Abstract
Definitive diagnosis of canine oral melanocytic neoplasms is often difficult because of variability in pigmentation and cellular pleomorphism. These neoplasms can resemble carcinomas, sarcomas, and round cell neoplasms, which differ in prognosis and treatment. A variety of immunohistochemical antibodies have been used for diagnosis of melanocytic neoplasms in humans and dogs; however, sensitivity and specificity of many markers have not been determined in amelanotic melanocytic neoplasms in dogs. The authors investigated a comprehensive panel of immunohistochemical markers in 49 canine oral amelanotic melanocytic neoplasms—namely, Melan-A, PNL2, HMB-45, microphthalmia transcription factor (MiTF), S-100, tyrosine hydroxylase, tyrosinase, tyrosinase-related proteins 1 and 2 (TRP-1 and TRP-2), and CD34. Ten well-differentiated cutaneous soft tissue spindle cell sarcomas were negative controls. Melan-A, PNL2, TRP-1, and TRP-2 were highly sensitive and 100% specific for the diagnosis of canine oral amelanotic melanocytic neoplasms. S-100 and MiTF showed high sensitivity but were less specific; that is, they also labeled a proportion of the soft tissue spindle cell sarcomas. HMB-45, tyrosinase, and tyrosine hydroxylase were 100% specific but had low sensitivities. CD34 did not label any of the melanocytic neoplasms but did label 80% of the soft tissue spindle cell sarcomas. A cost-effective and efficient immunodiagnostic cocktail containing antibodies against PNL2, Melan-A, TRP-1, and TRP-2 was created that had 100% specificity and 93.9% sensitivity in identifying canine oral amelanotic melanocytic neoplasms. The spindloid variant was the variant with the lowest sensitivity to the cocktail. The likelihood of correctly diagnosing canine oral amelanotic melanocytic neoplasms was dramatically higher when biopsy samples contained ample overlying and adjacent epithelium.
Melanocytic neoplasms are one of the most frequently diagnosed neoplasms of the canine oral cavity, and the majority of these are malignant. 2,31,35,36 Oral malignant melanomas grow rapidly, are locally invasive, frequently metastasize to regional lymph nodes and distant sites, and commonly recur following surgical resection. 2,31,35 Poorly pigmented or amelanotic melanocytic neoplasms at any site can be diagnostically challenging. Melanocytes arise from embryonic neuroectoderm and commonly differentiate into spindloid or epithelioid cells. 20,35 The cellular features of neoplastic melanocytes may therefore closely mimic those of poorly differentiated malignant neoplasms, including carcinomas, soft tissue sarcomas, and round cell neoplasms such as lymphomas. 15,20,27,31,35,39 The combined lack of pigmentation and the indistinct cellular morphology of many oral malignant melanomas in particular often make definitive diagnosis difficult, if not impossible, when based on microscopic examination alone. It is, however, of the utmost importance to accurately diagnose and differentiate canine oral melanocytic neoplasms from other poorly differentiated neoplasms, given that prognosis and therapy vary greatly between the differentials. In these instances, ancillary diagnostic tests, such as immunohistochemistry (IHC), are required to confirm the diagnosis.
Based on the current literature, S-100, Melan-A, HMB-45, microphthalmia transcription factor (MiTF), PNL2, tyrosinase, and tyrosinase-related proteins 1 and 2 (TRP-1 and TRP-2) have been used for the immunohistochemical detection of melanocytic neoplasms in humans. * S-100, a calcium-binding protein, is widely distributed in the central and peripheral nervous systems of vertebrates 26 and can be detected in a variety of normal tissues and cells, including melanocytes, cutaneous sweat glands, myoepithelial cells, chondrocytes, glial cells, Langerhans cells, lymphoid tissue, and neurons, as well as many neoplasms, including schwannomas and various soft tissue spindle cell sarcomas. 8,33,35 MiTF has been identified as a nuclear transcription factor protein necessary for embryonic development and postnatal viability of melanocytes; however, the specificity of this marker is controversial. 5,14,18,24,27 Other markers, such as Melan-A, HMB-45, TRP-1, TRP-2, tyrosinase, tyrosine hydroxylase, and PNL2, are supposedly more specific for melanocytes and melanocytic neoplasms. 6-8,25,28,29,40,41,43,44 Melan-A is a product of the MART-1 gene, which is recognized as an antigen on neoplastic melanocytes by autologous cytotoxic T lymphocytes. 20,27,31,35 Monoclonal antibodies against HMB-45 recognize the melanosomal glycoprotein gp-100, which specifically defines a premelanosomal antigen in fetal melanocytes and melanocytic neoplasms. 43 TRP-1 and TRP-2 are glycoproteins that play a critical role in melanogenesis, 8,28,29,40,41 and TRP-1 has been found to be largely restricted to the early unmelanized melanogenic compartment of melanocytes (melanosomes). 28 Tyrosinase catalyzes the conversion of tyrosine to levodopa (L-dopa), which provides the substrate for the production of the pigment melanin. 28 Tyrosine hydroxylase can be detected within human epidermal cells, specifically within the cytosol of melanosomes. 22 PNL2, an antigen of melanocytes and granulocytes for which the role is unknown, has recently been demonstrated as a useful marker for human melanocyte differentiation. 6,25
Only a few of the previously described antibodies—including S-100, Melan-A, HMB-45, tyrosinase, TRP-2, and, most recently, PNL2—have been evaluated for the diagnosis of canine melanocytic neoplasms. 8,12,20,31,33,35,37 The reported specificities and sensitivities of these antibodies, however, are highly variable. In 2 studies, approximately 90% of canine amelanotic “melanomas” were found to be positive for S-100. 8,20 Antibodies against Melan-A have been reported as being highly sensitive and specific for the detection of canine melanocytic neoplasms. 7,8,12,20,31,35 In one small canine study (n = 7), Cangul et al stated that Melan-A expression was detected in a variety of epithelioid and spindloid melanocytic neoplasms. 7 In another study, Melan-A was not expressed in any of the canine amelanotic neoplasms examined. 20 However, others have demonstrated Melan-A labeling in both pigmented and nonpigmented canine melanocytic neoplasms. 7,31 A recent study reported that Melan-A labeled a higher percentage of malignant melanomas (83%) than did benign melanocytic neoplasms (32%); 12 however, the authors did not describe the criteria used to classify these neoplasms, and no follow-up information or survival data were provided. Only one study has reported HMB-45 as a sensitive (88% sensitivity) and specific marker for detecting canine melanocytic neoplasms. 37 Expression of TRP-2 in canine melanocytic neoplasms was examined in only one study and found to be less sensitive but more specific than S-100 and more sensitive than Melan-A in identifying canine amelanotic melanomas. 8 Two studies indicated that antibodies against tyrosinase did not yield adequate labeling of canine melanocytic neoplasms. 7,31 PNL2 has been demonstrated as a marker for normal and neoplastic melanocytes in albino rats 21 and, most recently, as a fairly sensitive and specific marker for canine melanocytic neoplasms. 12 In a study population of 69 melanocytic neoplasms, 43 (62%) were positive for PNL2, and there was no evidence of labeling in a variety of normal and nonmelanocytic neoplastic canine tissues. 12 Other antibodies, such as MiTF, TRP-1, and tyrosine hydroxylase, have not been evaluated for the diagnosis of canine melanocytic neoplasms.
Based on the current literature, each antibody discussed above varies in regard to sensitivity and specificity for the detection of neoplastic melanocytes, thereby limiting the use of a single antibody for the diagnosis of amelanotic melanocytic neoplasms. Furthermore, IHC, as based on a panel of individual antibodies for the diagnosis of these neoplasms, is expensive and labor intensive. A combination of antibodies in a “cocktail” form that contains multiple highly specific and sensitive antibodies would allow pathologists to accurately diagnose amelanotic melanocytic neoplasms in a cost-effective and efficient manner. Therefore, the goals of this study were (1) to determine the sensitivity and specificity of S-100, Melan A, HMB-45, TRP-1, TRP-2, MiTF, PNL2, tyrosinase, and tyrosine hydroxylase IHC in canine oral amelanotic melanocytic neoplasms as compared to canine subcutaneous soft tissue spindle cell sarcomas and (2) to develop a diagnostic immunohistochemical melanocytic neoplasm cocktail using a combination of the most sensitive and specific melanocytic antibodies listed above.
Materials and Methods
Case Selection
All cases in the study were submitted as surgical biopsies to the Diagnostic Center for Population and Animal Health between January 1996 and February 2009. In total, 49 canine melanocytic neoplasms were selected for the study on the basis of the following criteria: Neoplasms had to be located within the oral cavity or on the lip; neoplastic cells had to be present in small nests in the overlying epithelium and/or exhibit junctional activity (defined as proliferation of neoplastic cells at the dermoepidermal junction); 13 neoplastic cells had to have a morphology suggestive of melanocytic origin (polygonal to round to spindloid cells with round to oval nuclei containing fine chromatin and usually 1 or more distinct nucleoli); 13,35 and neoplastic cells either had to lack melanin pigment or only contain minimal amounts of melanin pigment.
All tissues from these cases were fixed in 10% neutral buffered formalin for less than 48 hours, routinely processed, and embedded in paraffin wax. Five-μm sections were cut and stained with hematoxylin and eosin for microscopic examination. All neoplasms were classified microscopically, on the basis of their cellular morphology: epithelioid (n = 28), spindloid (n = 12), and mixed (n = 9). Ten canine pigmented melanocytic neoplasms (9 from the oral cavity or lip and 1 from the eyelid) were selected to verify antibody labeling. Ten canine subcutaneous soft tissue spindle cell sarcomas, defined by infiltrative spindloid cells arranged in streaming bundles and whorls, were selected as negative controls. Neoplastic cells of these neoplasms did not contain melanin pigment, and there was no evidence of intraepithelial nests of neoplastic cells or junctional activity, given that all neoplasms were located in the subcutis.
Immunohistochemistry
IHC for Melan-A, HMB-45, S-100, MiTF, tyrosinase, tyrosine hydroxylase, TRP-1 (Novocastra, Newcastle, UK), TRP-2 (Abcam Inc, Cambridge, MA), PNL2 (Dako, Carpinteria, CA), and CD34 (Santa Cruz Biotechnology, Santa Cruz, CA) was performed on 5-μm serial sections from all cases included in the study. IHC for smooth muscle actin (SMA), muscle-specific actin (MSA), desmin, and glial fibrillary acidic protein (GFAP) (all Dako, Carpinteria, CA) was performed on 5µm serial sections from all of the negative control cases. These antibodies, along with CD34, were used to further classify these neoplasms and further confirm that the soft tissue spindle cell sarcomas were indeed not of melanocytic origin. IHC was performed on a Bond autostainer automated system (Leica Microsystems, Bannockburn, IL), using a biotin-free, polymeric alkaline phosphatase-linked antibody detection system with fast red chromogen and hematoxylin counterstain (Bond Polymer AP Red Detection Kit, Leica Microsystems). For detailed information about each antibody, see Table 1 . The 10 pigmented melanocytic neoplasms were used to confirm positive labeling with each antibody. For negative controls, antibodies were replaced with homologous immune sera.
Concentrations and Retrieval for Antibodies
Immunohistochemical Assessment
Immunoreactivity was assessed by light microscopy for each antibody. Immunoreactivity for Melan-A, HMB-45, S-100, tyrosinase, tyrosine hydroxylase, TRP-1, TRP-2, and PNL2 were scored according to percentage of positive cells as follows: 0 = less than 5% positive cells, 1 = 5 to 10%, 2 = 11 to 50%, 3 = 51 to 80%, 4 = 81 to 100%. All neoplasms with a score of 2, 3, or 4 were considered positive for that antibody. Neoplasms with a score of 1 were considered positive if intraepithelial nests consisting of at least 5 clustered neoplastic cells labeled positively or if there were aggregates of at least 20 cells that labeled positively anywhere within the neoplasm. If only individual scattered positive cells were observed in neoplasms with a score of 1, the neoplasm was considered negative for that antibody. Immunoreactivity for MiTF, SMA, MSA, desmin, and GFAP was reported as positive if greater than 15% of the neoplastic cells showed positive labeling or as negative if less than 15% of neoplastic cells showed positive labeling. Immunoreactivity for CD34 was considered positive if greater than 30% of neoplastic cells showed positive labeling.
Sensitivity and Specificity of Individual Melanocytic Antibodies
The sensitivity of the 9 melanocytic antibodies to detect canine oral amelanotic melanocytic neoplasms was determined on the basis of the antibody’s ability to positively label the 49 selected cases. The histologic diagnosis of amelanotic melanocytic neoplasm, based on the criteria described above, was used as the gold standard for the diagnosis of melanocytic origin. The specificity of each antibody to accurately diagnose these neoplasms was determined on the basis of the absence of labeling of the 10 canine subcutaneous soft tissue spindle cell sarcomas (negative controls).
Development of Melanocytic Neoplasm Immunodiagnostic Cocktail
Antibodies that had 100% specificity for detecting melanocytic neoplasms, as compared to the soft tissue spindle cell sarcomas, and that had a sensitivity of at least 80% for detecting the amelanotic melanocytic neoplasms were selected for the development of a melanocytic neoplasm immunodiagnostic cocktail. To improve the overall sensitivity of the cocktail, additional antibodies with lower sensitivities were included if they positively labeled neoplasms that were negative for the other antibodies or positive for only one other antibody. Based on these criteria, antibodies against PNL2, Melan-A, TRP-1, and TRP-2 were selected for inclusion in the cocktail to achieve the highest combined sensitivity for detection of oral amelanotic melanocytic neoplasms while maintaining 100% specificity in this study population.
The immunoreactivity of each neoplasm labeled with this immunodiagnostic cocktail was assessed as described above for the individual melanocytic antibodies. In addition, the labeling pattern of the cocktail was compared to the labeling pattern of the individual antibodies. The sensitivity and specificity of detecting canine oral amelanotic melanocytic neoplasms were determined for the immunodiagnostic cocktail as described above for the individual melanocytic markers.
Results
Immunohistochemical Assessment of Individual Antibodies
All 10 pigmented melanocytic neoplasms were positive for PNL2, Melan-A, TRP-2, MiTF, S-100, and HMB-45. Greater than 50% of these neoplasms had at least 50% of neoplastic cells positive for Melan-A, PNL2, MiTF, and S-100. Seven of the 10 pigmented melanocytic neoplasms were positive for TRP-1, and labeling in these neoplasms ranged from 11 to 80%. Six pigmented neoplasms were positive for tyrosinase, and all had 50% or less positive labeling with this antibody. Only 2 of the 10 pigmented melanocytic neoplasms were positive for tyrosine hydroxylase, and both had 10% or less positive labeling. As explained previously, this set of neoplasms was used only to establish that each antibody did label at least a proportion of canine melanocytic neoplasms. Additionally, all 10 neoplasms were negative for CD34.
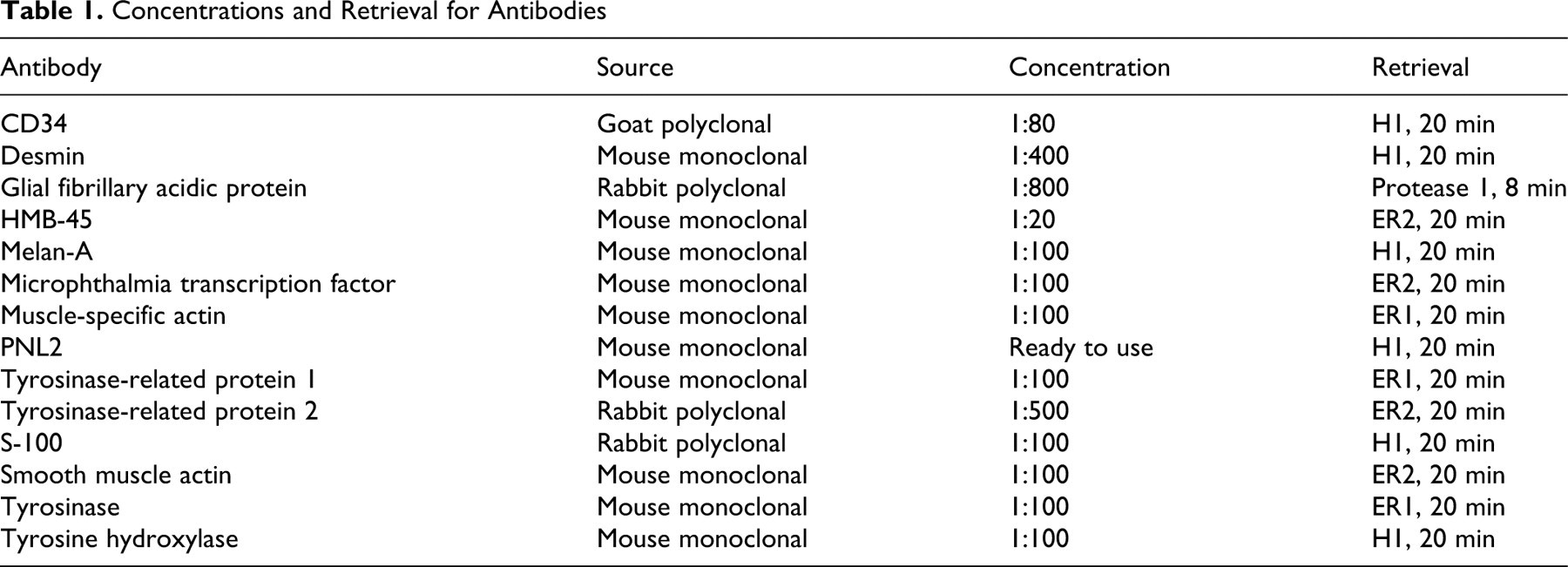
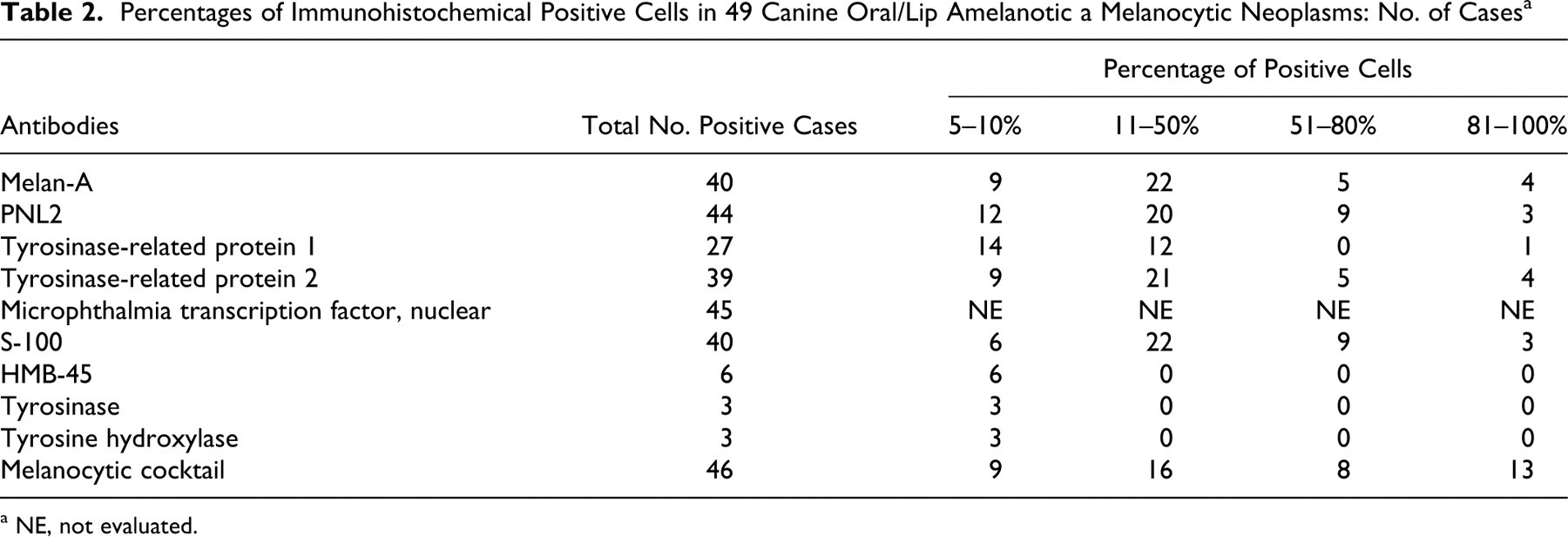
Table 2 shows the results of the immunohistochemical assessment of the 49 oral amelanotic melanocytic neoplasms. At least 48% of these neoplasms had more than 10% of neoplastic cells positive for Melan-A (Fig. 1a), PNL2 (Fig. 1b) TRP-1 (Fig. 1c), TRP-2 (Fig. 1d), and S-100. Thus, labeling with these markers was relatively easy to assess. Assessment of positive labeling for MiTF was also relatively easy; however, because neoplastic cells often showed either nuclear or cytoplasmic labeling or labeling of both, assessment of percentage of positive cells was difficult and not performed for all cases. All oral amelanotic melanocytic neoplasms that were scored as positive for HMB-45, tyrosine hydroxylase, and/or tyrosinase had less than 10% positive-labeled cells in those neoplasms. In general, IHC labeling was most commonly identified within nests of intraepithelial neoplastic cells, which were often located adjacent to the underlying mass (Fig. 2). Spindloid cell variant was the morphologic variant least likely to label with PNL2, Melan-A, or TRP-2. One of these 49 cases did not have enough tissue left to evaluate CD34 staining, but the remaining 48 neoplasms did not label for CD34.
Percentages of Immunohistochemical Positive Cells in 49 Canine Oral/Lip Amelanotic a Melanocytic Neoplasms: No. of Cases a
a NE, not evaluated.
Canine oral amelanotic melanocytic neoplasm. Immunohistochemistry demonstrated variable red cytoplasmic labeling of different areas for (a) Melan-A, (b) PNL2, (c) tyrosinase-related protein 1, (d) tyrosinase-related protein 2, and (e) the melanocytic neoplasm cocktail containing antibodies against Melan-A, PNL2, and tyrosinase-related proteins 1 and 2 in serial sections of a canine oral amelanotic melanocytic neoplasm. Immunohistochemical labeling with the melanocytic cocktail resulted in a higher overall percentage of positive neoplastic cells when compared with serial sections of the same tumor labeled with the individual antibodies. Immunohistochemistry using an alkaline phosphatase detection system with a vector red chromogen, counterstain, hematoxylin.
None of the 10 negative control cases (soft tissue spindle cell sarcomas) were positive for PNL2, Melan-A, HMB-45, tyrosine hydroxylase, tyrosinase, TRP-1, or TRP-2. Of the 10 neoplasms, 8 were positive for S-100 and 8 had positive nuclear labeling with MiTF. All 10 neoplasms showed positive cytoplasmic labeling for MSA, 9 for SMA, and 8 for CD34. None were positive for desmin or GFAP. These results support a muscle or perivascular wall origin for these neoplasms.
Sensitivity and Specificity of Individual Melanocytic Antibodies
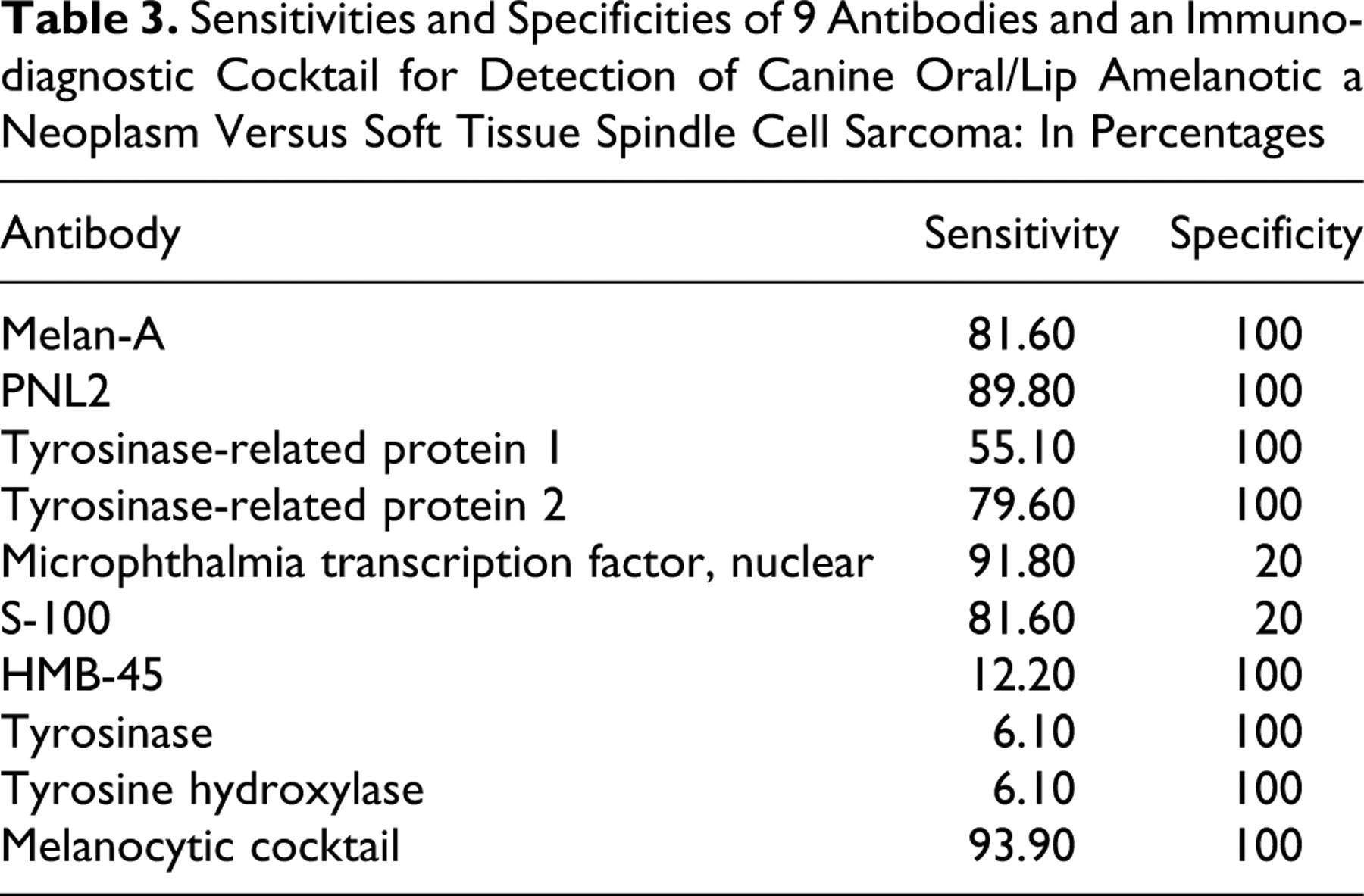
Table 3 shows the sensitivities and specificities of the individual antibodies in detecting amelanotic melanocytic neoplasms. Melan-A (40 of 49 = 81.6%), PNL2 (44 of 49 = 89.8%), TRP-1 (27 of 49 = 55.1%), and TRP-2 (39 of 49 = 79.6%) were highly sensitive and 100% specific for the detection of canine oral amelanotic melanocytic neoplasms. In contrast, S-100 and MiTF were highly sensitive but less specific (80% of the spindle cell sarcomas labeled with S-100; 80% had nuclear labeling with MiTF). HMB-45, tyrosinase, and tyrosine hydroxylase were 100% specific for the detection of canine oral amelanotic melanocytic neoplasms but had much lower sensitivities.
Sensitivities and Specificities of 9 Antibodies and an Immunodiagnostic Cocktail for Detection of Canine Oral/Lip Amelanotic a Neoplasm Versus Soft Tissue Spindle Cell Sarcoma: In Percentages
Melanocytic Neoplasm Immunodiagnostic Cocktail
All 10 pigmented melanocytic neoplasms had positive labeling with the immunodiagnostic cocktail that contained antibodies against Melan-A, PNL2, TRP-1, and TRP-2, and 9 of these neoplasms had 50% or greater positive labeling. None of the negative controls (soft tissue spindle cell sarcomas) had positive labeling with this immunodiagnostic cocktail.
Of the 49 amelanotic melanocytic neoplasms, 46 had positive cytoplasmic labeling with the immunodiagnostic cocktail (Fig. 1e). Of these 46 positive neoplasms, 37 (approximately 80%) were scored a 2, 3, or 4, indicating that at least 11% of the neoplastic cells were positive (Table 2). All 3 neoplasms that were negative with the immunodiagnostic cocktail were spindloid variants. The combination of Melan-A, PNL2, TRP-1, and TRP-2 in an immunodiagnostic cocktail resulted in 93.9% sensitivity and 100% specificity in this study population (Table 3) for the detection of canine oral amelanotic melanocytic neoplasms.
Discussion
Melan-A, PNL2, TRP-1, and TRP-2 were found to be highly sensitive and 100% specific for the detection of canine oral/lip amelanotic melanocytic neoplasms in this study population. S-100 and MiTF were found to be highly sensitive but less specific for the detection of melanocytic neoplasms, each labeling 8 of the 10 well-differentiated soft tissue spindle cell sarcomas. HMB-45, tyrosinase, and tyrosine hydroxylase showed 100% specificity for the detection of canine oral amelanotic melanocytic neoplasms but had low sensitivities. By combining Melan-A, PNL2, TRP-1, and TRP-2 into one cocktail, the overall sensitivity of this antibody cocktail (93.9%) for this study population was higher than that of any 1 antibody used alone. Furthermore, as shown in Table 2, the immunodiagnostic cocktail detected a larger number of positively labeled cells in many neoplasms when compared to the results based on individual antibody labeling, thereby making it easier for the pathologist to evaluate a slide for positive labeling. In addition, combining 4 antibodies into 1 cocktail dramatically decreases the overall cost and time of diagnostic testing as compared to running a panel of 4 individual antibodies.
A combination of location and microscopic diagnosis was used as the gold standard to classify the 49 neoplasms in this study as amelanotic melanocytic neoplasms. Additionally, all 49 neoplasms were negative for CD34, which further supports the diagnosis of melanocytic origin, as opposed to a soft tissue origin. This negative labeling was especially helpful for the spindloid variants. CD34, also known as the human hematopoietic progenitor cell antigen, is expressed by embryonic cells of the hematopoietic system and endothelial cells. 38,42 CD34 has been shown in human medicine to label various soft tissue neoplasms—including some peripheral nerve sheath tumors, neurofibromas, solitary fibrous tumors, dermatofibrosarcoma protuberans, leiomyomas, leiomyosarcomas, angiosarcomas, perivascular cell tumors, spindle cell lipomas, and epithelioid sarcomas, 4,9,38,42 among others—but it is generally not reported to label melanocytic neoplasms. 4,9 In veterinary medicine, CD34 has been shown to label a variety of canine perivascular wall tumors, including hemangiopericytomas, myopericytomas, angioleiomyomas, and angioleiomyosarcomas. 1,23 Granted, not all soft tissue sarcomas label with CD34; nonetheless, the negative labeling in these amelanotic melanocytic neoplasms supports our diagnosis. Unfortunately, there is no method available to unequivocally confirm the melanocytic origin of these neoplasms. Transmission electron microscopy could be used to confirm the presence of melanosomes in some neoplasms; however, in poorly differentiated amelanotic melanomas, melanosomes are often not identified with routine methods. 30,35 Intraepithelial nests and junctional activity are 2 commonly used criteria to identify melanocytic neoplasms, 35,36 and they were used to select our study population. These features are generally not present in soft tissue spindle cell sarcomas, which are one of the primary differentials for oral melanocytic neoplasms. Evaluation for intraepithelial nests or junctional activity does require intact epithelium within microscopic sections, which limits the use of these criteria in ulcerated neoplasms. Selection criteria for inclusion of cases in this study excluded suspected amelanotic oral melanocytic neoplasms that lacked appreciable epithelium.
Another important finding regarding intraepithelial nests of neoplastic cells was that IHC labeling was most consistently identified within these nests, which were often located superficial or adjacent to the underlying mass. Thus, labeling may not be identified in neoplasms that lack overlying or adjacent epithelium due to ulceration and/or sampling technique. Poorly differentiated oral melanocytic neoplasms that do not exhibit junctional activity or contain intraepithelial nests but are covered by intact epithelium may not be as likely to label with melanocytic specific antibodies or the immunodiagnostic cocktail. In the above 2 cases (lack of epithelium or no junctional activity and no intraepithelial nests), the sensitivity of the specific melanocytic markers and the immunodiagnostic cocktail would be expected to be lower than that in the current study population. However, in our experience, additional immunohistochemical testing has shown that most poorly differentiated oral neoplasms that do not exhibit junctional activity or contain intraepithelial nests are actually not of melanocytic origin, but instead most often are carcinomas or soft tissue sarcomas. To increase the likelihood of accurately differentiating amelanotic melanocytic neoplasms from spindle cell sarcomas by IHC, it is recommended that surgeons submit biopsy samples containing overlying epithelium along with some adjacent surface epithelium whenever possible.
Because soft tissue spindle cell sarcoma is one of the primary and most challenging differentials for oral melanocytic neoplasms—specifically, the spindloid variant of these neoplasms—10 soft tissue spindle cell sarcomas from the subcutis of dogs were chosen as negative controls. All 10 neoplasms were negative for the highly specific melanocytic markers Melan-A, HMB-45, PNL2, tyrosinase, TRP-1, TRP-2, and tyrosine hydroxylase and the melanocytic neoplasm immunodiagnostic cocktail. Additionally, 10 of 10 were positive for MSA, 9 for SMA, and 8 for CD34, which supports a muscle or perivascular wall origin for these neoplasms and further confirms that our negative controls were not of melanocytic origin. Positive S-100 and MiTF labeling of a large percentage of these soft tissue spindle cell sarcomas demonstrates the low diagnostic value of these antibodies for the diagnosis of amelanotic melanocytic neoplasms. Whereas the range of cells labeled by S-100 has been well documented, the specificity of MiTF has been highly controversial. In one study, IHC staining for MiTF was reported to have nearly 100% sensitivity for detection of human melanomas, including amelanotic melanomas, metastatic melanomas, spindloid variants of melanoma, and melanomas that failed to label with HMB-45 and S-100. 18 This study also suggested high specificity because there was no nuclear expression of MiTF in 60 nonmelanocytic neoplasms consisting of various types of carcinomas; however, 2 of these carcinomas showed cytoplasmic labeling, and soft tissue spindle cell sarcomas were not evaluated. 18 In other studies, MiTF expression was poor in desmoplastic melanomas but not significantly identified in nonmelanocytic neoplasms, which included some spindle cell sarcomas; thus, the authors suggested high specificity of this marker. 17,24 However, other studies have reported poor sensitivity and specificity and described MiTF expression in melanocytes of normal skin as well as various other cell types, including macrophages, lymphocytes, fibroblasts, Schwann cells, and smooth muscle cells, as well as the neoplasms derived from these cell types. 5,14 Angiomyolipomas, rare breast carcinomas, renal cell carcinomas, histiocytes, and mast cells have also been reported to express MiTF. 27 The low specificity of S-100 and MiTF antibodies in the current study must be remembered when evaluating labeling in undifferentiated neoplasms—especially, spindloid neoplasms. Also, labeling for MiTF in neoplasms with significant desmoplasia or in neoplasms that have been reexcised and contain significant fibroplasia must be interpreted with caution, given that it may be difficult to distinguish between positively labeled fibroblasts and spindloid neoplastic cells. Because MiTF is a nuclear transcription factor, only nuclear labeling has been reported for the diagnosis of melanomas in the current literature. 5,18,24,27 In the current study, we saw both nuclear and cytoplasmic labeling in many of the melanocytic neoplasms, as well as the soft tissue spindle cell sarcomas. The significance of this cytoplasmic labeling is unknown.
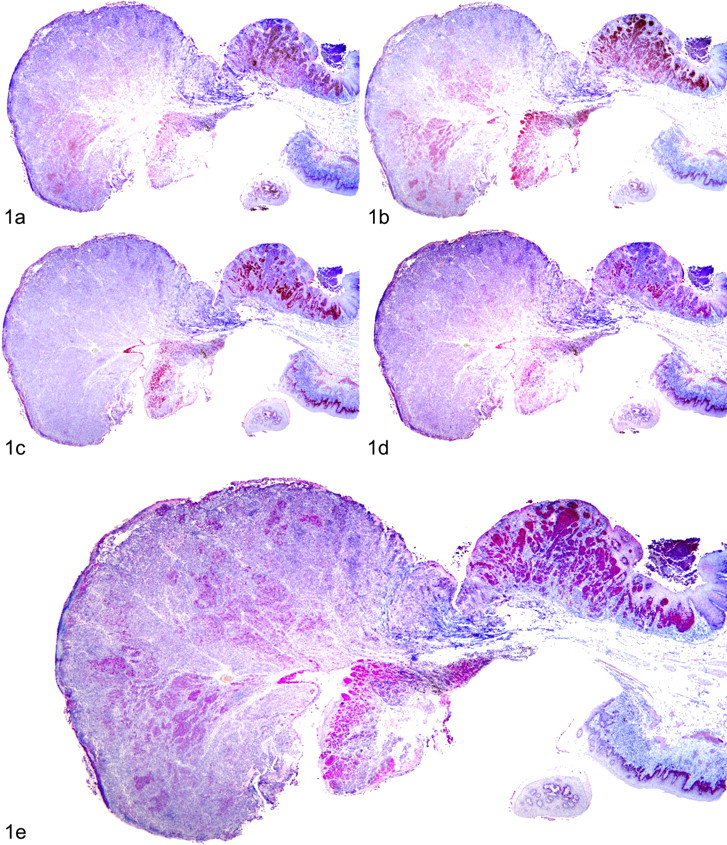
In humans, many of the melanocytic antibodies that have been reported to have high specificity in some studies (HMB-45, Melan-A, MiTF, tyrosinase, PNL2) have been reported to have poor sensitivity in detecting spindle cell and/or desmoplastic melanomas. 6,14,18,19,24,27,43 Similarly, in our study, the spindloid variant was the least likely variant to label with highly specific melanocytic antibodies, yet a significant number (6 of the 9 spindloid melanocytic neoplasms) did label with the immunodiagnostic melanocytic cocktail. The 3 amelanotic spindloid neoplasms that were negative with this cocktail did exhibit junctional activity (Fig. 3), and 1 of these 3 neoplasms contained intraepithelial nests, strongly supporting a melanocytic origin. Interestingly, all 3 neoplasms were positive for MiTF but negative for S-100, which has been thought to be a highly sensitive marker for even poorly differentiated melanomas. 20 Furthermore, these 3 neoplasms, along with all the melanocytic neoplasms in this study, were negative for CD34. This lack of CD34 labeling further supports a melanocytic, rather than soft tissue, origin of these spindloid neoplasms. Based on their location, microscopic features, and IHC labeling pattern, these 3 neoplasms are most likely of melanocytic origin but too undifferentiated to show positive labeling with specific melanocytic antibodies. Alternatively, these neoplasms may represent undifferentiated spindle cell sarcomas or some other type of undifferentiated neoplasm.
The age of the blocks did not affect the immunohistochemical labeling for any of the antibodies used. The labeling patterns of the oldest cases were similar to those of the more recent cases. In fact, 9 of the 10 oldest blocks all scored a 2 or more for Melan-A, PNL2, TRP-1, and TRP-2 IHC labeling.
In summary, an immunodiagnostic melanocytic neoplasm cocktail containing antibodies against PNL2, Melan-A, TRP-1, and TRP-2 is a cost-effective and efficient means of detecting canine oral/lip amelanotic melanocytic neoplasms, with high sensitivity and 100% specificity in relation to soft tissue spindle cell sarcomas. The likelihood of correctly diagnosing canine oral amelanotic melanocytic neoplasms dramatically increases when biopsy samples contain ample overlying and adjacent epithelium. Canine oral spindloid cell amelanotic melanocytic neoplasm is the least likely morphologic variant to label with common melanocytic markers.
Footnotes
Acknowledgements
We would like to thank Leica Microsystems, Inc, for its financial support and the Diagnostic Center for Population and Animal Health immunohistochemistry laboratory for its technical assistance.
Notes
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
Financial support to perform this project came from Leica Microsystems, Inc.
