of canine pooled plasma. g Dogs with FVIII activity <60% compared with canine pooled plasma were excluded from the study.
Blood samples
Blood samples were acquired from fasted, resting dogs immediately after placing an 18-gauge nonheparinized venous catheter in the cephalic vein. In staff-owned dogs and blood donors, the catheter was removed immediately after the blood sample was taken. In patients presented for ED or HD screening, anesthesia was necessary, and the catheters remained in place during radiological examination. When the blood samples were obtained, the first 2 ml were used for the CBC and biochemical profile to reduce the effects of tissue thromboplastin on the hemostasis tests. For measurement of hemostasis parameters, venous blood samples were drawn into siliconized vacutainer tubes containing 3.18% trisodium citrate anticoagulant. The ratio of whole blood to anticoagulant was 9:1 (vol/vol).
Blood was allowed to drop directly into the citrated tube or it was aspirated in a plastic syringe and gently transferred into the citrate-containing tube after the needle was removed from the syringe. The blood and anticoagulant subsequently were mixed. The blood samples were carefully checked for proper filling of the collection tubes; only specimens with a 9:1 ratio of blood to citrate anticoagulant were included in the study. Any contact with ethylenediamine tetra-acetic acid (EDTA) was strictly avoided. For TEG analysis, 1.17 ml of venous whole blood was placed in a 40 mm x 1.8 mm, silicone-lined microtube containing 0.13 ml of 3.18% trisodium sodium citrate and was allowed to equilibrate at room temperature for 1 hr.
TEG analysis
For establishment of reference intervals, TEG was performed with recalcified citrated whole blood using a commercial thromboelastograph h according to the manufacturers’ recommendations. Briefly, 1 ml of citrated whole blood was placed in a silicated vial i provided by the manufacturer. The vial contained kaolin, buffered stabilizers, and a blend of phospholipids. Mixing was ensured by gentle inversion of the kaolin-containing vials 5 times. Pins and cups j were placed in the TEG analyzer in accordance with the standard procedure recommended by the manufacturer. Each standard TEG cup was placed in the 37°C prewarmed instrument holder and was filled with 20 μl of calcium chloride (0.2 M). Then, 340 μl of kaolin-activated citrated whole blood was added for a total volume of 360 μl. Two levels of internal quality control materials (normal and abnormal) were assayed prior to each run. j An electrical internal quality control (so called e-test) was also performed.
Influence of the anticoagulant on TEG measurements
The comparison of the TEG results obtained with native whole blood or citrated whole blood was performed in 16 healthy dogs with a median age of 3 years (range: 2–6 years). The study group included 10 males, 3 females, and 3 spayed females. Sampling was performed as described above for the dogs used in the reference interval study, with the following modification: 1 ml of whole blood was placed in a silicated kaolin-containing tube in addition to a citrated vial. The first 2 ml of the blood obtained from the catheter was used for platelet count to reduce any effects of tissue thromboplastin contamination.
The native blood in the kaolin-containing tube was mixed by gentle inversion of the vial 5 times, 360 μl of blood was pipetted into a prewarmed TEG cup placed in the instrument holder, and the TEG tracing was initiated. With the native whole-blood specimens, TEG analysis was performed within 6 min of sampling. In contrast, the citrated whole-blood specimen was allowed to rest 1 hr at room temperature, and TEG was performed using recalcified blood as described for the samples used to establish the reference intervals.
Intra-assay repeatability
Sixty minutes after sampling, duplicate measurements were made from recalcified citrated blood with 1 TEG analyzer (2 channels per machine). A total of 6 healthy dogs with a median age of 2.5 years (range: 1–6 years) were studied. This group included 3 males, 2 neutered males, and 1 female. Intra-assay variation was assessed for TEG variables R, K, angle α, MA, and G by calculating the arithmetic mean and the pooled variance estimate based on the differences in the duplicate determinations. The standard deviation (SD) required for calculation of the coefficient of variation (CV) was consistent with the root of the pooled variance estimate.
Statistical analysis
Results were analyzed with Graph Pad Prism 1 and Analyse-it Method Evaluation Edition m statistical software. An Anderson-Darling test was performed to verify the assumption of normality. If the data was normally distributed, double-sided reference intervals were obtained by calculating the mean + 1.96 SD so that 95% of the reference population was included. The 90% confidence intervals (CIs) of the upper and lower reference limits were calculated automatically with the Analyse-it software. If the data were not normally distributed, a logarithmic transformation was performed, and the data were reanalyzed using the Anderson-Darling test. In case of normal distribution after transformation, logarithmic data were analyzed as described above. Data were depicted as histograms including the reference interval as well as the 90% CIs of the upper and lower reference limits as recommended by the IFCC. 23
The influence of the anticoagulant was assessed using a paired t-test. An unpaired t-test or a comparable nonparametric test (Mann-Whitney U test) was applied to investigate the effect of sex on TEG variables. After Bonferroni correction, significance was set at P < 0.01 for comparison of the influence of anticoagulant and sex.
Results
Reference intervals and intra-assay repeatability
Fifty-six dogs (19 German Shepherd Dogs, 15 Beagles, 8 Golden Retrievers, 4 Labrador Retrievers, 2 mixed-breed dogs, 1 Hovawart, 2 Great Danes, 1 Saint Bernard, 1 English Springer Spaniel, 1 Bernese Mountain Dog, 1 Boxer, and 1 Giant Schnauzer) with a median age of 2 years (range: 1–6 years) were included in the study. Sex was evenly distributed with 24 males, 20 females, 7 neutered males, and 5 spayed females. The dogs were clinically healthy based on physical examination as well as the results of the CBC, biochemical profile, and routine coagulation profile. The majority of the study variables were normally distributed, except for results of the K-value, which demonstrated a right-skewed distribution of data (Fig. 1A), which could be normalized after logarithmic transformation. Reference ranges and 90% CIs are displayed in Table 1 and Figure 1. As shown in Table 2, TEG variables did not differ significantly between male and female dogs. Intra-assay CVs for kaolin-activated TEG variables R, K, angle α, MA, and G were 7.6%, 17.7%, 7.4%, 2.9%, and 6.6%, respectively.
Influence of the anticoagulant on TEG measurements
The influence of the anticoagulant (native blood vs. recalcified citrate anticoagulated blood) on TEG R-value, angle α, K-value, MA, and G-value is depicted in Figure 2. As shown in the Figure, no significant effect of the anticoagulant was present, although the interindividual variation of results was lower for citrated samples.
Discussion
According to the proposals of the IFCC, at least 40 reference values are needed to obtain the 0.025 and 0.975 percentiles. 23 Given the proposal for establishment of human reference intervals, the number of values included should preferably be more than 120, but the quantity of samples required is strongly dependent on the confidence intervals. 23 The number of dogs included in the current study was lower than the optimal number recommended by the IFCC to establish reference intervals; however, even in humans, it is generally accepted that a smaller number of values may still be useful clinically for interpretation of patient test results. 23 Proposals for establishment of TEG reference intervals have not been published in the veterinary literature; however, a minimum of 40 animals has been recommended 12 for construction of reference intervals based on a previous publication. 23 The use of IFCC proposals in veterinary medicine is hampered by the fact that sample collection and laboratory analysis is expensive and time-consuming, and it is often difficult to identify enough healthy subjects. Therefore, alternative statistical methods have been evaluated to estimate reliable reference intervals even in data sets containing diseased animals. 8
Thromboelastography has rarely been performed in healthy dogs. The mean, SD, 25,27 and range 28 of TEG variables (R, K, angle α, MA, 27,28 and G 25 ) have been published for both TF-activated TEG (n = 18 dogs 25 ; n = 8 dogs 27 ) and TEG performed with native recalcified citrated whole blood (n = 15 dogs). 28 However, reference intervals have not been established by these authors. The current study is the first to provide reference intervals for kaolin-activated TEG in dogs. In humans, reference intervals for kaolin-activated TEG are as follows: R = 5.3–9.3 min, K = 1.4–3.5 min, angle α = 48.8–72.2 degrees, and MA = 55.3–69.3 mm (G was not evaluated). 5 Maximal amplitude and angle α were similar in humans (adults and children) and in the dogs investigated in the present study, but the lower reference limit of the R-value was considerably shorter in dogs (1.8 min) than in humans (5.3 min), indicating a higher activity of secondary hemostasis. 5 This is supported by previous reports that dogs have greater coagulation activity of the extrinsic and intrinsic pathways compared with humans. 8
Based on data obtained from 100 children of different age groups, age-related differences in kaolin-activated TEG were not indentified. 5 However, establishment of reference intervals for TEG variables in newborns revealed that TEG reaction time and G-value were significantly lower in neonates (P < 0.001) than in adults. 11 In the latter study, TEG was conducted without the use of an activator. Comparable studies have not been performed in dogs, so
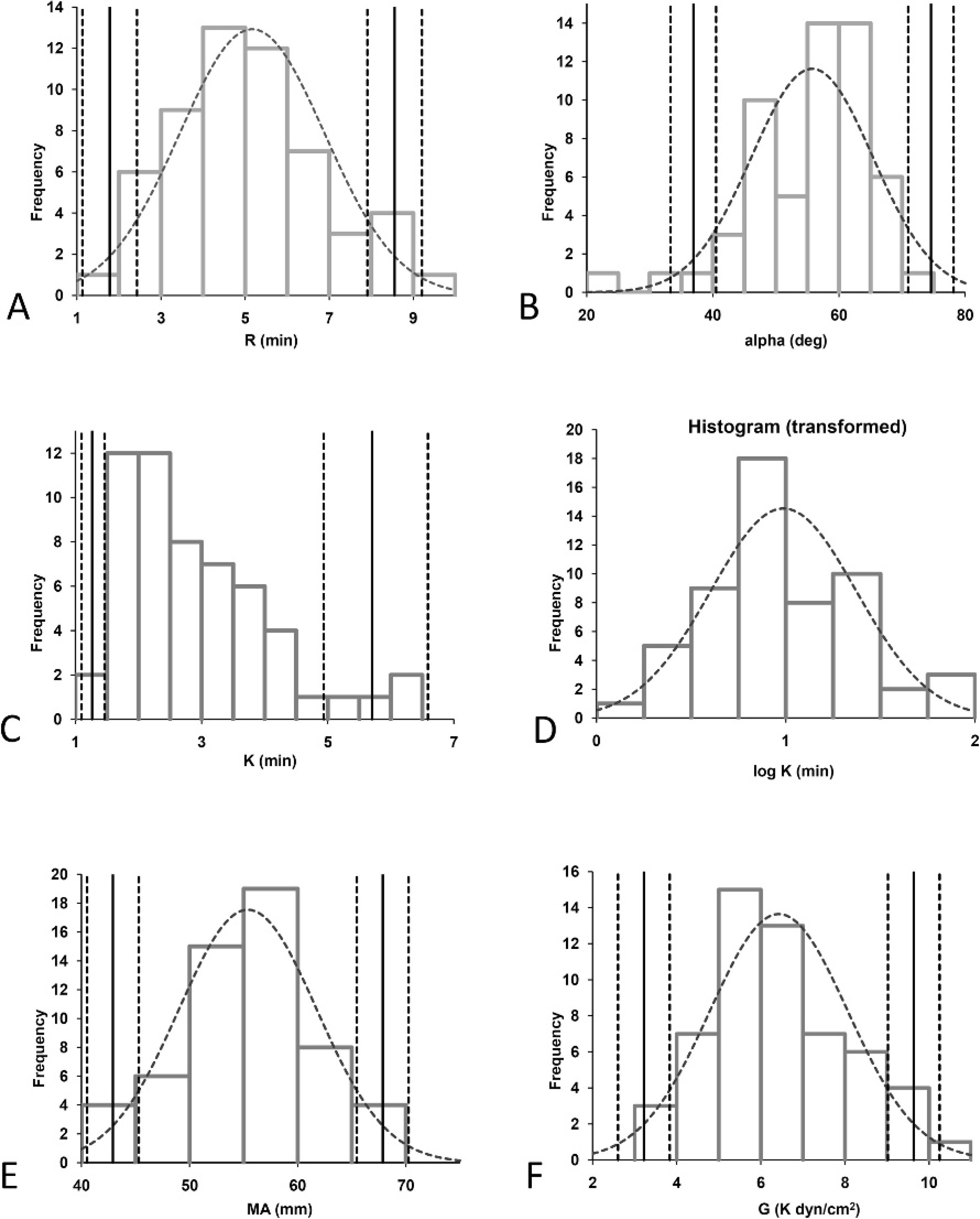
Histograms demonstrating the distribution of data obtained for calculation of reference intervals (n = 56 healthy dogs). The solid vertical line indicates the reference interval. The dotted lines indicate the 90% confidence interval (CI) of the upper and lower reference limits. The dotted curve indicates the Gaussian curve in case of normal distribution of the data. Log normal distribution is characterized by the lack of a Gaussian curve, but the CI is shown. In the case of nonnormal distribution of the data, only the upper and lower reference limits are shown. R = reaction time; K = coagulation time; α = angle α; MA = maximum amplitude; G = clot rigidity.
puppies or growing dogs have been excluded from the current investigation.
Tissue factor-, kaolin-, and celite-activated TEG have been conducted in human specimens to investigate the in vitro effect of the novel kallikrein inhibitor DX88 on TEG using different types of activation compared to control specimens. 24 R-time in the controls was shorter in kaolin-activated TEG than
Reference intervals for kaolin-activated thromboelastography variables and 90% confidence intervals (CIs) of upper and lower reference limits determined in recalcified citrated whole blood (n = 56 healthy dogs). *
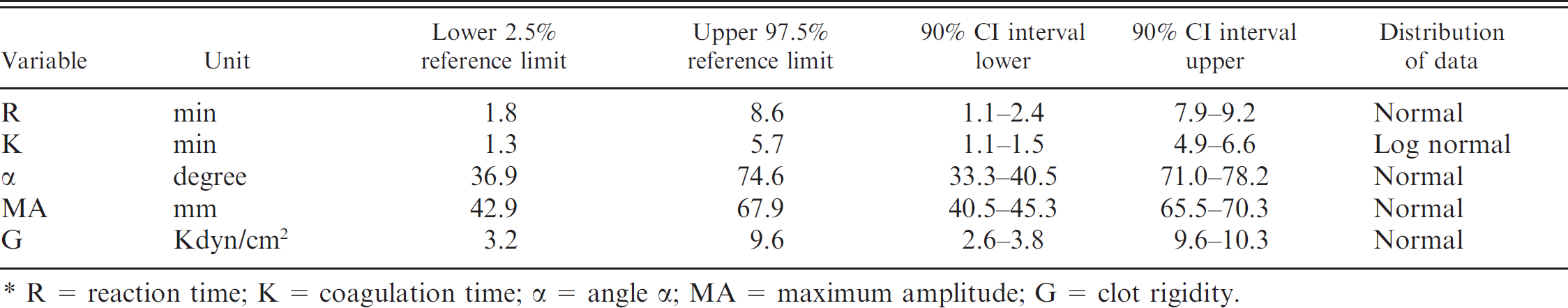
R = reaction time; K = coagulation time; α = angle α; MA = maximum amplitude; G = clot rigidity.
Influence of sex on results (mean + standard deviation) of different thromboelastography variables (n = 31 male dogs and 25 female dogs). *
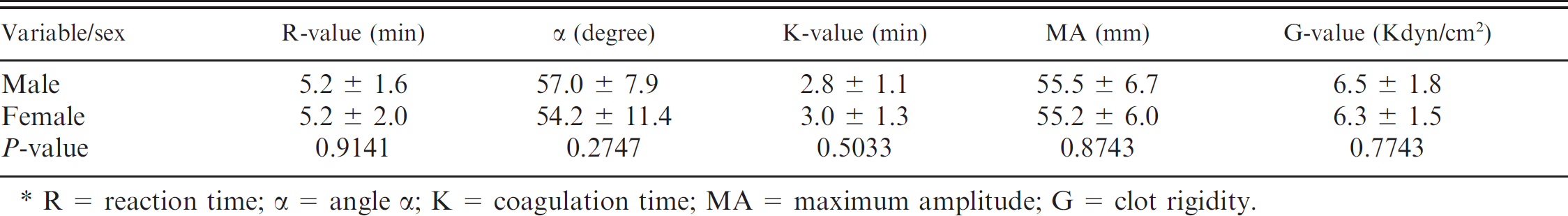
R = reaction time; α = angle α; K = coagulation time; MA = maximum amplitude; G = clot rigidity.
in TF-activated TEG. However, the direct comparison of kaolin- and TF-activated TEG in the control samples was not the aim of the study, so the possibility that the difference was significant was not investigated. 24 These results reinforce the importance of consulting activator-specific TEG reference intervals when interpreting patient test results.
The current study demonstrated that TEG parameters were similar in male and female dogs. In contrast, sex is known to have an impact on several variables of coagulation in humans. Males were reported to have higher plasma AT and protein C concentrations, 9 whereas females had higher plasma D-dimer concentrations. 15 An age-dependent decrease in PT was noted in males (r = −0.34; P < 0.001) but not in females (r = −0.04; P = 0.47). 22 However, information concerning the influence of sex on TEG variables apparently is lacking in humans and dogs.
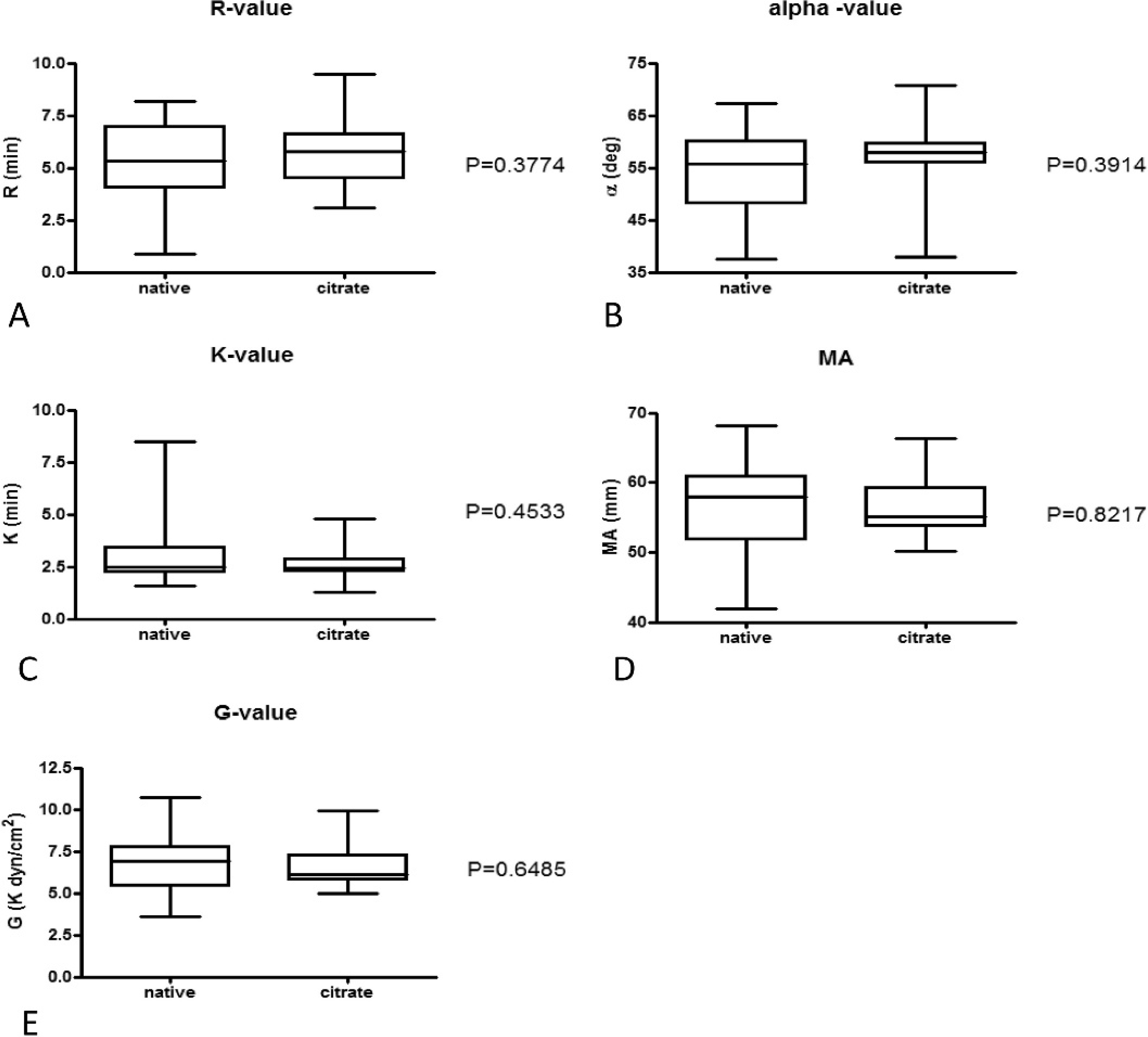
Box and whisker diagrams depicting thromboelastogram measurements performed using fresh whole blood without anticoagulant directly after sampling or using recalcified citrated whole blood 1 hr after sampling (n = 16 healthy dogs). The central box represents the values from the lower to upper quartiles. The middle line is the median. The horizontal lines indicate minimum to maximum values. The level of significance was P = 0.01 after Bonferroni correction. R = reaction time; K = coagulation time; α = angle α; MA = maximum amplitude; G = clot rigidity.
The castration status was not taken into account in the current study, as the number of neutered dogs was comparatively low.
A limitation of the current study was that most of the blood samples were obtained from Beagles and German Shepherd Dogs. These 2 breeds constituted most of the animals available at the university clinic that were either blood donors (mainly medium-sized to large breed dogs and Beagle dogs) or healthy dogs presented for routine radiological examination to screen for hip dysplasia or elbow dysplasia (which is known to affect large breed dogs). Because of the predominance of Beagles and German Shepherd Dogs in the reference population, the results obtained in the present study might not be representative for all dog breeds. Breed-specific differences in routine coagulation have rarely been investigated in dogs. One study compared TEG in 28 healthy Greyhounds and 15 non-Greyhound dogs. Clotting kinetics were significantly slower, and clot strength was significantly weaker in Greyhounds than in non-Greyhounds, except for R-time. 25 In the current study, Greyhounds were not included.
Previous investigations using canine citrated whole-blood specimens have shown a significant trend toward hypercoagulability indicated by a decrease in the R- and K-values, as well as an increase in angle α and MA when the storage time exceeded 120 min. 28 Thus, a fixed time point for measurements was recommended as performed in the current study. 28
Using canine citrated recalcified whole blood, coefficients of variation have been assessed for TF-activated TEG by calculating the arithmetic mean and the pooled variance estimate based on the differences of duplicate determinations after 30 min and 120 min, respectively. 28 Mean CVs for both time points for R, K, angle α, and MA were 6.46%, 15.71%, 7.86%, and 4.30%, respectively. 28 These results were similar to the CVs for kaolin-activated TEG obtained in the present study.
Based on the results of the current study, the anticoagulant did not have any significant impact on TEG measurements. In contrast, studies in adult healthy volunteers (n = 305 and n = 1030) and healthy children (n = 14) 21 undergoing minor surgical procedures demonstrated that TEG variables were significantly different when measured in recalcified citrated whole blood (analyzed 30 min after sampling) compared with native whole blood. In recalcified citrated whole blood, the R- and K-values were significantly shorter than in native blood whereas angle α and MA were significantly higher. 21 Other investigators did not demonstrate a significant difference in TEG between native whole-blood samples and citrated recalcified whole blood (n = 8) when assessed after storage for 30 min at room temperature. However, a significant decrease in K and an increase in angle α was observed after 120 min of storage. 3 These findings were in contrast to the results obtained in the current study. The number of dogs evaluated in the present study was low and significant effects of variables on TEG might have been missed, but the number of patients included was even lower in the majority of human studies demonstrating significant differences between native whole-blood samples and recalcified citrated blood. The most probable reason for the trend in hypercoagulation of citrated blood with respect to native blood has been highlighted in a previous study that found that citrate does not completely inhibit thrombin formation and activation of coagulation was not completely inhibited. 5 In the cited human investigations, celite was used for TEG analysis, 4 or thromboelastography was performed without any activating substances. 3,21,30 Therefore, it can be hypothesized that differences in activation of coagulation might be responsible for the different findings obtained in the current study. In humans, kaolin is known to act as a stronger activator compared with celite as reflected by a significantly shorter R (P = 0.0052) and K (P = 0.0004) times. 20 If kaolin is capable of inducing strong activation of clotting in native whole-blood samples, mild hyper-coagulability caused by the incomplete inhibition of thrombin activation in citrated specimens would not have been detected. This theory is supported by previous investigations in humans demonstrating that routine coagulation assays using potent activators of aPTT, PT, and TT are insensitive to minor thrombin generation during storage of citrated blood. 1,19 It is also possible that thrombin formation is completely inhibited in canine specimens as opposed to human specimens. However, there are no studies substantiating this hypothesis. In any event, the reference intervals reported in the present study may provide useful reference intervals for kaolin-activated TEG in canine blood specimens. Because of the higher interindividual variation in native blood samples, the use of citrated whole blood is preferred for TEG in dogs, especially for clinical studies.
Acknowledgements
The authors wish to thank Dirk Rohmann, P. J. Dahlhausen & Co. GmbH, Cologne, Germany, for kindly supporting this study, and Dr. Klaus Failing, Institute for Biomathematics and Data processing, Justus-Liebig University, Giessen, Germany, for his kind advice in statistical questions.
Footnotes
a.
STA Compactr, Roche Diagnostics GmbH, Mannheim, Germany.
b.
STA Neoplastinr Plus, Roche Diagnostics GmbH, Mannheim, Germany.
c.
STA APTT Kaolin, Roche Diagnostics GmbH, Mannheim, Germany.
d.
STA Fibrinogen, Roche Diagnostics GmbH, Mannheim, Germany.
e.
STA Unicalibrator, Roche Diagnostics GmbH, Mannheim, Germany.
f.
STA Factor VIII, Roche Diagnostics GmbH, Mannheim, Germany.
g.
Canine pooled plasma: Citrated blood was collected from 16 healthy adult dogs (8 females, 6 males, and 2 spayed females) with a median age of 3.5 years (range: 1–8 years) for harvesting of pooled plasma. The dog breeds sampled included 3 Beagles, 2 Belgian Malinoises, 2 Labrador Retrievers, 2 French Bulldogs, 2 Maremma Sheepdogs, 2 German Shepherd Dogs, 1 Rottweiler, 1 Staffordshire Bull Terrier, and 1 mixed breed. All dogs were healthy based on the medical history, physical examination, CBC, and biochemical profile. The dogs used as blood donors for pooled plasma preparation were not included in the study population used to establish the reference intervals.
h.
TEGr 5000 Thrombelastograph, Haemonetics Corporation (formerly Haemoscope Corporation), Braintree, MA.
i.
TEGr Hemostasis System Kaolin, Haemonetics Corporation, Braintree, MA.
j.
TEGr Hemostasis System Pins and Cups, Haemonetics Corporation, Braintree, MA.
k.
TEGr Coagulation Control Level I and II, Haemonetics Corporation, Braintree, MA.
l.
Graph Pad Software Inc., San Diego, CA.
m.
Analyse-itr Method Evaluation Edition for Microsoftr ExcelTM, Analyse-it Software Ltd., Leeds, UK.
