Abstract
Melanoma is the most common malignant oral tumor in dogs. It frequently presents a diagnostic challenge as many melanomas lack or contain scant melanin and may have a variable microscopic phenotype. Previous studies evaluating immunohistochemical markers for diagnosing melanoma have shown limited sensitivity and/or specificity for S-100, PNL2, melan A, TRP-1, TRP-2, and HMB-45. Sry-related HMG-box gene 10 (SOX-10) is a transcription factor associated with melanocytic, peripheral neural crest, and peripheral nervous system development. In humans, SOX-10 expression has been demonstrated in melanoma, breast carcinoma, glioma, and schwannoma, but has only recently been explored in veterinary species. In this study, 198 tumors comprised of 147 melanocytic neoplasms and 51 non-melanocytic neoplasms were evaluated by immunohistochemistry using a tissue microarray for SOX-10, PNL2, melan A, TRP-1, and TRP-2 expressions. The SOX-10 had the highest diagnostic sensitivity (96.7%) in melanomas. In addition, SOX-10 had the highest percentage (91.5%; 130/142) of melanomas label at least 75% of neoplastic cells. Of the 51 selected non-melanocytic tumors examined, SOX-10 labeling was observed in mammary carcinomas (6/6), gliomas (4/4), and oral soft tissue sarcomas (4/18). Of the 41 non-melanocytic oral neoplasms evaluated, SOX-10 had a specificity of 92.7%. Therefore, SOX-10 represents a useful immunohistochemical screening marker for the diagnosis of canine melanoma given its extremely high sensitivity and robust labeling intensity. The SOX-10 may have utility in diagnosing some non-melanocytic neoplasms in the dog, although this requires further investigation.
Melanoma is a common malignant neoplasm in the dog and is associated with an aggressive biological behavior.3,16 Common anatomic locations for canine melanoma include the oral cavity, nail bed, eye, and skin, although other anatomic sites have been reported. 16 Melanoma can present a diagnostic challenge in cases where tumors either lack or contain scant amounts of melanin, a key histologic diagnostic feature. 20 Moreover, melanomas are often morphologically heterogeneous and can be composed of variable cytologic phenotypes including polygonal and spindloid phenotypes, making distinguishing amelanotic melanomas from other neoplasms, such as sarcomas, difficult.19,20
In conjunction with histopathologic evaluation, immunohistochemistry (IHC) may facilitate the diagnosis of melanoma. Various IHC antibodies have been evaluated for the diagnosis of melanoma including melan A, PNL2, tyrosinase-related proteins 1 and 2 (TRP-1 and TRP-2), S-100, human melanoma black-45 (HMB-45), microphthalmia transcription factor (MITF), and tyrosinase.19–21 These antibodies have variable sensitivities and specificities, limiting the ability of a single antibody to confidently diagnose melanoma. At this time, the most comprehensive IHC panel used in the diagnosis of oral melanoma consists of a panel or cocktail of 4 IHC markers including melan A, PNL2, TRP-1, and TRP-2. With this panel, the reported sensitivity and specificity are 100% and 93.9%, respectively, when compared with subcutaneous soft tissue sarcomas. 20
Sry-related HMG-box gene 10 (SOX-10) is a transcription factor that acts as a nucleocytoplasmic shuttle protein and is important for melanocytic, peripheral neural crest, and peripheral nervous system development. 18 In humans, SOX-10 expression has been demonstrated in melanomas, breast carcinomas, gliomas, schwannomas, and granular cell tumors.5,10,15,17 For humans with malignant melanoma, SOX-10 IHC has a reported sensitivity of 100% and specificity of 93% with a positive predictive value of 87% and negative predictive value of 100% when compared with carcinomas and normal tissue. 15 In addition, SOX-10 has shown to be comparable or superior to S-100 in the identification of metastatic and micrometastatic melanoma in lymph nodes in humans.4,9
Although many reports have evaluated the efficacy of SOX-10 in humans, SOX-10 has only recently been evaluated in veterinary species, including the evaluation of a malignant nerve sheath tumor in a pig and to aid in the identification of neural crest stem cells in dogs undergoing spinal stem cell transplants.14,23 Furthermore, a study evaluating 20 oral melanomas indicated some, although limited, utility of SOX-10 in discerning melanoma from other oral neoplasms. 24 The SOX-10 has also been evaluated in screening for nodal metastasis of melanoma. 25 Given the nascent literature regarding the use of SOX-10 as a veterinary diagnostic immunomarker, we sought to comprehensively characterize its expression in both melanocytic and non-melanocytic canine neoplasms.
A tissue microarray (TMA) is a tissue evaluation system that allows for rapid histopathologic analysis of panels of dozens to hundreds of tissue samples while ensuring identical experimental conditions for each sample.12,13,22 With this technique, targeted or random tissue “cores” are collected from paraffin blocks and samples are re-embedded in a receiver block arranged in a grid layout. The prototypical use for TMAs has been IHC-based discovery and prognostic studies in cancer research and diagnostics. The TMA provides a cost-effective approach for immunomarker evaluation, as the compounding expense of evaluating individual slides for large study sets, such as is needed for powerful studies on sensitivity and specificity, may be prohibitive. Furthermore, the small sample size and arrangement of tissues on the array provides for rapid histopathologic evaluation in parallel. The processing of samples in parallel is also beneficial for ensuring consistent experimental conditions. Given the capability for screening large numbers of samples at a time, we used a TMA to screen for the expression of the diagnostic melanocytic immunomarkers melan A, PNL2, TRP-1, and TRP-2, and the candidate melanocytic marker SOX-10, comparing the expression between melanocytic and non-melanocytic tumors and various tissues. The aims of this study were to (1) verify the expression of SOX-10 in canine melanoma cell lines and (2) comprehensively determine the sensitivity and specificity of SOX-10 immunolabeling and diagnostic utility of this marker in the diagnosis of melanoma in the dog using the high-throughput TMA methodology.
Materials and Methods
Tumor Selection
Melanocytic neoplasms were selected by searching “melanoma” in 2 diagnostic laboratory case databases (a private diagnostic laboratory [Histotechniques] and an academic veterinary diagnostic laboratory [The Ohio State University]). The hematoxylin and eosin (HE)-stained slides were reviewed. Study inclusion for melanomas required one or more of the following criteria: junctional activity, intraepithelial proliferation (lentiginous or Pagetoid pattern), and melanin pigment. The pathologist also marked specific regions for TMA sampling, targeting subjectively representative regions lacking significant necrosis or hemorrhage. In addition, the academic diagnostic laboratory database was searched for a targeted selection of non-melanocytic tumors. Focus was given to non-melanocytic oral malignancies (eg, sarcoma and squamous cell carcinoma) as well as mammary neoplasms and gliomas (tumors reported to express SOX-10 in humans). 5 The inclusion criteria for oral soft tissue sarcomas included streaming and/or interwoven bundles of neoplastic spindloid cells without production of osteoid or cartilage, no vasoformative phenotype, and no presence of our inclusion criteria listed for melanomas. Non-melanocytic tumor types and case distributions are listed in Table 1.
Non-melanocytic neoplasms evaluated.

Tissue Microarray
All HE-stained slides were evaluated by a board-certified pathologist (RNJ). For each slide, a single region of interest was indicated with permanent marker by selecting regions considered most diagnostically representative of the tumor phenotype and absent of significant necrosis or hemorrhage. The HE-stained slide was then provided to the histology technician along with the original tissue block, and cores were obtained at the indicated region of interest using a 2-mm diameter punch needle and inserted into a recipient paraffin block using the Manual Tissue Arrayer MTA1 (Beecher Instruments, Sun Prairie, Wisconsin). TMA blocks were sectioned using a standard microtome at a thickness of 5 µm.
Immunohistochemistry
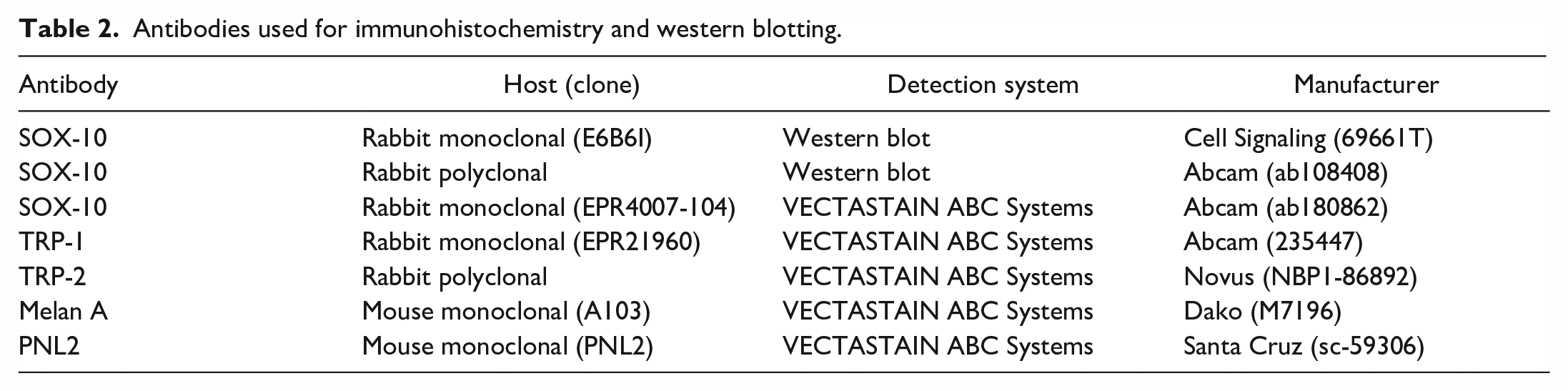
Details on primary antibodies, antigen retrieval, and detection procedures are summarized in Table 2. Following antigen retrieval with 1X Target Retrieval Solution (citrate; Agilent, Santa Clara, California) at 90°C to 95°C, sections were cooled for 15 minutes by immersing in room temperature water. Endogenous peroxidase was inactivated with 3% hydrogen peroxide diluted in methanol for 15 minutes. Sections were washed with 0.1% Tween-20 in Tris-buffered saline and protein blocked with 10% normal donkey serum (Jackson 017-000-121, Jackson ImmunoResearch Laboratories, Inc., West Grove, Pennsylvania) diluted in Tris-buffered saline for 15 minutes. Primary antibodies and sections were transferred to the autostainer (Lab Vision 360 Autostainer, Thermo Fisher Scientific, Waltham, Massachusetts) to incubate for 30 minutes at room temperature. Following rinsing with Tris-buffered saline, sections were incubated with secondary anti-mouse or anti-rabbit antibody (5 µg/ml) for 30 minutes at room temperature, rinsed again, and incubated in VECTASTAIN Elite ABC-HRP Reagent (Vector/PK-7100, Vector Laboratories Inc., Newark, California) for 30 minutes. Sections were rinsed, and labeled complexes were visualized with 3,3’-diaminobenzidine chromogen (Dako/K3468, Agilent, Santa Clara, California) and counterstained with Richard-Allan hematoxylin 2. A canine thyroid adenocarcinoma cell line (CTAC) was used as a negative control. 11 Four human melanoma cell lines (LM-MEL-1a, LM-MEL-19, LM-MEL-28, and LM-MEL-30 [ATCC, Manassas, Virginia]) were also evaluated and served as positive controls.
Antibodies used for immunohistochemistry and western blotting.
Qualitative and Semi-Quantitative Evaluation of Immunolabeling
TMAs were blindly evaluated independently by 1 board-certified anatomic pathologist (RNJ) and 1 anatomic pathology trainee (EK). Each sample was scored as either positive or negative for immunolabeling. If positive, the sample was then scored for the estimated percentage of neoplastic cells labeling. Labeling percentage scoring consisted of assigning the samples to one of five bins, representing immunolabeling of neoplastic cells equal to or more than the following percentages: 10%, 25%, 50%, 75%, or 90%. For example, if it was estimated that 15% of cells labeled for a marker, this would be assigned to the “10” category, given that the percentage was more than 10 and less than 25. Samples with less than 10% of neoplastic cells labeling were considered negative. If disagreements in scoring were identified, a consensus was achieved.
Cell Lines, Protein Extraction, and Western Blot Analysis
Three canine melanoma cell lines (Jones, Parks, and 17CM98) were obtained from the Animal Cancer Center at Colorado State University.1,8 The CTAC cell line, a thyroid adenocarcinoma cell line, was used as a negative control. 11 Four human melanoma cell lines (LM-MEL-1a, LM-MEL-19, LM-MEL-28, and LM-MEL-30) were also evaluated. Cell lines were cultured in Dulbecco’s Modified Eagle’s Medium (1×) (Life Technologies, Paisley, UK, catalog #11995-065) supplemented with 10% fetal bovine serum (BenchMark, Gemini, catalog # 100-106), GlutaMAX (Life Technologies, 35-050-061), Modified Eagle’s Medium Non-Essential Amino Acids Solution (Life Technologies, 11-140-050), and Antibiotic-Antimycotic (Life Technologies, 15-240-062). Cells were washed with 1× phosphate-buffered saline and incubated with gentle rocking at 4°C for 30 minutes in 0.5% NP-40 lysis buffer (150 mM NaCl, 0.5% NP-40, 50 mM Tris-HCL, pH 8.0) supplemented with protease inhibitors (cOmplete, Mini Protease Inhibitor Cocktail, Sigma-Aldrich, St. Louis,). Samples were spun at 15 000 rpm at 4°C for 10 minutes. The supernatant was collected and frozen at −80°C. Protein lysate concentrations were quantified using a BioRad Bradford Assay reagent (BioRad, Hercules, California, catalog # 500-0006). Thirty micrograms of each protein lysate were heat-denatured at 95°C for 5 minutes. Proteins and BenchMark pre-stained protein ladder (Invitrogen catalog no. 10748010) were then resolved by a Mini-Protean TGX (4%-20% gradient gel; BioRad) sodium dodecyl-sulfate polyacrylamide gel electrophoresis (SDS-PAGE) gel run at 100 V for 1 to 1.5 hours and then transferred to a nitrocellulose membrane (Amersham Protan 0.45 µm NC, GE Healthcare, Germany). Nonspecific binding sites were blocked for ≥30 minutes in 5% nonfat dry milk (bovine serum albumin [BSA] was used for Cell Signaling Antibody) in phosphate-buffered saline containing 0.1% Tween-20 at room temperature. Blots were then incubated with a primary antibody against SOX-10 overnight at 4°C on a rocker. The next day, blots were rinsed and incubated with a secondary antibody (Cy5-labeled goat anti-rabbit secondary antibody [1:1,250], GE Healthcare, PA45011) for 1 hour at room temperature. All antibodies were diluted in phosphate-buffered saline containing 0.1% Tween-20 containing 5% nonfat dry milk. After washing in phosphate-buffered saline containing 0.1% Tween-20, the membranes were visualized using an Amersham Typhoon imager (GE Healthcare). Images were collected using ImageQuant TL software (Cytvia, Marlborough, Massachusetts).
Evaluation of Sensitivity and Specificity
IHC sensitivity and specificity were based on original diagnoses of the HE-stained sections (considered “true positive” for this evaluation). Sensitivity was calculated as true positive melanomas/(true positive melanomas + false negative melanomas). Immunomarker specificity of SOX-10 was calculated as negative non-melanomas/(negative non-melanomas + positive non-melanomas).
Results
SOX-10 Protein Expression in Cell Lines
Three SOX-10 monoclonal antibodies were evaluated by western blot for immunoreactivity to cell lysates from 3 canine melanoma cell lines and 1 canine thyroid adenocarcinoma cell line (negative control). Specific immunoreactivity was found with 2 of the antibodies against 2 of the melanoma cell lines (Parks, 17CM98) and was negative in one of the melanoma cell lines (Jones) and the thyroid (CTAC) cell line. Two of the antibodies cross-reacted with specific bands of ~70 and ~100 kda, likely representing post-translationally modified SOX-10 protein, vs the calculated molecular weight of ~50 kda (Fig. 1). 6 One of these antibodies had a non-specific band present in all cell lines, and the third antibody did not cross-react with any cell lysate (Supplemental Figure S1). Based on these results, rabbit monoclonal clone EPR4007-104 was chosen for further evaluation. Human cell lines were then evaluated with this antibody, serving as positive controls. Strong and identical immunoreactivity was demonstrated with specific bands at ~70 and 100 kda in 3 human melanoma cell lines (LM-MEL-1a, LM-MEL-19, and LM-MEL-28), with weaker bands observed in a fourth human melanoma cell line (LM-MEL-30) (Supplemental Figure S2).
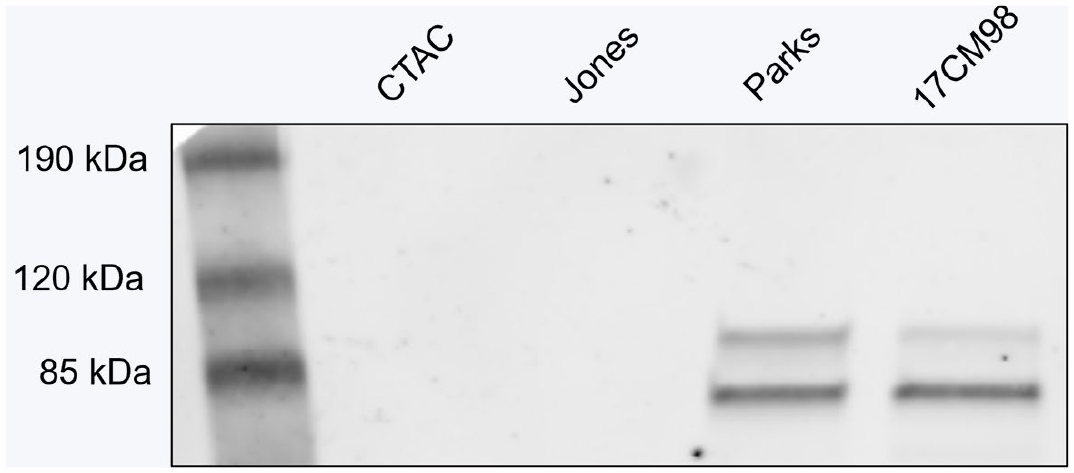
Western blot results for SOX-10 expression in 3 melanoma cell lines (Jones, Parks, and 17CM98) and control thyroid cell line (CTAC). Two of 3 melanoma cell lines (Parks and 17CM98) showed 2 bands at ~70 and ~100 kda.
Melanomas
A total of 157 melanomas met the inclusion criteria, were processed, and evaluated using TMA. Ten cases were excluded due to inadequate tissue being present for the evaluation of one or more immunomarkers in the TMA, precluding paired evaluation across all 5 immunomarkers. The final 147 cases included 103 oral melanomas and 44 non-oral melanomas.
HE and immunohistochemical expression of the melanocytic immunomarkers from a representative oral melanoma are shown in Figure 2. SOX-10 had the highest sensitivity in melanomas (Fig. 3a), with labeling in 142/147 of melanomas (sensitivity: 96.7%). TRP-2 had the second highest sensitivity, with labeling in 120/147 of melanomas (sensitivity: 84.5%). TRP-1 labeled the lowest number of melanomas, with labeling in 52/147 (sensitivity: 60.7%). Of total, 73.2% (104/142) of the SOX-10 positive melanomas had labeling of 90% or more of neoplastic cells within the TMA tissue, with only 2.8% (4/142) of SOX-10 positive tumors having less than 50% of tumor cells labeling (Fig. 3b). In comparing oral and non-oral melanocytic neoplasms, 95.1% (98/103) of oral melanocytic neoplasms and 100% (44/44) of non-oral melanocytic neoplasms labeled for SOX-10 (Figs. 3c, d).
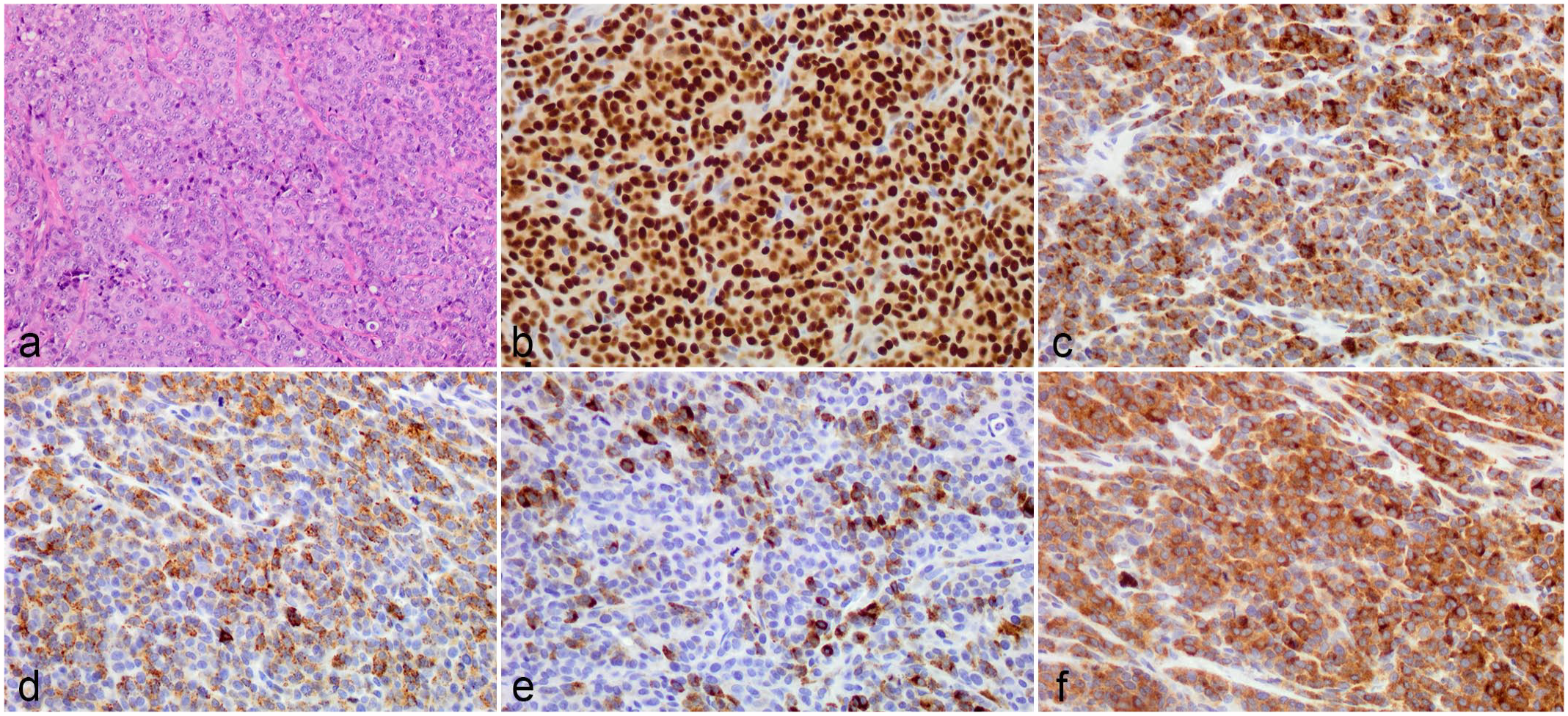
Histology and immunohistochemistry demonstrating representative morphology and positive immunolabeling for melanoma-associated immunomarkers in a canine single amelanotic oral melanoma. (a) Hematoxylin and eosin, (b) SOX-10 (nuclear), (c) melan A (cytoplasmic), (d) PNL2 (cytoplasmic), (e) TRP-1 (cytoplasmic), and (f) TRP-2 (cytoplasmic).
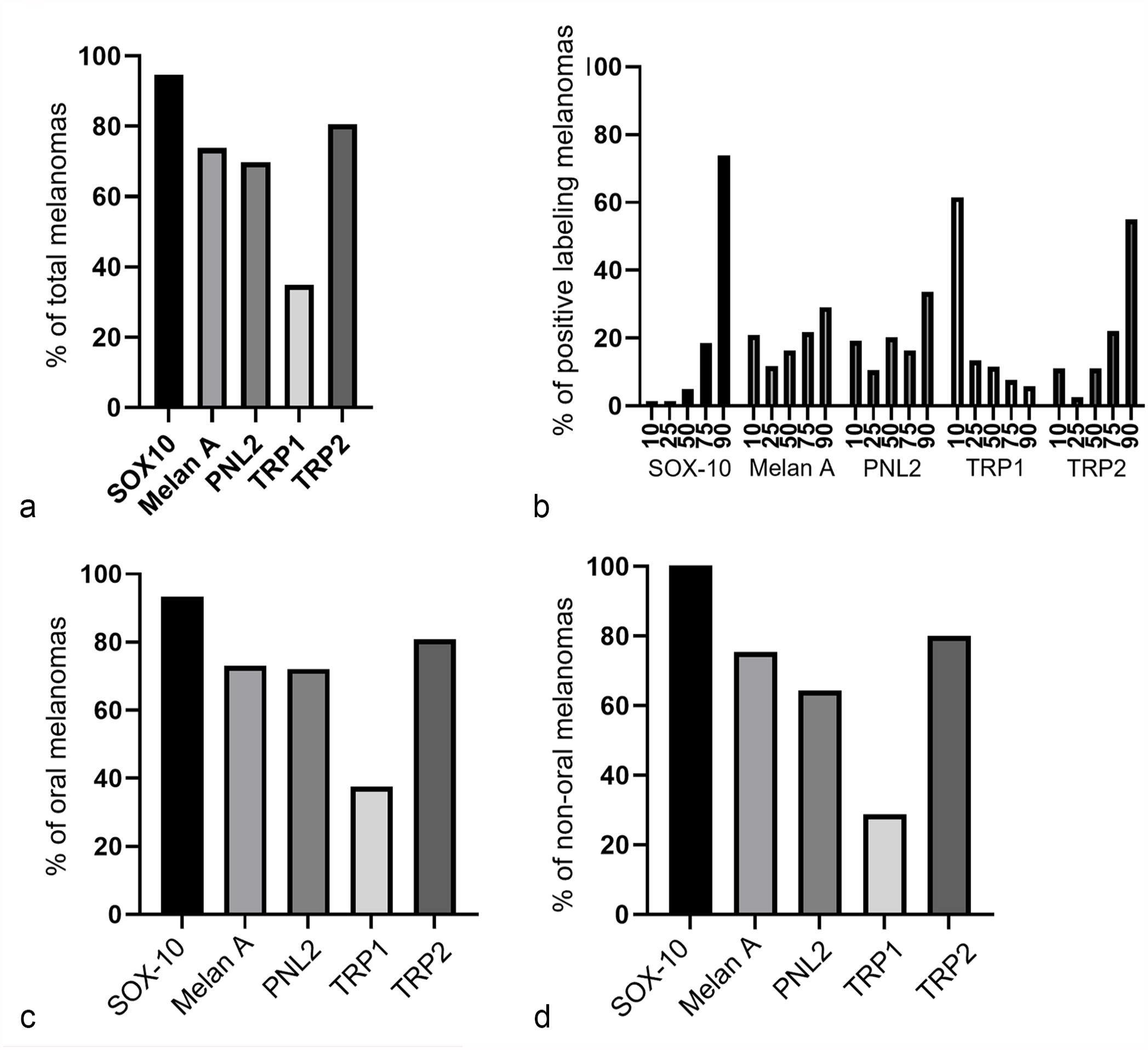
Immunohistochemical labeling in melanoma. (a) Percentage of immunopositive melanomas. (b) Percentage of cells labeling within immunopositive melanomas. (c) Percentage of oral melanomas with immunolabeling. (d) Percentage of non-oral melanomas with immunolabeling.
Non-Melanocytic Neoplasms
Fifty-one non-melanocytic tumors were evaluated. There were 14 SOX-10 positive non-melanocytic tumors, which represented 27% of non-melanocytic tumors evaluated and included the following: mammary carcinoma (6/6; 100%) (Fig. 4a), glioma (4/4; 100%) (Fig. 4b), and oral soft tissue sarcoma (4/18; 22%) (Fig. 4c). Of the 41 oral neoplasms examined (20 squamous cell carcinomas, 18 oral soft tissue sarcomas, and 3 peripheral odontogenic fibromas), 4/41 neoplasms were SOX-10 positive, all of which were oral soft tissue sarcomas. None of these SOX-10 positive non-melanocytic tumors labeled for melan A, PNL2, TRP-1, or TRP-2.
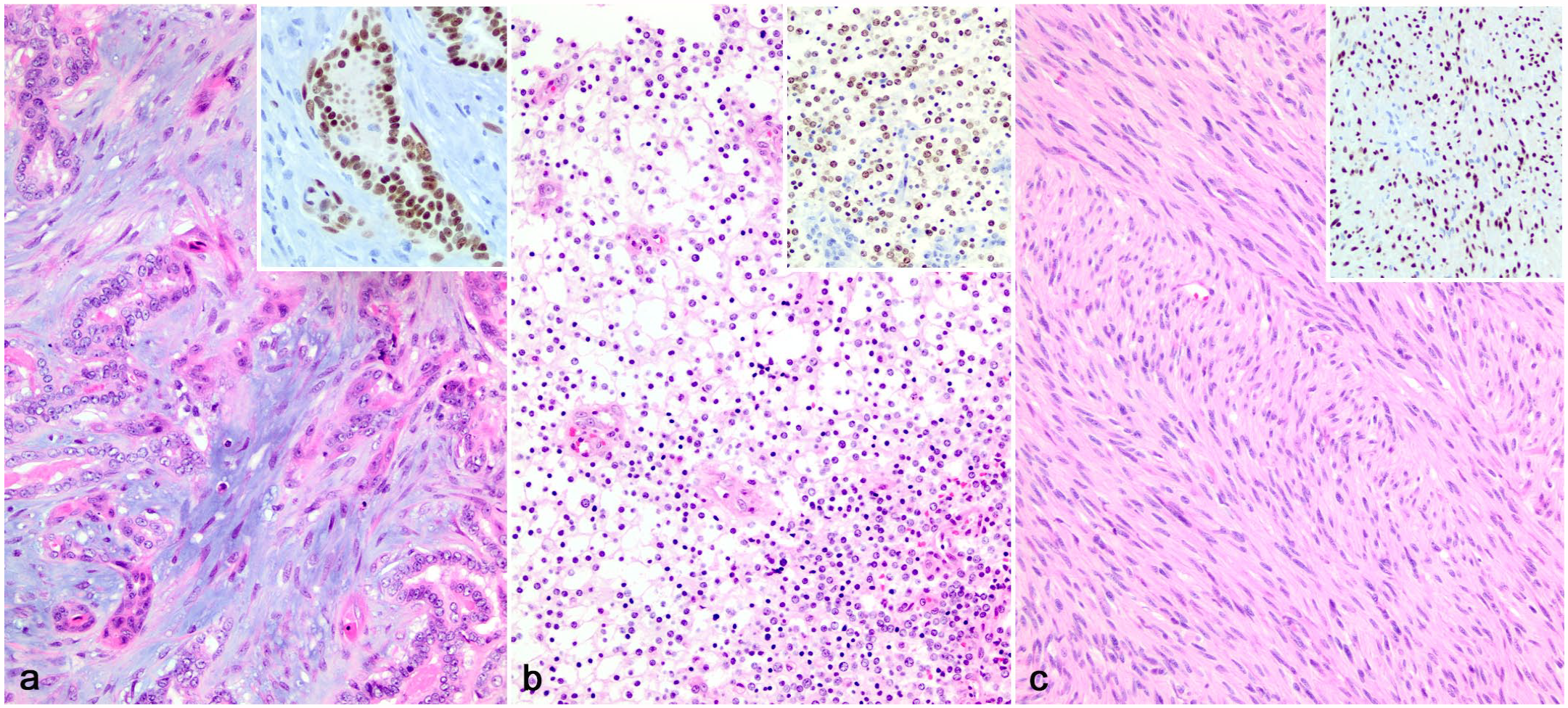
SOX-10 immunohistochemical labeling in non-melanocytic tumors. (a) Complex mammary carcinoma, dog. Hematoxylin and eosin (HE). Inset: strong nuclear immunolabeling for SOX-10. (b) Oligodendroglioma, dog. HE. Inset: moderate nuclear immunolabeling for SOX-10. (c) Oral soft tissue sarcoma, dog. HE. Inset: strong nuclear immunolabeling for SOX-10.
Discussion
Diagnosis of melanocytic neoplasms can be difficult due to the variable histologic appearance of melanomas, requiring the use of ancillary diagnostics such as IHC. Although an array of immunohistochemical markers has been proposed for the diagnosis of canine melanoma, each individual marker has its limitations, with varying sensitivities and specificities. A cocktail of multiple melanocytic markers has been shown to have a high sensitivity and specificity. 20
Here, we evaluated the utility of SOX-10 as a viable IHC marker for canine melanoma. Our results indicate that SOX-10 is a sensitive marker for canine melanomas, with a sensitivity of 96.7%, a similarly high sensitivity as that of SOX-10 in human melanoma. 15 Furthermore, immunolabeling was of robust intensity in positive tumors and was present in the majority of neoplastic cells, features that were superior to the other immunomarkers evaluated. Immunomarkers with unequivocal labeling are prioritized in diagnostics, and the results here support SOX-10 as an excellent screening marker for melanoma in ambiguous cases.
In our study, SOX-10 had a specificity of 92.7% in the oral non-melanocytic tumors evaluated. Immunolabeling was observed in oral soft tissue sarcomas, mammary carcinomas, and gliomas. In humans, SOX-10 immunolabeling is reported in mammary carcinomas, gliomas, and peripheral nerve sheath tumors, and we observed SOX-10 immunolabeling in both canine mammary carcinomas and gliomas in our study.2,10,15 We also observed SOX-10 labeling of 4/18 oral soft tissue sarcomas evaluated. In humans, peripheral nerve sheath tumors, which are considered within the category of soft tissue sarcomas in the canine classification system, are SOX-10 immunopositive. 10 Therefore, these SOX-10 positive oral sarcomas may represent peripheral nerve sheath tumors. None of the SOX-10-positive soft tissue sarcomas labeled for melan A, PNL2, TRP-1, or TRP-2. Interestingly, we subsequently evaluated S-100 labeling on original tissue blocks in these 4 SOX-10 positive soft tissue sarcomas; 4/4 were diffusely immunopositive for S-100 (data not shown), suggesting that they may have differentiation consistent with nerve sheath tumors. We conclude that tumors with a spindloid cell morphology that are SOX-10-positive require additional IHC (other melanocytic markers or markers specific to soft tissue sarcoma subtypes) to distinguish between spindloid cell-type melanoma and sarcoma subtypes. Importantly, distinction should also prioritize identification of distinguishing morphologic features of melanoma, such as pigmentation, intraepithelial neoplastic cells, and junctional activity.
Of the 3 canine melanoma cell lines we assessed by western blot, 2 expressed SOX-10. Loss of SOX-10 expression may be a tissue culture phenomenon in melanoma, as 3/12 human melanoma cell lines we tested did not express SOX-10 by western blot (data not shown), and SOX-10 has been shown to be integral to melanoma cell proliferation and survival, further suggesting that this absence of labeling in some cell lines may not be representative. 7
Of the 5 canine melanomas that were negative for SOX-10, all were negative for melan A, TRP-1, and TRP-2, with 1 tumor labeling diffusely positive for PNL2. We performed SOX-10 immunohistochemistry on the tissue blocks of all 5 of these melanomas. Four of 5 melanomas exhibited regional SOX-10 immunolabeling by whole-slide labeling, and in 3 of 4 of these, we suspect that tissue sampling was close but inaccurate for including diagnostic regions. Therefore, the true sensitivity for SOX-10 in our cases, by whole-slide evaluation, would be 99.3%. In addition, in 1 SOX-10 negative (TMA) melanoma, the SOX-10 labeling of the whole slide was widespread, putatively including the sampled region, despite being negative in the TMA. We are uncertain of the error that may have resulted in this being negative on the TMA. Finally, in 1 of 5 SOX-10 negative (TMA) melanomas, no SOX-10 immunolabeling was apparent in the whole slide; this melanoma was only positive for PNL2. We consider the possibility that this may represent an SOX-10 negative melanoma, something which appears to be rare. We conclude that accurate tissue sampling and quality control are crucial when using TMAs to screen immunomarkers, as any errors may confound results. In oral melanomas with diagnostically challenging morphologic phenotypes, the presence of intraepithelial neoplastic cells and/or junctional activity can be an important diagnostic feature. In this study, we did not specifically target regions of intraepithelial neoplastic cells or junctional activity in our region of interest selection, which may have impacted the sensitivity and specificity of our case evaluations. In addition, the sampling of non-diagnostic regions of the tissue blocks in several cases presents one of the technical confounding factors of TMAs. TMAs sample a small portion of the tissue block (typically 0.6-2 mm in diameter); therefore, evaluation of the TMA samples relies on accurate sampling of diagnostic regions of the original tissue block. Regions of inflammation and/or necrosis or variability in consistency of regions due to tissue depth, and so on may be causes of confounding factors. One potential downside of using TMAs for the validation or screening of immunomarkers is the small tissue size that is evaluated. Some neoplasms exhibit heterogeneous immunolabeling or immunolabeling of only certain populations of cells. Indeed, expression of certain melanocytic markers, such as melan A, has been shown to vary regionally, throughout a given melanoma and may be associated with morphologic phenotype. 19 This may limit capturing the full immunolabeling characteristics of certain tumors and should be considered when using a TMA to evaluate immunomarkers. Finally, tissue sampling may be random or targeted. The risks of random tissue sampling of tissue blocks include sampling of non-neoplastic tissue and necrotic (non-diagnostic) regions. To this point, we used a targeted TMA sampling approach where we identified the regions of interest prior to sampling, increasing the accurate representation of the tumors in the TMA. Finally, it should be noted that all of the melanomas evaluated here were diagnosed as melanoma based on specific diagnostic morphologic features on HE-stained sections. Therefore, the sensitivity and specificity that we report here is representative for the melanomas in this group (presumably well-differentiated melanomas) and may not be representative of all melanomas, such as poorly differentiated melanomas.
The results of this study support the utility of SOX-10 as a sensitive screening marker in cases of suspected melanoma. Owing to the immunolabeling of various other neoplasms, such as oral soft tissue sarcomas, with some confounding morphologic features, the specificity of SOX-10 should be considered when evaluating tumors with a spindloid cell morphology and lacking features such as pigment, junctional activity, and intraepithelial nests. Additional studies may be beneficial in identifying the diagnostic utility of SOX-10 expression in non-melanocytic tumors including glial, mammary, and peripheral nerve sheath tumors.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858241273318 – Supplemental material for Evaluation of SOX-10 immunohistochemical expression in canine melanoma and non-melanocytic tumors by tissue microarray
Supplemental material, sj-pdf-1-vet-10.1177_03009858241273318 for Evaluation of SOX-10 immunohistochemical expression in canine melanoma and non-melanocytic tumors by tissue microarray by Emily King, Matthew Cook, Hannah Wittorff, Wessel Dirksen, William C. Kisseberth and Ryan N. Jennings in Veterinary Pathology
Footnotes
Acknowledgements
We acknowledge Dr Rachel Cianciolo for her assistance in selection of regions of interest for the study set. We also thank Kevin Thorburn and the Comparative Pathology & Digital Imaging Shared Resource, Department of Veterinary Biosciences and the Comprehensive Cancer Center, The Ohio State University, Columbus, OH, and supported in part by grant P30 CA16058, National Cancer Institute, Bethesda, MD.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
