Abstract
Vascular hamartomas are considered developmental lesions rather than true neoplasms. Reports of such anomalies in the canine brain are scarce, and their classification is confusing. This case series of vascular hamartomas from the brains of five dogs was characterized using histochemistry and immunohistochemistry, in addition to gross and microscopic findings. All five hamartomas were located in the telencephalon, three in the pyriform lobe, without any predilection for the left or right side. Each hamartoma consisted of a proliferation of thin-walled vessels which varied in caliber. These vessels were elastin-negative, with varying amounts of collagen and no muscular component. In four of the five hamartomas, lining cells were actin- and factor VIII-positive. All five hamartomas contained glial fibrillary acid protein (GFAP)-positive parenchyma at moderate to high frequency, and four contained neurofilament-positive axons between component vessels. This report shows that vascular hamartomas in the canine brain are structural malformations for which immunohistochemistry is useful for accurate classification.
Vascular hamartomas are defined as disorganized and excessive proliferations of vascular tissue 11 which, due to their limited growth, are considered developmental malformations rather than true neoplasms. Usually, the use of the term “hamartoma” implies the presence of the lesion at birth, with subsequent growth paralleling that of the animal but ceasing with maturity. 10 In the human brain, vascular hamartomas are classified as capillary telangiectasis, cavernous angiomas, and arteriovenous or venous malformations. 16 In the veterinary literature, vascular hamartomas have been rarely described in the canine, equine, and bovine brain, 2 8 whereas single cases of such malformations in the spinal cord of a dog, 7 a Hereford calf, 6 a goat, 12 and a foal 9 have been reported. Classification of such lesions remains confusing, and terminology between reports tends to vary; designations such as angiomatous hamartoma, telangiectatic hamartoma, and cerebral angiomatosis have been used to describe morphologically similar lesions. 8 15 In human medicine, the clinical significance of hamartomas is generally attributed to their potential to spontaneously hemorrhage, their ability to act as space-occupying lesions, and their association with seizure activity. 16 The objective of this study was to characterize a case series of vascular lesions of the canine brain that were originally diagnosed as vascular hamartomas. Histochemistry and immunohistochemistry were used as additional tools to further define vessel structure and the relationship between the vessels of each hamartoma with the surrounding brain parenchyma.
Formalin-fixed, paraffin-embedded brain tissue from five dogs diagnosed histologically as having vascular hamartomas were obtained from the necropsy (n = 4) and biopsy (n = 1) services at the University of Pennsylvania School of Veterinary Medicine. Material had been stored in paraffin blocks for up to 7 years. Sections were cut 5-µm thick and stained with hematoxylin and eosin. Selected representative sections from each case were also stained with Masson's trichrome stain for collagen and smooth muscle and Miller's elastin stain for elastic fibers and collagen. Further sections were mounted on Probe-On Plus slides (Fisher) for immunostaining using an indirect peroxidase method. Primary mouse monoclonal antibodies were used against human neurofilament at 1:200 dilution (Dako) and muscle-specific actin (BioGenex, prediluted antibody), whereas rabbit polyclonal antibodies were used against bovine glial fibrillary acid protein (GFAP) at 1:2,000 dilution (Dako) and factor VIII at 1:1,000 dilution (Dako). Sections stained with antibodies to GFAP and factor VIII were treated with Citra solution (BioGenex) and heated in a microwave for 7 minutes at 200 W. Prior to staining with antibodies against neurofilament, sections were pretreated with trypsin (1 mg/ml, pH 7.7) for 10 minutes, and prior to staining with antibodies against actin, sections received no pretreatment. All sections were blocked with 3% H2O2 for 10 minutes, incubated with the relevant primary antibody for 30 minutes at room temperature, followed by biotinylated anti-mouse or anti-rabbit secondary antibodies (Dako) for 15 minutes. Finally, sections were incubated with a peroxidase label for 15 minutes, and its presence was visualized using a 4-minute incubation at room temperature with 3,3′-diaminobenzidine (Dako). For each set, a negative control consisted of substituting the primary antibody with one that was inapplicable to the tissues concerned, in this case, cytokeratin AE1/AE3 (Dako) at 1:100 dilution. Normal brain parenchyma and blood vessels served as internal controls for the anti-GFAP, -actin, –factor VIII, and -neurofilament antibodies. The frequency of staining for each antibody was evaluated subjectively by the ability to see positive cells at different levels of magnification. Positive staining was considered highly frequent when positive cells could be seen at low magnification (2× or 4×), moderately frequent when they could be seen at intermediate magnification (10×), and rare when they were only evident at high levels of magnification (40×).
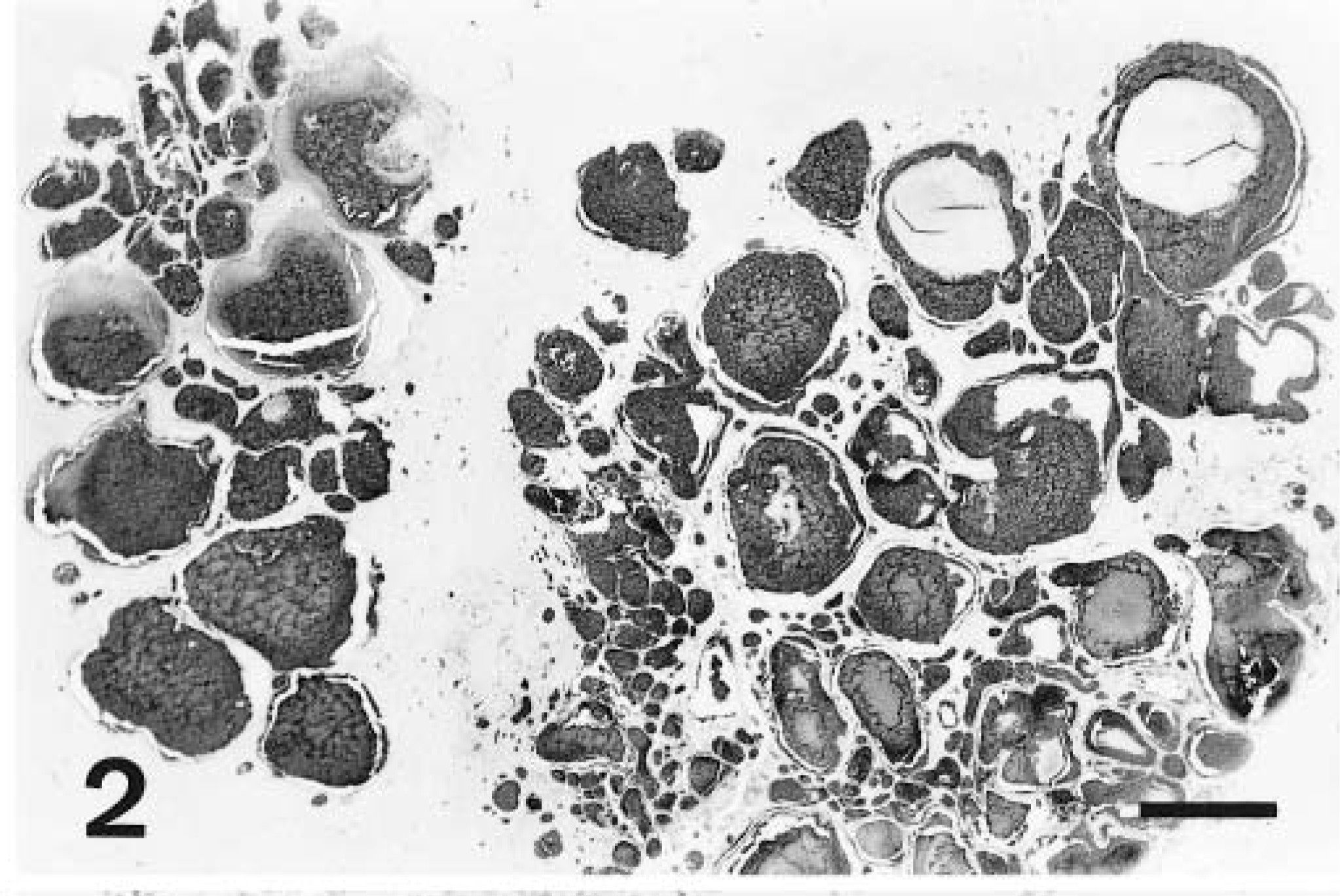
The case signalment, size, and location of the lesion and the presence or absence of hemorrhage are summarized for each case in Table 1. Age at time of diagnosis ranged from 4 to 15 years, and in the small number of cases available, no sex or breed trend was seen. In all four necropsy cases, the dogs had been euthanatized, but the initial reason for presentation varied. Dog No. 1 was a brain collected at necropsy and submitted to us by a referring veterinarian through our biopsy service. The clinical history is no longer available for this case. Dog No. 2 had been diagnosed with idiopathic epilepsy and had a 2-month history of generalized weakness attributed to polyneuritis, which was confirmed histologically in the radial, tibial, and sciatic nerves. Dog No. 4 had a 1-day history of shaking, abnormal mentation, seizure activity, and constant head tremors. Dog Nos. 3 and 5 were considered incidental hamartomas. In the former, there was histologic evidence of thyroiditis and atherosclerosis, with a 5-day history of hindlimb ataxia due to the presence of a thromboembolus at the aortic bifurcation. In the latter, the main presenting condition was thrombocytopenia, secondary to megakaryocytic aplasia. Full necropsies had been performed on dog Nos. 2–5, and none had any evidence of vascular neoplasia or congenital lesions elsewhere in the body. The hamartomas were located in the telencephalon in all five cases, and the pyriform lobe was involved in three of the cases. There was no propensity for one side of the brain to be affected more frequently than the other. In one dog (No. 4), multiple hamartomas were diagnosed, involving the right thalamus and the hippocampus and caudate nuclei, bilaterally. Grossly, the hamartomas presented as discrete areas of softening in the brain parenchyma with brown or black discoloration (Fig. 1) or as raised, multilobulated, purple/red masses. One had a cavitated center, which exuded brown fluid on sectioning. The lesions ranged from 0.4 to 2.0 cm in diameter. Microscopically, each consisted of focal or multifocal, nonencapsulated proliferations of thin-walled, blood-filled channels, involving both grey and white matter. The channels varied in caliber and were lined by flattened endothelial cells. Variably, there was minimal to mild compression of the surrounding parenchyma, and in one hamartoma (dog No. 3), numerous vessels were partially occluded by fibrin thrombi, which were infiltrated by low numbers of plump, spindle-shaped cells. The blood-filled channels occasionally abutted each other, but most were separated by intervening trabeculae of brain parenchyma (Fig. 2).
Characterization of vascular hamartomas from dog Nos. 1–5.
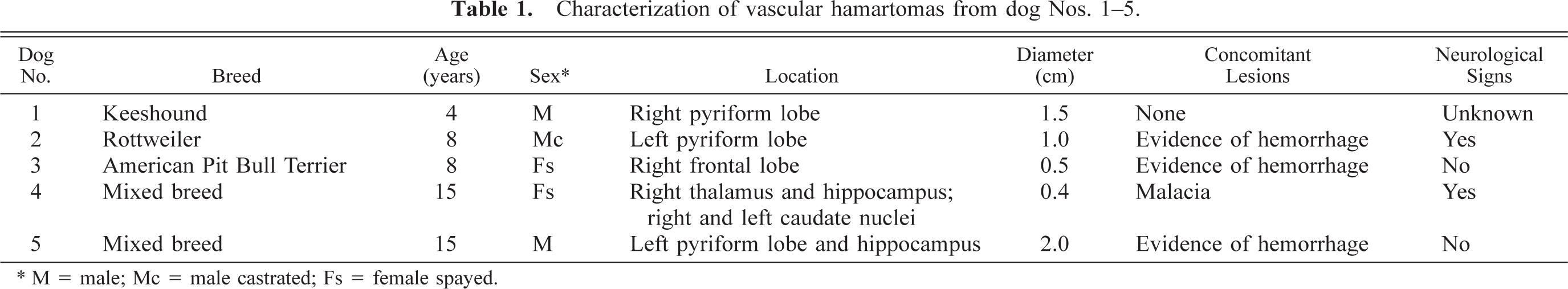
M = male; Mc = male castrated; Fs = female spayed.
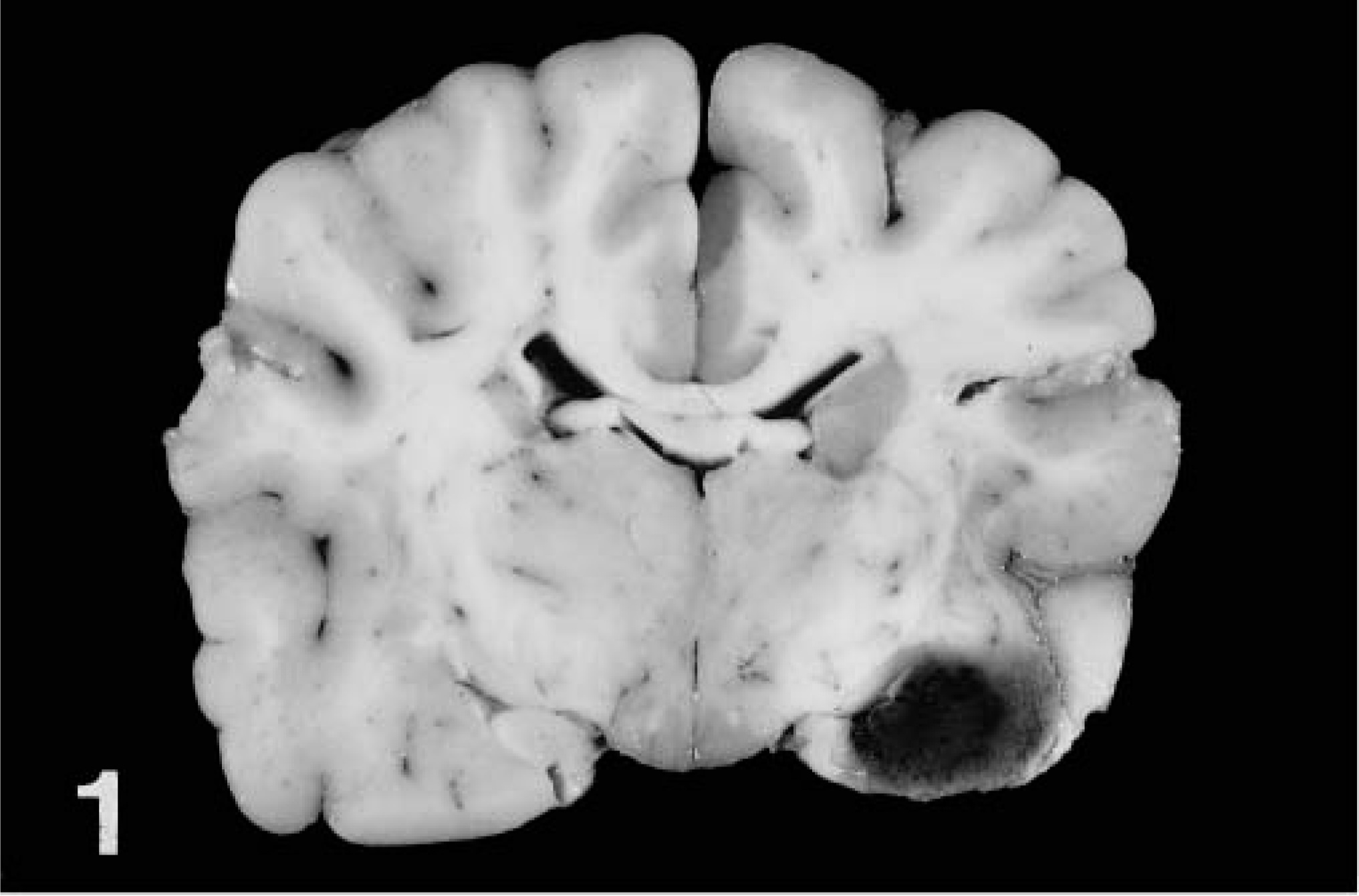
Vascular hamartoma, dog No. 1. Sagittal section of brain at level of thalamus, demonstrating the gross appearance of the vascular hamartoma in the left pyriform lobe.
In three of the hamartomas, the parenchyma between and adjacent to the proliferating vessels was infiltrated by moderate numbers of hemosiderin-laden macrophages, with or without hematoidin deposition and variable degrees of hemorrhage, which was most extensive in dog No. 3. Consistently, the hamartomas were surrounded by more distantly located proliferating capillaries. It was difficult to be certain whether these were part of the hamartoma or a reaction to it. All cases had associated gliosis. In one brain (dog No. 4), which had no evidence of hemorrhage, there was one perivascular focus of moderate numbers of lymphocytes and plasma cells in the meninges and multifocal areas of malacia, with clusters of chromatin debris and karyorrhectic cells surrounding the hamartoma.
In all brains, normal parenchymal and meningeal vessels stained positively with Miller's elastin and trichrome stain. The endothelium of all normal vessels was intensely factor VIII–positive and the tunica media was intensely and diffusely actin-positive. No endothelial cells in normal vessels were actin-positive. In the brain parenchyma, all astrocytes stained intensely GFAP-positive, whereas many axons and neuronal cell bodies were neurofilament-positive. For each set of sections, there was no specific staining in the negative controls. Vessels within the hamartoma were elastin-negative, and the amount of collagen present in the vessels walls varied between cases. In all hamartomas, factor VIII– and actin-specific staining were restricted to the endothelial lining cells of the vessels (Figs. 3, 4), except in dog No. 3, where cells infiltrating the thrombi were also actin-positive, and in dog No. 4, where lining cells were only factor VIII–positive. In four cases, factor VIII and actin staining were present at moderate to high frequency. In dog No. 2, factor VIII–positive cells were present, although rare. In all five hamartomas, GFAP-positive astrocytic processes were observed at moderate to high frequency between vessels of the hamartoma (Fig. 5). Neurofilament-positive axons were noted at moderate to high frequency between the vessels of the hamartoma in four cases and were rare in one case (Fig. 6). In the surrounding proliferating capillaries associated with all hamartomas, the endothelial cells and tunica media were uniformly and strongly positive for factor VIII and actin, respectively. In dog No. 5, the extensive hemorrhage obscured immunostaining to some degree, but it was still possible to discern specific staining.
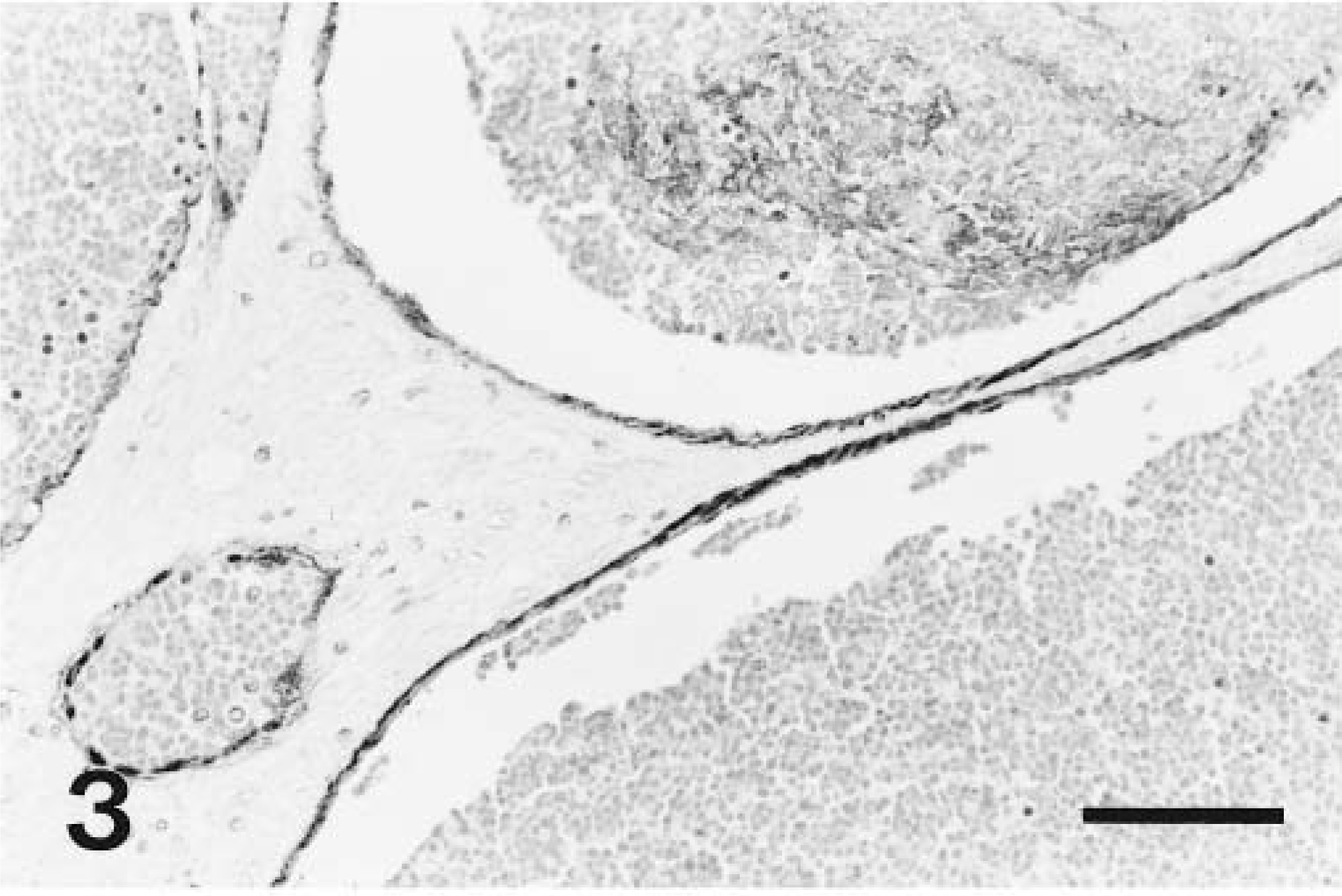
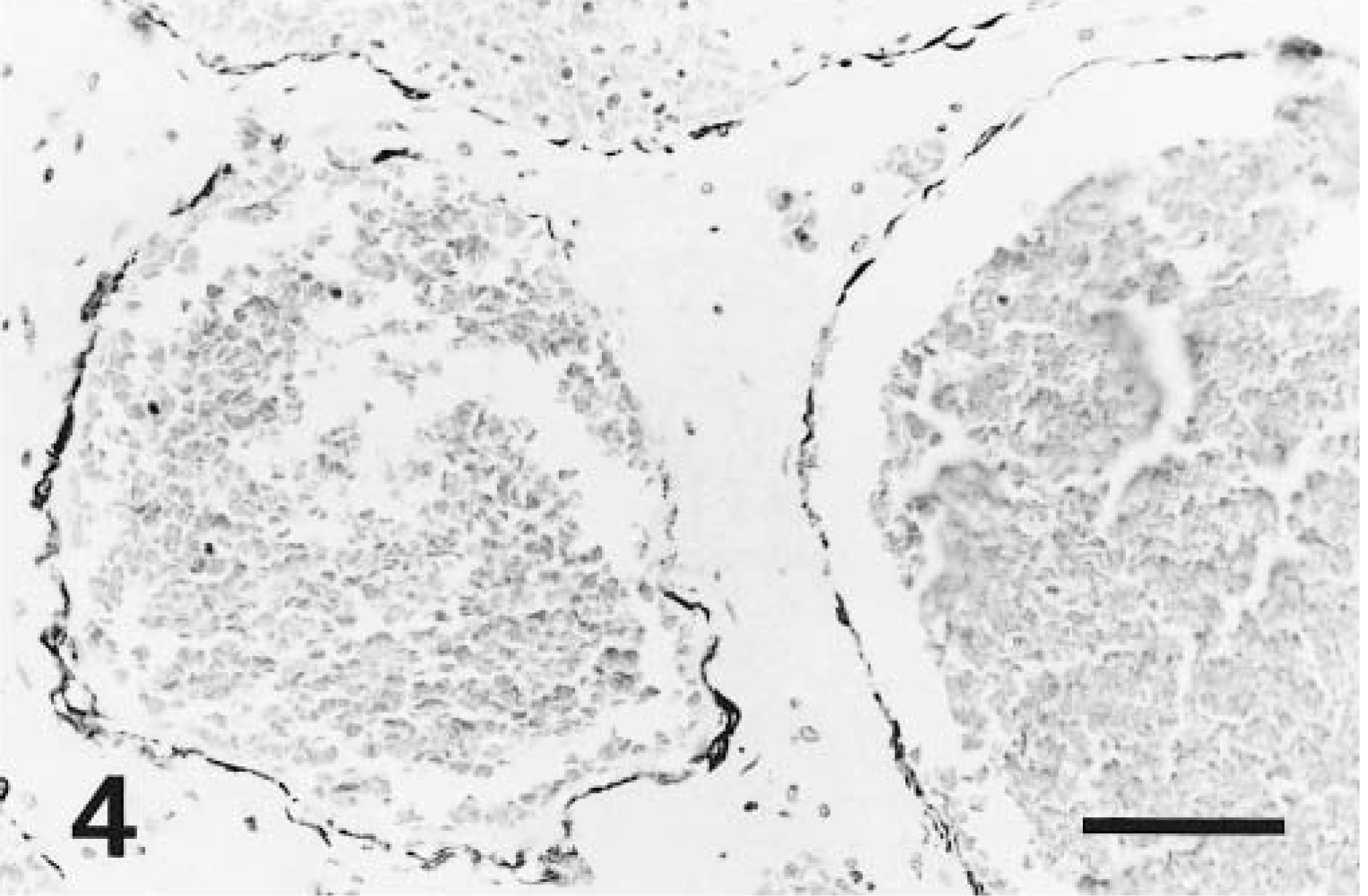
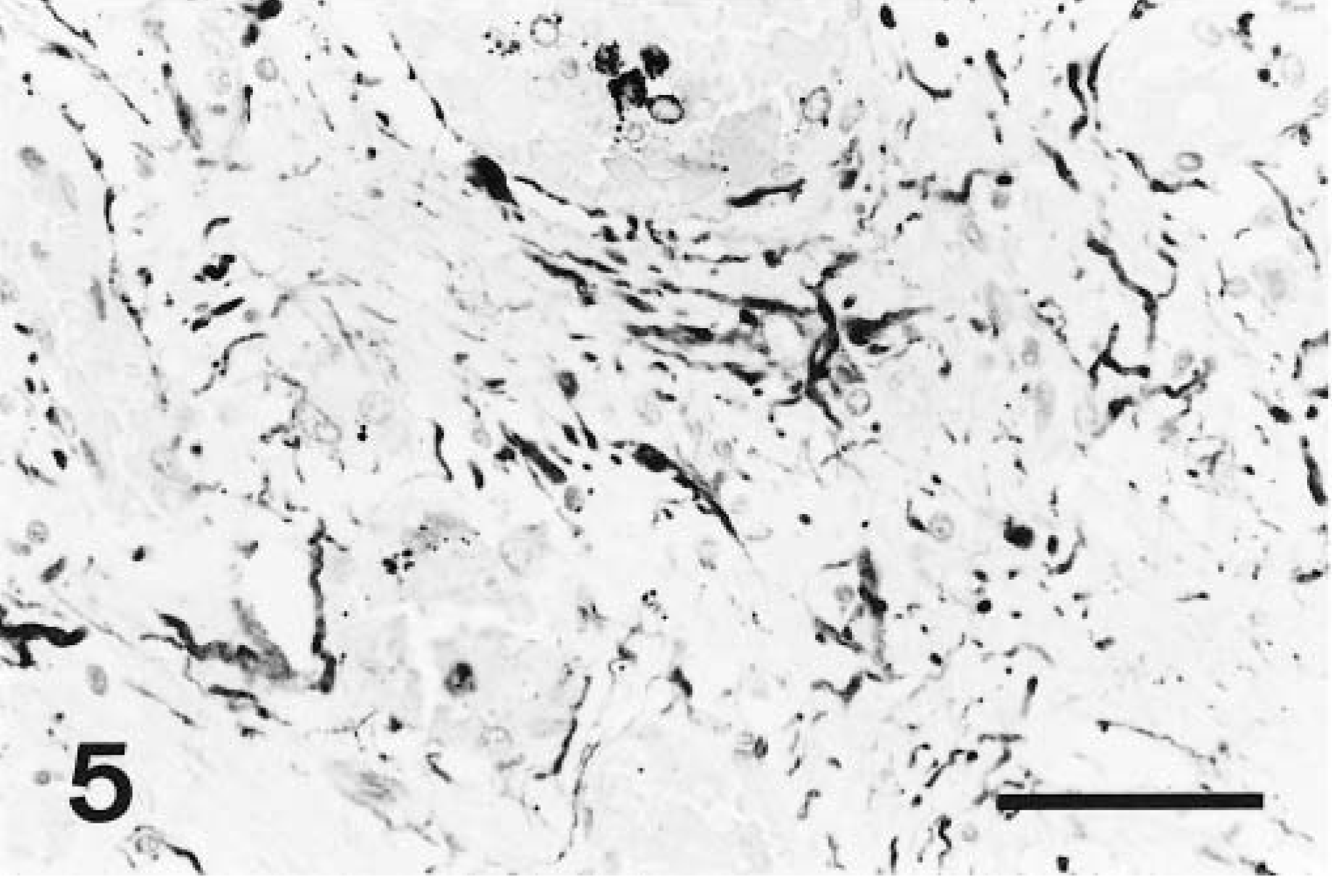
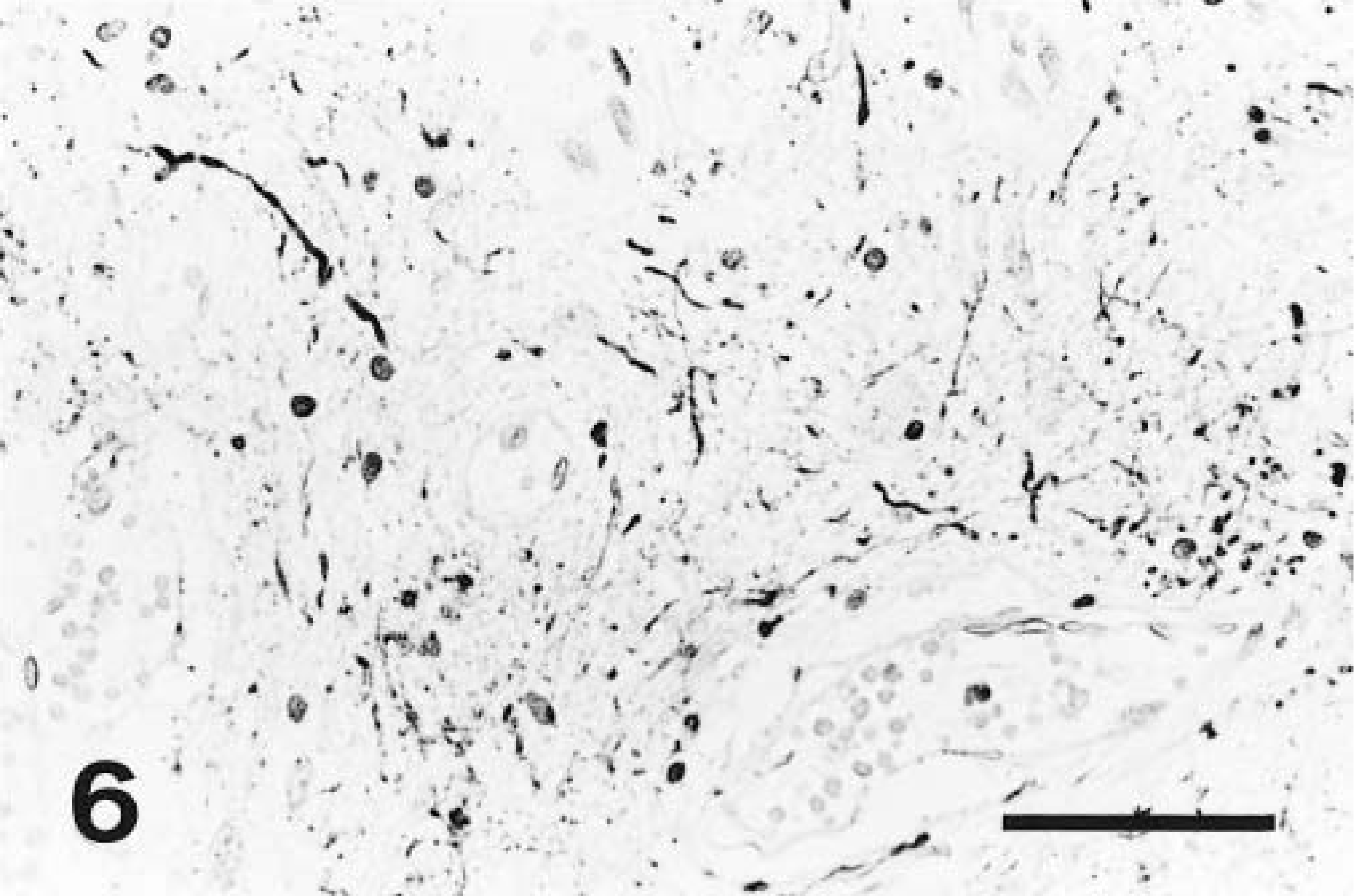
In this case series of vascular hamartomas of the canine brain, no age, breed, or sex predisposition was noted, but hamartomas were consistently located in the telencephalon, with a preponderance for the pyriform lobe—an observation made previously by Frankhauser et al. (1965). The hamartomas were structurally very similar to and most consistent with the capillary telangiectatic form described in human medicine. 16 This conclusion is based on two common features; specifically, the composition of the vessel walls within the hamartomas and the presence of intervening normal brain parenchyma between hamartoma vessels.
Vessels had thin walls lined by factor VIII–positive endothelial cells, with small to moderate amounts of collagen, no muscular component, and no elastic membranes. Factor VIII is a universally accepted marker for endothelial cells in many species. 1 14 The presence of factor VIII–specific staining confirmed the endothelial nature of the vessel-lining cells in all of our hamartomas. Middleton et al. (1999) recently demonstrated similar factor VIII positivity in endothelial cells lining a spinal cord vascular hamartoma in a goat. 12 As in this study, many of these lining cells were also actin-positive. Actin is the constituent element of microfilaments, which control cell movement, contractility, and conformation. 4 Endothelial cells can be actin-positive, depending on their original location, 1 indicating that the vessel-lining cells in this series of hamartomas may have been exclusively endothelial. However, some of these cells may also represent pericytes, modified smooth muscle cells that surround capillaries and venules 5 and are actin-positive. 17 Pericytes are primitive mesenchymal cells that have been identified at the leading edge of normal sprouting capillaries, where they are thought to be responsible for directing the growth of new vessels. 13 If these cells are indeed pericytes, then their presence in this series of hamartomas, together with the lack of muscular and elastic components, may simply point to the failure of vessels to develop beyond their primitive components.
The presence of normal interstitium between the hamartoma vessels is considered the main criterion for differentiating capillary telangiectasis from another subtype of vascular hamartoma, cavernous hemangioma. The latter has very little or no intervening neural tissue. 3 Antiglial fibrillary acid protein and neurofilament antibodies unequivocally confirmed the presence of normal brain parenchyma between the constituent vessels of all five hamartomas, again consistent with a diagnosis of the capillary telangiectasis subtype of vascular hamartoma. The presence of normal interstitial tissue in the hamartomas of this series may reflect the true nonneoplastic nature of such lesions, in that it demonstrates the apparent synchronous growth of normal parenchyma and anomalous vessels, with minimal compression of the surrounding architecture.
Footnotes
Acknowledgements
We thank Ms Jackie Ferracone and Ms Barbara Hall for their technical skills and assistance with the immunohistochemistry in this project and Mr. James Hayden for his assistance with photomicrographs.
