Abstract
A 7-year-old female spayed Golden Retriever dog presented with fever and a 10-day history of neurological signs, including ambulatory paraparesis and pelvic limb ataxia. Neurological examination initially revealed a T3-L3 myelopathy. Thoracic radiographs revealed a diffuse miliary pulmonary pattern. Endotracheal washes and fine-needle aspirates from several organs aimed at identifying a potential infectious agent or neoplastic process were all unsuccessful. Due to worsening of the clinical signs, euthanasia was elected. Necropsy findings included multifocal, pale to dark red, firm nodules infiltrating the lungs, heart, mesentery, pancreas, small intestine, brain, and spinal cord. Cytological examination of impression smears obtained from the pulmonary nodules during necropsy revealed clusters of epithelioid cells admixed with fewer spindle cells, erythrocytes, and scattered leukocytes. Clinical signs and cytological findings initially suggested the possibility of a widespread granulomatous disease or a metastatic epithelial neoplasm as possible clinical differentials in this case. The final diagnosis was based on the gross and histological findings, with confirmation following histochemistry and immunohistochemistry.
History, Clinical, and Pathological Findings
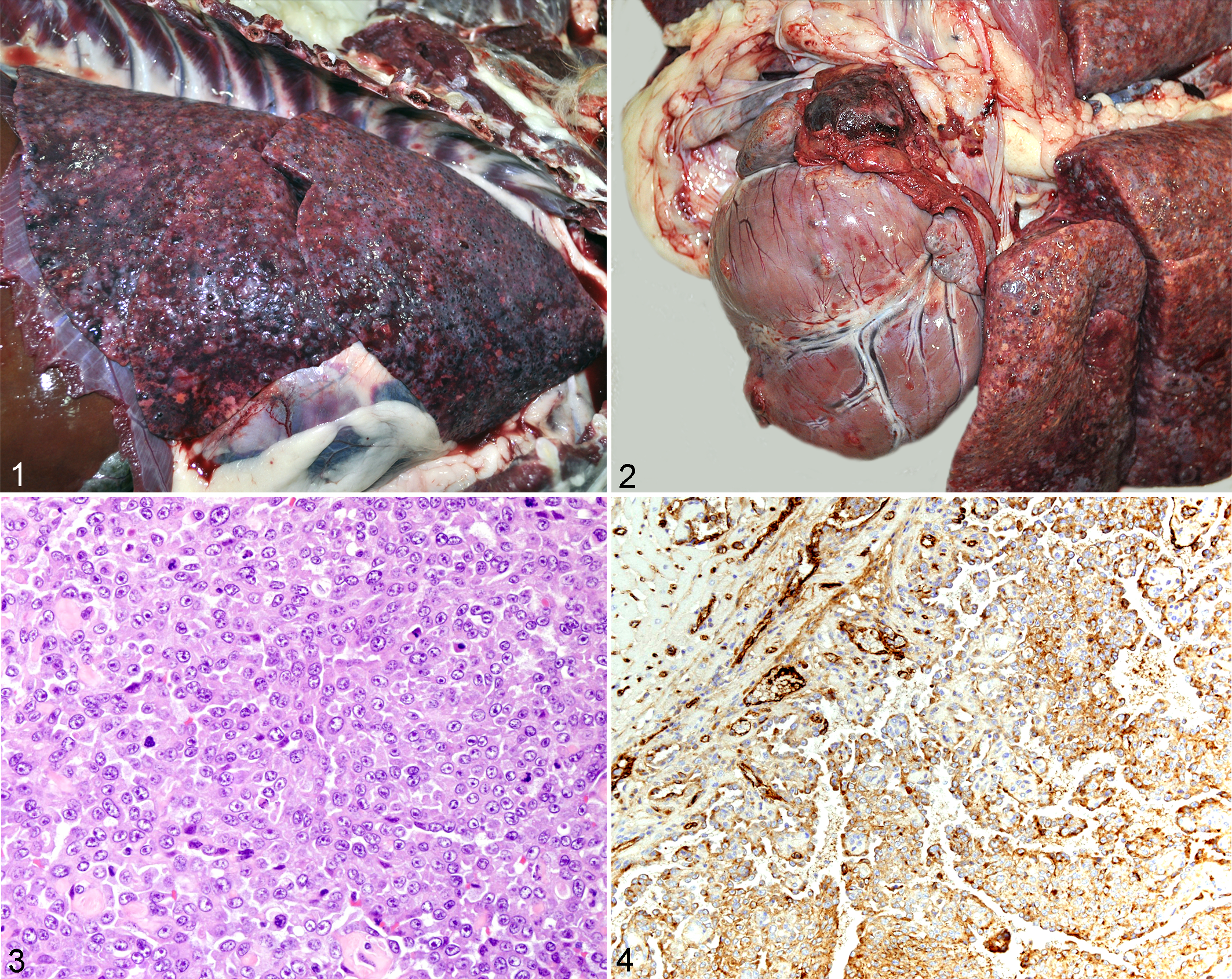
A 7-year-old female spayed Golden Retriever dog was referred to the University of Georgia Veterinary Teaching Hospital with fever and a 10-day history of neurological deficits. Initial neurological examination revealed a T3-L3 myelopathy characterized by progressive paraparesis and symmetrical pelvic limb ataxia, with delayed postural reaction in the pelvic limbs. Thoracic radiographs evidenced a diffuse miliary pulmonary pattern suggestive of fungal pneumonia or metastatic neoplasia. Endotracheal washes and fine-needle aspirates from lungs, liver, spleen, and peripheral lymph nodes were unsuccessful in detecting a potential infectious agent or neoplasia. A complete blood count, chemistry profile, urinalysis, and abdominal ultrasound were also performed and did not reveal any significant changes. Serology and polymerase chain reaction of blood and urine samples were also negative for potential fungal organisms. Euthanasia was elected shortly after presentation due to rapid clinical deterioration, with severe dyspnea, decompensatory hypoxemia, and worsening of neurological signs, which progressed to include a right thoracic limb nerve root signature characterized by a toe-touching lameness. Necropsy findings included multifocal, dark red, firm, 3 to 5 mm in diameter, homogeneous nodules expanding the lungs (Fig. 1), heart, mesentery, pancreas, small intestinal serosa, brain, and cervical and thoracic spinal cord. The pericardial sac contained 25 ml of serosanguineous fluid and was focally adhered to a dark red, firm, 3 cm in diameter, irregular nodule arising from the right auricle (Fig. 2). The myocardium was also infiltrated by similar masses (Fig. 2). Cytological examination of impression smears obtained from the pulmonary nodules during necropsy was performed by one of the authors (A.W.), and results revealed clusters of epithelioid cells admixed with fewer spindle cells, erythrocytes, and scattered leukocytes.
Differential Diagnoses
Based on the presence of fever, neurological signs, respiratory signs, and radiographic findings consistent with a miliary pattern in the lungs, initial clinical differentials in this case included widespread granulomatous disease or a possible metastatic carcinoma affecting the spinal cord. Clinical differentials for disseminated granulomatous disease included systemic mycosis caused by Blastomyces dermatitidis, Coccidioides immitis, Histoplasma capsulatum, Cryptococcus neoformans, or Aspergillus spp; algal infection by Prototheca zopfii, Prototheca wickerhamii, or Chlorella spp; and systemic bacterial infection by Nocardia asteroides or Mycobacterium spp. Differentials for metastatic carcinoma included mammary carcinoma, pulmonary adenocarcinoma, thyroid adenocarcinoma, renal adenocarcinoma, pancreatic adenocarcinoma, and salivary gland adenocarcinoma. 1
Histological, Histochemical, and Immunohistochemical Findings
Histological findings in sections of lung, heart, mesentery, pancreas, small intestine, brain, and spinal cord revealed an infiltrative neoplasm composed of 2 distinct populations of neoplastic cells. Within the pulmonary parenchyma and myocardium, groups of closely packed epithelioid cells were arranged in sheets or less often irregular vascular-like structures that multifocally infiltrated and effaced adjacent tissues (Fig. 3). Neoplastic epithelioid cells were moderately pleomorphic with polygonal, abundant, eosinophilic cytoplasm showing distinct cell borders. Scattered cells had intracytoplasmic vacuoles that were often filled with a single erythrocyte. Nuclei were round, with stippled chromatin, 1 or 2 nucleoli, and had moderate anisokaryosis. Mitotic activity ranged from 1 to 2 per high-power field (HPF) at 400×. In contrast, neoplastic cells within the spinal cord, brain, mesentery, pancreas, and small intestine were elongate and arranged in well-demarcated groups forming variable-sized vascular channels filled with erythrocytes, neutrophils, macrophages, and fibrin. These neoplastic cells were supported by small amounts of a dense collagenous stroma. Neoplastic cells lining the vascular channels were markedly pleomorphic and showed moderate amounts of stellate to elongated, eosinophilic cytoplasm with indistinct cell borders. Nuclei were markedly pleomorphic, elongated, and irregular and composed of roughly aggregated chromatin with 1 to 2 nucleoli. Mitoses ranged from 5 to 7 per HPF at 400×. Areas of necrosis and hemorrhage were scattered throughout these neoplastic foci. In sections of lung and heart, Gomori’s reticulin (GR) special stain highlighted a prominent GR-positive basement membrane outlining groups of epithelioid neoplastic cells. In these sections, neoplastic cells showed strong positive cytoplasmic immunostaining for factor VIII–related antigen (Fig. 4).
Diagnosis
The gross, histological, histochemical, and immunohistochemical findings in this case were consistent with a diagnosis of disseminated hemangiosarcoma of both epithelioid and vascular morphologic variants.
Discussion
Hemangiosarcomas (HSAs) are neoplasms of vascular endothelial origin that are commonly diagnosed in dogs and less often in other domestic species. 2,5,9 Canine HSAs tend to affect adult to older large-breed dogs, particularly German Shepherds. 5 They are considered highly metastatic tumors based on the presence of disseminated neoplasia in more than 80% of the patients at the time of diagnosis. 5,9 A strong predilection site for canine HSA includes the skin, particularly sparsely coated areas such as the ventral abdomen, for which there appears to be an association with chronic ultraviolet light exposure. 5 Visceral HSAs do not have a predilection site and usually affect the spleen, right atrium, and liver. 9 Similar to what we observed in this case, the coexistence of neoplasms within multiple organs at clinical presentation or postmortem examination makes the differentiation between a multicentric or metastatic process difficult in most cases. 5
Hemangiosarcoma can vary in its histological appearance, with epithelioid HSA being an uncommon morphologic variant described in dogs, horses, cattle, and humans. 2 –4,6 –8,10 Most epithelioid HSAs in veterinary species are reported to affect the integument. 2,10 Including the present case, only 4 cases of canine epithelioid HSA have been diagnosed in the Department of Pathology at the University of Georgia, between 2005 and 2013, with 3 of 4 cases reported to be confined to the skin. Accordingly, there are only a few case reports of epithelioid HSA documenting focal or disseminated extra-cutaneous or visceral involvement, such as lung, tendon, and skeletal muscle, 2 –4,6 –8,10 with only 2 cases concurrently involving multiple organs. 3,8 The present case is unique in its visceral and disseminated presentation, with involvement of the spinal cord and concomitant presence of epithelioid and vascular morphologic variants. While the well-documented vascular variant of HSA usually presents little to no diagnostic difficulty, epithelioid HSA has been previously confused with epithelial and/or histiocytic neoplasms in veterinary and human medicine, in both clinical and pathology settings. 6,8,10 Although the gross findings observed in this dog were consistent with HSA, cytological evidence of epithelioid neoplastic cells noted on impression smears taken from the pulmonary nodules raised the suspicion of a possible disseminated granulomatous disease or epithelial neoplasia. A similar scenario has also been reported previously in a dog with epithelioid HSA mimicking metastatic epithelial prostatic neoplasia. 8 Given the potential challenges associated with the diagnosis of epithelioid HSA, immunohistochemistry is a valuable tool in obtaining a definitive diagnosis. Positive cytoplasmic immunostaining of neoplastic cells for factor VIII–related antigen (von Willebrand factor) and/or CD31 has been demonstrated to be extremely valuable in the final diagnosis of epithelioid HSA, especially in cases where the classic vascular morphology is not present. 2,7,8,10 Additional immunohistochemical stains such as cytokeratin can also be performed to rule out a suspected epithelial neoplasm but are often not necessary. 2,10 Although CD31 or cytokeratin immunohistochemistry were not performed in this case, GR-positive fibers outlining groups of neoplastic cells and strong intratumoral immunostaining for factor VIII–related antigen, in combination with the epithelioid morphology of the neoplastic cells, sufficed to confirm the diagnosis of epithelioid HSA in this dog. 2,10
The present case is unique in the concurrent presence of epithelioid and vascular-type HSA, which to the best of our knowledge has not been previously reported. In this case, the final diagnosis of an epithelioid HSA was based on the gross and histological findings, as well as on the characteristic positive GR staining and positive immunostaining of epithelioid neoplastic cells for factor VIII–related antigen.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
