Abstract
Oral focal epithelial hyperplasia is a rare and seldom reported disease in animals and humans induced by a papillomavirus. The present report is the first description of this disease in a Neotropical primate, a howler monkey (Alouatta fusca). The diagnosis was based on gross and microscopic findings. The generic papillomavirus antigen was identified by immunohistochemistry and was found not to be related to any human papillomavirus DNA tested by in situ hybridization. This virus is probably a specific papillomavirus of the howler monkey (HMPV).
Keywords
Focal epithelial hyperplasia (FEH) is a rare contagious disease caused by a papillomavirus (PV) that affects mainly Native Americans and Eskimos. FEH was first described in 1965 from observations of several soft sessile to papular nodular elevations of the buccal mucosa in Navajo, Chavante Indian, and Alaskan Eskimo children. 3 These lesions were painless and tended to disappear spontaneously. 3 Human papillomavirus (HPV) from FEH was first characterized in 1983 and designated HPV-13, which is related to HPV-6 and HPV-11. 13 Years later, another type of HPV obtained from FEH was identified and referred to as HPV-32, which was also related to HPV-6 and HPV-13. 4
Since 1965, FEH has been detected in human beings in several parts of the world and in animals, 9 including chimpanzees (Pan troglodytes), 1 10 16 18 pygmy chimpanzees (Pan paniscus), 19 20 and Asian lions (Panthera leo persica). 15 The present report is the first description of FEH in a howler monkey (Alouatta fusca), a Neotropical primate.
An adult male howler monkey captured in São Paulo was kept in captivity for 6 months with another young individual of the same species at the Technical Veterinary Medicine and Fauna Biology Division of Parks and Green Areas Department of São Paulo City, Brazil, for rehabilitation. There was no clinical evidence of FEH in the two monkeys during this period; the last physical examination was performed 8 days before the end of the captivity. The two animals were then transferred for 20 days to an adaptation area, where the monkeys were reintroduced in a remnant Neotropical forest in the São Paulo metropolitan area. About 10 days after the reintroduction, the adult howler monkey was found on the ground after being attacked by dogs and died minutes later.
A complete necropsy was performed, and samples of the lower lip and several organs were collected, fixed in 10% neutral buffered formalin, processed routinely with an automatic tissue processor, embedded in paraffin, cut into 3–5-μm sections, and stained with hematoxylin and eosin (HE) for light microscopy. Immunohistochemical evaluation of sections from the lower lip was done with a rabbit polyclonal antibody (cod. B508, 1/8000, Dako, USA) for the detection of papillomavirus group-specific epitopes and monoclonal anti-HPV type 6, 11, and 18 antibodies (clone 4C4/F10/H7/83, 1/40, Novocastra, USA) for detection of HPV antigens. The monoclonal antigens were recovered by heating with a microwave oven (10 mM citrate buffer, pH 6.0, twice for 10 minutes each time). The spretavidin–biotin–peroxidase complex method (cod. K0492, 1/300, Dako) was used with diaminobenzidine as chromogen and Harris' hematoxylin as a counterstain. Positive controls included a human cervical papilloma.
In situ hybridization (ISH) was performed with biotinylated probes of HPV types 6, 11, 16, 18, 30, 31, 33, 35, 45, 51 and 52 (cod. K0012, Dako) on the paraffin-embedded sections of the lower lip. The sections were deparaffinized in xylene, rehydrated in graded alcohols, and washed in twice-distilled water. The antigen was recovered by heating with a microwave oven (10 mM citrate buffer, pH 6.0, for 5 minutes each) and washing in twice-distilled water. Sections were treated with proteinase K (25 μg/ml in phosphate-buffered saline, pH 7.4) for 5 minutes at 37 C. DNA was denatured with the HPV DNA probe for 10 minutes at 95 C and chilled quickly on ice. Hybridization was performed overnight at 37 C in a moist chamber. Two posthybridization stringent washes were performed in 0.1× standard sodium citrate at 37 C for 15 minutes, and the catalyzed signal amplification system (CSA cod. K1500, 1/400, Dako) was used for hybridization detection with diaminobenzidine as chromogen and Harris' hematoxylin as a counterstain. Sections from paraffin-embedded HPV-infected human cervical cells were used as positive controls, and reaction with a negative DNA probe provided with the kit was used as a negative control.
Gross examination indicated that the howler monkey died of multiple dog bites, which produced extensive hemorrhage and pulmonary edema. In the oral cavity, there were multiple 2–5-mm-diameter well-circumscribed soft mucosal elevations of the lower lip (Fig. 1) and the anterolateral region of the tongue. The lesions were isolated or coalescent, and their color was whitish or indistinct from the adjacent normal mucosa. Other incidental findings were focal ulcerative colitis and pinworms in the large intestine.
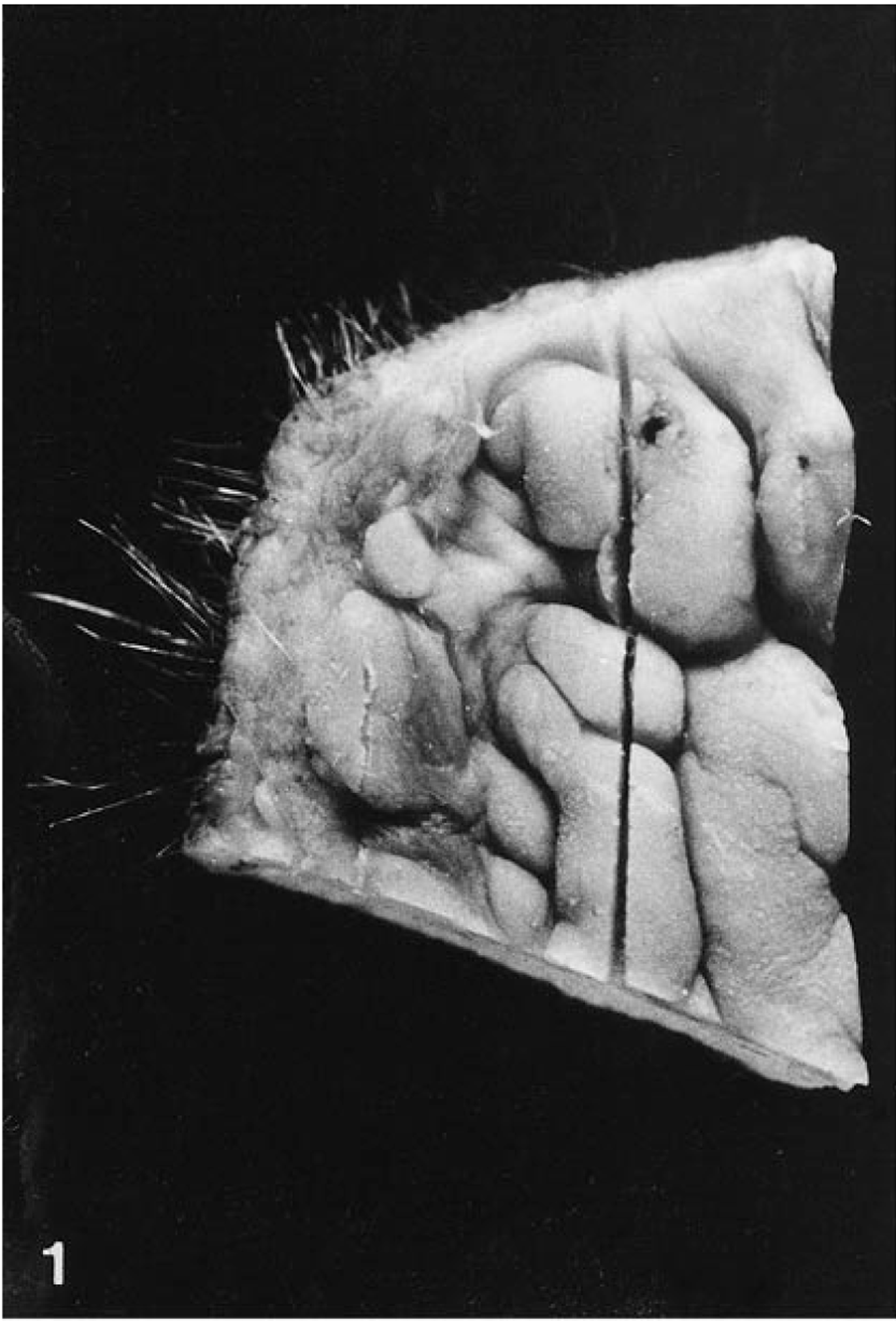
Lower lip; howler monkey. Elevated coalescing mucosal plaques.
Microscopically, the lower lip lesions consisted of marked hyperplasia of the prickle-cell layer, resulting in thickening, elongation, and fusion of the rete ridges and minimal parakeratosis (Fig. 2). The superficial keratinocytes were markedly vacuolized, with single or double nuclei centrally or eccentrically located, showing nuclear degeneration and marginated chromatin. These degenerating cells had features consistent with koilocytes (Fig. 3). Additional histologic findings were hyperplasia of the basal layer, numerous dyskeratotic cells, and coarse keratohyaline granules in the upper keratinocytes. Congested capillaries and minimal perivascular lymphocyte infiltration were present in the superficial stroma.
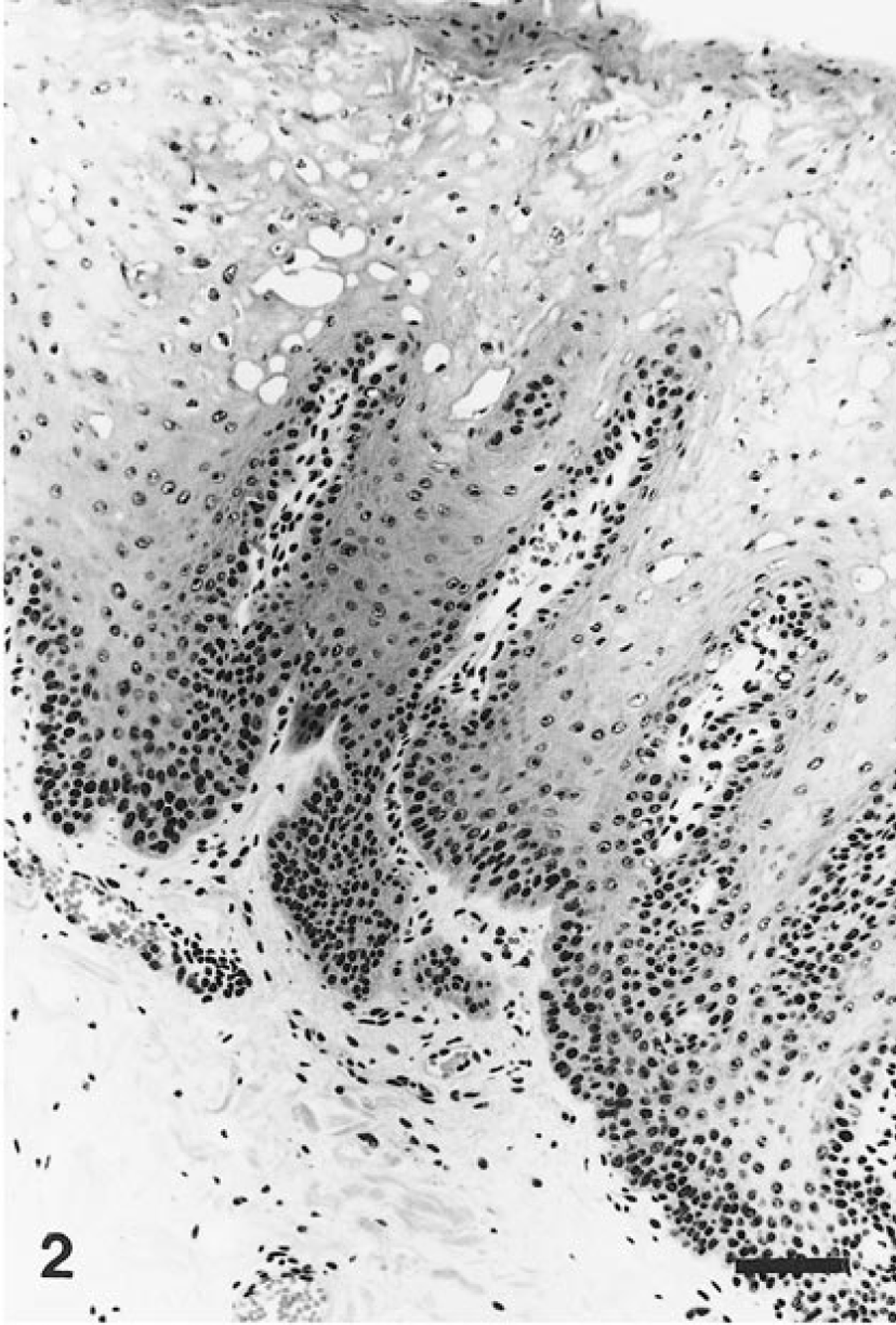
Lower lip; howler monkey. Hyperplasia of the prickle-cell layer, resulting in thickening, elogation, and fusion of the rete ridges. Note the numerous typical koilocytes, the minimal parakeratosis, and the hyperplasia of the basal layer. HE. Bar = 50 μm.
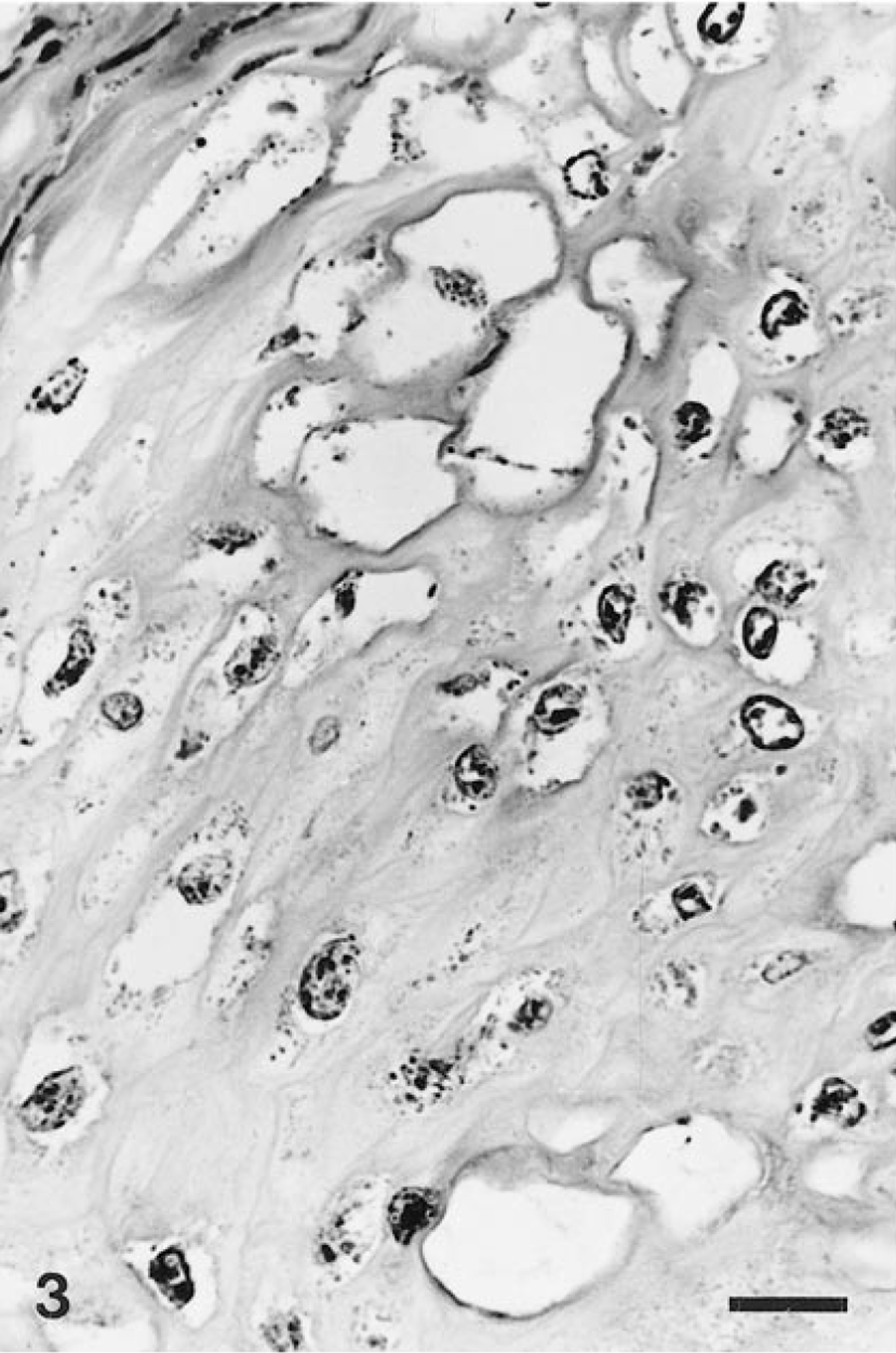
Lower lip; howler monkey. Prominent vacuolar changes of the keratinocytes, with central or eccentric single nuclei, showing nuclear degeneration (koilocytes). HE. Bar = 10 μm.
Positive signals for generic PV antigens were present only within the nuclei of the koilocytes (Fig. 4). The specific reactions for the detection of HPV-6, HPV-11, and HPV-18 capsid antigens were negative, and there was no cross-hybridization with any of the 11 HPV probes tested.
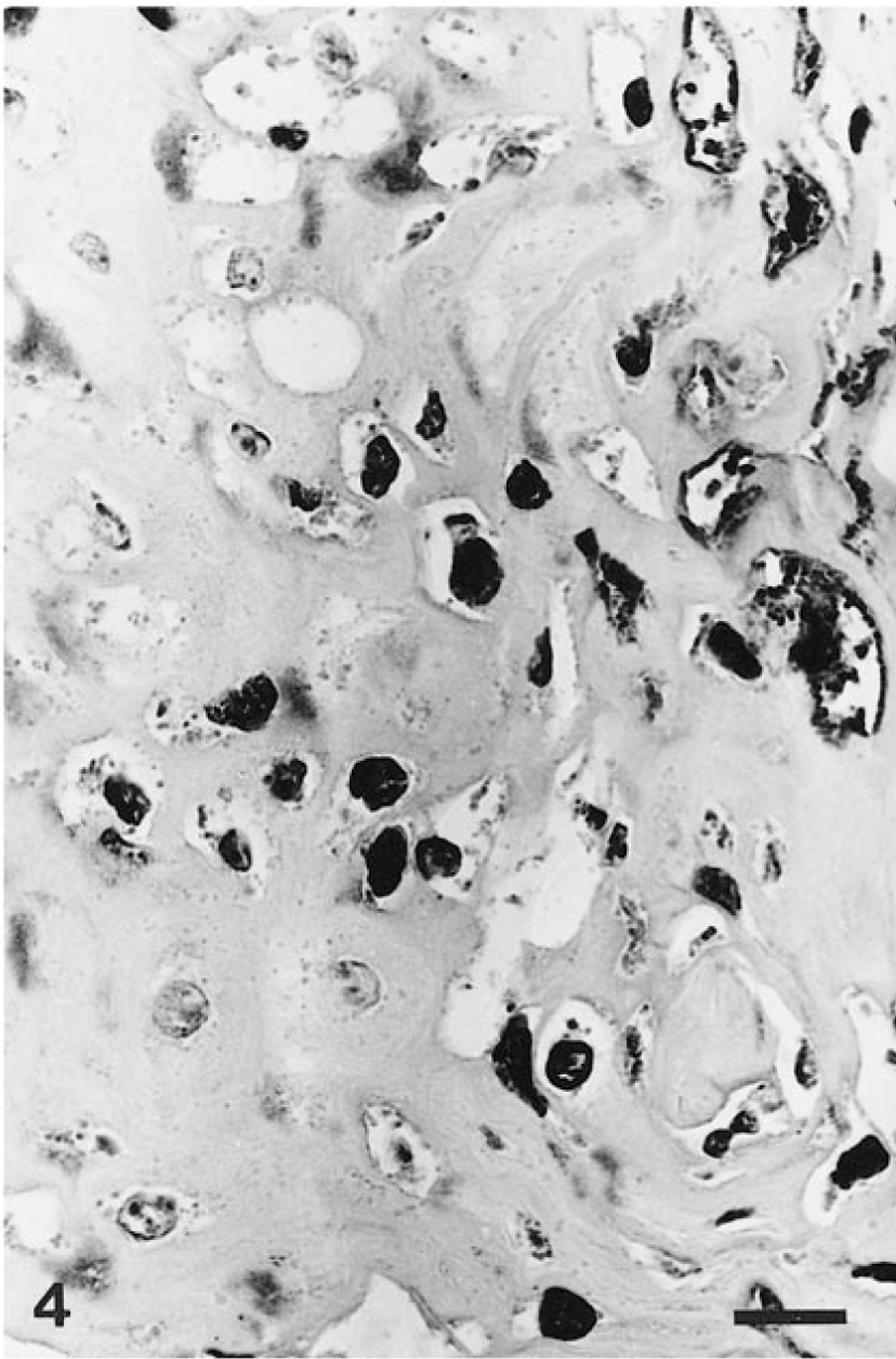
Lower lip; howler monkey. Numerous cells are positive for papillomavirus group-specific antigens. Positive staining was restricted to koilocyte nuclei. Streptavidin–biotin–peroxidase complex method, Harris' hematoxylin counterstain. Bar = 10 μm.
The gross and microscopic features found in this New World monkey were similar to those of oral focal epithelial hyperplasia PV-induced disease described in humans, 3 chimpanzees, 1 10 16 18 20 and Asian lions. 15
The negative ISH results eliminated the possibility of the howler monkey PV being closely related to any HPV types tested. This PV is probably not related to HPV-13 and pygmy chimpanzee PV-1 (PCPV-1) 9 13 19 20 because those two types showed partial homology with HPV-6 and HPV-11.
PVs are known to be transmitted by direct contact. 14 The origin of the contamination, however, could not be established in the present case. Immunosuppression is often associated with infections caused by PVs. 14 Although this animal was probably under the stress of poor adaptation to the environment, there was no evidence of lymphocyte depletion in the lymphoid tissues.
The absence of a significant inflammatory process in the lesions indicates that they were not regressing. 14 These data may favor the hypothesis that the infection and the appearance of the lesion occurred within 38 days.
PVs are a heterogeneous group of DNA viruses that cause benign or malignant epithelial proliferations in a wide variety of vertebrates. 14 All PVs have similar structure and genetic organization, which allow the study of molecular relationships and evolution among them and with their hosts and the biology of natural disease. 5 Lineages and supergroups of PVs have been identified. All genital and mucosal HPVs belong to supergroup A, including the rhesus monkey PV-1 (RhPV-1), colobus monkey PV-1 (CgPV-1), and PCPV-1. 7 Therefore, we suggest that this mucosotropic howler monkey papillomavirus (HMPV) belongs to supergroup A.
PVs are highly species specific, and there are few reports of productive lesions being caused by a PV from closely related species, as occurs in dogs and coyotes. 17 PVs and their hosts have a linked evolution, which implies that speciation events of the host lead to differentiation and isolation of PV populations. 6 Phylogenetic analysis has suggested that the ancestor of the modern PV of Old World monkeys and human beings already existed before the monkey–human branching event. 5 6 8
In view of the evolutionary distance between Old World primates (Catarrhini) and New World primates (Platyrrhini), 12 there may also be some distance among their PVs. We also know that the Catarrhini are more closely related to humans than are the Platyrrhini. 2 11 Thus, the HMPV is probably not closely related to HPV or to any other monkey PV described thus far.
Cloning and sequencing of DNA from this HMPV might elucidate its phylogenetic relationship to HPVs and other monkey PVs and its association with oral FEH.
Footnotes
Acknowledgements
We thank the veterinary and biological staff from the Divisão Técnica de Medicina Veterinária e Biologia da Fauna, Departamento de Parques e Áreas Verdes da Prefeitura de São Paulo, for submitting the clinical case. We also thank Dr. Idércio Luiz Sinhorini, Shirlei Meire da Silva, and Marco Aurélio Gattamorta for assistance with the photomicrographs. This work was supported financially by FAPESP with grants 95/3621-6, and the study was part of the master project of L. R. M. de Sá, who was supported by FAPESP grant 97/4815-4.
