Abstract
Neuromuscular and vascular hamartoma (NMVH) is an infrequent gastrointestinal lesion described in human and veterinary medical literature. The histologic features of this entity are haphazardly arranged fascicles of smooth muscle, nerve fibers, scattered ganglion cells, and hemangiomatous blood vessels. Here we describe 2 putative cases of NMVH in a 1.7-y-old, intact female Anatolian mixed-breed dog and a 4-mo-old intact male Akita dog. Both animals had gastrointestinal clinical signs, including hematochezia, and on exploratory laparotomy, intussusception was confirmed. Histologic examination confirmed NMVH within the cecal wall in both cases using a panel of immunohistochemical (IHC) markers for vascular structures (CD31), smooth muscle (alpha–smooth muscle actin [α-SMA]), and nerves (glial fibrillary acidic protein [GFAP] and S100). The complete surgical excision of the lesion in both animals was considered curative without persistent clinical signs 14 mo and 12 mo, respectively, after surgery.
Neuromuscular and vascular hamartoma (NMVH) of the gastrointestinal tract has been described in human literature since 1982. 8 It consists of a hamartomatous proliferation of the mesenchymal components of the intestine that leads to stenosis. 8 Histologically, NMVH consists of haphazardly arranged fascicles of smooth muscle, nerve fibers, scattered ganglion cells, and hemangiomatous blood vessels; however, other mesenchymal components, including fibrous connective tissue and adipose tissue have been described in this condition, changing the name to neuromesenchymal hamartoma. 16 Histologic changes have been reported in the mucosa, submucosa, muscle, and serosal layers of the intestine. 1 A 2020 retrospective study of 4 human cases made a detailed description of the vasculopathic changes found in NMVH, including vessel-in-vessel appearance, obliterative venopathy, concentric myohypertrophy, and vasculitis. 1
NMVH has been poorly documented in the veterinary literature.20,22,23 We retrieved only 2 cases of NMVH diagnosed in animals after a comprehensive search of Google, PubMed, CAB Direct, Web of Science, and Scopus, using the search terms “neuromuscular and vascular hamartoma”, “dog”, “animal”, “intestinal”, and “neuromesenchymal hamartoma”. Here we describe the histologic features of a putative intestinal neuromuscular and vascular hamartoma in 2 dogs using a panel of histochemical (Masson trichrome, Verhoeff–van Gieson [VVG]) and immunohistochemical (CD31, alpha–smooth muscle actin [α-SMA], glial fibrillary acidic protein [GFAP], S100) stains (Table 1).
Technical details of the antibodies (source, clone, manufacturer, concentration used, epitope retrieval, and detection method) used for immunohistochemical stains on sections of putative neuromuscular and vascular hamartoma in 2 dogs.

Heat-induced epitope retrieval in a DeCloaker (pressure cooker) for 16 min at 110°C Diva DeCloaker 10X, DV2004MX, pH 6.25.
Case 1. A 1.7-y-old, 24.4-kg, intact female Anatolian mixed-breed dog was presented to the veterinary hospital due to a 2-wk history of weight loss, hyporexia, lethargy, hematochezia, and mucus in its stool. On physical examination, the patient was panting and had pale and tacky mucous membranes, 120 bpm, 4 of 9 body condition score, 38.9°C body temperature, and lacked enlargement of peripheral lymph nodes. CBC and serum biochemistry findings yielded leukocytosis (19.2 × 109/L; RI: 5.0–16.8 × 109/L) caused by neutrophilia (15.5 × 109/L; RI: 3.0–11.6 × 109/L). After 3 d, the patient was nonresponsive to fluid therapy, antibiotics (ampicillin 20 mg/kg SC q8h; metronidazole 25 mg/kg PO q12h), antiemetic (maropitant [Cerenia] 2 mg/kg PO q24h), and gabapentin (10 mg/kg PO q12h). No ova or parasites were retrieved during zinc sulfate centrifugation, intestinal nematode antigen was not detected, and the real-time PCR panel was negative for the following infectious agents: Cryptosporidium spp., Giardia spp., Salmonella spp., canine circovirus, canine enteric coronavirus, canine parvovirus 2, canine distemper virus, Campylobacter jejuni, C. coli, Clostridium difficile toxin A/B genes, C. perfringens alpha toxin (CPA) gene, C. perfringens enterotoxin (CPE) gene, and C. perfringens CPnet/E/F toxin genes. An abdominal ultrasound identified an intussusception at the proximity of the ileocecal-colonic junction. An exploratory laparotomy and enteroanastomosis were performed, and the intestinal segment containing a 5-cm diameter intraluminal cecal mass was submitted for histologic examination. Post-surgical recovery was uneventful. The patient was in good health at a follow-up 14 mo post-surgery.
The intestinal segment was fixed with 10% neutral-buffered formalin and processed routinely to produce 4-µm, H&E-stained slides. The cecal mass submitted was firm, and gray after fixation, with approximately the same dimensions described during surgery. On histologic examination, expanding the wall of the cecum from the submucosa to the serosal layer was a non-encapsulated and poorly demarcated mass composed of a mixture of haphazardly arranged smooth muscle fascicles (highlighted with α-SMA), nerve bundles (highlighted with GFAP and S100), fibrous connective tissue (highlighted with Masson trichrome stain), adipose cells, and numerous vascular structures (Fig. 1). The proliferative vascular structures included arteries, veins, and capillaries distributed from the mucosa to the serosal layer lined by a single layer of CD31-immunolabeled endothelial cells and surrounded by α-SMA–immunolabeled spindle cells (smooth muscle cells and pericytes; Fig. 1C, inset). Multifocally, the blood vessels have irregularly thickened walls with hypertrophy of the tunica media and tortuous-to-irregular lumens. The mucosa was multifocally ulcerated and covered with fibrin and karyorrhectic cellular debris (Fig. 1A). The non-ulcerated mucosa had multifocal dilated intestinal glands filled by glandular secretion, necrotic sloughed epithelial cells, and karyorrhectic debris.
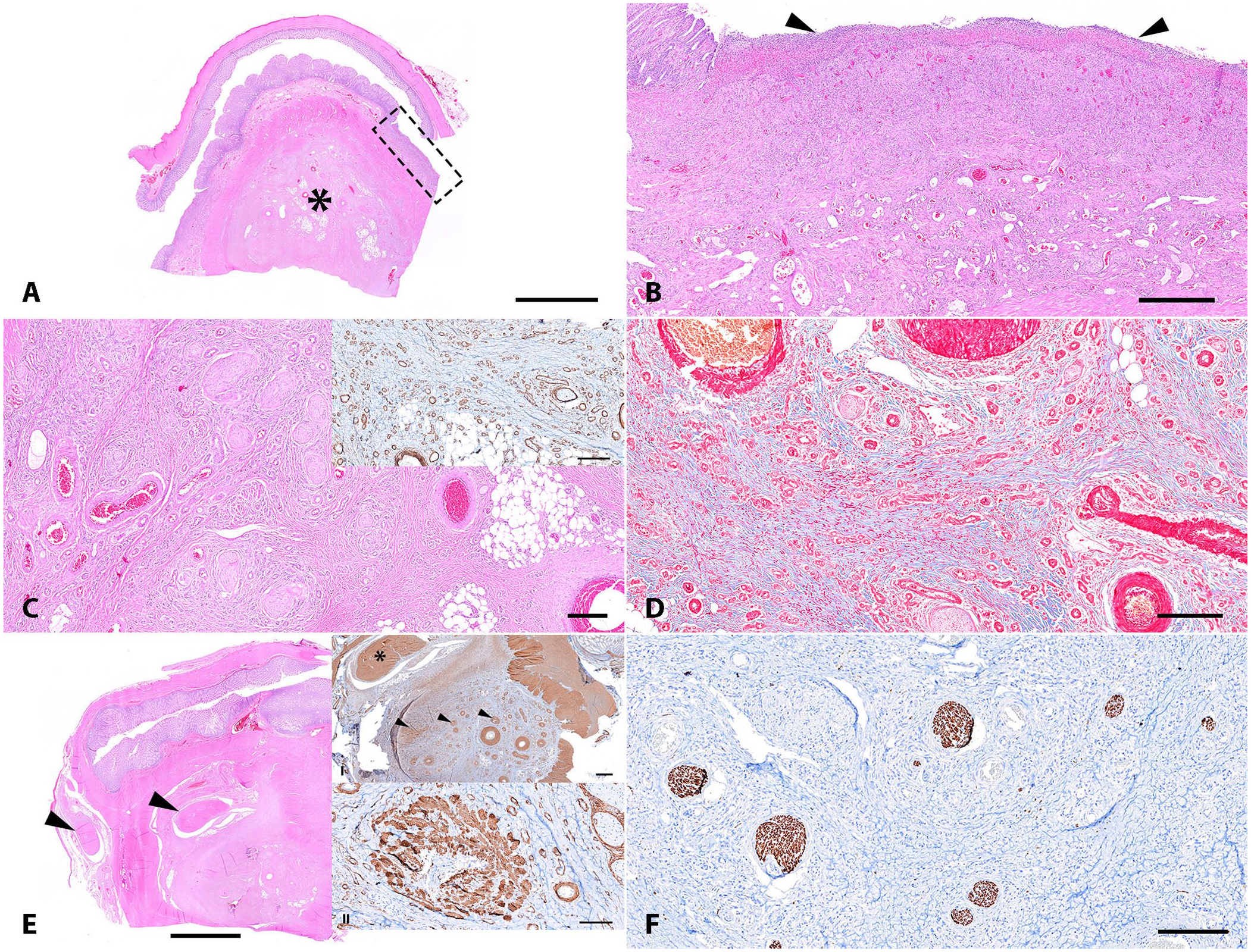
Putative neuromuscular and vascular hamartoma (NMVH) in the cecum of a 1-y and 8-mo-old, 24.4-kg, intact female Anatolian mixed-breed dog (case 1).
Case 2. A 4-mo-old, 12-kg, intact male Akita dog was presented to the veterinary hospital due to a 1-wk history of vomiting, bloody diarrhea, and hyporexia. On physical examination, mucous membranes were light pink and tacky, 134 bpm, 34 rpm (eupneic), 3 of 9 body condition score, and 38.1°C body temperature. The abdomen was compliant and painful on palpation, with a mid-abdominal mass and hematochezia. An abdominal ultrasound was performed and identified a jejunal intussusception and mesenteric lymphadenopathy; thus, surgical intervention was recommended. Preoperative CBC results were consistent with microcytic (MCV 52.7 fL; RI: 61.6–73.5 fL) and hypochromic (MCH 19.5 pg; RI: 21.2–25.9 pg) non-regenerative anemia (Hct 0.31 L/L, RI: 0.37–0.61 L/L; Hb 115 g/L, RI: 131–205 g/L), leukocytosis (29.0 × 109/L; RI: 5.0–16.8 × 109/L), neutrophilia (19.9 × 109/L; RI: 3.0–11.6 × 109/L), lymphocytosis (5.4 × 109/L; RI: 1.0–5.1 × 109/L), monocytosis (3.4 × 109/L; RI: 0.2–1.1 × 109/L), and thrombocytopenia (125 × 109/L; RI: 148–484 × 109/L). Serum biochemistry profile yielded low creatinine (35 µmol/L; RI: 44–159 µmol/L), hyponatremia (140 mmol/L; RI: 144–160 mmol/L), hypochloridemia (108 mmol/L; RI: 109–122 mmol/L), hypoproteinemia (50 g/L; RI: 52–82 g/L), and hypoalbuminemia (20 g/L; RI: 23–40 g/L). Exploratory laparotomy revealed a non-reducible intussusception involving the distal jejunum, ileum, cecum, ileocecal junction, ascending colon, transverse colon, and 50% of the descending colon, entrapping the mesenteric root. The intestinal segment was removed, and enteroanastomosis was performed. A sample of the gastric content was submitted for aerobic and anaerobic bacterial culture and antimicrobial susceptibility testing. Klebsiella spp. resistant to amoxicillin, amoxicillin–clavulanic acid, cephalexin, and doxycycline grew on aerobic culture. Anaerobic culture did not yield bacterial growth. The patient was discharged with a 7-d prescription of antiacid (1.5 mg/kg PO q12h), antimicrobials (amoxicillin–clavulanic acid 25 mg/kg PO q12h; metronidazole 25 mg/kg PO q12h), and gabapentin (10 mg/kg PO q12h). Post-surgical recovery was uneventful. The patient was euthanized 12 mo after the surgery for causes unrelated to the gastrointestinal disorder.
The intestinal segment was fixed in 10% neutral-buffered formalin and processed routinely to produce 4-µm, H&E-stained slides. On histologic examination, the small intestine formed the outer layer of the intussusception (intussuscipiens) that enveloped the large intestine (colon, cecum intussusceptum; Fig. 2A). Extending from the submucosa to the serosa, within the small and inner large intestines and the bridging mesenteric adipose tissue were disordered and vaguely nodular, markedly increased numbers of well-differentiated and blood-filled vascular channels (Fig. 2B). These vascular channels were lined by a single layer of CD31-immunoreactive endothelial cells and, in the more nodular proliferations, were surrounded and separated by α-SMA–immunolabeled spindle cells (smooth muscle), fibrous connective tissue (highlighted with Masson trichrome stain), and scattered nerve fibers (highlighted with S100 and GFAP; Fig. 2B–F). Multifocally, the blood vessels have irregular, tortuous vascular lumens, with fibrointimal hyperplasia and medial hypertrophy. The jejunal mucosa was eroded with partially lost mucosal epithelium and crypts spaced by moderately neutrophilic infiltrates, fibrin, and hemorrhage. The neutrophils extended into the small intestinal submucosa in a gradually diminishing severity. Ectatic lymphatic vessels contained numerous neutrophils. No fungal organisms were evident with GMS staining.
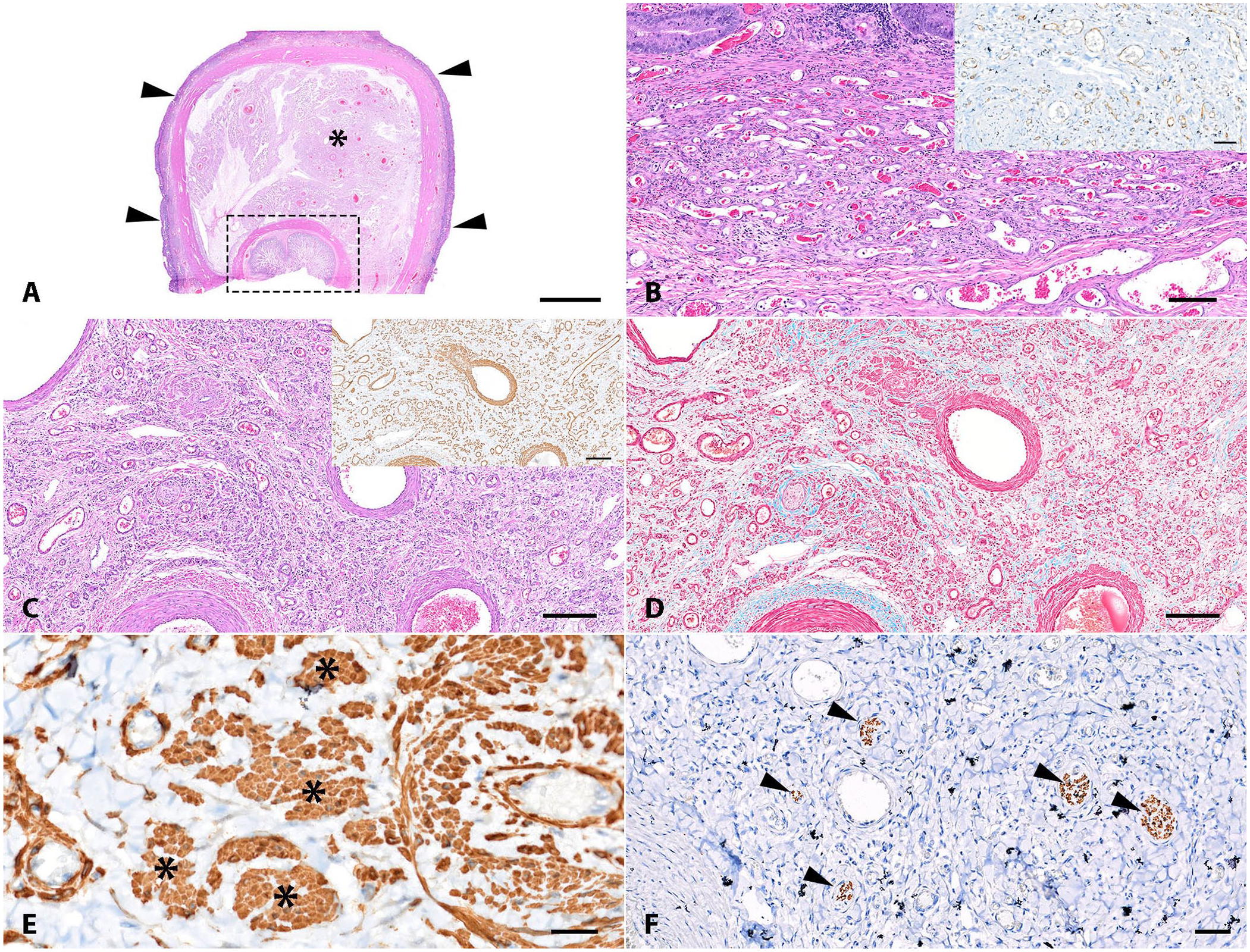
Putative neuromuscular and vascular hamartoma (NMVH) within a jejunal-colonic intussusception in a 4-mo-old, 12-kg, intact male Akita dog (case 2).
NMVH is a lesion poorly described in veterinary medical literature. The discussion regarding clinical and pathologic findings is limited. The low number of reported cases precludes the identification of relevant patterns and predisposing factors. Retrieved cases in animals are limited to an NMVH in a cecal-colonic intussusception in an 8-y-old male West Highland White Terrier with intermittent hematochezia, and a spontaneous NMVH in the border area of the jejunum and ileum in a 110-wk-old F344 rat without clinical signs (Table 2).20,22,23 Our cases had gastrointestinal clinical signs, including hyporexia, weight loss, vomiting, abdominal pain, and, as in the previously reported case, hematochezia. 23 The only clinicopathologic finding in case 1 was neutrophilia. In contrast, case 2 had bloodwork abnormalities related to chronic blood loss (non-regenerative anemia, hypoproteinemia, leukocytosis, thrombocytopenia) and diarrhea (hyponatremia, hypochloridemia). Only a moderate increase in liver function enzyme activities (alkaline phosphatase, aspartate aminotransferase, alanine aminotransferase) was reported in a dog with NMVH. 20 The anatomical location was the cecum in both cases, with a concurrent intussusception as in the previously reported canine case.20,23 Intussusception is the invagination of an intestinal segment into another segment, usually aboral, typically caused by the presence of foreign bodies, intestinal parasites, surgical interventions of the intestine, inflammation of the intestinal mucosa, and intramural inflammatory (abscess) or neoplastic lesions. 21 The presence of the NMVH within the intestinal layers could lead to an intussusception; however, the number of cases is too low to confirm this hypothesis.
Clinicopathologic and histologic features of neuromuscular and vascular intestinal hamartomas reported in animals.

ALP = alkaline phosphatase; ALT = alanine aminotransferase; α-SMA = alpha–smooth muscle actin; AST = aspartate aminotransferase; CK AE1/AE3 = cytokeratin AE1/AE3 (pancytokeratin); Cl = chloride; GFAP = glial fibrillary acidic protein; IHC = immunohistochemistry; Na = sodium; NF = neurofilament; vWF = von Willebrand factor; WHWT = West Highland White Terrier.
NMVH is an extremely infrequent lesion in human and veterinary medicine.17,19,23 The definition exists within a spectrum of histologic findings recognized simultaneously. This poses a challenge for the diagnostic pathologist. We support our putative diagnosis of NMVH in these 2 cases based on our histologic findings, most of which were consistent with the ones described in the human literature, including haphazardly arranged smooth muscle fascicles, nerve fibers, and bundles, proliferative vascular profiles in addition to connective fibrous tissue, and adipose cells.1,19 However, no ganglion cells were found in either case, unlike in a previous canine report.20,23 In both of our cases, the lesions extended from the submucosa to the serosa, unlike the previous report in which only the muscular layer and serosa were affected. 23 As in previous reports, the diagnosis of NMVH in veterinary cases was confirmed using one IHC marker for smooth muscle (α-SMA, desmin), one for nervous tissue (S100, neurofilament, GFAP), and one vascular marker (CD34, CD31, von Willebrand factor).20,22,23
Several differential diagnoses must be considered when evaluating a cecal mass associated with intussusception with the histologic features aforementioned. Leiomyoma and leiomyosarcoma are benign and malignant proliferations of smooth muscle cells and are commonly encountered in the intestine of dogs, associated with anemia, melena, hypoglycemia, tenesmus, obstruction, weight loss, and perforation of the intestine. 11 Leiomyosarcoma has been reported within an intussusception. 11 The cellular proliferation was not uniform in our 2 cases, thus neoplasm was not favored. Chronic inflammation can induce the proliferation of granulation tissue, with fibrosis, hypertrophic and hyperplastic fibroblasts, and neovascularization. 13 In both of our cases, mucosal ulceration was present, and neovascularization could be attributed to a healing ulcer; however, adipose and nervous components are not features of granulation tissue. The nerve bundles could be derived from the myenteric plexuses, with a normal anatomic arrangement in cases of inversion of the cecum. 23 However, in both of our cases, the nerve bundles were found within the masses.
Finally, the differential diagnoses list should include vascular abnormalities within the gastrointestinal tract in dogs. These lesions are seldom reported and comprise scattered case reports describing a spectrum of vascular malformations, including Dieulafoy lesion in the gastric mucosa, jejunal arteriovenous fistula, and small intestinal, cecal, and colonic vascular ectasia (angiodysplasia).2,3,5,7,9,15 In humans, vascular abnormalities in the gastrointestinal tract are classified as arteriovenous malformations, multiple phlebectasia, telangiectasia, hemangioma, and blood vessel abnormalities related to disorders in connective tissue. 10 Arteriovenous malformations are further subclassified into angiodysplasias and vascular ectasias, which occur more commonly in the colonic mucosa and small intestine, respectively, without any predisposing factor aside from the age of the patient. 10 In our cases, the blood vessels were irregular and tortuous with medial hypertrophy; however, convincing evidence of arteriovenous fistula was not evident. Hypertrophy of the tunica media was the only vascular finding previously reported in NMVH. 1
In humans, NMVHs are infrequent lesions reported to occur in the small and large intestines.14,17 This condition has been described in male and female patients ranging from 12 to 91 y of age, and the lesion is associated with gastrointestinal bleeding, abdominal pain, and intestinal obstruction.8,17 Histologic features of NMVH in humans overlap with chronic Crohn disease, ischemia, and enteritis secondary to radiation therapy. 19 The etiology of NMVH is not fully understood and is controversial in human medicine. Some authors claim that rather than a separate entity, NMVH represents a chronic phase of an inflammatory process such as Crohn disease.4,12,18 In contrast, other published cases classify NMVH as separate from a chronic inflammatory process.6,17 In our cases, there was evidence of mucosal ulceration due to the intussusception; however, an underlying comorbidity, such as inflammatory bowel disease, was not identified in the histologic sections examined. Ancillary testing failed to identify a relevant infectious agent in either case, and surgical removal was considered curative with a good prognosis (12 mo at minimum).
NMVH is a rare condition in dogs, associated with hematochezia and intussusception. Surgical removal was considered curative in our 2 cases. Our putative diagnoses of NMHV were based on histologic findings. IHC stains for vascular, smooth muscle, and nervous histologic structures are necessary to diagnose the hamartomatous mesenchymal components. NMVH etiology and pathogenesis remain obscure in animals and humans.
Footnotes
Acknowledgements
We thank Lisa Parsons, Lisa Jolly, and Keisha Snerling from the Histology Laboratory of Auburn University for technical assistance and slide preparation for the immunohistochemistry procedures.
Declaration of conflicting interests
Tatiane Terumi Negrão Watanabe and Linda Huang are employed by Antech Diagnostics, Mars Petcare Science & Diagnostics. The remaining authors declared no potential conflicts of interest concerning the research, authorship, and/or publication of this article.
Funding
We did not receive any specific grant from public, commercial, or not-for-profit funding agencies.
