Abstract
A 12-y-old, male Dachshund was presented for elective orchiectomy. The testes were of normal size. The left testis had numerous dark-red, blood clot–like foci within the vaginal tunic over the pampiniform plexus, epididymis, and testis. Histologically, the red foci were limited to the vaginal tunic and consisted of disorderly growing, variably sized, thin-walled blood vessels lined by a single layer of endothelial cells without mitoses and supported by a thin layer of pericytes. The blood vessels were distended by erythrocytes without thrombus formation. Endothelial cells had cytoplasmic immunolabeling for CD31; pericytes had strong cytoplasmic immunolabeling for α–smooth muscle actin. Our case of subclinical unilateral vascular hamartomas of the vaginal tunic in a dog has not been reported previously in domestic animals or humans, to our knowledge.
A 12-y-old, 12.5 kg, intact male Dachshund canine was presented to the veterinary clinic for elective orchiectomy. Preoperative CBC and serum biochemistry profile results were normal, except for mild lymphopenia. The patient was induced with Telazol–Torbugesic–Dexdomitor (TTDex, 0.02 mL/kg; Pfizer) IV and intubated. Anesthesia was maintained with isoflurane. Routine castration was performed using a pre-scrotal incision without scrotal ablation. When the left testis was removed, soft, dark-red, foci similar in appearance to blood clots were noted in the vaginal tunic over the pampiniform plexus and extending over the testis. The lesions did not extend along the spermatic cord beyond the external inguinal canal, and laparotomy was not deemed necessary. All of the visible, abnormal tissue over the vaginal tunic and the left testicle were submitted for histologic evaluation. The patient was clinically normal 6 mo post-surgery.
The testis within the vaginal tunic was 5.5 × 2.5 × 1.5 cm, with a segment of spermatic cord attached. The vaginal tunic had many, 0.2–0.3 cm, round, or ovoid up to 0.3 × 0.5 cm, slightly raised, plaque-like to nodular, dark-red foci over the epididymis, testis, and proximal spermatic cord (Fig. 1A). The testicular parenchyma was grossly normal. Representative sections of tissue were fixed in 10% neutral-buffered formalin and processed routinely to produce 4-µm, H&E-stained slides. Immunohistochemical labeling was performed for alpha–smooth muscle actin (α-SMA; monoclonal mouse, prediluted; BioCare Medical) and CD31 (monoclonal mouse, 1:200 dilution; Dako), as smooth muscle and vascular biomarkers, respectively.
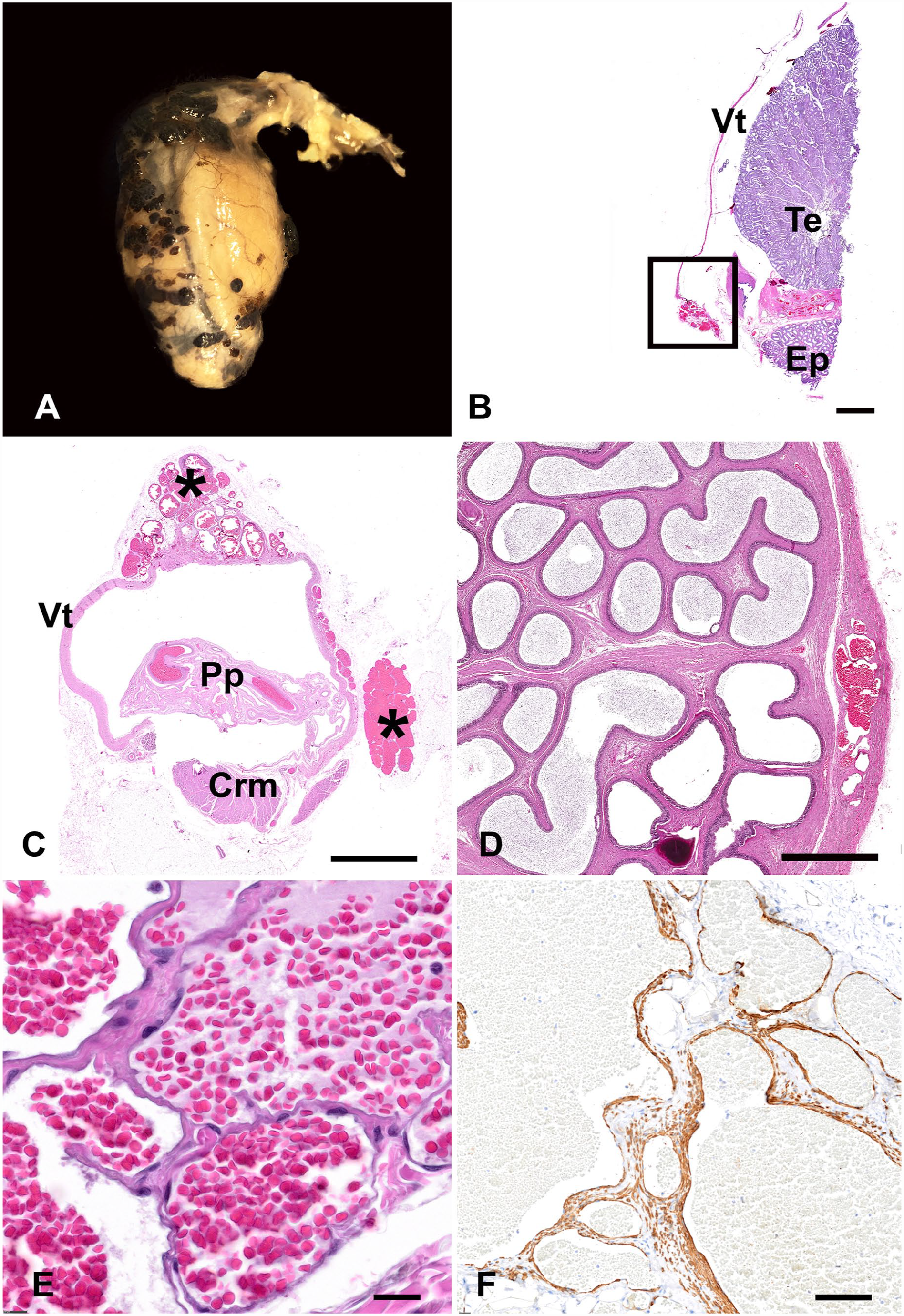
Unilateral vascular hamartomas of the vaginal tunic in a 12-y-old, male, Dachshund dog.
Histologically, within the fibrous stroma of the visceral vaginal tunic, were randomly spaced, variable-sized clusters of thin-walled blood vessels lined by a single layer of endothelial cells with an ovoid nucleus, coarse chromatin, and an indistinct nucleolus (Fig. 1B–D). The endothelium was supported on a thin layer of pericytes (Fig. 1E). The blood vessels were filled by erythrocytes without thrombus formation. Endothelial cells had cytoplasmic immunolabeling against CD31; pericytes had strong cytoplasmic immunolabeling against α-SMA (Fig. 1F). Vascular proliferation was limited to the vaginal tunic, and we diagnosed the lesion as a unilateral, multilocular vascular hamartoma. Testicular parenchyma, epididymis, testicular veins, and arteries had no other histologic lesions.
Vascular malformations, or hamartomas, specifically located in the vaginal tunic have not been reported in humans or animals, to our knowledge. We retrieved no cases of vaginal tunic vascular hamartomas in a search of Google, PubMed, CAB Direct, Web of Science, and Scopus using the search terms: vascular hamartoma, vaginal tunic, tunica vaginalis, dog, canine, domestic animals, humans. Additionally, we retrieved all other cases with a lesion of the vaginal tunic submitted to the Auburn Pathobiology Diagnostic Pathology Service (Auburn, AL, USA) from 2002–2022 (Table 1).
Vaginal tunic samples submitted to the Auburn Pathobiology Diagnostic Pathology Service, 2002–2022.
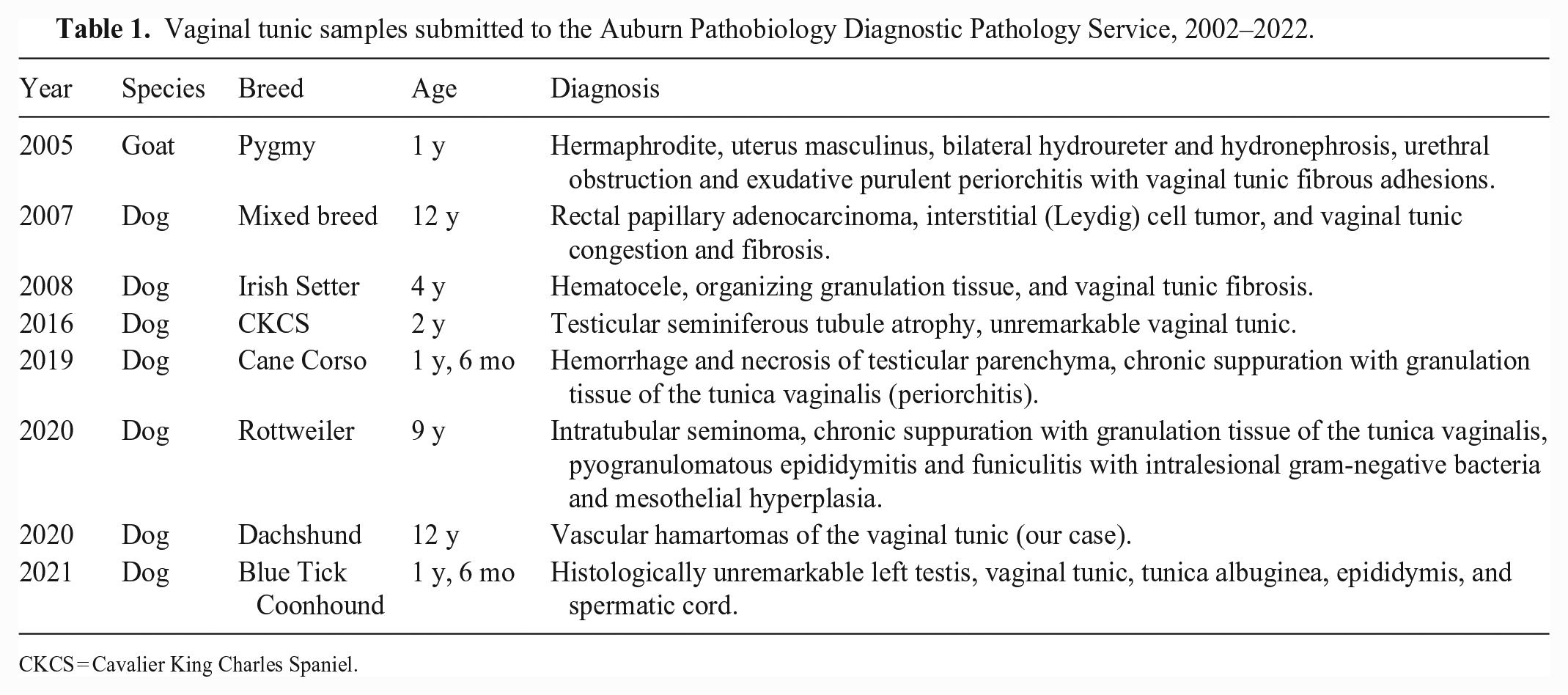
CKCS = Cavalier King Charles Spaniel.
Hamartoma is the term used to describe a focal, disorganized, overgrowth of well-differentiated tissue in an organ, in which the tissue constitutes a normal component.5,21,26,28 Hamartomas may be present at birth, and their growth is driven by the organ of origin.21,28 Epithelial, mesenchymal, or a combination of both cell types can form a hamartoma. 31 Vascular hamartoma is a non-neoplastic proliferation of disorganized, but well-differentiated vasculature, forming a focal developmental abnormality. 7 The focal growth of vascular hamartomas within the tissue of origin supports a diagnosis of a malformation over a true neoplasm. 23 The term vascular hamartoma is widely used in veterinary medicine and is also referred to as vascular malformation in human pathology.6,24 In humans, the identification of a genetic basis of vascular development has aided comprehension of the signaling pathways involved in the pathogenesis of vascular malformation. 18 Specifically, vascular malformation has been attributed to a gain-of-function somatic mutation in the GNA11 gene. 18 Identification of affected genes represents a potential pharmaceutical target for the treatment of vascular anomalies in humans. 18
Given that blood vessels are in all organs, vascular hamartomas can arise in any anatomical location.21,26 Vascular hamartomas described in domestic animals are classified into 3 categories: 1) dysplastic vessels: tortuous, thick-walled, vascular structures with smooth muscle, 2) redundant capillaries: capillary buds to large arteries with well-organized vessel structures, or 3) numerous, thin-walled, endothelial-lined vessels separated by edematous fibrostroma. 26 Vascular hamartomas reported in the veterinary literature have had secondary effects caused by local obstruction or compression of the adjacent anatomic structures, making them clinically relevant, benign lesions. 7 Vascular hamartomas have been reported in dogs, cats, horses, cows, and rats, in several anatomic locations including the skin, CNS, heart, and genitourinary tract. 24
Vascular hamartoma from the scrotum in dogs is a well-characterized benign lesion, comprised of single or multifocal proliferations of lobules of capillaries surrounding larger central vessels, with absent atypia and rare mitoses.10,22 In some areas, spindle cells proliferate between the capillary structures.10,22 The lesion has been described also in pigs; complete excision is curative.10,22 Differential diagnoses include cutaneous well-differentiated hemangiosarcomas and hemangiomas. Well-differentiated hemangiosarcomas are easily distinguished from scrotal vascular hamartomas, given their invasive pattern, high mitotic index, and cellular atypia. 22 Hemangiomas, unlike scrotal vascular hamartomas, lack central larger vessels, are exophytic, single, and do not have spindle-cell proliferation between the capillaries. 22 Angiomatoses are vascular proliferations with well-described clinical manifestations in cattle, dogs, and cats. 10 Syndromes include bovine cutaneous angiomatosis and progressive angiomatosis of dogs and cats. 10 Characteristic histologic features including intraluminal endothelial hyperplasia and interconnecting vessels, and anastomosing channels are not observed in canine scrotal vascular hamartomas. 22 We did not observe these typical angiomatosis histologic features in our case.
The vaginal tunic is a dense, collagen-fiber membrane lined by a single layer of mesothelial cells, and it is histologically and anatomically continuous with the peritoneum. It is comprised of visceral and parietal layers. 9 Specific conditions affecting the vaginal tunic alone in animals are uncommon. Reported conditions involving the vaginal tunic in animals include mesothelioma in dogs, rats, stallions, bulls, and calves, and leiomyoma in a dog.4,13,14,16,17,19,27,30 Non-neoplastic conditions affecting the vaginal tunics include hydrocele and hematocele. 9 Inflammatory conditions involving the testicular tunics are commonly reported and can be secondary to a disseminated infection caused by agents involved in polyserositis or extension of peritonitis in domestic animals. 9 Inflammatory processes, such as epididymitis and periorchitis, can lead to pyocele, and increased intratesticular pressure can cause testicular degeneration and atrophy. 9 However, we did not observe testicular degeneration in our case. No adhesions were between the vaginal tunic and the testis. No cysts formed in the vaginal tunic. Histologic alterations associated with the presence of vascular hamartoma were absent within the epididymis, testis, and pampiniform plexus.
Vascular abnormalities include vascular malformations and neoplasia arising from vascular structures. 15 Vascular hamartomas can occur in any tissue and are present at birth.3,21 The spectrum of vascular abnormalities reported in the testicular structures in animals includes neoplastic and non-neoplastic conditions. Many of these conditions are idiopathic with a poorly understood pathogenesis. Vascular lesions within the testicular structures can be further classified into developmental malformations or hamartomas, and benign neoplasms. Vascular hamartomas within the ovaries are reported in mares, cows, and sows.2,12,20,25 A testicular vascular hamartoma has been described in a calf. 29
Vascular abnormalities reported in humans in the testes and scrotum include traumatic lesions, congenital malformations, and neoplasia. Identifying non-neoplastic lesions mimicking neoplasms is important because these lesions can distort the normal structure of the testes and are potential differential diagnoses. 1 The vascular lesions reported in this group include intratesticular hemorrhage, segmental testicular infarction, and organized testicular hematocele. 1 The testicular tunics (tunica albuginea, vaginal tunic) can have non-neoplastic lesions within the mesothelium, such as mesothelial cysts and mesothelial hyperplasia. 1 Vascular neoplasms involving the testis and testicular tunics reported in humans are cavernous hemangioma of the tunica albuginea and testicular hemangioma.8,11
Footnotes
Acknowledgements
We thank the technical staff of the Auburn University College of Veterinary Medicine Histology Laboratory.
Declaration of conflicting interests
The authors declared no potential conflicts of interest concerning the research, authorship, and/or publication of this article.
Funding
We did not receive any specific grant from the public, commercial, or not-for-profit funding agencies.
