Abstract
The immunoreactivity of PNL2 and antityrosinase in formalin-fixed, paraffin-embedded canine melanocytic neoplasms (n = 101) was compared with that of Melan A. Of the 113 samples overall, 106 were positive for PNL2, 101 for Melan A, and 90 for tyrosinase. Six melanomas that were positive for PNL2 were negative for Melan A; 1 melanoma that was negative for PNL2 was positive for Melan A. Eighty tumors were positive for all 3 markers; 111 reacted with at least 1 the 3 antibodies. Decalcification with formic acid for up to 1 week did not affect immunoreactivity of any of the markers; however, decalcification with HCl for 1 day or 1 week notably decreased or completely abrogated immunoreactivity for Melan A and PNL2. There was only minor loss of immunoreactivity for tyrosinase in tissues decalcified with HCl for 1 week. Prolonged fixation (up to 2 months) did not affect PNL2 or tyrosinase immunoreactivity; however, Melan A immunoreactivity was reduced after 1 month of fixation. PNL2 was not expressed in 120 nonmelanocytic tumors (carcinomas, sarcomas, steroid-producing tumors, and leukocytic tumors). In summary, antibody PNL2 is slightly more sensitive than Melan A and more sensitive than tyrosinase in the identification of canine melanocytic neoplasms. Furthermore, PNL2 does not appear to cross-react with nonmelanocytic neoplasms. PNL2 is resistant to prolonged fixation but sensitive to strong decalcification. Results indicate that PNL2 is an excellent marker in the identification of canine melanomas and that the sensitivity is close to 100% when used in conjunction with Melan A and tyrosinase.
The diagnosis of melanoma may be challenging, particularly in tumors without appreciable melanin. 27,35 In addition, the variety of histologic appearances of canine melanocytic neoplasms makes them difficult to distinguish from sarcomas, carcinomas, or leukocytic tumors. 27 Immunohistochemistry is widely used to confirm a diagnosis of melanoma in human and veterinary medicine. 5,8,10,16,18,24,27,28,31,35 There are numerous melanoma markers with variable sensitivity and specificity. 10 Commonly used melanocytic markers in humans include Melan A, PNL2, S100 protein, and tyrosinase. 5,10,35
S100 protein, a calcium-binding protein with an unknown function in melanogenesis, is a widely used melanocytic marker with high sensitivity (defined as percentage of positive melanomas) for human melanomas (97 to 100%) and with slightly lower sensitivity (76%) for canine melanomas. 24,27 S100 has been the standard melanoma marker for many years, but its specificity is poor in human and animal tissues; numerous nonmelanocytic tumors and normal tissues express this marker, including but not limited to nerve sheath cells, myoepithelial cells, adipocytes, and leukocytes, thereby making it unsuitable as a sole test for the diagnosis of melanoma. 24,27
Melan A is a cytoplasmic protein recognized by cytotoxic T cells. 24 It is a specific melanocytic differentiation antigen (defined as percentage of Melan A–negative nonmelanocytic tumors examined; 95 to 100% in human melanomas) and highly sensitive (75 to 92%). 24 Melan A has been used for more than a decade in the diagnosis of melanomas from animals—particularly, dogs but also other species. 8,15,18,27,28 Its sensitivity in the dog is high (positive in more than 92% of melanomas), 27 but it is also expressed in nonmelanocytic steroid-producing tumors of the adrenal gland and gonads. 26
PNL2 is a monoclonal antibody originally produced against a subtype of human somastotatin receptor; its function in melanogenesis is unknown. 30 However, this monoclonal antibody recognizes a fixative-resistant melanocytic antigen, 6,30 the exact structure of which has yet to be identified. 24 In humans, PNL2 also labels angiomyolipomas and mature myeloid cells, including neutrophils but not other nonmelanocytic tumors, including various carcinomas and sarcomas. 6 The melanocytic antigen targeted by monoclonal PNL2 can still be detected after melanin bleaching or decalcification. 30 The sensitivity of PNL2 in human melanomas is comparable to that of Melan A, particularly in the epithelioid variants, but it is very low for desmoplastic melanomas. 6,30 In one study, 6 PNL2 detected metastatic melanomas in a higher percentage than did Melan A, although when compared with the primary tumor, the number of positive cells was somewhat lower. 30 In another study, PNL2 was less sensitive than Melan A or HMB-45, an antibody recognizing the premelanosomal glycoprotein 100. 22,24 Human mucosal melanomas appear to be more readily detected with antibodies to PNL2 than to Melan A. 21
Tyrosinase is a cytoplasmic melanocyte differentiation protein, with an essential role in the synthesis of melanin. 4,7,23 Tyrosinase is present in human cells and tumors of melanocytic lineage. 16 Tyrosinase sensitivity for human melanomas is very high (84 to 94%) but diminishes with increasing clinical stage and in metastatic lesions. 24 The most commonly used monoclonal antibody to tyrosinase (clone T311) is poorly reactive in desmoplastic and spindle cell variants of human melanomas. 5,16,24
The aims of this study were to (1) document the immunoreactivity of monoclonal antibody to melanocytic marker PNL2 and a monoclonal antibody to tyrosinase in canine melanomas; (2) compare the immunoreactivity with that of Melan A; (3) determine PNL2 cross-reactivity with nonmelanocytic tumors; (4) examine the effects of prolonged formalin fixation on the immunoreactivity of PNL2, Melan A, and tyrosinase antigens; (5) determine the effects of mild and strong decalcification on the immunohistochemical detection of Melan A, PNL2, and tyrosinase.
Methods
The Purdue University Animal Disease Diagnostic Laboratory database was searched for canine melanocytic neoplasms; 122 cases were selected, and histologic sections of each case were reviewed by one author (J.A.R.-V.) to confirm the diagnosis. The number of cases selected, based on the diagnosis and the presence of enough tissue for immunohistochemical procedures, was 101 (total number of samples, 113). The samples in this study were mostly from the oral cavity (n = 53) or the skin (n = 26). Fewer melanomas were in other organs, including bone (n = 7), heart (n = 1), eye (n = 8), lip (n = 9), lung (n = 2), and lymph node (n = 7). Nineteen specimens—mostly, melanomas from the foot or oral cavity—had been decalcified; in most instances, the exact type of decalcifying solution and duration of decalcification were unknown.
Three melanocytic markers were used (Table 1 ): mouse monoclonal antibody anti-melanoma marker, clone PNL2 (Santa Cruz Biotechnology, Santa Cruz, CA); mouse monoclonal antibody anti–Melan A, clone A 103 (Dako, Carpinteria, CA); mouse monoconal antibody anti-tyrosinase, clone SPM360 (Affinity Bioreagents, Golden, CO). Antibody PNL2 was diluted 1:150 and incubated for 2 hours; anti-Melan A antibody was diluted 1:160 and incubated for 90 minutes; and antibody to tyrosinase was diluted 1:25 and incubated for 2 hours. All reagent incubations were at room temperature. Heat-induced epitope retrieval with a decloaker was used for all 3 markers (citrate buffer [pH, 6.0] for PNL2 and tyrosinase; EDTA buffer [pH, 8.0] for Melan A). To avoid day-to-day variations in intensity of the reaction, all 3 markers were simultaneously tested for a given sample. A non-avidin-biotin immunoperoxidase-diaminobenzidine (DAB) detection method (EnVision+, Dako) was used for all markers. Optimization and validation of Melan A in canine tissues have been reported. 26,27 For PNL2 and tyrosinase, we optimized the immunohistochemical technique following a previously reported protocol. 25 In addition to these 3 melanocytic markers, one or more of the following markers were used to confirm or rule out a diagnosis of melanoma in specific cases: S100, vimentin, cytokeratins, CD18, MUM1. 25,29 Counterstaining with Azure B was done in cases with moderate to abundant melanin that was difficult to distinguish from DAB precipitate. Melanin stained green and DAB remained brown, although it darkened. Cells containing melanin and DAB stained dark brown to black. 17,27 Normal tissues and 120 nonmelanocytic tumors, including various carcinomas, sarcomas, leukocytic tumors, and steroid-producing tumors, were also evaluated with monoclonal antibody PNL2.
Antibody Reagents, Antigen Retrieval, and Detection Systems Used in Immunohistochemistry

a HIER, heat-induced epitope retrieval with decloaker, 20–24 psi, 125°C for 30 seconds.
When possible, the immunoreactivity was compared between nondecalcified and decalcified samples from the same tumor (10 cases). To further evaluate the effects of decalcification on immunoreactivity for the 3 melanocytic markers, formalin-fixed samples from 2 large melanomas were used. Five paraffin blocks were produced from each tumor; one was not exposed to decalcifying solution, and the other 4 were immersed in decalcifying solution (either formic acid or HCl solutions; US BioTex Corporation, Webbville, KY) for 1 day or 1 week.
The exact fixation time for each sample in this case series was unknown, although based on our experience, it ranged from 10 hours to 3 days. To determine possible effects of prolonged fixation on immunoreactivity, samples from one melanoma were fixed in 10% neutral buffered formalin for 1 day and 1, 2, 4, 6, 8, and 10 weeks. Paraffin sections from each sample were simultaneously reacted with the 3 melanocytic markers used in this study. 27
Immunoreactivity was semiquantitatively evaluated in a 4-grade scale (0 = < 5% positive cells; 1 = 5 to 10% positive cells; 2 = 11 to 50% positive cells; 3 = > 50% positive cells) modified from a previous study. 27 This grading system was used to compare reactivity among different melanocytic markers in the same tumor and the effects of decalcifying solutions and duration of formalin fixation.
Results
The melanocytic tumors in this study were clasified by the prevalent cell type. 13 A desmoplastic melanoma type was added to this classification, based on the tumor definition in human pathology. Desmoplastic melanoma is characterized by the presence of abundant stroma admixed with neoplastic cells. 12 The most common phenotype in this series was epithelioid/polygonal (47 samples), followed by mixed (32 samples) and spindle (24 samples). Less common subtypes included desmoplastic (7 samples) and balloon (3 samples). Of the 113 samples, 88 (77.7%) were melanotic melanomas and 25 (22.3%) were amelanotic. Twenty-three melanocytic tumors were considered benign; the rest were malignant on the basis of nuclear features and mitotic index or the presence of metastasis. 32
Immunoreactivity of Melanomas for Melan A, PNL2, and Tyrosinase
All 3 melanocytic antigens were located in the cytoplasm. For PNL2, immunoreactivity was usually granular and diffuse; for Melan A, granular and diffuse or polar; 27 for tyrosinase, diffuse and usually dull (Fig. 1 ). Immunoreactivity was strong (3+) in 77 (68%), 62 (55%), and 57 (50%) samples for PNL2, Melan A, and tyrosinase, respectively. Overall, the number of positive cells and intensity of reactivity was higher for PNL2 than for Melan A, although in some cases, Melan A reactivity was stronger than for PNL2. In most melanocytic tumors, the number of immunoreactive cells for PNL2 was similar and/or lower than that for tyrosinase; however, with few exceptions, intensity of reaction for tyrosinase was weaker than for PNL2 and, in some heavily pigmented neoplasms, difficult to distinguish from melanin, even after counterstaining with Azure B (Fig. 1C, inset). The number of samples positive for at least 1 of the 3 melanocytic markers was 111 (98%); the 2 negative melanomas were amelanotic, of spindle cell type, and S100 positive. Of 88 melanotic tumors, 85 (96.6%), 87 (98.8%), and 69 (78.4%) were positive for Melan A, PNL2, and tyrosinase, respectively; of 25 amelanotic melanomas, 16 (64%), 19 (75%), and 21 (84%) were positive for Melan A, PNL2, and tyrosinase, respectively.
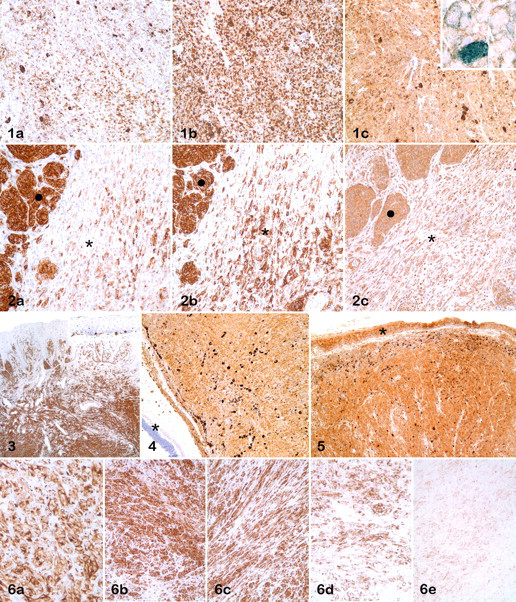
When immunohistochemical results were grouped by phenotype (Table 2 ), Melan A was positive in 41 (87%), 30 (94%), and 21 (88%) of epithelioid/polygonal, mixed, and spindle melanomas, respectively; PNL2 was positive in 45 (96%), 31 (97%), and 21 (88%) samples of epithelial/polygonal, mixed, and spindle melanomas, respectively. The most common positive phenotype for tyrosinase was epithelioid/polygonal, with 40 (85%) samples, followed by mixed (27 samples, 84%) and spindle (16 samples, 67%) cell types. The number of spindle cells positive for Melan A was, in many spindle or mixed melanomas, lower than that for PNL2; tyrosinase reactivity in spindle cells was higher than that for the other markers (Fig. 2 ). In 1 mixed melanoma, the spindle cell component was negative for all 3 melanocytic markers. In addition, in comparison of the number and/or intensity of labeling between the spindle and polygonal phenotypes of a given mixed melanoma, spindle cells were labeled more weakly than polygonal cells. Of 7 desmoplastic melanoma samples, 6 (86%) were positive for Melan A and PNL2 and 4 (57%) for tyrosinase. When the cell type of desmoplastic melanomas was considered, those with the polygonal type (2 samples) were always positive for all markers, whereas the spindle cell type was positive in 2 samples (66%) for Melan A and PNL2 and in 1 sample (33%) for tyrosinase. In most melanomas, there was no significant difference in the labeling for any of the 3 melanocytic markers based on the location of neoplastic cells (superficial or deep); in only 2 samples, cells in the deepest portion of the mass were nonreactive or less reactive than those near the surface (Fig. 3 ).
Immunohistochemical Results by Phenotype a
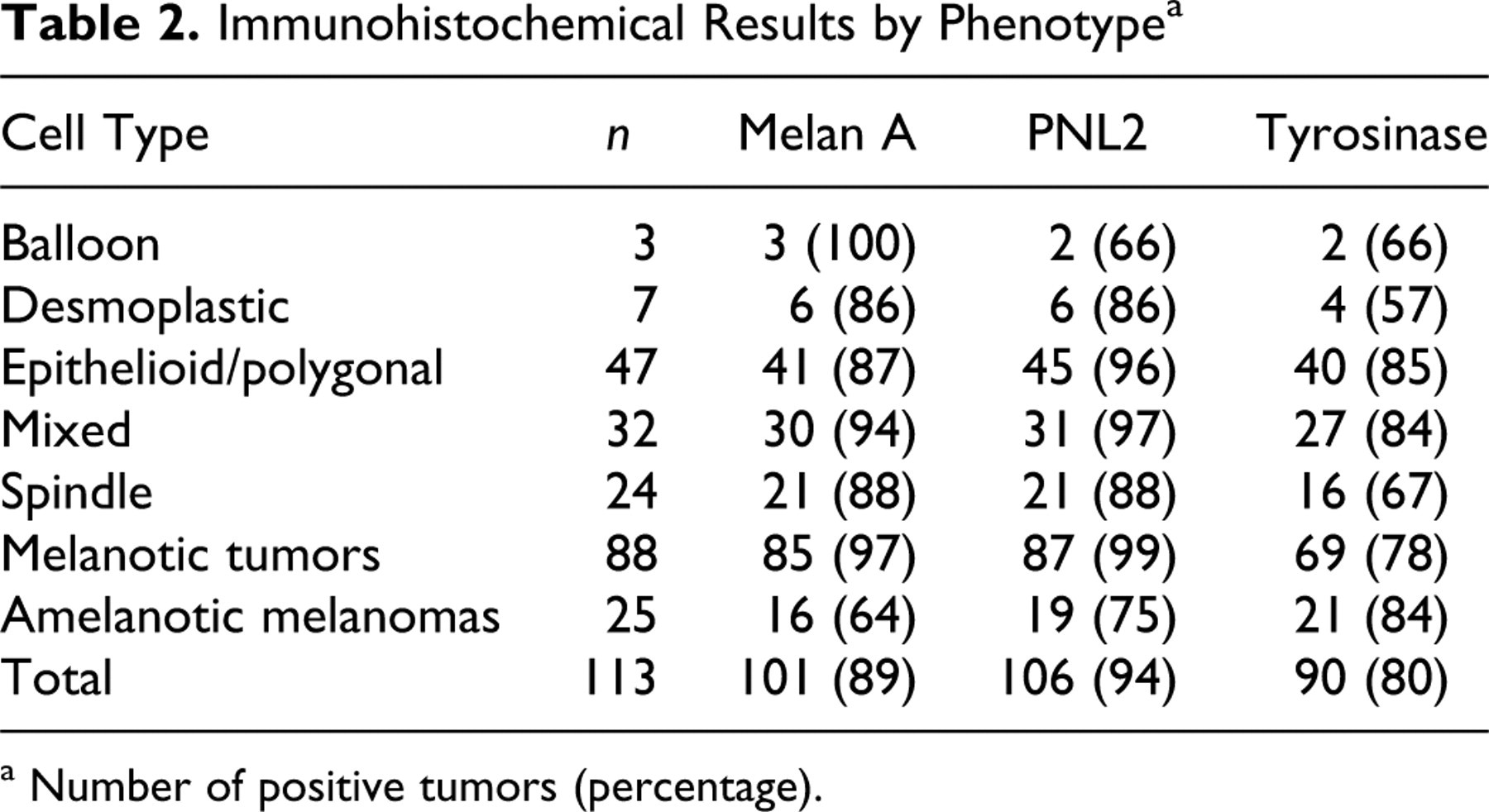
a Number of positive tumors (percentage).
Although the majority of tumors were from the skin or the oral cavity, there were no apparent differences in the immunoreactivity of examined markers based on organ affected (Fig. 4 ). Tyrosinase immunohistochemistry resulted in nonspecific background staining of the epidermis, adnexal epithelium, and mucosal epithelium. This background staining was sometimes as intense as the specific immunoreactivity of melanocytic tumors (Fig. 5 ).
Decalcification Effects on Immunoreactivity
The effects of decalcification on immunoreactivity was evaluated in 10 cases in which both decalcified and nondecalcified tumor samples were available. In 6 of 10 cases, there was reduced immunoreactivity in decalcified samples for Melan A and PNL2; of these 6 cases, 2 were negative for Melan A. For tyrosinase, 4 of 8 cases had mild reduction in immunoreactivity. Two samples of melanomas were used in a controlled experiment of decalcification effects. When formic acid was used as decalcifying solution, there were no negative effects in immunoreactivity for any of the 3 melanocytic markers, except for a slight decrease in reactivity for PNL2-labeled tissues in 1 sample decalcified for 1 week. There was major immunoreactivity reduction for Melan A and PNL2 in samples decalcified with HCl after 1 day; samples were nonreactive after 1 week of decalcification (both samples for Melan A and 1 sample for PNL2) (Fig. 6 ). Loss of immunoreactivity was mild to moderate for tyrosinase and observed after only 1 week of decalcification in HCl. In addition, the intensity for tyrosinase was higher in tissues decalcified with HCl for 1 day than in nondecalcified tissue from the same specimen.
Effects of Prolonged Fixation on Immunoreactivity
Slight to moderate Melan A immunoreactivity reduction (2+) was observed in tissues fixed for 1 month or longer and was more apparent (1+) at 10 weeks' fixation. Prolonged fixation (up to 10 weeks) did not affect immunoreactivity for PNL2. Tyrosinase immunoreactivity was slightly reduced in a melanoma fixed for 10 weeks.
PNL2 and Nonmelanocytic Neoplasms
None of the normal tissues (except for epidermal melanocytes and late spermatids of seminiferous tubules) or nonmelanocytic neoplasms (including steroid-producing tumors) reacted with antibody PNL2 (Table 3 ). These tumors were not examined with the other 2 melanocytic markers. Canine neutrophils were not labeled with PNL2 antibody.
PNL2 Immunohistochemistry in Nonmelanocytic Neoplasms

a Number of positive cases / total number of cases examined.
Discussion
In this study, antibody PNL2 was highly sensitive and specific for canine melanocytic neoplasms. Although PNL2 antibody has been successfully used for human and rodent melanocytic proliferations, 6,19,30 to our knowledge this is the first report of PNL2 immunohistochemistry in canine melanocytic neoplasms. The number of melanomas that were positive for PNL2 was equal to, or slightly higher than, that of Melan A for the different phenotypes. PNL2 reactivity is low or undetectable in human spindle and desmoplastic melanomas. 6,30 In our study, although spindle cell melanoma was less reactive to PNL2 than were other phenotypes, the number of positive cases for this phenotype was high (86%) and identical to that for Melan A. Other authors have not noticed a significant reduction of PNL2 reactivity in rodent spindle cell melanomas. 19,20
None of the nonmelanocytic tumors or normal tissues (except for normal melanocytes and late spermatids of the seminiferous tubules) reacted with antibody PNL2. Based on these results, the specificity of PNL2 for canine melanocytic tumors appears to be 100%. In contrast, Melan A is expressed in canine steroid-producing tumors. 26 No attempt was made to examine cross-reactivity of tyrosinase with nonmelanocytic neoplasms.
Melan A is one of the most commonly used melanocytic differentiation markers for human and canine melanomas. 16,27 As with other melanocytic markers, Melan A is poorly reactive with many human spindle or desmoplastic tumors. 16 Melan A sensitivity in this study was slightly lower (89%) than previously reported and lower than that for PNL2. 27 However, Melan A was positive in one sample that was negative for PNL2 and tyrosinase. Melan A reactivity for spindle and desmoplastic melanomas was identical to that for PNL2 and higher than that for tyrosinase.
The reported sensitivity of tyrosinase in human melanomas is comparable to that for other melanocytic markers (84 to 97%) but slightly lower in metastatic lesions and poorly reactive in desmoplastic and spindle melanomas. 4,5,9,11,14,16,22,24 A monoclonal antibody to tyrosinase commonly used in human studies (clone T311) has been used in canine melanomas with poor results owing to cross-reactivity with normal mucosal epithelium, epidermis, adnexal structures, and skeletal muscle. 27 In the current study, a different anti-tyrosinase monoclonal antibody was used, one reported by the manufacturer to be reactive on human, canine, and feline samples, and the number of positive melanomas was lower than that for Melan A or PNL2. In addition, tyrosinase immunoreactivity was usually less intense than that for the other markers and sometimes difficult to distinguish from background reaction or melanin, even after Azure B counterstain. Also, we experienced cross-reactivity issues similar to those previously reported. 27 In this study, the main advantage of using tyrosinase as a melanocytic marker was that decalcification with HCl apparently did not reduce its antigenicity, as opposed to that for the other 2 markers. Interestingly, decalcification, particularly with HCl, seemed to increase the intensity of the reaction for tyrosinase when compared with that of the nondecalcified samples. Moreover, the percentage of amelanotic melanomas positive for tyrosinase was higher than that for melanotic tumors (23 of 25, 92%) and higher than that for Melan A or PNL2 combined (20 of 25, 80%), which makes tyrosinase a valuable marker in amelanotic melanomas. However, the apparent background produced by the tyrosinase antibody will require evaluation of nonmelanocytic tumors with this marker to validate its use in melanocytic neoplasms. The reduced number of melanotic melanomas positive for tyrosinase might have resulted from difficulties evaluating the reactivity in heavily pigmented tumors.
The combined use of antibodies to Melan A, PNL2, and tyrosinase in this study provided a high sensitivity (98%) for melanocytic tumors. The combined use of Melan A and PNL2 slightly increased the sensitivity when compared with that of PNL2 alone (95% versus 94%). Based on the phenotype (spindle amelanotic) of the 2 neoplasms negative for these 3 markers and their positivity for S100, we cannot completely rule out other tumor types, including malignant peripheral nerve sheath tumor.
The antigen recognized by monoclonal antibody PNL2 is considered resistant to melanin bleaching with KMnO4 and nitric acid decalcification, although the duration of this treatment was not indicated; in the same study, Melan A immunoreactivity was abolished after these treaments. 30 We attempted to bleach several melanotic melanomas, but during the immunohistochemical procedure, most of the tissue was lifted off the slide or was severely damaged, precluding interpretation of results. Two different decalcifying solutions were used in this study—one solution considered to have mild decalcifying action with good preservation of morphology and hematoxylin and eosin staining (formic acid) and one solution with HCl considered to be a strong decalcifying agent with alterations in the hematoxylin and eosin staining. Results of the controlled experiment with decalcifying solutions indicate that the antigens recognized by all 3 melanocytic markers did not lose immunoreactivity after 1 day of treatment with formic acid, and only PNL2 had minor loss of immunoreactivity after 1 week of decalcification. However, when HCl was used, there was major loss of immunoreactivity for Melan A and PNL2 and complete loss after 1 week of decalcification. Immunoreactivity for tyrosinase was better preserved than it was for the other 2 antigens after decalcification with HCl. These results suggest that when stringent decalcification is used in a suspected melanoma, tyrosinase should be included in the diagnostic panel because it appears to be more resistant than Melan A or PNL2 to decalcification-induced antigenicity loss. Because decalcification with nitric acid is not a regular procedure in our laboratory, we did not examine its effects in this study.
In most samples, location of the tumor cells (superficial or deep) within the mass did not significantly affect the labeling intensity of any of the 3 melanocytic markers. Only in 2 cases were cells in the deep portion of the mass either not labeled or only weakly labeled. Immunoreactivity for antibody HMB45, commonly used to characterize human melanomas, is progressively lost with increasing depth from the tumor surface. 24
Prolonged fixation results in mild to moderate reduction in immunoreactivity for Melan A. 27 To our knowledge, the effects of prolonged fixation on PNL2 and tyrosinase immunoreactivity have not been reported. In this study, fixation up to 10 weeks did not affect the immunoreactivity for PNL2. Tyrosinase immunoreactivity was slightly affected after 8 weeks of fixation; Melan A immunoreactivity was slightly or moderately affected after 4 weeks of fixation. Historically, prolonged formalin fixation was believed to reduce tissue antigenicity owing to the formation of cross-links, 1 –3 altering or hiding epitopes and preventing immune recognition. 33 However, we evaluated 82 cellular and infectious disease antigens of various animal species and found that 93% of the antigens were adequately preserved after 7 weeks of fixation. 33,34 Results of the current study corroborate those findings and support the idea that for most antigens, with optimal antigen retrieval procedures, prolonged formalin fixation does not significantly alter immunoreactivity.
Given the results of this study, we conclude the following: First, immunohistochemistry on formalin-fixed, paraffin-embedded tissues with antibody to melanoma marker PNL2 is highly sensitive and specific for canine melanocytic neoplasms. Second, antibody PNL2 sensitivity is slightly superior to that of Melan A or tyrosinase. Third, decalcification with HCl markedly reduces immunoreactivity for Melan A and PNL2. Fourth, prolonged formalin fixation does not affect PNL2 immunoreactivity and only slightly reduces Melan A and tyrosinase immunoreactivities. Fifth, the combined use of antibodies to PNL2, Melan A, and tyrosinase provides almost 100% sensitivity for canine melanocytic neoplasms.
Footnotes
Acknowledgements
We appreciate the technical assistance of Dee DuSold and the Animal Disease Diagnostic Laboratory histology staff. Preliminary results from this study were presented as a poster in the 2009 annual meeting of the American College of Veterinary Pathologists (abstract No. 83).
The authors declared no potential conflicts of interests with respect to the authorship and/or publication of this article.
The authors received no financial support for the research and/or authorship of this article.
