Abstract
Papillomaviruses (PV) are associated with benign mucosal and cutaneous epithelial proliferations. In dogs, PV-associated pigmented plaques and papillomas can undergo malignant transformation, but this is rare, and most cases of canine squamous cell carcinoma do not arise from PV-induced precursor lesions. We describe herein the progression of pigmented plaques to invasive and metastatic squamous cell carcinoma associated with 2 canine papillomaviruses (CPV) in 2 related Basenji dogs. Immunohistochemistry for PV antigen revealed strong nuclear immunoreactivity within keratinocytes from pigmented plaques from both dogs, consistent with a productive viral infection. Polymerase chain reaction (PCR) using degenerate primers for the L1 gene revealed PV DNA sequences from 2 different CPVs. In situ hybridization for CPV revealed strong hybridization signals within the pigmented plaques and neoplastic squamous epithelial cells from both dogs. We report here progression of PV-associated pigmented plaques to metastatic squamous cell carcinoma within 2 Basenji dogs associated with 2 different CPVs.
Papillomaviruses (PV) are DNA viruses associated with various epithelial proliferations. In humans, certain mucosal PVs are causally associated with cervical cancer and a subset of head and neck cancers. 10 The causal association between cutaneous PVs and skin cancer is less established. However, patients with the genetic disease epidermodysplasia verruciformis (EV) are at increased risk for PV-associated skin cancers, suggesting that PVs may play a role in carcinogenesis. 1 In dogs, PV-induced pigmented plaques and papillomas can undergo malignant transformation, but this is rare, and most cases of squamous cell carcinoma (SCC) do not arise from PV-induced precursor lesions. 8 One exception includes research dogs with a mutation within the common γ-chain. These dogs develop papillomas that progress to metastatic SCC associated with canine papillomavirus (CPV)-2. 4 Apart from these research dogs, there have been no reports of dogs developing metastatic SCC from PV-induced precursor lesions. The present study describes 2 related Basenji dogs that developed metastatic SCC arising from cutaneous pigmented plaques associated with 2 different CPVs.
Dog No. 1 was a 9-year-old Basenji dog that developed over 30 verrucous epithelial lesions on all 4 limbs, head and face, neck, and body. The dog had been diagnosed with intestinal lymphoma 2 years prior and was currently in remission and receiving monthly administration of asparaginase and vinblastine. He was otherwise in good health with normal complete blood count and chemistry panels, which were performed throughout his therapy. Skin biopsies were obtained, fixed in 10% buffered formalin, and submitted for routine processing. Histologically, the lesions were consistent with viral pigmented plaques, characterized by epithelial hyperplasia, orthokeratotic hyperkeratosis, and increased keratohyalin granules (Supplemental Fig. 1). Within the center of 2 plaques, there was increasing dysplasia progressing to in situ SCC; within 2 lesions, there was progression to invasive SCC.
The patient was weaned off chemotherapy over 2 months, but within 2 years, the skin lesions progressed, and the dog developed enlarged draining peripheral lymph nodes and intrathoracic masses. Biopsy samples of skin revealed additional plaques with progression to in situ and invasive SCC (Supplemental Fig. 2). In total, there were 4 different plaques biopsied on 3 separate occasions that had progressed to SCC, often with the characteristic viral plaque flanking the margins of SCC. Due to decreased quality of life, the dog was euthanized, and a limited necropsy performed to obtain samples of skin, enlarged draining lymph nodes, and lung. Neoplastic islands of squamous epithelial cells effaced portions of the lymph node (Supplemental Fig. 3) and portions of the pulmonary parenchyma (Supplemental Fig. 4).
Dog No. 2, a 13-year-old Basenji dog and the biological mother of dog No. 1, presented with similar cutaneous verrucae approximately 2 months following development of initial clinical signs in dog No. 1. Biopsies of several masses revealed lesions consistent with pigmented plaques, similar to those described for dog No. 1. Three of the pigmented plaques had regions of increased dysplasia and SCC in situ; in the center of 1 plaque, there was progression to invasive SCC. Within a year, the dog developed subcutaneous nodules at 1 of the biopsy sites and enlarged draining lymph nodes. Biopsies of the subcutaneous nodules and draining lymph nodes were consistent with SCC. The dog was euthanized 2.5 years after development of cutaneous plaques.
Immunohistochemistry (IHC) was performed on formalin-fixed paraffin-embedded (FFPE) tissue samples from pigmented plaques from both dogs using an anti-bovine papillomavirus antibody as previously described. 7 A non-reacting antibody was used as the negative control. Strong nuclear immunoreactivity was evident within small numbers of keratinocytes in the spinous layer of the hyperplastic epithelium, often evident along the junction with normal skin in both dogs. In 3 biopsies from dog No. 1, immunoreactive keratinocytes were detected along the hyperplastic margins (plaque) immediately adjacent to areas of epithelial dysplasia and SCC (Supplemental Figs. 5a and 5b).
To further evaluate which specific CPVs were present, polymerase chain reaction (PCR) was performed using extracted genomic DNA from FFPE samples. The degenerate primers CanPV/FAP64 were used to amplify an approximately 400 base-pair (bp) region of the L1 gene as previously described. 5 Amplicons were purified, sequenced, and aligned to known papillomavirus sequences in GenBank as previously described. 7 The PV DNA sequence obtained from multiple lesions from dog No. 1 was 99% identical to CPV12 9 over 315-319 bp. The PV DNA sequence from a pigmented plaque from dog No. 2 was 99% identical to CPV12 over 315 bp; the PV DNA sequence from additional pigmented plaques revealed a putatively novel PV over 314 bp. Viral genome sequencing was performed on samples from dog No. 2, and the viral genome, designated CPV16, is published as a Genome Announcement and deposited in GenBank, accession number KP099966. 6 Identification of the CPV16 viral genome enabled us to generate specific primers and hybridization probes to distinguish between CPV12 and CPV16 infection.
PCR using specific primers for the E6/E7 oncogenes for CPV12 and CPV16 was performed on samples of pigmented plaques and SCC from both dogs. All samples from dog No. 2 were positive and all samples from dog No. 1 were negative using the CPV16-specific primers (Supplemental Fig. 6). Using the CPV12 primers, the pigmented plaques from dog No. 2 were positive while the SCC samples were negative; all samples from dog No. 1 except the lung were positive using the CPV12-specific primers (Supplemental Fig. 7).
Since CPVs can be amplified from normal skin, 5 we wanted to determine if the PV DNA identified with PCR was present within neoplastic cells or represented a surface contaminant. Colorimetric in situ hybridization (ISH) was performed on FFPE sections using the highly sensitive RNAscope ISH method (Advanced Cellular Diagnostics [ACD], Inc, Hayward, CA). Twenty probe pairs spanning the E6 and E7 genes for both CPV12 9 (Genbank JQ754321) and CPV16 6 (Genbank KP099966) were designed and synthesized (ACD). Control probes included a predesigned reference gene (Cl-Porlr2a) and a bacterial gene DapB (negative control) (ACD). In situ hybridization was performed following the manufacturer’s recommended protocol. Evans et al 3 has characterized the in situ hybridization signal patterns in human cervical biopsies using the RNAscope method and probes for HPV E6/E7. Intense diffuse nuclear signals represented detection of both viral DNA as well as viral mRNA; dot-like nuclear and cytoplasmic hybridization signals represented viral mRNA. 3
Pigmented plaques from dog No. 1 revealed strong diffuse nuclear hybridization signals within keratinocytes in the upper epithelial layers and dot-like cytoplasmic and nuclear hybridization signals within the basal and suprabasal keratinocytes using the CPV12 probe; signal was limited to the pigmented plaque, and adjacent normal epithelium was negative. Strong diffuse nuclear hybridization signals were noted within keratinocytes in the pigmented plaques using the CPV16 probe. Given the negative PCR result for these samples from dog No. 1 and the similar sequence identity (78%) between the E7 genes of CPV12 and CPV16, the CPV16 probe likely hybridizes with CPV12 nucleic acid that is abundant during productive infection. No hybridization signal was noted using the negative control probe.
Sections of invasive SCC (Fig. 1a) and metastatic islands within lymph nodes and lungs (Supplemental Fig. 8a) from Dog No. 1 revealed strong dot-like cytoplasmic and nuclear hybridization signals within tumor cells using the CPV12 probe. The probe for CPV16 revealed extremely faint cytoplasmic and nuclear hybridization signals (Fig. 1b and Supplemental Fig. 8b). Surrounding normal tissues did not react with any probe. No hybridization signal was noted using the negative control probe.
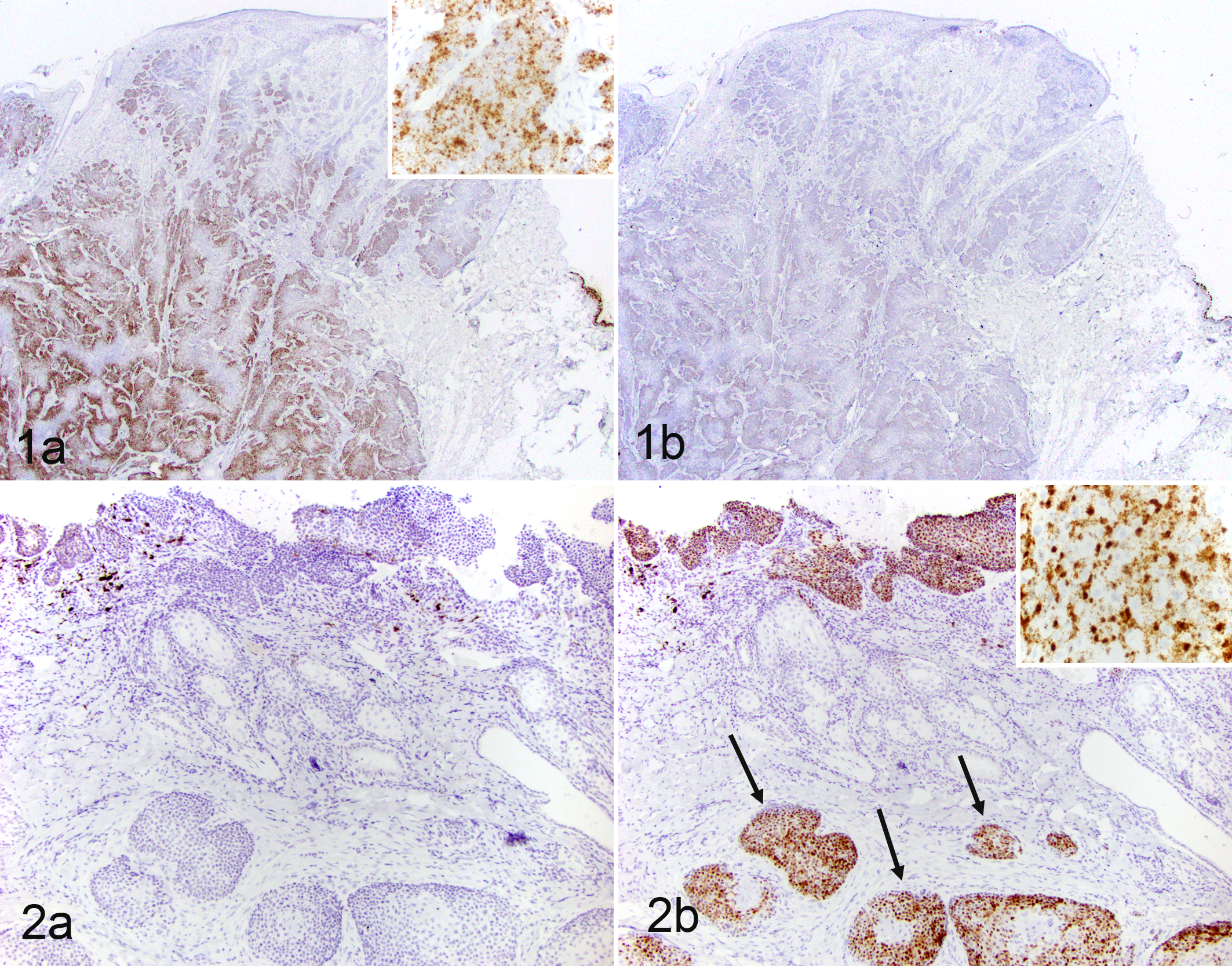
Multiple pigmented plaques from dog No. 2 revealed strong diffuse nuclear and dot-like cytoplasmic hybridization signals within the hyperplastic keratinocytes using the CPV12 probe. Other pigmented plaques from dog No. 2, sections of invasive SCC (Fig. 2b), and islands of metastatic SCC within lymph nodes (Supplemental Fig. 9b) revealed dot-like nuclear and cytoplasmic hybridization signals using the CPV16 probe. No hybridization signal was noted using the probe for CPV12 (Fig. 2a and Supplemental Fig. 9a) on sections of SCC, and no hybridization signal was noted using the negative control probe.
The ISH clearly identified PV nucleic acid within neoplastic cells in both dogs, supporting a causative role for CPV in carcinogenesis. Additionally, the ISH demonstrated that the SCC most likely arose from PV-induced lesions associated with 2 different CPVs.
We have described in this report malignant transformation and progression of pigmented plaques associated with CPV12 in dog No. 1 and CPV16 in dog No. 2. In human cervical cancer, progression to malignancy is associated with overexpression of the oncogenes E6 and E7 and dysregulation of tumor suppressor genes. 2 However, since only a small subset of patients develop cancer in association with PV infections, 2 it is likely that host, viral, as well as other external factors contribute to cancer development. Immunosuppression is one factor that can predispose individuals to PV infections, as seen in patients on immunosuppressive therapies or patients with certain genetically based diseases. 1,2
The 2 dogs in this report were closely related, and an underlying genetic predisposition is possible. Currently, there is no reported predisposition for Basenji dogs to develop PV infections. This may reflect under-recognition in this somewhat uncommon breed or indicate a non-Basenji breed–associated genetic predisposition within these 2 dogs. An underlying immunosuppression is also possible. However, while dog No. 1 was receiving chemotherapy at the time of initial diagnosis, the treatment was stopped after 2 months, and dog No. 2 was not receiving any immunosuppressive drugs. Immunological functional assays, though, were not performed on either dog, and an underlying immunosuppression cannot be ruled out.
In addition to host factors, there are likely viral factors associated with risk for malignant transformation. In human EV patients, infections with specific PV types are associated with progression to cancer. 1,2 Since this is the only dog reported with CPV16 infection 6 and CPV12 has only been described in one other dog, 9 further studies are necessary to determine if these viruses are casually associated with some cases of SCC in our canine population.
In conclusion, we described malignant transformation and progression of PV-associated pigmented plaques to metastatic SCC associated with 2 different CPVs in 2 related Basenji dogs. While these results are suggestive of a causal association between CPV infection and malignant transformation in these dogs, it is certainly not conclusive. Further evidence regarding the transforming abilities of the viral oncogenes would be necessary to yield more conclusive evidence for a causal association.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by the Bernice Barbour Foundation and research funds provided by North Carolina State University, College of Veterinary Medicine.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
