Abstract
The relationship between a retroviral infection and the development of nonviral intracytoplasmic inclusion bodies was studied in a Boa constrictor model. Twelve juvenile age- and size-matched inclusion body disease (IBD)-negative boas were randomly divided into three groups. Each group was inoculated intraperitoneally with 1 ml of an IBD virus (IBDV)-infected liver homogenate or 1 ml of normal boa liver homogenate (sham-inoculated control) or was left untreated. All boas were monitored for development of IBD by daily examination and serial liver biopsy over 1 year. The 4 IBDV-inoculated boas became IBDV and inclusion positive by 10 weeks postinoculation. The average size and density of inclusion bodies increased with the duration of infection. Ultrastructurally, inclusion bodies, 2 mm in diameter consisted of intracytoplasmic aggregates of granular electron-dense material that were not membrane limited. Larger inclusions (3–6 μm in diameter) were characterized as membrane-bound aggregates of amorphous to granular electron-dense material admixed with membranelike fragments. The sham-inoculated and untreated control snakes did not become inclusion or IBDV positive. Direct comparison of the protein electrophoretograms of IBDV-infected and normal boa tissues demonstrated a prominent 68-kd protein band unique to infected inclusion-positive tissues. Monoclonal antibodies directed against the 68-kd protein band specifically labeled inclusion bodies. The results of this study demonstrate that IBD inclusions represent an intracytoplasmic accumulation of an antigenically distinct IBDV-associated protein.
A disease entity named for its association with intracytoplasmic inclusion bodies of various sizes within the epithelial cells of visceral organs and neurons has been recognized in snakes of the family Boidae since the 1970s. 25 Clinical signs are variable and include chronic regurgitation, neurologic disease characterized by star gazing, dysequilibrium, tremors, disorientation, and paresis, increased susceptibility to secondary infections, and neoplasia. 25 In 1994, a C-type retrovirus-like agent that buds from both the endoplasmic reticulum and plasma membrane was identified as the etiologic agent. 25 Experimental transmission studies have shown that inclusion body disease (IBD) can be effectively transmitted by intraperitoneal and subcutaneous injection of fluids containing mature viral particles. 25 Very little is known about the natural history of the virus and how IBD is transmitted in nature. 25 It has been speculated that the blood-feeding macronyssid mite Ophionyssus natricis may be involved in the transmission cycle, but vector incrimination studies are lacking. 25 The inclusions after which the disease was named were initially described as non-membrane-bound aggregates of granular electron-dense material without further characterization. 25 The morphology of the described inclusion bodies most closely resembles that described for proteinaceous nonviral inclusions. 7 9 11 15 20 23
Nonviral inclusions characterized as amorphous intracytoplasmic eosinophilic hyaline bodies have been demonstrated in human and animal tissues afflicted with a diverse array of disease conditions, including alcoholism, neoplasia, morbid obesity, Indian childhood cirrhosis, pyrrolizidine alkaloid and copper exposure, and prolonged griseofulvin treatment. 2 7–9, 11 15 26 These inclusion bodies represent intracytoplasmic aggregates of various intermediate filaments, including cytokeratins, vimentin, and neurofilaments. 8 10 11 20 In some cases, the intermediate filaments within non-membrane-bound inclusions were thickened by a diffuse coating of ubiqutin. 18 20
Nonviral inclusions consisting of unidentified granular to fibrillar electron-dense material have been demonstrated in cells infected with several retroviruses, including avian leukosis virus, visna virus, human immunodeficency virus (HIV), and simian immunodeficiency virus (SIV). 1 6 12 21 32 The ultrastructural morphology of retrovirus-associated inclusions are variable. One variant known as tubuloreticular inclusions was described as fine tubular meshworks of lipid-rich membranes complexed with unidentified proteins that distended the endoplasmic reticulum. 13 21 Other retrovirus-associated inclusions were characterized as basophilic intracytoplasmic bodies of various sizes composed of non-membrane-limited aggregates of viral protein and ribosomes. 12 Large eosinophilic intracytoplasmic bodies that approximate those observed with IBD of boid snakes have not been described in association with any other retroviral infection.
In the original description of IBD, the virus and inclusion bodies were usually found together in the same animal and often within the same cell, but a clear causal relationship between the two entities was not established. Despite the lack of an established casual relationship between the virus and the inclusion body, the current recommendation is to euthanatize all boid snakes exhibiting signs compatible with IBD when the characteristic inclusion bodies in biopsy specimens are revealed by light microscopy. 26 Because of the morphologic and histochemical similarities between IBD inclusions and other types of nonviral intracytoplasmic bodies, we decided to study and better characterize the IBD virus (IBDV)-associated inclusions. 2 7–9, 11 15 25 26 The objectives of the study were to examine the relationship between the retrovirus-like agent and its associated nonviral inclusion bodies and to identify and characterize the material within the inclusion bodies.
Materials and Methods
Source of IBDV
An adult female Boa constrictor amarali with a history of chronic regurgitation, recurrent mouth rot, shedding difficulties, and posterior paresis was euthanatized with pentobarbitol, and a complete necropsy examination was performed at the owner's request. Samples of liver, lung, kidney, splenopancreas, brain, and spinal cord were collected and processed for paraffin sectioning. Additional specimens of liver and kidney were either processed for transmission electron microscopy (TEM) or frozen at −80 C. Light microscopy revealed numerous intracytoplasmic eosinophilic inclusion bodies (up to 140/high-power field) within hepatocytes, neurons, pancreatic acinar cells, and lymphocytes. Ultrastructural evaluation of the liver by TEM revealed abundant C-type retrovirus-like particles that were 100–110 nm in diameter with an electron-dense core enclosed by a hexagonal capsid. A membranous envelope bearing short peplomers covered the external surface. The size, morphology, and budding properties of the viruslike particles were all consistent with IBDV. 25 No other lesions or pathogens were identified. The signalment, history, clinical syndrome, histopathology, and ultrastructural findings all supported a final diagnosis of IBD. The frozen liver and kidney were labeled as IBDV and inclusion positive and retained as a future source of virus and proteins.
Experimental infection
A group of 12 clinically normal age- and size-matched juvenile boa constrictors were obtained through a donation from the Birmingham Zoo. All boas were individually housed in appropriately sized polypropylene cages equipped with a water bowl, hide box, a rock, and a focal heat source to allow for behavioral thermoregulation. All cages were maintained in University of California (UC)–Berkeley animal rooms with an ambient temperature of 24–28 C and a 12:12 hr light–dark cycle. Routine care was provided through the UC–Berkeley Office of Laboratory Animal Care. After a 1-month period of acclimation, each snake was anesthetized with isofluorane, a celiotomy was performed, and a baseline wedge biopsy of liver approximately 6 × 4 × 2 mm was collected from each snake. Each biopsy specimen was halved upon collection and processed for both light and electron microscopy. Bleeding from the biopsy site was controlled with sterile gel foam. The body wall and skin were closed with absorbable monofilament suture material. All snakes were monitored closely for evidence of infection or other postsurgical complications for 10 days. All 12 snakes remained clinically and behaviorally normal throughout the acclimation and postoperative healing period. No inclusions or retrovirus-like particles were demonstrated in any of the baseline biopsy specimens. The persistent clinical normalcy and negative biopsy findings indicated that all 12 boas were free of IBD.
Approximately 1 g of the frozen IBDV-positive boa liver was thawed, diluted 1:10 in sterile serum free minimum essential medium, and homogenized in a ground glass tissue homogenizer to prepare an inoculum for experimental transmission. The resulting homogenate was clairified by centrifugation at 1,000 × g for 10 minutes, and the supernatant was pushed through a 0.45-μm filter under positive pressure. A sham inoculum prepared from a clinically normal B. constrictor liver that was determined to be free of inclusions and IBDV by light microscopy and TEM was prepared by identical methods.
The group of 12 IBDV-free boas was randomly split into three groups of four snakes each. In one group, the four snakes were each given 1 ml of the filtered normal boa liver homogenate by intraperitoneal (IP) injection (sham-inoculated controls). 25 To eliminate the possibility of accidental infection, the sham inoculation was prepared and administered before any IBDV-infected material was handled. In the second group, each snake was inoculated with 1 ml of the filtered IBDV–infected liver homogenate by IP injection. 25 The third group was left untreated (untreated controls). All three groups of boas were monitored daily for clinical evidence of IBDV infection following a strict clean-to-dirty order of operation. The experimentally infected and control animals were housed in different rooms and were never cared for by the same technician.
All 12 boas were serially monitored for histopathologic changes and ultrastructural evidence of IBDV infection by repeated hepatic wedge biopsies taken at 10, 20, and 32 weeks postinoculation (PI). Each biopsy specimen was divided into three pieces upon collection and processed for paraffin sectioning, frozen sectioning, and TEM. At 52 weeks PI, all 12 boas were euthanatized with intracardiac pentobarbital (100 mg/kg) while under a surgical plane of isofluorane anesthesia, and complete necropsies were performed. Samples of liver, gall bladder, lung, kidney, splenopancreas, brain, and spinal cord were collected and processed for paraffin sectioning. Additional specimens of liver and kidney from each snake were processed for TEM (liver and kidney), immunoelectron microscopy (liver), frozen sectioning (liver), or were frozen at −80 C (liver and kidney). All procedures involving the use of animals were conducted in accordance with an approved animal protocol and the National Institutes of Health Guide for the Care and Use of Laboratory Animals. Special permission to conduct multiple celiotomies and liver biopsies was granted and overseen by the UC–Berkeley Institutional Animal Care and Use Committee.
Tissue processing
Tissue specimens for light microscopy were fixed for 24 hours in 10% neutral buffered formalin, embedded in paraffin, sectioned at 3–5 μm, and mounted on glass slides. Sections were stained with Harris hematoxylin and eosin (HE), toludine blue (T-blue), Congo red, thioflaven-T, phosphotungstic acid hematoxylin (PTAH), and periodic acid–Schiff (PAS) using standard histochemical methods. 19 28
Specimens for TEM were cut into 1-mm cubes, fixed in 2% glutaraldehyde for 24 hours at 4 C, embedded in EPON, sectioned at 1 μm, mounted on glass slides, and stained with T-blue. 22 Selected blocks were sectioned at 60 nm, mounted on copper grids, and stained with lead citrate and uranyl acetate using standard electron microscopy laboratory methods. 22
Liver biopsy analysis
Histopathologic changes within the liver, including inclusion body development, were monitored over time in the series of HE-stained liver sections collected from each of the experimentally infected and control boas. The average size of inclusion bodies was determined by measuring 50 randomly selected inclusions in each specimen with a standard ocular micrometer and calculating the mean diameter for each time point. The density of inclusion bodies was determined by counting all inclusions in 30 high-power field for each specimen. Both the inclusion body measurements and the density figures were collected using a structured slide review format to minimize the counting of duplicate and/or overlapping fields. To further minimize duplicate counts or measurements, each section was represented only once in each data set. The reported values for inclusion body sizes and densities represent the calculated mean ± 2 SD. The average size and density of inclusion bodies at each time point were compared directly. The ultrastructural characteristics of inclusion bodies and the presence or absence of retrovirus-like particles were determined from the biopsy specimen halves processed for TEM.
Protein electrophoresis
Specimens of liver and kidney collected from normal (IBDV-free) control boas and IBDV-infected, inclusion-positive boas (the original donor snake and experimentally infected snakes at 1 year PI) were homogenized in 1% sodium dodecyl sulfate (SDS) using a Dounce tissue homogenizer. The total protein in each homogenate was quantified using a BCA protein assay kit in accordance with the manufacturer's protocol (Peirce Biochemicals, Rockford, IL). The initial analysis of proteins in the crude extracts was accomplished with SDS polyacrylamide gel electrophoresis (SDS-PAGE) using an 8- × 8-cm minigel system in accordance with the manufacturer's protocol (Bio-Rad, Richmond, CA). The gels were run under reducing conditions using a discontinuous buffer system with a 10% resolving gel overlay and a 4% stacking gel. 17 Aliquots of each tissue homogenate were diluted 1:4 in reducing sample buffer and heated to 95 C for 5 minutes, and each lane was individually loaded with 15 μg of protein. One lane of molecular mass standards (Bio-Rad) was run on each gel. Minigels were run at 200 V until the bromophenol blue dye front had migrated to approximately 5 mm from the bottom of the gel. Gels were stained for total protein with either Coomassie brilliant blue R-250 or silver following the manufacturer's protocols (Bio-Rad). The protein band profiles of infected and normal tissue extracts were compared directly by visual inspection. Bands unique to IBDV-infected, inclusion-positive tissue were tentatively referred to as IBD proteins and given a specific designation number that corresponded to the molecular mass.
Aliquots of IBD proteins were purified by preparative SDS-PAGE. Batches of IBDV-infected, inclusion-positive boa liver homogenate containing 700 μg of protein were prepared as described above and loaded onto a 20- × 20- × 1.5-mm 10% acrylamide gel overlay with a 4% stacking gel layer and run at 35 mA/gel following the manufacturer's protocol (Bio-Rad). One lane of molecular mass standards (Bio-Rad) was run on each gel. The gels were negatively stained for protein using a copper stain kit in accordance with the manufacturer's protocol (Bio-Rad). Protein bands of interest (IBD proteins) were excised with a clean scalpel blade, destained, minced, and electroeluted into a dialysis membrane–covered cap with a molecular mass cutoff of 12–15 kd using Tris-glycine buffer and a current setting of 10 mA/tube (Bio-Rad). The purity of each lot of protein was assessed on a 10% acrylamide minigel tube (Bio-Rad). Samples appearing as single bands on Coomassie brilliant blue R-250–stained minigels were dialyzed against 0.9 % NaCl, sterile filtered, and used to immunize mice and as antigens in enyzyme-linked immunosorbent assays (ELISAs).
Mouse immunization and hybridoma derivation
Blood was collected from three adult female BALB/c mice to obtain baseline serum. The mice were then immunized with electrophoretically purified inclusion protein (100 μg protein/dose) at 2-week intervals. Each mouse was immunized three times by subcutaneous injection, and blood was collected from the retro-orbital space 10 days after the third immunization at a surgical plane of methoxyflurane anesthesia. The initial injection was emulsified with an equal volume of Freund's complete adjuvant. 29 The two subsequent booster immunizations were made with incomplete Freund's adjuvant. 29 All baseline and postimmunization antisera specimens were assayed for specific antibody by solid phase plate ELISA and western blot. After confirming seroconversion to the immunogen, one of the mice was given 100 μg of inclusion protein dissolved in 0.2 ml 0.9 % NaCl intravenously and euthanatized by carbon dioxide asphyxiation 72 hours later.
The freshly euthanatized mouse was exsanguinated by cardiocentesis for antisera preparation, and the spleen was removed with sterile surgical instruments. The distal end of the spleen was incised and the parenchymal contents were flushed from the cut end with sterile phosphate-buffered saline. The splenic capsule and its remaining contents were then macerated with sterile scissors and added to the cell suspension. Splenic lymphocytes were counted and fused with P3X myeloma cells with polyethylene glycol following the manufacturer's protocol (PEG 1500; Boehringer Mannheim Biochemicals, Indianapolis, IN) at a ratio of 7:1. 16 The resulting hybridomas were grown in Dulbecco's modified Eagles medium supplemented with 10% fetal bovine serum, penicillin, streptomycin amphotericin-B, and hypoxanthine aminopterin and thymidine selective medium supplement (Sigma Chemical Co., St. Louis, MO). 4 16 All hybridomas were screened for antibody production by ELISA and western blot and cloned by limiting dilution using standard laboratory procedures. 4 16 Hybridoma clones producing anti–IBD protein antibody were selected, expanded in flasks, and grown in vitro for monoclonal antibody production. 4 All mouse hybridoma and myeloma cells were grown in a humidified 37 C incubator under an atmosphere of 5% CO2. 4
ELISA
All mouse serum samples and hybridoma supernatants were screened by solid phase ELISA using a standard protocol. 4 Dynatech Immulon II plates (Dynatech Laboratories, Chantilly, VA) were coated with 0.5 μg of electrophoretically purified inclusion protein diluted in 50 μl of 100 mM sodium carbonate/bicarbonate buffer and incubated overnight at 4 C. Unbound sites were blocked with 2% nonfat milk (2 hours at room temperature), and the plates were washed three times with Tris-buffered saline (TBS) (pH 8.0), containing 0.05% Tween-20 detergent (TBS-T) (Sigma Chemical Co.). The plates were then reacted with a source of primary antibody followed by alkaline phosphatase–labeled goat anti-mouse IgG (γ-chain-specific antibody, diluted 1:1,000; Kirkegaard & Perry, Gaithersburg, MD) and 5-bromo, 4-chloro, 3-indolyl phosphate (BCIP) microwell substrate (Kirkegaard & Perry). All antibody incubations were for 1 hour at room temperature with three washes in TBS-T (5 minutes/wash) after each incubation. The ELISA results were recorded as direct optical density values obtained from an automated ELISA plate reader (Dynatech MR5000, Dynatech Laboratories) with test and reference wavelength settings of 630 nm and 550 nm, respectfully. The serum antibody titer was approximated by serial dilution using TBS as a diluent. 4 The endpoint antiserum titer was defined as the highest dilution that yielded an optical density twice that achieved with the same dilution of baseline mouse serum. 4
Hybridoma supernatants were diluted 1:1 in TBS and assayed in duplicate wells with and without antigen using the ELISA protocol. Negative controls for all hybridoma supernatants consisted of P3X myeloma cell–conditioned medium diluted 1:1 in TBS. 30 Serum from an immunized mouse diluted to its midpoint titer with TBS was used as a positive control for all hybridoma supernatant assays. 4 The results for each well were recorded as a binding ratio. Binding ratios were calculated by subtracting the average background optical density figures (wells without antigen that were treated with the test supernatant or conditioned medium) from the average test and negative control figures to obtain corrected values. 30 31 The average corrected test value was then divided by the corrected control value. 30 Any ratio ≥2 was considered positive. 30
Western blot
Proteins separated by SDS-PAGE were transferred from the gels to nitrocellulose membranes for western blot analysis of mouse sera and hybridoma supernatants. 27 Transfers were made in 1 hour at 100 V using a Tris-glycine tank buffer containing 20% (vol/vol) methanol. Immediately following transfer, each blot was washed three times in TBS-T, reversibly stained for total protein with Ponceau red stain following the manufacturer's protocol (Bio-Rad), and cut into strips that were two lanes wide. Each resulting strip contained one lane of IBDV-infected, inclusion-positive boa liver proteins and one lane of normal boa liver proteins. All blots were blocked in 2% nonfat milk followed by sequential incubations with mouse anti-inclusion antibody and alkaline phosphatase–conjugated goat anti-mouse IgG (γ-chain-specific antibody, diluted 1:1,000; Kirkegaard & Perry). 31 All antibody incubations were for 2 hours at room temperature with three washes in TBS-T (5 minutes/wash) between incubations and blot color development. Antibody labels were visualized by incubation of the blots with a precipitating alkaline phosphatase substrate (50 μg/ml BCIP) and 100 μg/ml nitroblue tetrazolium (Kirkegaard & Perry). 31 Band development was visually monitored, and the reaction was stopped by rinsing with deionized water. 31 Negative controls for the hybridoma supernatants and mouse antisera consisted of P3X myeloma cell–conditioned medium diluted 1:1 in TBS and baseline mouse serum diluted 1:10 with TBS, respectfully. 30 The mouse antiserum was diluted to its midpoint ELISA titer with TBS before being employed as a source of antibody in western blot procedures. 4 Once characterized, the diluted antiserum was employed as a positive control in all subsequent blotting procedures.
Immunohistochemistry and immunoelectron microscopy
Immunohistochemical staining was done on both paraffin and frozen sections of normal and IBDV-infected, inclusion-positive boa liver. All slides were immunohistochemically stained using a modified avidin–biotin–peroxidase complex (ABC) method and kit (Vectastain ABC Kit, Vector Laboratories, Burlingame, CA). 14 28 Paraffin sections were deparaffinized in xylene, rehydrated in a graded ethanol series, rinsed in TBS, treated for 10 minutes in 0.05% protease type XIV (Sigma Chemical Co.) at 37 C, and rinsed again for 10 minutes in TBS. 14 Specimens for frozen sectioning were embedded in O.C.T. (Miles, Inc., Elkhart, IN), snap frozen in 2-methylbutane chilled to its freezing point with liquid nitrogen cut on a cryostat set at −20 C, air dried, fixed in chilled absolute acetone for 30 seconds, and air dried a second time. All sections were mounted on poly-lysine–treated slides (Fisher Scientific, Pittsburgh, PA) and encircled with a wax pen (PAP pen) to create a hydrophobic well. Endogenous peroxidase activity was blocked by emersion in 0.3% H2O2 in methanol for 30 minutes. 14 All sections were then washed in TBS for 5 minutes and blocked for 30 minutes in 10% normal horse serum. Sets of paraffin and frozen sections were stained with anti–intermediate filament antibodies (high and low molecular mass cytokeratins, pancytokeratin [AE1/AE3], neurofilament triplet, and vimentin) or anti–IBD protein antibodies followed by biotinylated horse anti-mouse IgG (γ-chain-specific antibody diluted 1:500; Vector Laboratories) and preformed ABCs. All incubations were for 1 hour at room temperature with three 10-minute washes in TBS between each step. Baseline mouse serum and conditioned medium were used as negative control primary antibodies for the mouse antisera and hybridoma supernatants, respectfully. Positive control tissues for the intermediate filament stains consisted of normal canine tissues known to contain the intermediate filaments of interest (UC–Davis VMTH control tissues) and sets of normal Boa constrictor tissues that were predicted to contain intermediate filaments. Intermediate filament antibody labels were visualized by incubating the slides with aminoethylcarbozole (AEC) (Zymed kit, Zymed Laboratories, South San Franciso, CA). The anti–IBD protein labels were developed with diaminobenzidine (DAB). Color development was visually monitored and stopped by washing with deionized water (dH2O). All slides were counterstained with Mayer's hematoxylin. The AEC-stained slides were washed in dH2O and heat polymerized in Crystal mount (Biomedia Corp., Foster City, CA) before being coverslipped. The DAB-stained slides were coverslipped using standard laboratory methods.
Samples of IBDV-infected, inclusion-positive and samples of normal boa liver were processed for immunoelectron microscopy. All specimens were fixed in 4% paraformaledehyde in 0.1 M phosphate buffer (pH 7.3) for 4 hours, embedded in LR white resin, thin sectioned, and mounted on nickel grids in accordance with the manufacturer's protocol (Polysciences, Warrington, PA). All sections were blocked in heat-inactivated goat serum for 1 hour and incubated with either monoclonal anti–IBD protein antibodies, polyclonal mouse anti–IBD protein antisera (positive control), or P3X myeloma cell–conditioned medium (negative control). Bound antibody was visualized by incubation with goat anti-mouse IgG (heavy- and light-chain specific) conjugated to 10-nm colloidal gold particles (1:50; Vector Laboratories) following the manufacturer's protocol (BBI Publication G1, Vector Laboratories). All antibody incubations were for 2 hours at room temperature with five washes in TBS (5 minutes/wash) between steps. All immunogold-stained sections were washed in TBS for 5 minutes and lightly counterstained with uranyl acetate and lead citrate prior to examination. 22
Results
All four of the boas inoculated with the potentially infectious liver homogenate prepared from the IBD-positive boa developed intracytoplasmic inclusions within hepatocytes by 10 weeks PI (Figs. 1–4). At 10 weeks PI, the affected hepatocytes were vacuolated and contained low to moderate numbers of small (1–2 μm) round eosinophilic hyaline intracytoplasmic inclusion bodies. Sequential biopsies demonstrated that both the number and average diameter of inclusion bodies increased over time (Table 1). The degree of cytoplasmic vacuolation in affected hepatocytes also increased with the duration of IBDV infection (Figs. 1–4). The maximal inclusion body diameter of 5–6 μm and density of ≥120/high-power field was first demonstrated at 32 weeks PI (Table 1). The inclusions were histochemically characterized as being PTAH positive, PAS negative, and Congo red negative and exhibited orthochromatic staining with T-blue. None of the anti–intermediate filament antibodies labeled the inclusions despite an apparent affinity for the endogenous filaments in snake tissues (data not shown). No inclusion bodies were demonstrated in any of the preinoculation biopsy specimens or in any of the tissues taken from the sham-inoculated or uninoculated control snakes.
IBD inclusion body kinetics.

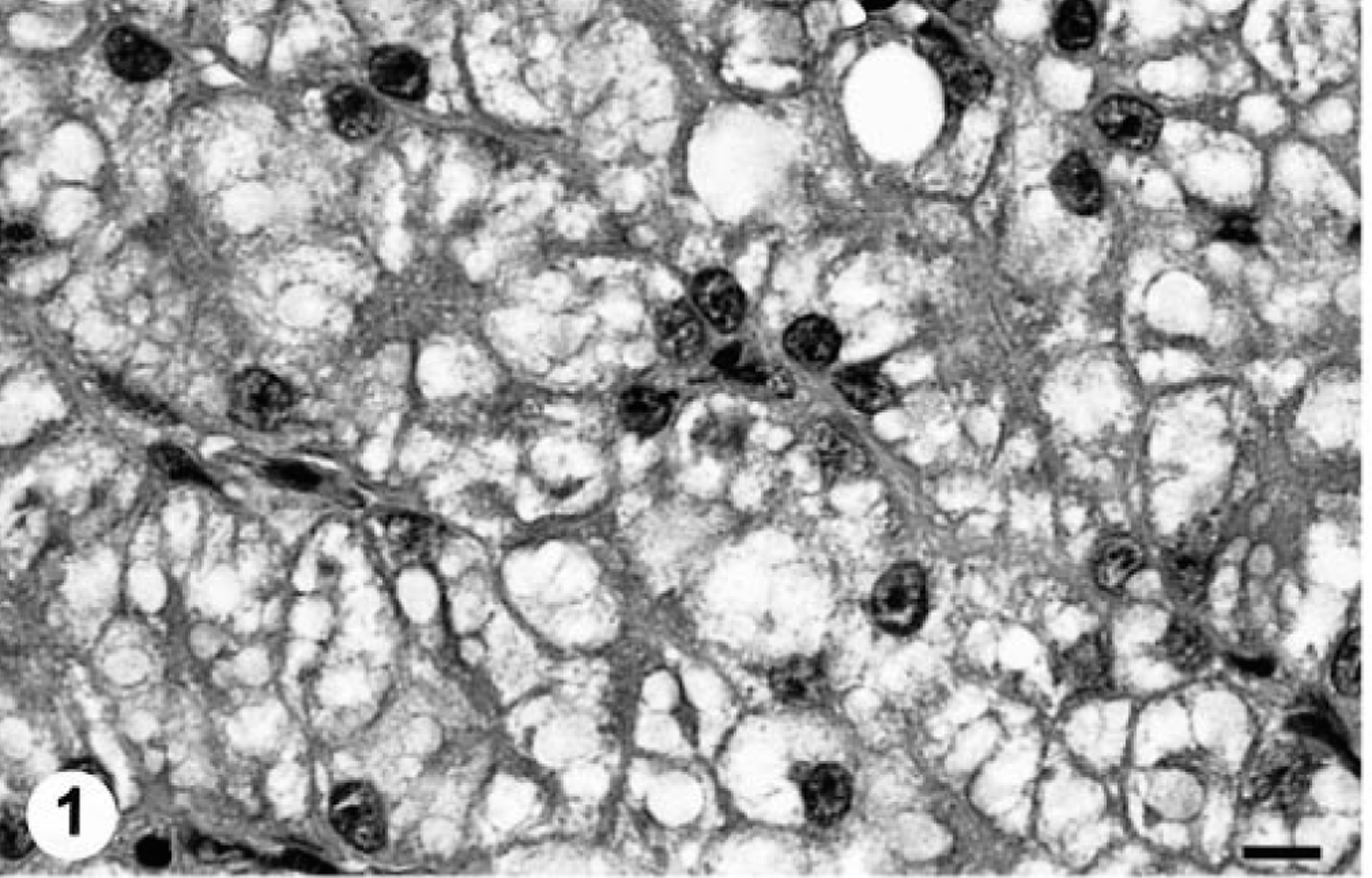
Liver; Boa constrictor. Serial liver biopsy specimens from a Boa constrictor experimentally infected with IBDV, showing an increase in the average size and density of intracytoplasmic inclusions (arrows) with increased duration of infection. Baseline specimen.
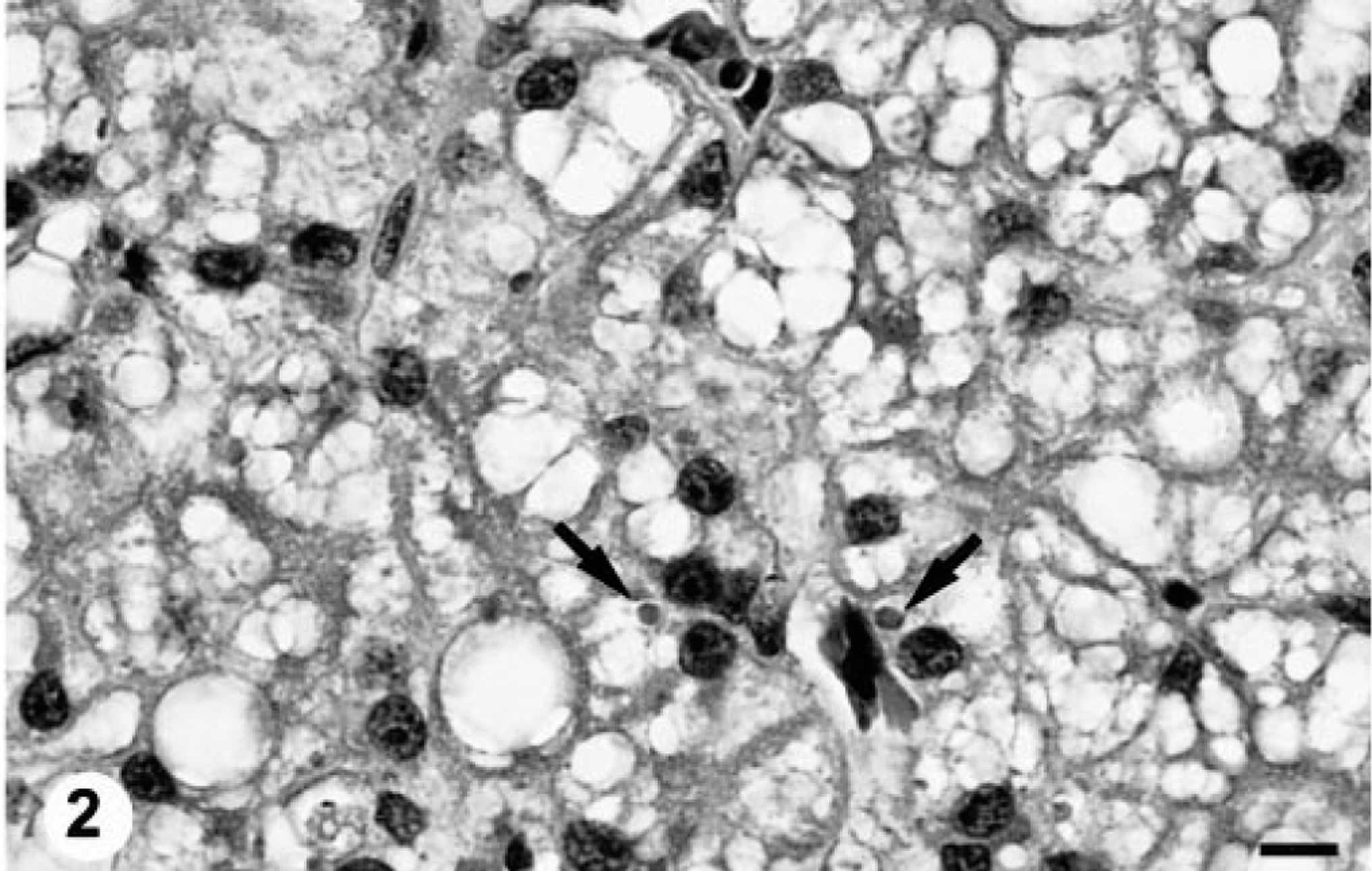
Ten weeks PI.
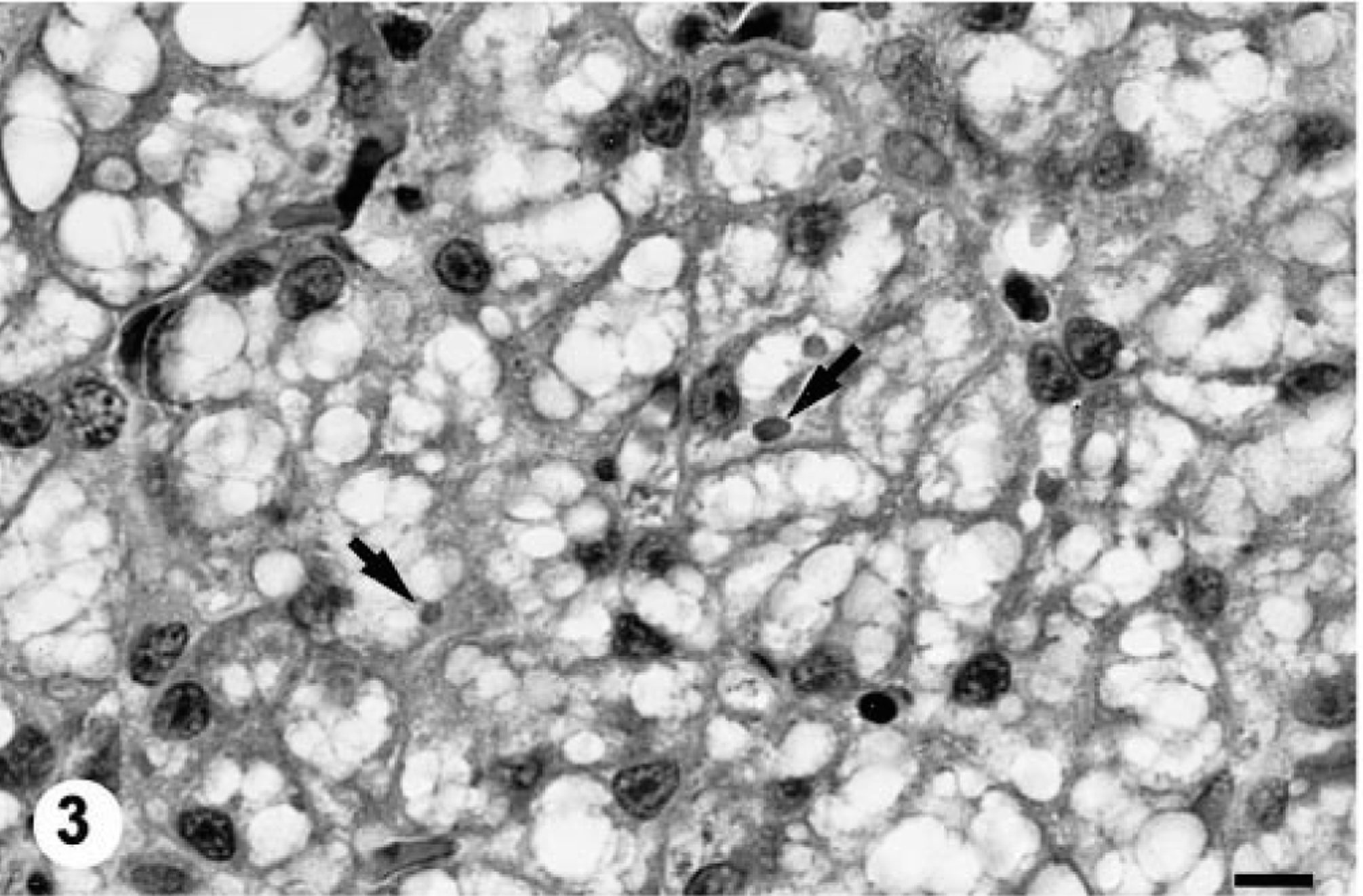
Twenty weeks PI.
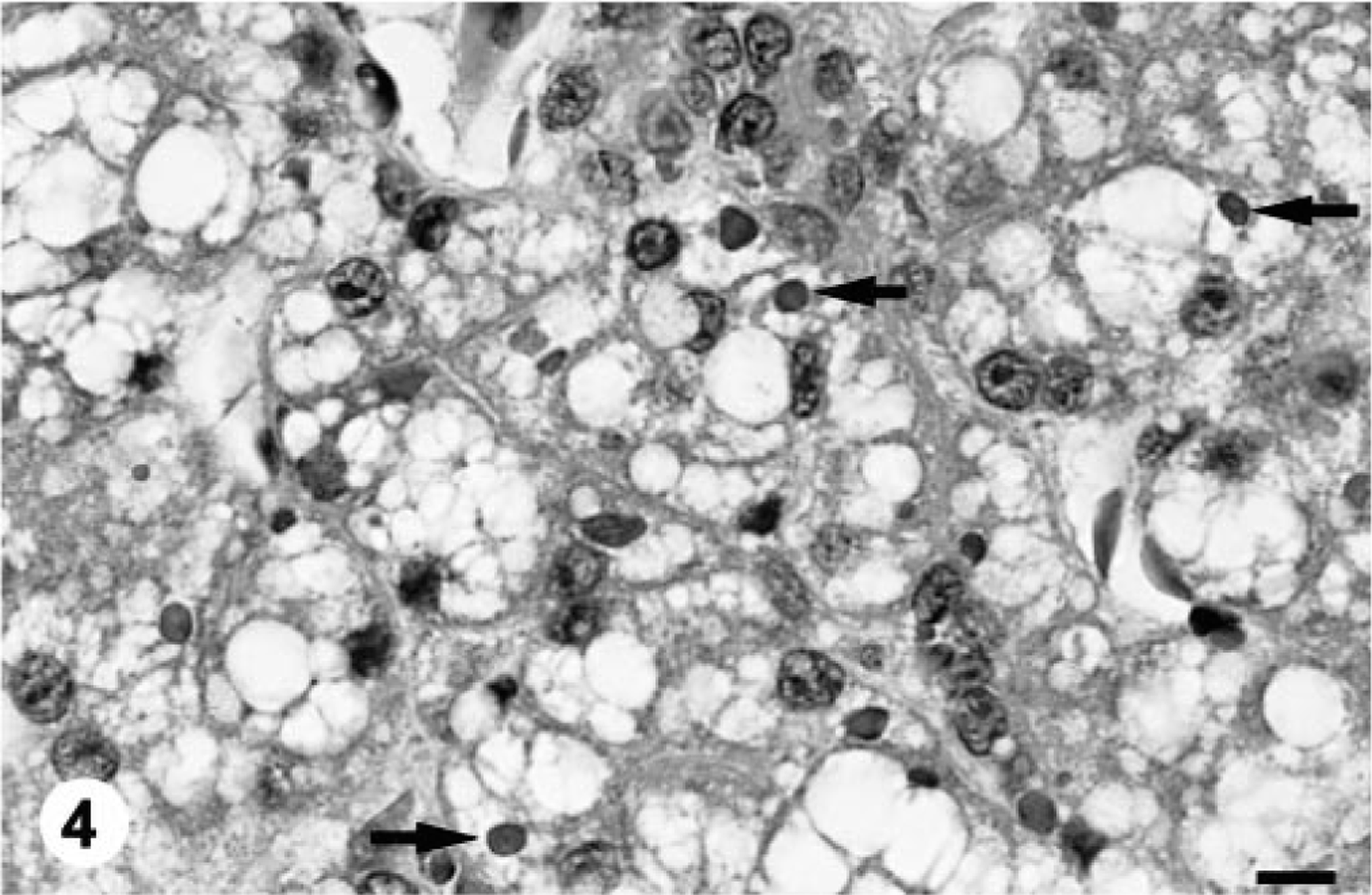
Thirty-two weeks PI. HE. Bar = 10 μm.
Electron microscopic evaluation of liver specimens from all four of the experimentally inoculated inclusion-positive boas revealed budding C-type retrovirus-like particles morphologically and morphometrically identical to those described in the donated animal by 10 weeks PI (Fig. 6). The retrovirus-like particles were observed to bud from both cytoplasmic and plasma membranes of hepatocytes. The assumed mature particle was an enveloped intercellular structure 100–110 nm in diameter with an electron-dense core enclosed by a hexagonal capsid and a membranous envelope bearing short spikelike peplomers on its external surface. The membranous envelope on the budding particles appeared slightly thicker and was more electron dense than the adjacent plasma membrane (Fig. 6). Occasional intracellular particles with electron-lucent cores were budding from dilated regions of smooth endoplasmic reticulum. The size, morphology, and budding properties of the viruslike particles were all consistent with the IBDV. 23 No retrovirus-like particles were demonstrated in any of the preinoculation biopsy specimens or in any tissues taken from the sham-inoculated or uninoculated control snakes. None of the boas developed any clinically apparent signs of IBD by 1 year PI.
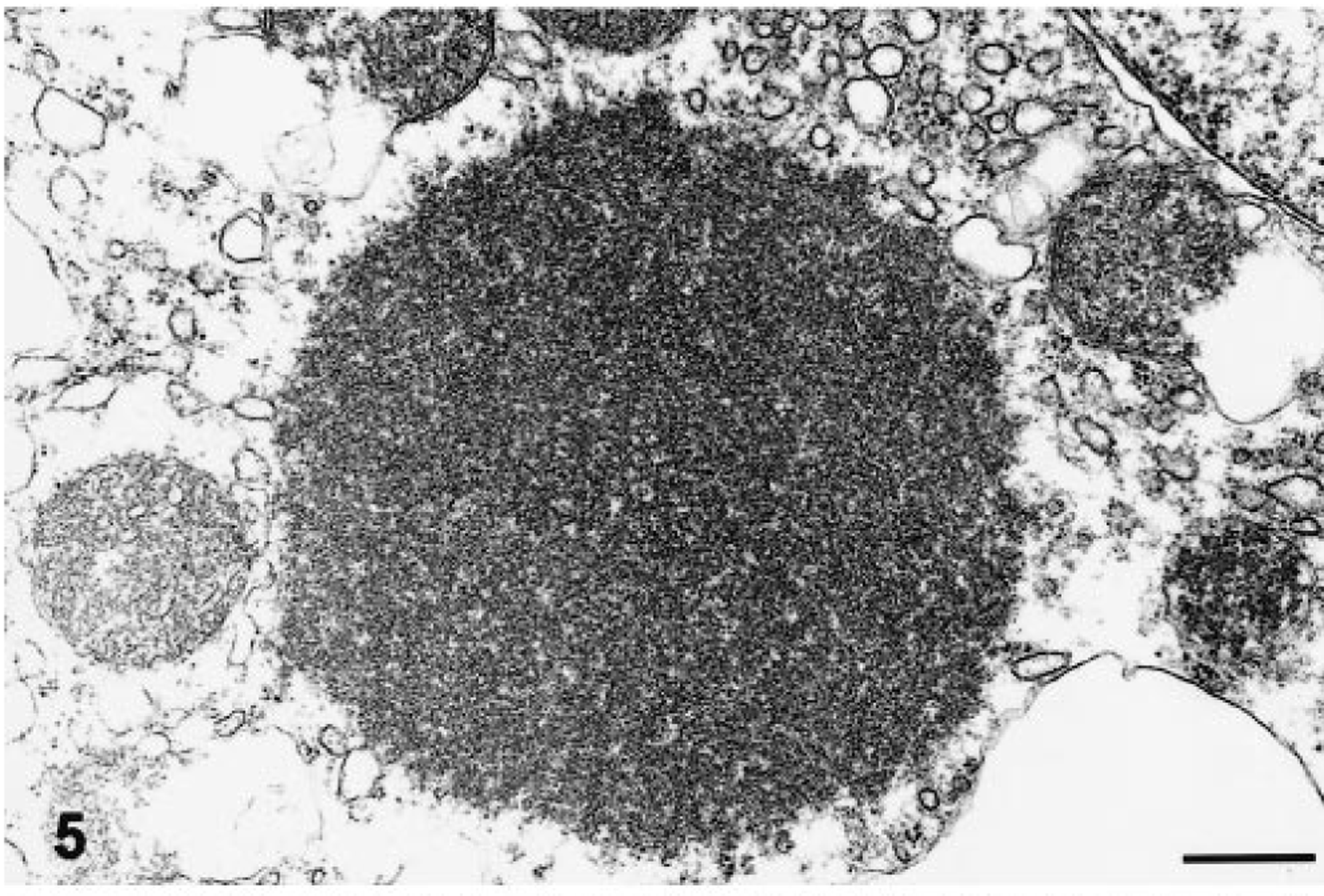
Electron micrographs. Liver; Boa constrictor. Three morphologic variants of intracytoplasmic inclusion bodies found in IBDV-infected snakes. Fig. 5. A small round inclusion similar to those illustrated in Fig. 2. The inclusion is characterized as an intracytoplasmic aggregate of granular electron-dense material. No limiting membrane is identified.
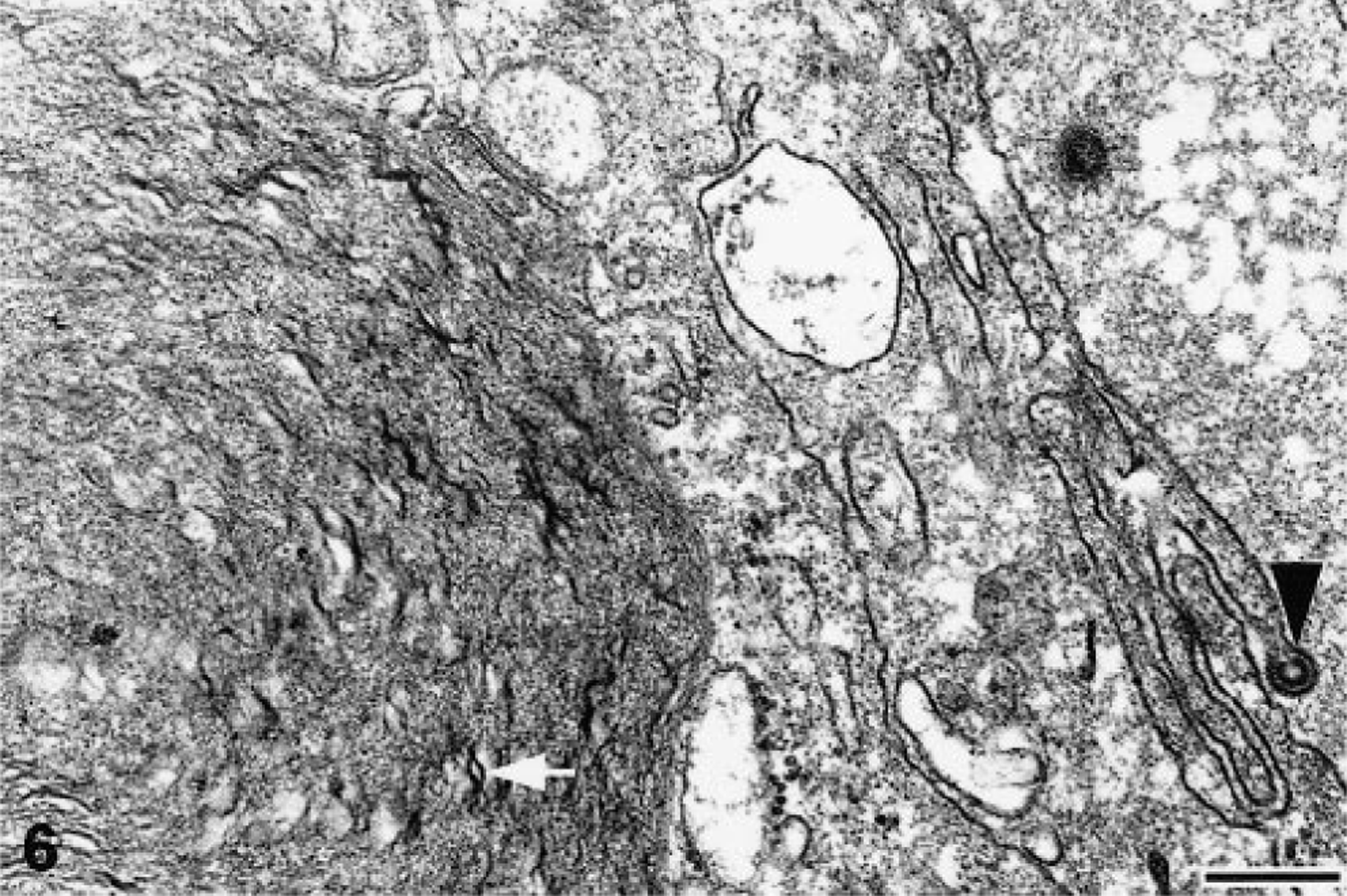
Large (>3 μm) inclusion body. A C-type retrovirus-like particle (arrowhead) in the process of budding from the plasma membrane of the inclusion-bearing hepatocyte. Large inclusions are characterized as membrane bound intracytoplasmic structures that are composed of granular to amorphous electron-dense material admixed with membranelike fragments (arrows). The bilayered structure of the membranelike fragments and the irregular deposits of granular electron-dense material on the surfaces is evident in several regions.
The ultrastructural morphology of the inclusions differed with size. The small round punctate bodies <2 μm in diameter consisted of intracytoplasmic aggregates of granular electron-dense material that were not membrane limited (Fig. 5). The larger, more conspicuous inclusions 3–6 μm in diameter were characterized as membrane-bound aggregates of granular electron-dense material admixed with membranelike fragments (Figs. 6, 7). The membranelike fragments within inclusions had a typical bilayered morphology with irregular deposits of electron-dense material on the surfaces (Figs. 6, 7).
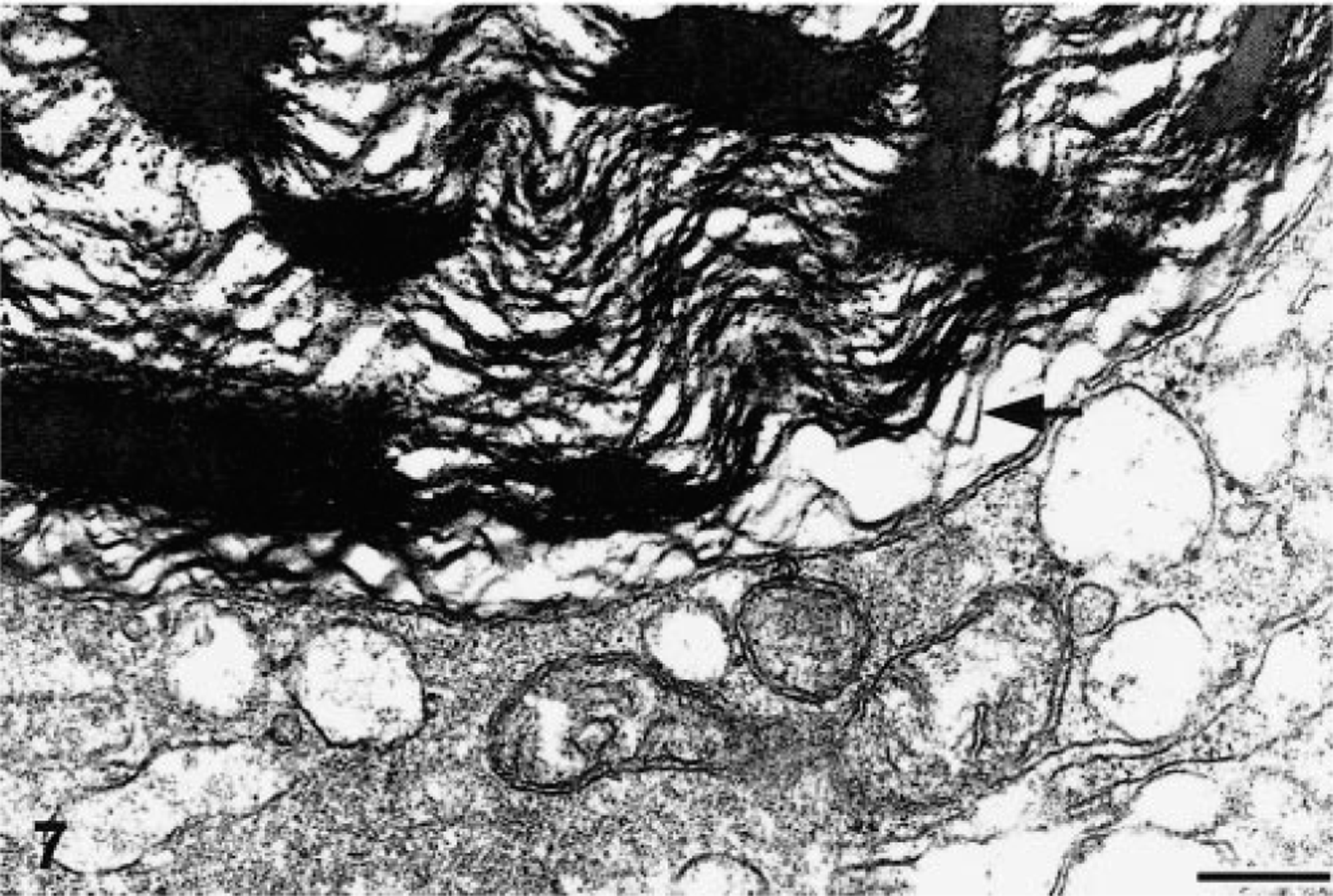
Large inclusion body. Multifocal solid densities of electron-dense material containing membrane profiles are present. Uranyl acetate and lead citrate. Bar = 250 nm.
Comparative analysis of the proteins in infected boa liver and kidney specimens with high density of intracytoplasmic inclusions (≤140/high-power field) with normal boa tissues demonstrated a prominent 68-kd protein band that was unique to infected tissues (Fig. 8). The band appeared as a singlet in both Comassie blue–stained and silver-stained acrylamide gels but was tightly opposed to an overlying 69–70-kd band that was present in both infected and normal tissue (Fig. 8).
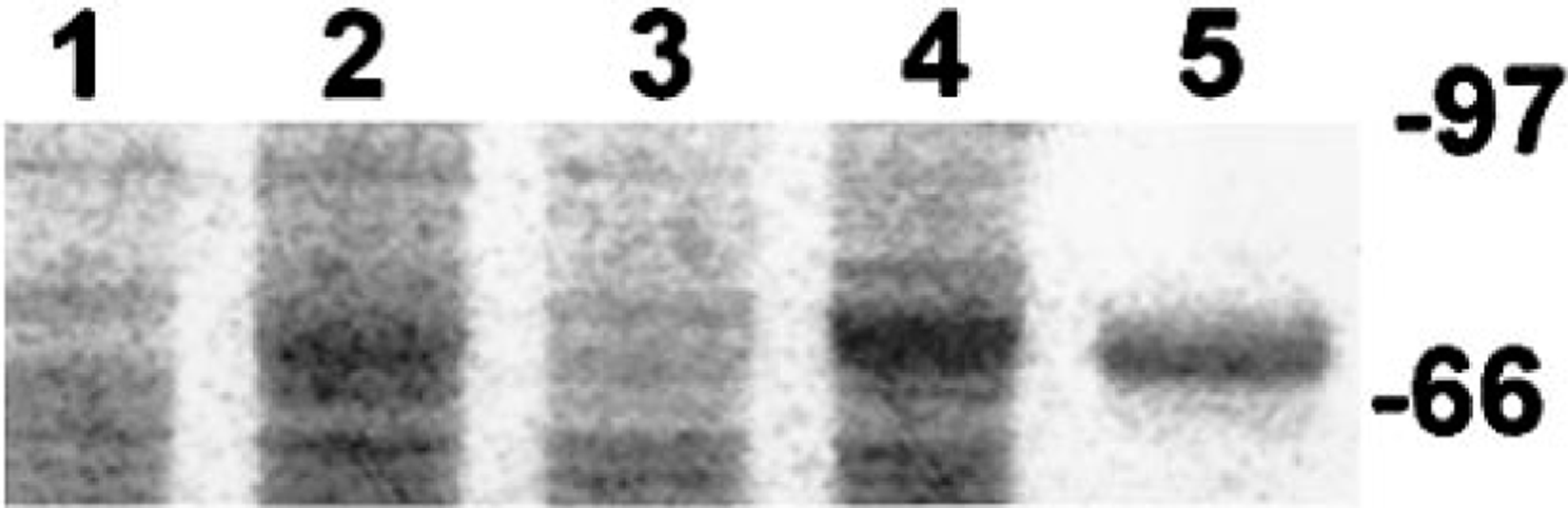
Polyacrylamide gel containing electrophoretically spearated proteins extracted from normal and IBDV-infected B. constrictor liver and kidney. Lanes 1 and 3 contain normal boa liver and kidney homogenates, respectfully. Lanes 2 and 4 contain IBDV-infected liver and kidney homogenates, respectfully. Both of the infected tissues have a high density of large inclusion bodies. A prominent 68-kd protein band is present in both IBDV-infected, inclusion-positive tissues; this band is not present in normal boa tissues. An aliquot the electrophoretically purified protein fraction is shown in lane 5. Coomassie brilliant blue R-250 total protein stain. The markers represent the molecular masses in kilodaltons.
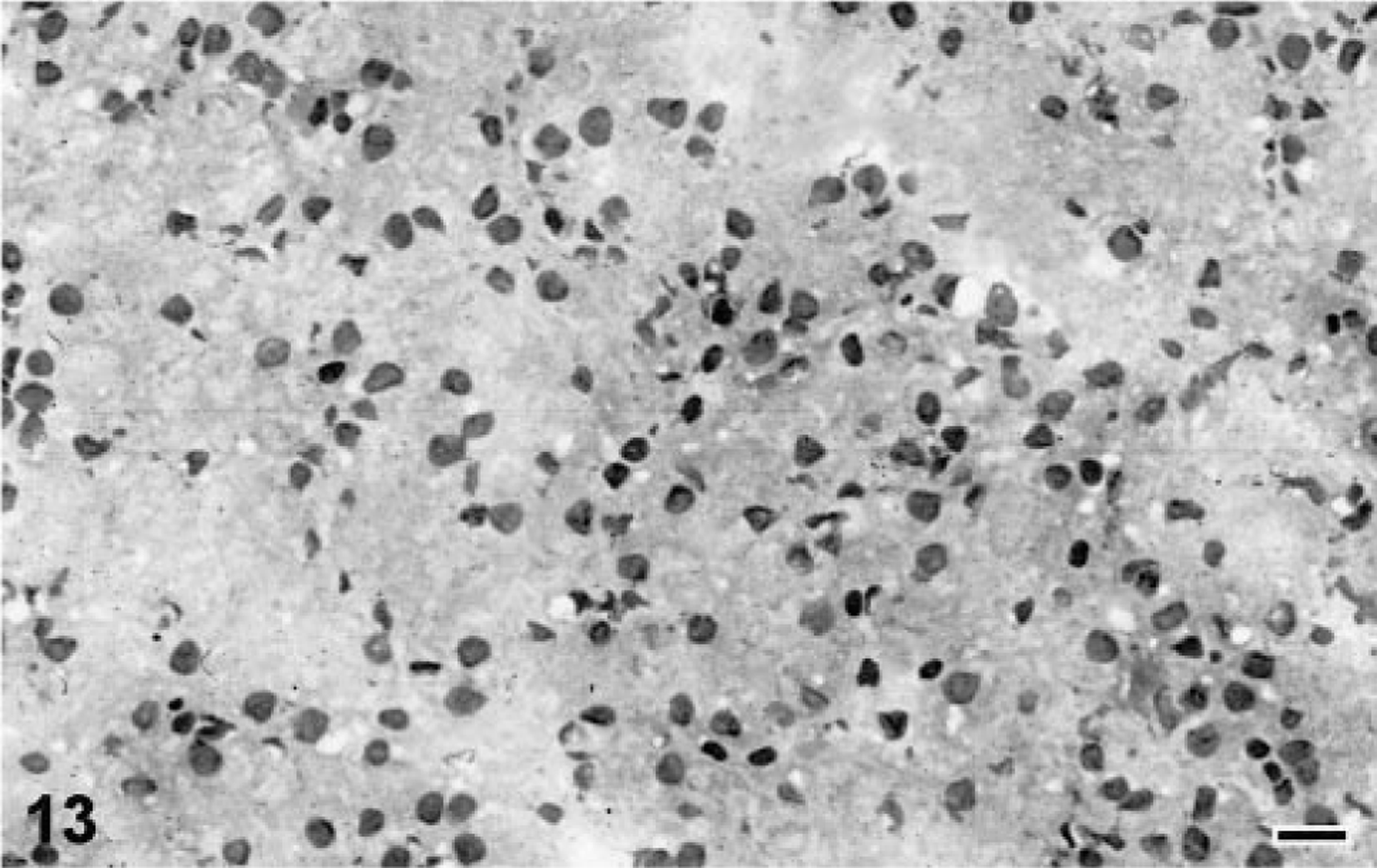
Mouse antiserum prepared against samples of the electrophoretically isolated 68-kd IBD protein exhibited affinity for both infected and normal boa liver (Fig. 9). Careful screening of a library of hybridomas by ELISA and western blot resulted in the isolation of a clone (2H2) producing a monoclonal IgG antibody directed against the 68-kd IBD protein (Fig. 9). Immunohistochemical staining of frozen sections of IBDV-infected, inclusion-positive boa liver demonstrated that the monoclonal antibody exhibited affinity for IBD inclusion bodies (Figs. 10, 11). Numerous smaller and less conspicuous intracytoplasmic aggregates of antigenically related material were demonstrated throughout the cytoplasm of some inclusion-bearing cells (Fig. 10). No reactivity was exhibited for normal boa liver homogenates or sections (Figs. 9, 11). All attempts at labeling structures in formalin- or paraformaledehyde-fixed B. constrictor tissues with the monoclonal antibody (2H2) were unsuccessful. Neither baseline mouse serum or conditioned medium exhibited any affinity for B. constrictor tissues.
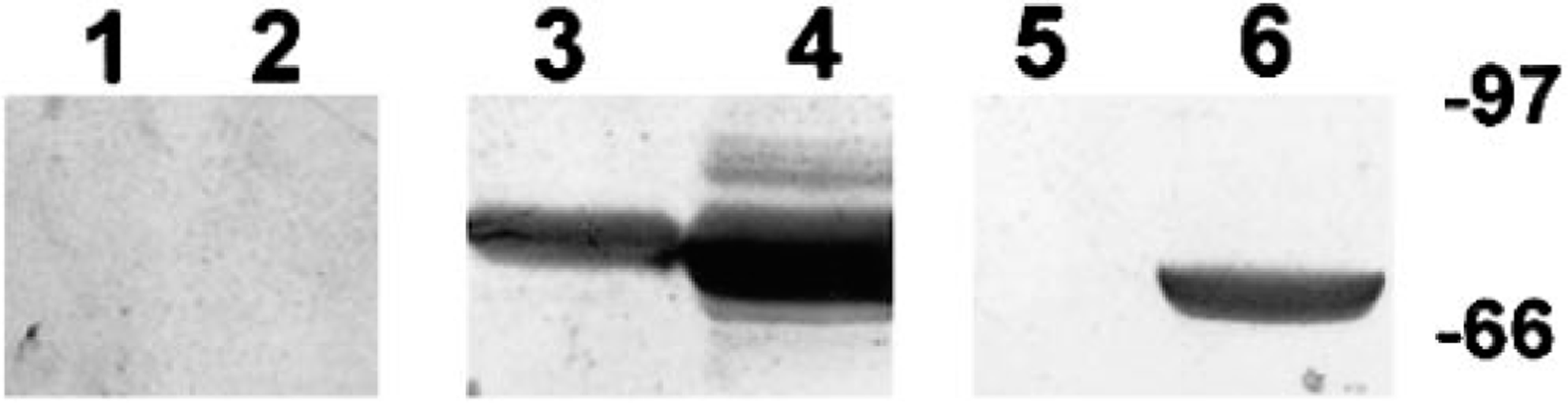
Western blot of normal and IBDV-infected, inclusion-positive B. constrictor liver proteins. Lanes 1, 3, and 5 contain normal boa liver. Lanes 2, 4, and 6 contain IBDV-infected boa constrictor liver with a high density of large inclusion bodies. Lanes 1 and 2 were stained with baseline mouse serum. Lanes 3 and 4 were stained with polyclonal mouse antisera raised against the 68-kd IBD protein. Lanes 5 and 6 were stained with monoclonal antibody 2H2, which demonstrates specific affinity for the 68-kd IBD protein band. The markers represent the molecular masses in kilodaltons.
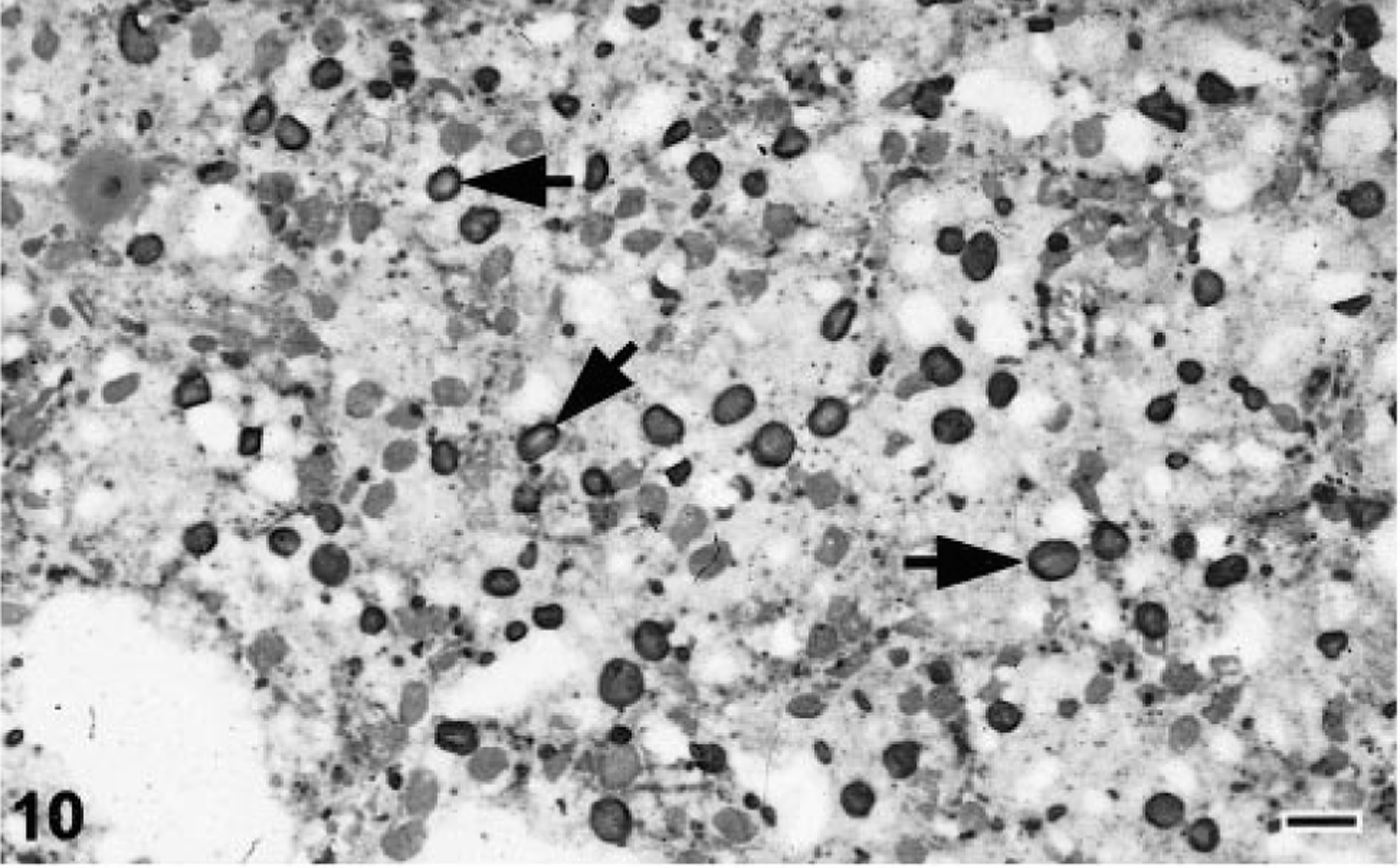
Liver; Boa constrictor. Frozen sections of IBDV-infected, inclusion-positive and normal B. constrictor liver stained with the anti-IBDV monoclonal antibody 2H2.
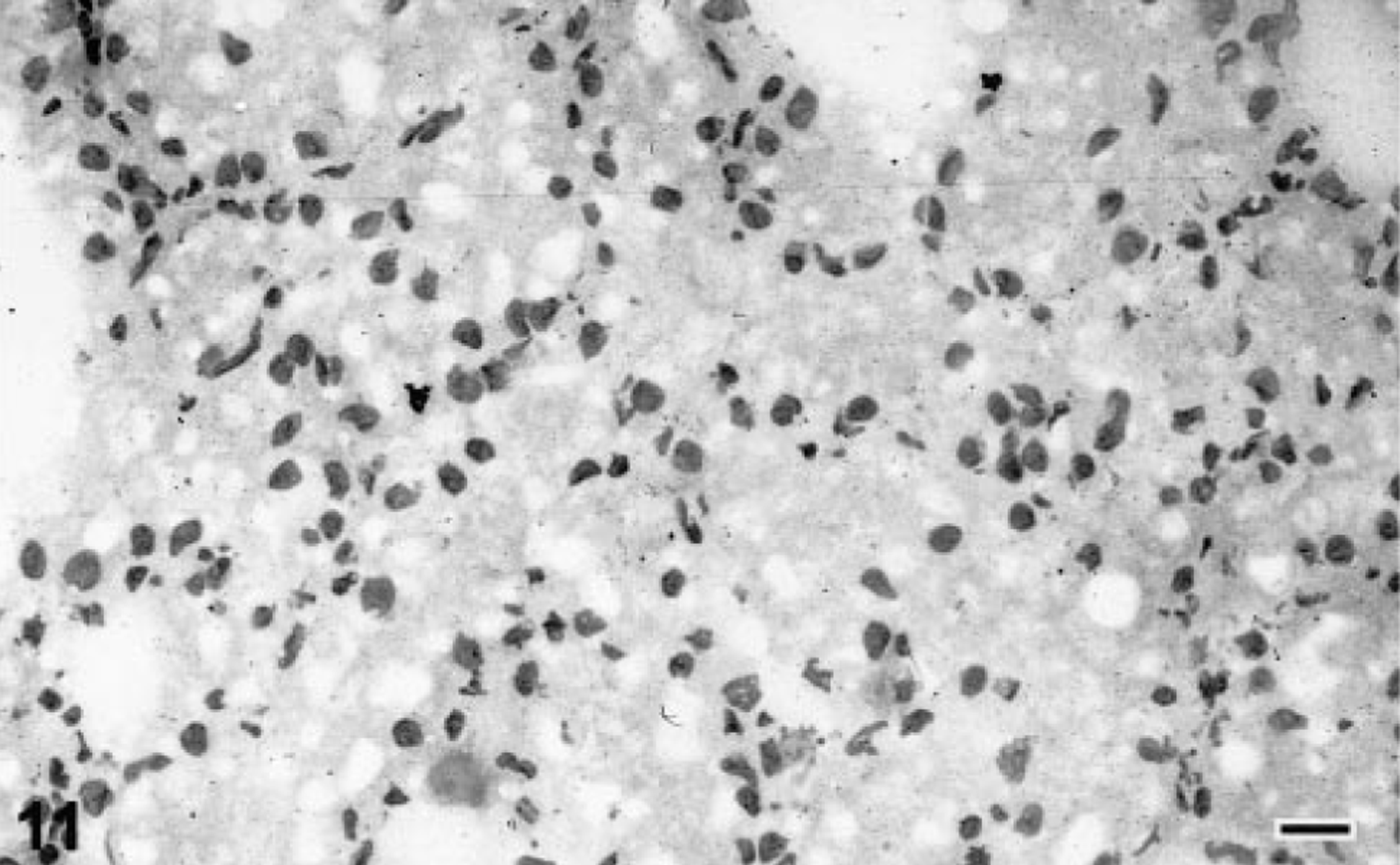
Liver; Boa constrictor. Frozen sections of IBDV-infected, inclusion-positive and normal B. constrictor liver stained with the anti-IBDV monoclonal antibody 2H2
Discussion
All four boas inoculated with the filtered liver homogenate prepared from IBDV-infected boa liver became infected with a budding retrovirus-like agent morphologically and morphometrically consistent with the IBDV and developed the characteristic intracytoplasmic inclusion bodies by 10 weeks PI. Increased cytoplasmic vacuolation was evident in IBDV-infected hepatocytes by 10 weeks PI and appeared to increase over time. Cytoplasmic vacuolation has been described as a cytopathic effect observed with IBDV infection. 25 Unlike previous studies, no clinical evidence of inclusion body disease was demonstrated in any of the experimentally infected boas during the 1-year study. 25 This lack could be attributable to strain differences in the pathogen, host coadaptation, or other unidentified factors. Also, the incubation period for the onset of clinically apparent disease may exceed that for the production of demonstrable inclusion bodies. It has been suggested that B. constrictor has a greater resistance to clinically evident IBD than do other boid species and may be the natural host to some strains of IBDV. 25 Further work on the pathogenesis of IBDV infections in boas and the pathogenic properties of different boa- and python-derived strains is needed before any definitive conclusions can be made in reference to the disease process.
Budding retrovirus-like particles were not demonstrated in all cells bearing the characteristic inclusion bodies, but all snakes with inclusion bodies were demonstrated by electron microscopy to be infected with the retrovirus-like agent. The presence of inclusions without demonstrable viral replication within a given cell could indicate that the inclusions persist in cells beyond the period of active replication but could also be attributable to timing or simple sectional geometry. Similar results were reported in the original description of IBD and with HIV-infected lymphocytes bearing tubuloreticular inclusions. 25 32
Histochemically, IBD inclusions were PTAH positive, orthochromatic with T-blue, PAS negative, and eosinophilic with HE. This pattern of staining suggests that the material within the inclusion body is proteinaceous. 19 28 The negative results obtained with Congo red and thioflaven-T suggest that the proteinaceous material is not in a beta-pleated sheet (amyloid-like) conformation. 19 28 The Mallory body, a well-characterized member of the nonviral inclusion family, yields a similar histochemical profile. 23 Mallory bodies stain eosinophilic with HE and are PAS negative, PTAH positive, Congo red negative, oil red O negative, orthochromatic with T-blue, and are metenamine silver reduction negative. 23 Many of the well-characterized proteinaceous intracytoplasmic nonviral inclusion bodies are composed of aggregates of intermediate filaments. 8 10 11 20 Intermediate filaments are cytoskeletal components that range from 6 to 11 μm in diameter and include vimentin, cytokeratins, tonofilaments, and neurofilaments. 10 In the case of the Mallory body, the intermediate filaments within the inclusions have been identified as high- and low-molecular-mass cytokeratin filaments. 2 8 9 11 15 Ultrastructurally similar aggregates of vimentin and neurofilaments have been identified in other variants of intracytoplasmic nonviral inclusion bodies. 20 The lack of affinity of anti-cytokeratin, -neurofilament, and -vimentin antibodies for IBD inclusions suggests that they do not contain intermediate filament proteins. Although a dense coat of ubiquitin could have interfered with antibody binding, this protein has not been demonstrated in membrane-bound variants of intracytoplasmic inclusion bodies. 20
The kinetics of IBD inclusion formation and the antigenically unique properties of the 68-kd IBD inclusion protein suggest that it may be an overproduced and/or poorly degraded component of the virus that accumulates in the host cell cytoplasm. Both the average size and the number of inclusions per high-power field increased with the duration of the infection in all four experimentally infected snakes. The ultrastructural morphology of inclusion bodies varied somewhat with the size of the inclusion body. The small round punctate bodies <2 μm in diameter were composed of intracytoplasmic aggregates of granular electron-dense material that were not membrane limited, whereas the larger inclusions appeared as membrane-bound structures filled with granular electron-dense material admixed with filamentous membranelike structures. Proteins that are synthesized within a cell that are not destined for secretion can accumulate intracytoplasmically as non-membrane bound aggregates. 24 Membrane-bound inclusions, however, often represent either an accumulation of an undigested substrate in phagolysosomes or an accumulation of synthesized proteins inside cisterns of rough endoplasmic reticulum. 24 The presence of granular electron-dense material admixed with membranelike fragments in the same structure would support a phagolysosomal origin for the larger variants of IBD inclusions. Intracytoplasmic accumulation of unidentified granular electron-dense material has been demonstrated with other retroviruses. 6 12 In some cases, the production and accumulation of retroviral proteins results in intracytoplasmic aggregates admixed with ribosomes that are detectable by light microscopy as basophilic inclusion bodies of various sizes. 12
A direct comparison of the proteins in normal and IBDV-infected, inclusion-positive boa livers by SDS-PAGE demonstrated the presence of an abundant IBD-specific 68-kd protein band. Western blots stained with monoclonal antibody 2H2 demonstrated epitopes of the 68-kd IBD protein that appeared to be IBD specific. Immunohistochemical staining of frozen sections of IBDV-infected, inclusion-positive liver sections with 2H2 resulted in specific labeling of IBD inclusions. Antigenically similar material was demonstrated in all size categories of IBD inclusion bodies. None of the baseline biopsy specimens and none of the tissues collected from control or sham-inoculated snakes had inclusions or retrovirus-like particles. No proteins antigenically related to the 68-kd IBD protein were demonstrated in any noninfected boa tissues. The 68-kd IBD protein was thereafter referred to as the IBD inclusion protein.
The genome of retroviruses encodes for several products referred to as PRO, POL, and GAG. The GAG region is transcribed and translated as a 68-kd polyprotein that is posttranslationally cleaved into the matrix (MA), capsid, nucleic acid binding, and other structural components of the mature particle. The MA protein associates with and can covalently bind to cell membrane lipids via a sequence of cationic amino acid residues that is believed to be a highly conserved region among retroviruses. 5 The MA protein is believed to both direct membrane localization of viral particle assembly and form a stable shell-associated layer on the inner surface of the membrane envelope. 5 Recent studies have shown that retroviruses can complete the budding process without the transmembrane envelope proteins, suggesting that it is the core proteins that drive the budding process. 3
The finding of an antigenically unique 68-kd protein within inclusion bodies that appears to be associated with membranes is interesting. In the original description of IBD, the inclusions were identified as aggregates of electron-dense material. 25 Unidentified material characterized as electron-dense subunits were shown budding from cytoplasmic membranes of IBDV-infected cells. 25 The size and morphology of the filamentous structures identified within the larger variants of IBD inclusions characterized in this study closely approximated cytoplasmic membranes. A distinct bilayered structure was evident on close examination. Irregular deposits of granular electron-dense material were evident on the surfaces of the membranelike filaments. The larger variant of the IBD inclusion body may represent an intraphagolysosomal accumulation of IBDV GAG polyprotein and GAG-coated cytoplasmic membranes. If the membranelike structures observed within the large variants of IBD inclusions are indeed GAG-coated membranes, IBDV-infected boa constrictors may be a good alternative (lower vertebrate) animal model for the study of GAG–membrane interactions and the role of these complexes in viral replication and inclusion development. Further work would be required to more specifically identify the 68-kd IBD inclusion protein in IBDV-infected boas.
Because of the potential for IBDV-infected boas to persist as inapparent carriers and the lack of IBDV natural history transmission data, all newly acquired specimens should be quarantined and checked for IBD by liver biopsy before being placed in collections. Minimal biopsy analysis should include histopathology with a thorough examination of the specimen for eosinophilic intracytoplamsic inclusion bodies and cytoplasmic vacuolation. Because many different insults to tissue can cause the formation of similar intracytoplasmic inclusions, inclusion-positive specimens should be subjected to additional diagnostic measures such as TEM before a diagnosis of IBD is made. The highly specific nature of the anti-68-kd IBD inclusion protein monoclonal antibody may make it a useful reagent in the immunohistochemical differentiation of IBD inclusions from other types of nonviral inclusion bodies.
Footnotes
Acknowledgements
We thank K. and C. Pazzi and the Birmingham Zoo for their donation of the boa constrictors employed in this study. We also thank D. Naydan of the UC Davis VMTH Histopathology Laboratory for her assistance and technical expertise with the immunohistochemical procedures. Special thanks is also extended to K. Schwendinger (Consolidated Veterinary Diagnostics, Sacramento, CA) for the production of exceptional slides and histochemical stains. We are grateful to the UC Berkeley animal care staff for the excellent level of animal care and record keeping and to Tom Bednarek at the UTMB, Department of Pathology Imaging Laboratory, for all for his assistance with the illustrations. This project was funded in part by a UC Davis Laboratory Animal Medicine Training Grant awarded by the National Institutes of Health.
