Abstract
Congenital hepatic fibrosis was observed in a newborn calf. Light microscopy revealed that periportal areas were linked via connective tissue to the central vein regions and to other periportal areas. Hyperplastic fibers were positive for type I collagen. A remarkable increase in the number of myofibroblasts that were positive for α-smooth muscle actin and vimentin was observed in the inner wall of the sinusoids, indicating the occurrence of various fibrogenesis. Ultrastractually, foci of cells resembling cholangiole epithelium cells were observed within the sinusoids, thereby suggesting either ductal plate dysplasia or a bile duct anomaly.
Congenital hepatic fibrosis, which was described in 1961, 5 is a fibroplastic disease affiliated with anomalous disorders of the liver, such as Meckel's syndrome 7 Wilson's disease. 10 This disease is relatively rare in humans and has been reported to resemble biliary cirrhosis in infants and young adults. 1,4
At the time of birth, a 1-day-old male Holstein–Friesian calf showed marked abdominal swelling, and the animal died soon after birth. Necropsy revealed marked enlargement of the liver (approximately 30 × 20 × 10 cm) and extreme irregularity and increased hardness of the surface (Fig. 1). The cut surface revealed a lobular structure partitioned by connective tissue and widespread dilatation of the intrahepatic veins. In addition, a cyst about 20 cm in diameter had formed in the left pulmonary posterior lobe.
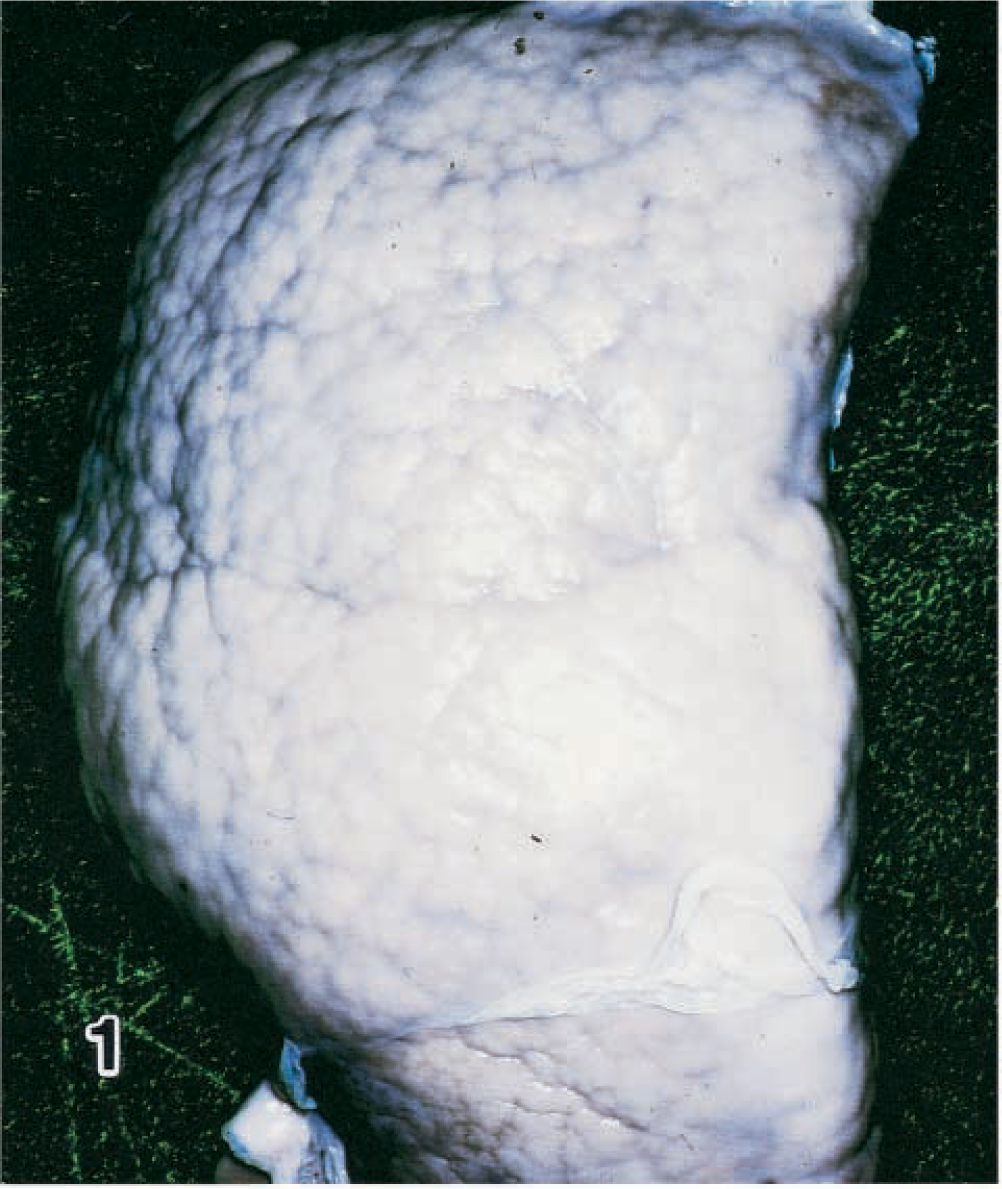
Liver; calf. Congenital hepatic fibrosis. Note extreme irregularity and consistency of the suface.
Tissue samples were fixed in 10% neutral buffered formalin and processed routinely for hematoxylin and eosin (HE), Azan, and Elastic van Gieson stains. Liver samples prepared in liquid nitrogen and cut by a freezing cryostat were used for immunohistochemical analysis by the avidin–biotin–peroxidase complex (ABC) procedure (Histofine SAB-PO Kit, Nichirei Co., Tokyo, Japan). The antisera used in this study were specific for type I at a dilution of 1:80, type III at 1:80, and type IV collagen at 1:160 (Chemicon, International ICN, Temecula, CA), cytokeratin (AE-1) prediluted (YLEM, Roma, Italy), vimentin prediluted (Biomedica Co., Foster City, CA), desmin prediluted (Biomedica), and α-smooth muscle actin (SMA) prediluted (Biomedica Co.). Formalin-fixed liver samples were postfixed with 1% osmic acid, embedded in Epok 812 resin, cut at 69 nm, stained with uranyl acetate and lead nitrate, and observed with a Hitachi H-7000 electron microscope.
Histologically, the liver findings included widespread fibrosis and linkage of periportal areas to the central vein region and to other periportal areas via connective tissue, which stained strongly with Azan stain (Fig. 2). The sinusoids were markedly enlarged because of hyperplastic proliferation of the fine fibers, whereas the hepatic cords showed signs of atrophy. Hepatocytes frequently formed foci and occasionally formed glandular cavities (Fig. 3). Moreover, foci of cells resembling cholangiole epithelial cells with a bright nucleus and dark cytoplasm were observed within the sinusoids, and frequent formation of cholangiole structures similar to the canal of Hering was also noted. Numerous fusiform cells covered the inner wall of the sinusoids.
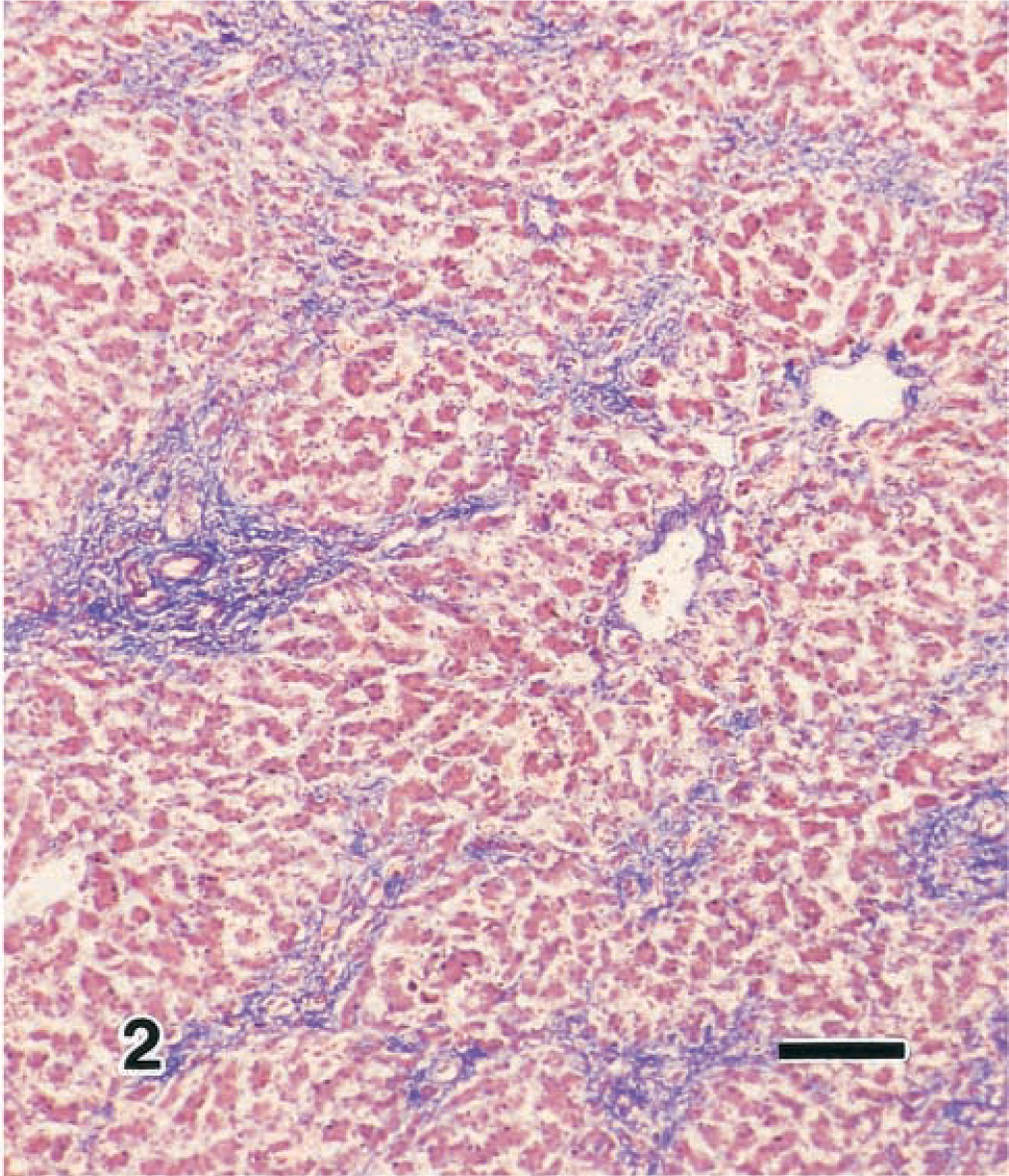
Liver; calf. Congenital hepatic fibrosis. Note the increased amount of fibrous conective tissue in the perilobular area. Azan. Bar = 120 µm.
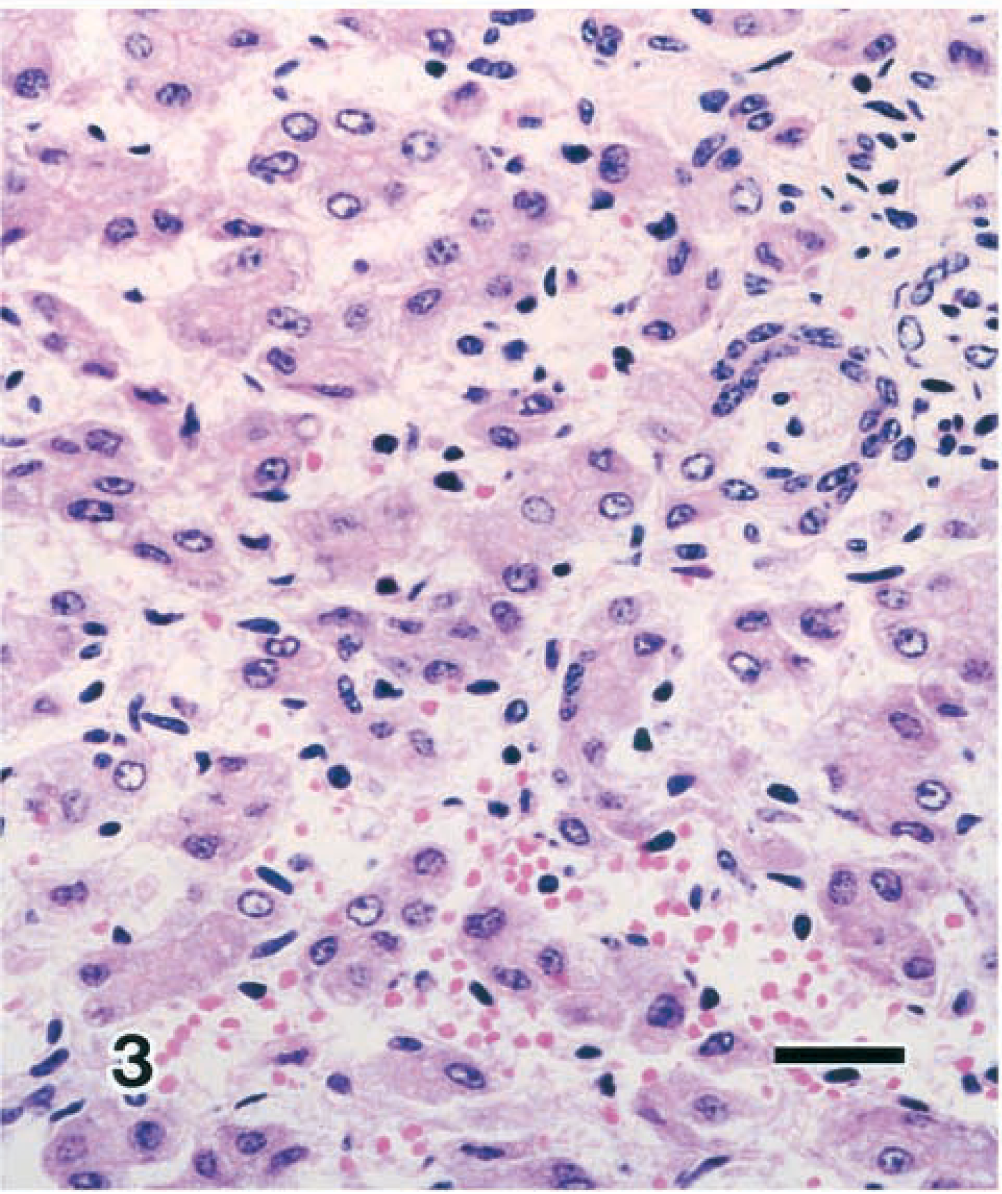
Liver; calf. Congenital hepatic fibrosis. Hepatocytes constitute a cholangiole structure similar to the Hering canal. HE. Bar = 25 µm.
Immunohistologic investigation revealed that the hyperplastic fibers present in the portal area, around the central vein, and in the sinusoid were positive for type I collagen. There was an increased level of type IV collagen in the outer wall of the sinusoids, and some periportal areas were positive for type III collagen. The cholangiole epithelium discovered within the sisusoids showed a positive response to anti-AE1 antibody (Fig. 4). Furthermore, hepatocytes showing weak positivity were either scattered singly or in groups within the hepatocyte foci. The fusiform cells that covered the inner wall of the sinusoids showed positive responses to both anti-α-SMA and vimentin (Fig. 4). Ito cells showing desmin positivity were not observed.
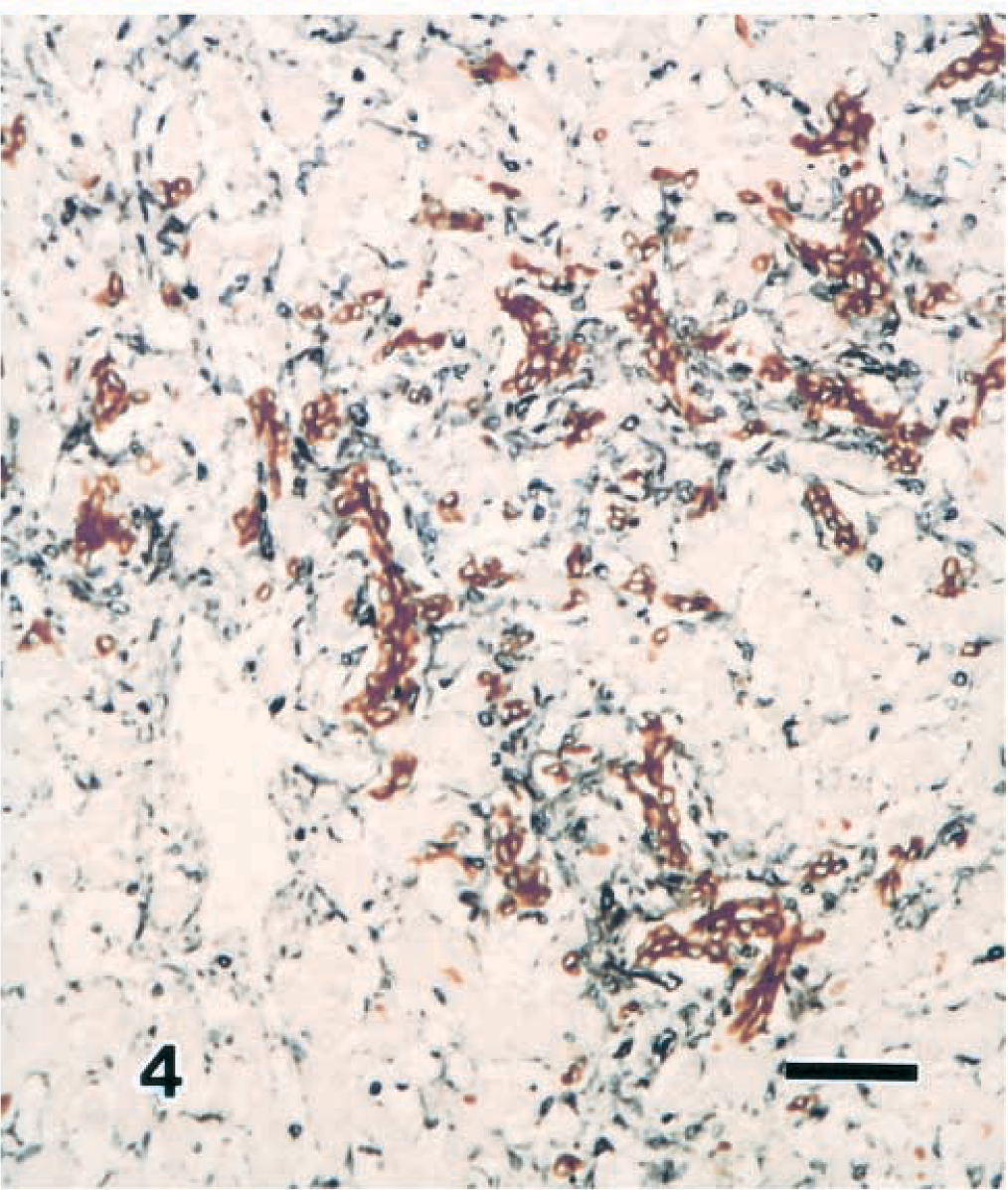
Liver; calf. Congenital hepatic fibrosis. The cholangiole epithelium cells stain positive with anti-AE1 (brown color), and the myofibroblasts stain positive with anti-vimentin (blue color). ABC method. Bar = 45 µm.
Electron microscopic examination revealed that hepatocyte foci with incompletely formed lumina were connected to the sinusoids and discharging bile. These hepatocytes had a situated nucleus, granular endoplasmic reticulum, free ribosomes, and mitochondria with microvilli projecting from the free edge. This morphology contains features of cholangiole epithelial cells and is an intermediate form that resembles cells of the canal of Hering. The endoplasmic reticula and mitochondria in the cytoplasm of the peripheral hepatocytes were dilated, and a substance of homogeneously low–medium electron density and a myelin-like structure were present within the cristae of the mitochondria.
Many of the glomeruli and proximal tubules of the kidneys displayed an immature morphology, small cysts were observed at various sites. The large cyst in the lung consisted of a cluster of small cysts up to 1 cm in diameter. These small cysts were lined with columnar cells, covering elastic and fibrous connective tissue with a little muscle.
Although the specific morbidity associated with this disease was hyperplasia of the bile duct with type I collagen fibroplasia and an abnormal morphology, cyst formation was also observed in the kidney and lung; thus, this disease closely resembles congenital hepatic fibrosis affiliated with polycystic disease reported in humans. 1 Ductal plate malformation arising from incomplete ductal plate formation or reconstruction abnormality during early embryonal bile duct formation has been implicated in the pathologic onset of congenital hepatic fibrosis. 2 Developmentally, the liver forms from the ductal plate as a primordium from the early to mid-embryonal period, after which the liver parenchyma and bile duct form as the result of reconstruction. 9
The cells that reacted positively to anti-vimentin and anti-α-SMA antibodies within the sinusoid of the fibroplastic lesion displayed the features of myofibroblasts, thereby suggesting their active participation in the process of fibrogenesis. The myofibroblastic cells transformed from Ito cells are positive for anti-α-SMA and anti-desmin. 3,8 In the present study, the antibody for desmin did not stain myoepithelial cells. However, Ito cells were less frequently and less intensely stained with anti-desmin when compared with the reaction for anti-α-SMA antibody. 3 Further studies are needed to clarify the relation of the myoepithelial cell and the Ito cell.
The present case resembled cases of human congenital hepatic fibrosis, but an important feature of fibroplasia of the liver is the appearance of myofibroblasts derived from abnormal formation of the cholangiole epithelium, suggesting that cholangiofibrosis, in which myofibroblasts play the major role, is a noteworthy feature underlying the onset of this disease. 6
